Dichlorofluoromethane: A Chemical at the Crossroads
Historical Development
Dichlorofluoromethane, which many chemists know as R-21 and some still call Freon-21, walks a line between groundbreaking invention and environmental challenge. Engineers in the first half of the twentieth century were hungry for safer, more stable refrigerants. Before chlorofluorocarbons hit the scene, refrigeration used compounds like ammonia and sulfur dioxide—reliable, but not exactly user-friendly or safe. With the arrival of CFCs and HCFCs, of which Dichlorofluoromethane is a member, refrigeration grew both easier and more widespread. Its role in the rise of affordable cooling cannot be understated. By the 1960s, air conditioners, refrigerators, and even fire suppression systems leaned heavily on compounds like Dichlorofluoromethane. The chemical’s story speaks to the double-edged sword of technological progress—every benefit seems to trail some consequence.
Product Overview: More Than a Refrigerant
Looking at Dichlorofluoromethane, it’s tempting to label it as a mere refrigerant. But that misses the breadth of its contributions. Besides cooling systems, it carved out roles as a propellant and as a chemical intermediate. The versatility comes down to stable bonds between carbon, chlorine, and fluorine. That blend made for a substance tough enough to get the job done under a range of pressures and temperatures, without breaking down or catching fire easily. Its volatility and non-flammability led to years of commercial use—demonstrating how chemicals create solutions for safety concerns as well as comfort.
Physical & Chemical Properties
Dichlorofluoromethane shows up as a colorless gas at room temperature, with a faintly sweet smell that’s common among lighter halocarbons. It carries a boiling point just south of room temperature and can transition to a liquid with moderate pressure. In my time handling pressurized cylinders, you notice how these gases chill instantly on release—clearly a sign of the rapid expansion and evaporative cooling that make them so effective for heat transfer. The chemical itself refuses to support flames and doesn’t corrode ordinary metals in its pure form. Its low toxicity in short-term exposures led factory workers to treat it with less caution than many harsher solvents.
Technical Specifications & Labeling
On the technical side, guidance assigns Dichlorofluoromethane the identifier R-21 in the refrigeration world. Labels often carry a warning about ozone depletion and the need for responsible containment. Industry standards go beyond just listing hazards; they highlight handling protocols, emphasizing the need for proper ventilation and the hazards posed by pressurized cylinders. Large storage tanks or drums must employ sturdy valves and pressure relief features to keep shipping safe. International regulation has forced stricter oversight, especially after evidence mounted of the compound’s impact on stratospheric ozone.
Preparation Method
For decades, chemical companies relied on reacting chloroform with hydrogen fluoride while managing reaction temperatures and byproducts very carefully. This process, not exactly benign, ran in reactor vessels lined to resist the corrosive effects of halogens and acids. The production released hydrochloric acid, which adds another layer of complexity in making and handling the substance. My own experience in research settings showed how much effort goes into keeping halogenated intermediates contained and neutralized—not just for worker safety, but for environmental concerns.
Chemical Reactions & Modifications
As a starting point in organic synthesis, Dichlorofluoromethane is like a blank canvas, open to modifications through halogenation or elimination reactions. Chemists have used it to forge more complicated molecules that show up in polymer manufacturing and specialty refrigerants. The attraction, at least before the regulatory clampdown, came from the way this molecule can add or lose atoms under precise conditions, giving rise to either new refrigerants or intermediates for plastics. Those reactions remind me every time how industrial chemistry juggles stability with the need to create materials that are both tough and manufacturable.
Synonyms & Product Names
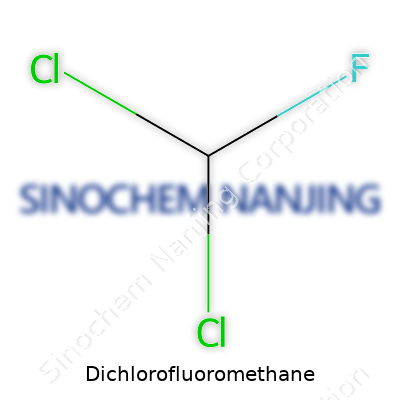
Anyone poking through technical catalogs or regulatory lists might trip across several names for Dichlorofluoromethane. Some trade names feel historic—Freon-21, for example—while the simple R-21 code continues to pop up in HVAC references. Synthetic chemists lean toward the chemical formula, CHCl2F, especially in research papers. Flipping through safety sheets, one finds almost poetic variations on the theme, but they all refer to the same molecular structure and hazards.
