An Editorial Look at Dichloroacetic Acid: Legacy, Science, and the Road Ahead
Historical Development
Long before the world started searching for alternatives to petroleum-based chemicals or looking for clever ways to tweak molecules for new cancer therapies, chemists took a close look at compounds like dichloroacetic acid (DCA). Early records point to the late 19th and early 20th centuries as the time DCA first emerged through the work of organic chemists searching for new structures by chlorinating acetic acid. Back then, synthetic chemistry often churned out curiosities before uncovering practical uses. DCA played a backstage role as one of those molecules sitting at the crossroads of industrial chemistry and laboratory experiments. Over time, the industrial age and developments in chemical manufacturing pushed forward more robust methods of producing simple chloro acids, making DCA not just a laboratory milestone, but part of the broader story of how organic chemistry became industrial reality. As scientific focus shifted to understanding biochemical processes, DCA caught attention again, this time as researchers noticed its effect on lactate metabolism, pushing it beyond the boundaries of basic chemical curiosity.
Product Overview
Dichloroacetic acid is a small molecule with a simple structure — two chlorine atoms attached to the methyl group of acetic acid. At first glance, it seems plain compared to more exotic compounds, but its simplicity hides a flexibility that cuts across fields. In labs, technicians have used its sodium salt for decades to adjust pH levels, interfere with certain enzymes, and tweak small molecule properties. This has carried over to industry, where the acid and its salts slip into textile bleaching, pharmaceuticals, and even water treatment circles. It turns out that a stable carboxylic acid like DCA, resistant to some types of breakdown yet reactive enough for chemical transformations, can find itself pulled in many directions. Rather than making bold headlines, DCA quietly factors into protocols and research plans worldwide.
Physical & Chemical Properties
Dichloroacetic acid comes off as a colorless, pungent liquid or a white solid at room temperature, depending on its degree of purity and storage. It carries that characteristic smell, something like vinegar but much sharper — a reminder that chemical composition does more than determine reactivity; it dictates how we experience a substance physically. Its relatively low boiling and melting points, coupled with high solubility in water and many organic solvents, make it easy to handle in reaction vessels. These practical features speak volumes for chemists focused on process efficiency. The chlorine atoms on the molecule crank up its acidity far above that of acetic acid, bringing its pKa down, a detail that sits front and center during buffer preparation and enzyme inhibition work.
Technical Specifications & Labeling
Scientists working with DCA usually demand high purity, especially in research or healthcare settings. Labels on containers list assay percentages, trace impurities, handling recommendations, and container materials. Reputable chemical suppliers continuously analyze batch-to-batch purity and water content, as moisture can alter reaction outcomes. In the industrial world, those technical points translate into specific requirements for safe storage, typically in cool, well-ventilated areas, well away from common reactants like metals and oxidizers. Regulators across different regions often reference international standards, underscoring the significance of clear, accurate labeling.
Preparation Method
Industrially, most DCA comes from the chlorination of acetic acid or its derivatives. Despite sounding straightforward, the process tends to demand precise control over reaction conditions. Over-chlorination risks forming trichloroacetic acid, while under-chlorination leaves unreacted starting material. Commercial producers tweak temperature, chlorine flow rates, and sometimes employ catalysts to hit the sweet spot. This process must also account for the byproducts — they affect purity and, ultimately, which applications the final acid suits best. The story of DCA’s preparation, from early bench-top work to streamlined industrial production, highlights an often overlooked truth: chemistry relies as much on control and reproducibility as it does on discovery.
Chemical Reactions & Modifications
Dichloroacetic acid stands out for the way those two chlorines onboard make the molecule primed for substitution reactions. Chemists often replace the chlorines or use DCA as a building block to create other chlorinated compounds. In the lab, the acid finds itself used as a precursor or a reagent in condensation reactions, esterifications, and more. The strong electron-withdrawing effect of the chlorines influences any reaction that involves the molecule, whether it’s breaking it down or combining it with something new. This is where DCA’s character becomes evident — not just as a static product but as a tool for creative molecular engineering. These transformations open doors to customized chemical syntheses, pharmaceuticals, and advanced materials research.
Synonyms & Product Names
DCA goes by many names, and that sometimes creates confusion for newcomers or students starting out. “Dichloroacetic acid” serves as the systematic label. Sometimes, folks in the lab call it “DCA,” while chemical catalogs list its sodium salt as “sodium dichloroacetate.” Older literature may use “bichloroacetic acid,” though this has fallen out of fashion. Regardless of the name, the molecule remains the same — a reminder that communication and clarity matter as much as chemical skills in science.