Safety & Operational Standards
Safety culture has shifted a lot since the early days of CFCs. Modern protocols demand strong ventilation, sealed systems, and leak detection to avoid anything escaping into the workspace—or worse, outside. I’ve learned firsthand that leaks, even small ones, can trigger alarms or force building evacuations. Direct contact brings risks of mild anesthesia and, at high concentrations, heart irregularities or suffocation. Large-scale operations require strict equipment checks, pressure controls, and mitigation plans for accidental releases. International agreements like the Montreal Protocol have driven companies to scrutinize every step of the supply chain and to certify only those who meet both health and environmental guidelines.
Application Area
Most folks recognize Dichlorofluoromethane from the HVAC industry. Factories also used it as a feedstock for more stable fluorinated chemicals, and spray-can manufacturers once chose it for its tame fire risk and steady pressure. Before global treaties started to phase out ozone-depleting substances, it snuck into an astonishing array of products—from fire extinguishers to blown foam insulation. That tells me just how integral certain molecules can become in daily life. But with that widespread usage comes responsibility for what leaks into the air as those appliances age or break.
Research & Development
R&D doesn’t stand still, even for legacy chemicals. Scientists dug into alternative synthesis pathways and investigated degradation products to determine the least harmful byproducts. The last decades have seen a shift—teams now search for replacements that keep performance but lose the environmental risk. My own university days included studies on new refrigerant classes and testing performance head-to-head, learning just how tough it is to balance thermal capacity, non-toxicity, and environmental safety all at once. Even as regulatory pressure mounts, researchers continue to probe which molecules could do the job without legacy baggage.
Toxicity Research
Early regulatory documents reported low acute toxicity at workplace exposure levels. That encouraged widespread use but led to complacency about chronic impacts and environmental buildup. Over time, studies linked atmospheric breakdown products to ozone layer thinning, allowing more ultraviolet radiation to reach the surface of the earth. My mentor in toxicology hammered home the idea that today’s “safe” may well be tomorrow’s crisis. Chronic exposure, especially in confined spaces, can cause central nervous system effects and alter cardiac rhythms—risks amplified for workers in poorly ventilated areas. Long-term environmental monitoring now pulls back the curtain on previously undetected impacts, feeding back into regulatory cycles and influencing policy.
Future Prospects
Dichlorofluoromethane won’t fade from memory, even if its market shrinks. Stockpiles and aging products still carry traces, creating management responsibilities for old machines and infrastructure. Policy leaders hunt for phase-out paths that don’t force hardship on low-income populations or disrupt critical refrigeration in medical settings. From a chemistry standpoint, the molecule’s design spurs ongoing efforts to use similar compounds as learning tools rather than end-point solutions. The legacy of Dichlorofluoromethane lies as much in what not to do as it does in what can be accomplished. Today, research circles focus on both removing past threats and designing each next generation of refrigerants with full lifecycle scrutiny, blending chemical ingenuity with hard-won environmental humility. The hopes rest on international cooperation, clear-eyed research, and a willingness to overhaul both habits and supply chains.
What is dichlorofluoromethane used for?
What Dichlorofluoromethane Does
Dichlorofluoromethane shows up in a lot of places, but you won’t find it on most household labels these days. Old refrigerators, freezers, and air conditioners used to run thanks to this stuff, known to scientists as R-21, which belongs to the group called hydrochlorofluorocarbons, or HCFCs. It helped cool things down without the same risk of explosions as plain old ammonia or even early chlorofluorocarbons (CFCs).
I grew up in a house where the AC always hummed in the summer—its cool, recycled air carried a particular, almost sweet chill. Many older appliances I tinkered with in college repair shops relied on HCFCs like dichlorofluoromethane. At the time, most people trusted big brands to balance safety and efficiency. Few asked what chemicals cooled the coils.
The Problem with R-21
Today, knowing what we do about climate change and pollution, the story looks different. Some refrigerants, such as CFCs and HCFCs, rise up and break down ozone high in the atmosphere. UV rays start pouring through holes left behind, raising cancer risk, damaging crops, and threatening ocean life. Dichlorofluoromethane sounds technical, but what it really meant was: easy on machines, tough on the environment.
The Montreal Protocol turned this story around. Countries lined up to say enough with ozone-destroyers. According to the United Nations Environment Programme, global consumption of ozone-depleting substances dropped by 98% since 1987. Cleaners, chillers, and aerosols went through an overhaul. In my own backyard, old fridges got hauled off and new, climate-friendlier units rolled in.
Is It Still in Use?
Some industries stuck with dichlorofluoromethane after the risks became clear, because switching to new cooling formulas takes cash and testing. Factories, especially in older buildings or in parts of the world where replacement comes slow, might still have equipment with HCFCs. So there’s a phaseout process, not an overnight switch. The U.S. Environmental Protection Agency tracked leaks and disposal for years to make sure nobody dumped the old refrigerant into landfills or released it in wide open air.
Better Ways Forward
Safer, smarter options exist. Companies push for hydrofluorocarbons (HFCs), which save the ozone but still trap heat in the atmosphere, making them potent greenhouse gases. New ideas, such as using carbon dioxide, ammonia, or even hydrocarbons, trade higher risks for lower emissions if handled right—each brings upsides and new handling rules. Most modern fridges in stores rely on blends that balance safety and climate impact. I’ve repaired enough of them to see the difference—a new compressor with a lower global warming potential (GWP) label usually means less worry, both for people and the planet.
Getting Rid of the Old Stuff
Fixing this isn’t just about swapping one chemical for another. Recycling workers, repair techs, and homeowners need reminders about collecting and disposing of old refrigerants. Laws matter, but training and practical awareness move the dial most. In my experience, jobs that offer the right tools, such as recovery tanks and leak detectors, make it more likely the job’s done safe. Public pressure plays a role, too: more people ask tough questions now, even before buying appliances or hiring HVAC techs.
Staying Aware
Dichlorofluoromethane had its moment because it worked well and seemed safe for years. But the real lesson is about thinking ahead. Whether fixing old equipment or buying new, everyone plays a part by asking what cools their homes—or what lingers in that garage mini-fridge from the 1970s. Each choice adds up, nudging us toward healthier air and climate under the same sky.
Is dichlorofluoromethane hazardous to health?
What Really Lies Behind Dichlorofluoromethane
Anyone familiar with old air conditioning units or vintage fridges has probably encountered dichlorofluoromethane, better known as R-21. This gas once seemed like a miracle — keeping food cold, homes cool, and factories humming. No one wants to demonize the past, but science caught up and forced us to reassess what we breathe and touch. Personal experience tells me, after years working in an HVAC repair shop, the chemicals we once handled without gloves can come around to bite us decades later.
Hazards: More than Just Environmental
A lot of people focus on ozone damage when they talk about refrigerants, but breathing in dichlorofluoromethane or standing in a leaky room does real damage to people. Inhaling this stuff in high concentrations leads to dizziness, headaches, and—on bad days—heart palpitations. The colorless nature of the gas tricks the senses: the room looks safe, the discomfort sneaks up. Chronic exposure over months or years puts extra stress on your liver and central nervous system. Colleagues back in the eighties sometimes shrugged off nosebleeds and nausea, chalking it up to “just part of the job,” until regulations forced protective gear for everyone.
The stats back up our lived experience. The National Institute for Occupational Safety and Health (NIOSH) recognized dichlorofluoromethane as a respiratory hazard, flagging its potential to irritate the lungs. In larger doses, it even pushes oxygen out of the air, which creates a suffocation risk with almost no warning. Some countries banned it outright in consumer products after studies found links to chemical-induced hepatitis and irregular heart rhythms. Even the smallest exposures build up over time, especially for people who handle machinery or chemicals for a living.
Regulation Has Improved Safety—But Not Everywhere
Stronger rules since the late nineties, especially through the Montreal Protocol, cut down on how much of this refrigerant makes its way into homes. Here’s the issue: old buildings and imported equipment still leak. The black market in parts means mechanics sometimes come across vials of R-21 with none of the safety warnings we expect these days. My own shop keeps old warning posters as reminders. Safe handling should never rely on luck.