Safety & Operational Standards
Working with DCA calls for vigilance, especially given its corrosive nature and its ability to irritate the respiratory tract, skin, and eyes. Most laboratories rely on gloves, eye protection, and fume hoods as routine barriers against accidental exposure. In high-volume industrial settings, additional containment — like local exhaust ventilation and closed systems — cuts down on the potential for leaks or vapor build-up. Regulators require safety data, and in research, safety protocols nearly always get reviewed before a project gets underway. Long ago, such caution wasn’t automatic — it came from hard lessons and long experience.
Application Area
DCA’s reach crosses into several sectors. In the clinic, it’s been tested for rare metabolic disorders and as a candidate for cancer therapies focused on shifting cell metabolism. Though not a silver bullet and often kept to experimental contexts, researchers cling to optimism, thanks to DCA’s ability to tip the balance between lactic acid production and aerobic metabolism. The chemical industry uses it in synthesis — a stepping stone for pharmaceutical compounds and specialty chemicals. Environmental applications include water treatment, where its strong acidity and reactivity can play a role, although byproduct toxicity and regulations demand strict oversight. DCA also appears as a trace disinfection byproduct in chlorinated drinking water, bringing attention to public health monitoring efforts.
Research & Development
Researchers continue to test the boundaries of what DCA can do. Universities and private labs look hard at DCA as a metabolic regulator, especially in animal models of cancer and metabolic disease. Biochemists drill into the way DCA inhibits specific enzymes like pyruvate dehydrogenase kinase, swinging energy production pathways back toward normal in dysfunctional cells. Some teams experiment with modifications — swapping functional groups, changing salt forms — hoping for molecules with stronger therapeutic impact and fewer side effects. Beyond medicine, synthetic chemists explore novel reactions, pushing DCA’s role as a reagent. Some attention has turned to greener synthetic routes, lowering waste and minimizing energy use in chloride chemistry.
Toxicity Research
Toxicologists have scrutinized DCA from multiple angles, particularly given its appearance as a contaminant in chlorinated water supplies. Studies show chronic exposure links to liver toxicity and even potential carcinogenicity in animal models. Regulatory agencies set exposure limits, especially in drinking water contexts, to reduce risks to human health. Some evidence connects high doses to nerve and liver problems, driving calls for tight control in both clinical tests and environmental policy. This underscores a key principle — risk always follows utility, and ongoing toxicological research remains essential as new uses for DCA get proposed.
Future Prospects
Looking toward the future, the fate of dichloroacetic acid will probably hinge on balancing its practical strengths with responsible management of risks. Most excitement in recent years comes from emerging medical research — if DCA-based treatments prove safe in humans, that could unlock therapies for metabolic diseases and certain cancers. On another front, industrial chemists continue to seek cleaner production routes and safer byproduct handling, aligning with the push for sustainable chemistry. Monitoring in water systems will grow tighter as standards change. The story of DCA shows that even simple compounds, once overlooked, can pivot toward new importance as science and industry evolve together. Careful attention to health, safety, and environmental stewardship will shape whether DCA remains just another tool, or proves pivotal in solving modern challenges.
What is dichloroacetic acid used for?
A Closer Look at Dichloroacetic Acid's Roles
Dichloroacetic acid caught my attention during my early days in medical research labs. It’s not a name you hear every day, but its uses pop up in some pretty important places. The compound’s primary hook? Its ability to alter how cells make and use energy. That property sits at the center of why so many scientists, doctors, and engineers want to learn what it can do.
Medical Research and Experimental Treatments
Years of research have pointed to dichloroacetic acid for potential cancer treatments. Early studies suggest it may prompt certain cancer cells to self-destruct by shifting their metabolism back to a more “normal” state. Cancer cells tend to thrive by burning glucose in a way healthy cells don’t, using a process called the Warburg effect. This chemical seems to push them out of that state, making it harder for tumors to grow as aggressively. Researchers at several universities have run clinical trials and lab tests, but the road to using this compound as an approved medicine includes hurdles over safety, dosing, and potential long-term effects.
Industrial and Laboratory Applications
Outside the medical spotlight, dichloroacetic acid plays a part in organic synthesis. Chemists reach for it during the creation of certain drugs, pesticides, or specialty chemicals. Its strength as an acid and its unique structure mean it can help build more complex molecules. In my experience running industrial chemical tests, we used dichloroacetic acid to adjust pH in reactions where other acids just didn’t hit the mark. The ability to fine-tune conditions during synthesis can make a huge difference between a clean reaction and a wasted batch.
Environmental and Analytical Uses
This compound also shows up in water testing labs. Measurement of dichloroacetic acid usually serves as a flag that chlorinated drinking water could contain byproducts you might not want to see. In the process of disinfecting drinking water, especially with chlorine, a variety of new chemicals form. Some of these, like dichloroacetic acid, end up as part of the residue. Regulatory bodies track such compounds closely, both to protect public health and to measure the effectiveness of water treatment plants.