Protecting Health in Real-World Situations
The answer to the hazard question looks pretty clear once you strip away the jargon. Direct contact or heavy exposure will hurt the body, and even trace amounts aren’t worth the risk. Practical changes start at home and the workplace. People should replace aging air conditioners with modern, safer refrigerants. Companies need to train workers to spot leaks, handle chemicals with the right gear, and never cut corners to save a few bucks. Local governments must fund inspections for public housing and schools, making sure they aren’t hiding leaking pipes behind the walls.
DIY culture makes sense for small repairs, but refrigerants belong in the hands of professionals. Certification programs help, giving workers the know-how to protect themselves and their families. Every year brings a new lesson: short-term savings never justify long-term health problems. Listening to both science and lived experience steers us away from repeating mistakes. This might not make headlines, but it matters a lot.
What are the storage requirements for dichlorofluoromethane?
Looking Beyond the Drum: Safe Storage for an Unusual Chemical
Storing chemicals like dichlorofluoromethane often looks easy on paper, but one misstep can turn costly or dangerous. My work in industrial supply taught me that many folks still underestimate just how unpredictable these compounds get when stored in less-than-ideal spaces. We’re not talking about common solvents here. Dichlorofluoromethane, or R-21, needs a little more respect than a rusty shed or a neglected corner in the warehouse.
Why Storage Rules Matter
Dichlorofluoromethane usually comes in pressurized cylinders or tanks. It’s a colorless, nonflammable gas, which sounds harmless, but leaks often go unnoticed until someone feels dizzy or worse. I’ve seen a seasoned technician open a cylinder too fast without checking for leaks; a sharp, choking odor spread in seconds. That single slip forced an evacuation.
This gas reacts poorly with moisture and high temperatures. Tanks ought to stand in a cool, dry spot, shielded from sunlight and away from any ignition sources even if the fire risk looks low. My old warehouse manager drilled it into us: never store cylinders where heat or mechanical shocks could jostle or over-pressurize them. These basic habits keep both property and health intact.
Letting temperatures swing up and down spells trouble. Hot summers tend to build up cylinder pressure, sometimes beyond safety limits. Direct sunlight does no favors, either. At home, most folks steer clear of leaving gas canisters in the garage for this reason. In industry, the stakes grow; a bursting tank can turn a storage room into a disaster scene.
Ventilation Is No Luxury
Breathing apparatus and leak detectors aren’t just boxes to tick on a checklist. They’ve kept my colleagues and I out of the hospital more than once. Poorly ventilated storage causes gas to collect in low-lying areas, especially since dichlorofluoromethane is heavier than air. The first sign of a problem might be someone fainting. I always favor mechanical ventilation, even if the space “seems big enough.” Relying on open doors or occasional window breeze just invites risk.
Regulatory Guidance: Not Just Red Tape
Occupational health and environmental agencies operate for a reason. OSHA and EPA rules set precise thresholds for safe storage because people have learned the hard way. In my experience, following these requirements keeps surprise inspections uneventful and insurance premiums down. Dedicated signage, regular leak checks, and pressure monitoring bring peace of mind, especially for those working night shifts or off-hours. Keeping safety data sheets (SDS) close at hand isn’t just a formality. More than once, I’ve flipped one open in a hurry.
Finding Safer Ground
Facilities ought to segregate dichlorofluoromethane from incompatible chemicals. Strong acids, alkalis, or oxidizers don’t belong nearby. Solid barricades or dedicated storage cages offer more than security against theft; they create a real barrier when things go wrong. I’ve seen companies cut corners—storing incompatible materials together led, at one site, to a pressurized leak corroding metal shelving and endangering everyone in the vicinity.
Improving on Experience
Better training, regular inspections, and honest hazard communication build the backbone of smart storage culture. Old-timers may say they’ve seen it all, but chemical properties never change to accommodate carelessness. Every time a workplace takes a shortcut, they trade safety for convenience—and if something goes wrong, the bill comes due in injury or lost product.
How should dichlorofluoromethane be handled safely?
Why Dichlorofluoromethane Needs Respect
Dichlorofluoromethane, often called R-21, pops up in refrigeration, air conditioning, and some specialized industrial processes. Using it without a plan risks health and can put the environment on the line. Breathing it in doesn’t just cause minor discomfort—it can result in dizziness, headaches, or much worse. Any kind of chemical with the power to harm air quality and damage the ozone layer deserves careful attention.