Risks and Responsible Handling
While dichloroacetic acid holds promise and utility, it also brings risks. It can cause burns or respiratory irritation, and animal studies point to possible liver and nerve side effects. In any lab or plant setting, gloves, goggles, and a ventilated workspace shape safe habits. On top of the usual lab protocols, researchers always track exposure levels and document symptoms. There’s been some online chatter about this chemical as a “miracle cure,” but the science doesn’t support unsupervised use. Anyone feeling tempted to try it for health reasons needs to talk to a qualified professional, not just trust Internet rumors.
Finding the Right Balance
Dichloroacetic acid sits at an interesting crossroad between science and safety. The biggest challenge comes down to drawing clear lines around where it helps and where it can harm. As deeper studies unfold, researchers should keep safety front and center, and regulators must update rules as new facts surface. In practice, that means more independent trials, better ways of monitoring exposure in labs, and honest communication about what this compound can and can’t do. The promise of dichloroacetic acid lies in its potential, but the value depends on how thoughtfully people put it to use.
Is dichloroacetic acid safe to use?
Understanding What Dichloroacetic Acid Is
Dichloroacetic acid (DCA) sounds technical, but it’s a small molecule, close cousin to acetic acid—the stuff in vinegar. Chemists first started paying attention to DCA because it breaks down easily in water and doesn’t stick around in the environment like other chemicals. That fact alone grabs the attention of researchers interested in less persistent pollutants. Over recent years, it’s made headlines in scientific circles for potential roles in medicine, but the safety story deserves a closer look.
What Science Says About Exposure Risks
DCA is no stranger to labs and water treatment plants. It pops up as a byproduct of chlorinating drinking water, so trace levels end up in tap water. Studies by the U.S. Environmental Protection Agency flagged DCA alongside other disinfection byproducts as a possible health risk with long-term exposure. In animals, long-term high-dose DCA exposure can cause liver toxicity, nerve damage, and even cancer. Researchers watched for changes in liver enzymes, shifts in metabolism, and wear-and-tear on heart tissue. At lower doses, results become less clear-cut, but caution wins out—as with anything that messes with key organs.
In medical experiments, some scientists tested DCA as a drug to alter metabolism in cancer cells. Those studies involved people with serious illnesses, not the general public, and under strict supervision. Patients dealt with potential nerve problems, upset stomach, and, at high doses, signs of liver stress.
Why This Matters for Everyday Life
For most folks, DCA shows up in life through tap water. Municipal utilities keep a close watch on levels, setting strict safety thresholds based on data from animal studies and human monitoring. The EPA capped it at 0.07 milligrams per liter for total haloacetic acids, DCA included. Most cities come in under that number. Still, people in areas with older water systems sometimes get a spike in these byproducts, especially if heavy chlorination kicks in during the summer. It makes sense to check your local water report, especially if you’re on a private well or using an old plumbing system.
Practical Steps and Talking Solutions
Home filters using activated carbon step in to remove much of the DCA and other byproducts from drinking water. Regular maintenance matters—old filters harbor their own problems. Some people opt for reverse osmosis filters for a bigger safety net, though this approach costs more and wastes some water. From a wider perspective, utilities continue to refine water treatment methods to chop down on unwanted chemicals, experimenting with alternative disinfectants or changing the point in the process when chlorine gets added.
On the medical front, no over-the-counter DCA products have the green light from the FDA or other regulators. Any seller making big claims about DCA cures risks legal trouble and, even more, dangers for unsuspecting buyers. If a doctor isn’t involved, don’t trust DCA as safe. That lesson goes for most substances under medical study—lab results don’t always make a safe, effective product for everyday folks.
Staying Informed and Making Safe Choices
Learning more about what comes out of the tap brings peace of mind. Checking local reports or asking questions at your water utility opens the door for informed decisions. If odd flavors, colors, or smells show up, don’t guess—ask for a test. Reliable information beats rumor every time. For any health treatment, nothing replaces professional advice and scientific studies reviewed by experts.
Safety with DCA, as with many chemicals in the modern world, comes down to respecting limits, listening to unbiased science, and asking the right questions—whether you’re pouring a glass of water or considering something riskier.
What are the side effects of dichloroacetic acid?
What Happens After Exposure
Dichloroacetic acid (DCA) found its way into scientific circles mostly for its potential in cancer treatment research. You hear about it now and then, mostly out of curiosity, but few people really sit with the side effects and consider what it means for people’s bodies. Both laboratories and some hopeful patients tried DCA for various reasons, hoping the benefits would outweigh what comes after swallowing or injecting it.
Short-Term Reactions: No Hiding from the Basics
On the surface, you’ll notice a few immediate reactions after DCA enters the body. Many report stomach pain, nausea, and sometimes diarrhea. That’s pretty typical with chemicals that interact with digestion. DCA isn’t candy. Some people talk about feeling tired, almost as if the energy gets sapped right out of their muscles. Studies trace this to DCA interfering with normal metabolism, leading to that fatigue.