Thinking Safety: It’s Not a Guessing Game
Handling this chemical means taking the responsibility seriously from the moment a tank is delivered. Good ventilation stands as the first line of defense. In a cramped or stuffy area, vapors settle in quickly. Workers who use extractor fans or keep air flowing have fewer problems. At my old workplace, we kept garage doors open and maintained exhaust fans—simple steps that let you breathe easier and sleep better at night.
Wearing the right gear counts for a lot. Tough gloves and goggles give decent coverage. Not every job needs a bulky hazmat suit, but skipping protection invites trouble. Dichlorofluoromethane burns skin if released as a liquid, so nobody wants to handle it bare-handed. I’ve seen folks skip goggles, brush their eyes, and regret it minutes later. Always keep a pair of safety glasses on hand—there’s no good excuse for risking your eyesight.
Leaks, Storage, and Spill Response
Leaks happen—valves fail, fittings loosen. The faster you spot a problem, the less damage it causes. Regular checks keep everyone ahead of the danger. Sniffers and electronic detectors work far better than waiting for a whiff of a strange smell. No leak stays small forever if ignored. My old supervisor taught by example: ask questions if you hear a hiss or spot frost, and trust your gut over routine.
Storage gets overlooked far too often. Cool rooms away from sunlight and heat keep the pressure under control. Some forget that a hot day in a metal shed can force tanks to burst. This isn’t melodramatic—it’s happened in shops and warehouses across the country. Bolted racks stop tanks from tipping, and clear labels make it obvious where things sit. Folks who tape over markings or stack cylinders in a corner soon learn why the rules exist.
Training and Accountability
Training works best face to face. Working with a real instructor, learning to fit the regulator, reading the pressure gauge, and tracing a leak beats one-size-fits-all online instructions every time. Rules posted on a wall get ignored if nobody talks about them or explains why they matter. A good foreman knows which person never misses a detail and checks in with new hires.
Disposing of dichlorofluoromethane isn’t just a chore for “someone else.” Cans and tanks must go to certified processors who reclaim or destroy the gas without releasing toxins. Cutting corners leads to fines and worse—chemical leaks cause lasting harm. Local waste authorities give clear directions, so there’s no excuse for dumping.
Better Practices Protect More Than People
The story of dichlorofluoromethane isn’t just about rules on paper—it’s about taking pride in work and caring for both coworkers and the planet. Risking health for a shortcut doesn’t pay off. Simple habits, honest training, and clean storage make a real difference. Concrete action beats wishful thinking every time. Safe handling isn’t fancy; it’s just smart.
What are the environmental impacts of dichlorofluoromethane?
Looking Past the Chemistry: Why Dichlorofluoromethane Matters
Once you start looking into the history of refrigeration, dichlorofluoromethane, better known as HCFC-21 or R-21, shows up pretty often. This chemical helped cool houses, offices, and supermarket freezers for decades. On the surface, it kept food safe and modern systems running smoothly. Underneath, it caused a fair amount of trouble for the atmosphere and more than a few headaches for people invested in long-term environmental health.
Chlorine, Ozone, and Us: The Chemistry Meets the Sky
The main problem comes from chlorine in the molecule. When dichlorofluoromethane escapes into the air, sunlight eventually breaks it apart. Chlorine atoms jump out, reaching up into the stratosphere and tearing apart ozone molecules. Ozone up high absorbs the harshest bits of solar ultraviolet light, protecting crops, animals, and everyone who remembers sunburns that lasted days. The Montreal Protocol recognized this damage in the 1980s, and countries started phasing out many ozone-damaging chemicals, including dichlorofluoromethane and its cousins.
Climate Pressure: Global Warming Hits Home
The trouble doesn’t stop with ozone. Dichlorofluoromethane also traps heat in the atmosphere. Measured over a hundred years, its global warming impact is thousands of times stronger than carbon dioxide, according to the Intergovernmental Panel on Climate Change (IPCC). Factories, repair shops, and even old air conditioners can leak small amounts, adding up to a bigger problem. I’ve seen stacks of worn-out appliances behind local refrigeration shops, waiting for “proper disposal” while gases slowly seep out. It sounds minor until you realize the pound of coolant in a single unit could linger in the air for ten or fifteen years. Multiply that by thousands, and it puts local waste in a global spotlight.
Real People, Real Choices
Communities near manufacturing plants and disposal centers sometimes deal with more than just headlines. Leaked refrigerants can settle in low-lying areas, raising the risk for people with asthma or other conditions. In my own neighborhood, concerns about chemical spills made folks think twice about where to send old freezers and air conditioners. It became clear that handling doesn’t stop at the curb; a safer path means paying attention to the whole life of these materials, from creation through disposal and recycling.
Better Paths and Hard Lessons
Strict enforcement of phase-out deadlines proves essential. The U.S. Environmental Protection Agency banned new production and import of most HCFCs, including dichlorofluoromethane, for uses like new air conditioning equipment. Improved leak detection, incentives for safer alternatives, and technician training all help. Recovery and recycling programs did not get as much attention as front-line regulation, but programs like those by the EPA or the United Nations Environment Programme support safe removal and destruction of gases still out there in old machines.
The story of dichlorofluoromethane is a reminder that even hidden chemicals have long shadows. Real solutions mean thinking about the lifespan of every product, encouraging accountability at every step, and keeping people’s health in the picture. It comes down to smarter design and shared responsibility, because the air over our heads doesn’t belong to any one country, company, or person.
| Names | |
| Preferred IUPAC name | Chlorodifluoromethane |
| Other names |
Freon 21 R-21 Ucon 21 Fluorocarbon 21 |
| Pronunciation | /daɪˌklɔːroʊˌflʊəroʊˈmiːθeɪn/ |
| Identifiers | |
| CAS Number | 75-43-4 |
| 3D model (JSmol) | `3D model (JSmol) string for Dichlorofluoromethane`: `ClC(F)Cl` |
| Beilstein Reference | 1201170 |
| ChEBI | CHEBI:39097 |
| ChEMBL | CHEMBL1376 |
| ChemSpider | 12398 |
| DrugBank | DB14096 |
| ECHA InfoCard | ECHA InfoCard: **100.003.125** |
| EC Number | 200-871-9 |
| Gmelin Reference | 7836 |
| KEGG | C14359 |
| MeSH | Dichlorofluoromethane |
| PubChem CID | 6367 |
| RTECS number | PA7525000 |
| UNII | K50XQU1029 |
| UN number | UN1029 |
| Properties | |
| Chemical formula | CHCl2F |
| Molar mass | 86.468 g/mol |
| Appearance | Colorless gas |
| Odor | Faintly ether-like |
| Density | 1.48 g/cm³ |
| Solubility in water | 16.9 g/L (20 °C) |
| log P | 0.79 |
| Vapor pressure | 3060 mmHg (at 21.1°C) |
| Acidity (pKa) | 15.2 |
| Basicity (pKb) | 13.7 |
| Magnetic susceptibility (χ) | '-81.7·10⁻⁶ cgs' |
| Refractive index (nD) | 1.4242 |
| Viscosity | Viscosity: 0.139 cP (at 25 °C) |
| Dipole moment | 1.60 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 279.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –275.2 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -677.8 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | R03BB01 |
| Hazards | |
| Main hazards | Harmful if inhaled, may cause damage to organs through prolonged or repeated exposure, causes serious eye irritation, may cause respiratory irritation, decomposes on heating to emit toxic fumes. |
| GHS labelling | GHS02, GHS04 |
| Pictograms | GHS04 |
| Signal word | Warning |
| Hazard statements | H220, H280 |
| Precautionary statements | P210, P261, P271, P403 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Autoignition temperature | 648°C |
| Explosive limits | 10.5–15.2% |
| Lethal dose or concentration | Inhalation-rat LC50: 567000 ppm/4H |
| NIOSH | RS1475000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Dichlorofluoromethane is "1000 ppm (parts per million)". |
| REL (Recommended) | 50 ppm (190 mg/m3) |
| IDLH (Immediate danger) | 2000 ppm |
| Related compounds | |
| Related compounds |
Chlorofluorocarbons Difluorochloromethane Chlorodifluoromethane Trichlorofluoromethane Dichloromethane Chloromethane Fluoromethane Carbon tetrachloride |