As with any acidic substance, a taste or accidental skin contact can burn. If someone breathes in vapors accidentally in a lab setting, coughing or a scratchy throat follows, showing the body quickly reacts to foreign chemicals.
Nerve Trouble Over Time
The longer a person takes DCA, the more complicated things get. One main concern: nerve problems. There’s a medical term for it—peripheral neuropathy. Picture numb toes, fingers that tingle for no reason, or a burning pain in hands and feet. People using DCA long-term, even at doses much lower than many cancer trials, have run into this problem.
Researchers from the University of Alberta noticed this in their clinical studies. Volunteers who took DCA over weeks began losing sensation or coordination. The nerves seemed slower to heal, and sometimes the symptoms stuck around even after people stopped using the chemical. For me, seeing this side effect hit patients who started optimistic about experimental therapy hammers in how all medications bring unpredictable trade-offs.
Liver Concerns: Silent but Serious
DCA travels to the liver almost instantly, where it gets broken down. Not everyone responds the same. Some people’s livers work harder and start showing signs of distress: elevated enzyme levels in bloodwork—early warning signs for bigger liver problems. Jaundice, though rare from DCA alone, can develop if use goes unchecked or the body lacks robust detox systems.
Anyone thinking about experimental treatments has to face the ugly truth: the liver rarely complains right away. But months down the line, damage can sneak up, especially when the drug isn’t regulated or the user isn’t under close doctor watch.
What the Data Shows
The World Health Organization and several clinical trial summaries document that DCA use can compromise brain function, too, especially when dosed too high. Forgetfulness, trouble speaking, or trouble focusing crept into patients’ reports. Researchers propose that this happens because of DCA’s effect on how nerve cells process energy.
Making Safer Choices
If someone asks me about DCA, I tell them to treat it with the same caution they’d show any substance that changes how their body works. Only take it under medical supervision. Watch for numbness or unexplained pain, and don’t brush off a sour stomach or fatigue as just a bad day. Focusing on regular blood tests and nerve checks can catch trouble before it turns serious. If you ever consider DCA after seeing an online ad or reading the next “miracle cure” story, ask for proof and talk face-to-face with a doctor. Only then can you really weigh the benefits and risks for your own health.
How is dichloroacetic acid stored and handled?
Why Storage Matters
Any time you deal with dichloroacetic acid, you realize instantly that safe practices aren’t optional. One careless slip can leave you with burns or set off a release that lingers in the lab. This acid comes with a bit of attitude — it’s volatile, loves moisture, and gives off fumes you really don’t want hanging in the air. My years around chemical stockrooms have shown me that the smallest misstep—like leaving the container even a little loose—invites headaches nobody wants. Proper storage saves skin, air, and peace of mind.
The Reality of Safe Storage
Glass and plastic containers both see use, but glass wins out since dichloroacetic acid doesn't always play nice with plastics over time. Screw tops with Teflon-lined caps keep the fumes and acid where they belong. I’ve seen colleagues improvise with parafilm, which only works as a last resort. My advice? Don’t skimp on the real supplies. Those fumes can corrode cheap hardware and make things go south fast. Keep the containers upright, labelled in bold, and set above drip trays, just in case.
No acid should ever cozy up to a sunny window or a heat source. Store it below 25°C, or lower in warm climates. Out-of-control temperatures lead to pressure build-up and, sometimes, pops you’d rather avoid. I tell new staff: check that chemical fridge every day, and don’t ever let food sneak in. Cross-contamination wrecks fridges and reputations alike.
Handling and Personal Safety
Grab a bottle, and already you’re eyeing the safety goggles, face shields, and gloves. I’ve felt that stinging fog from a careless pour, and trust me, once is enough. Nitrile or neoprene gloves outlast latex here. Long sleeves help, but even the steadiest hands slip now and then, so always tie up sleeves and double-check for holes. Fume hoods are your friend. Ventilation pulls those harsh fumes away from faces, and I’ve watched lab mates get lazy and regret it later. Even a small spill on the skin can turn painful in under a minute, so a safety shower or eyewash within arm’s reach isn’t optional.
Transferring acid into smaller bottles starts with planning. I’ve seen more than one rookie rush this and splash it everywhere. Move slow, pour steady, and always—always—label everything immediately. Confusing one clear liquid for another sets the stage for trouble down the line.
Disposal Doesn’t End at the Sink
Ditching old dichloroacetic acid can’t happen in ordinary drains. I once had to deal with fumes backing up into a lab after a careless pour; it nearly triggered an evacuation. Neutralize the acid in a dedicated container, then send it to a chemical disposal team. Waste logs matter a lot. Regulators don’t take kindly to forgetting signatures or missing information.
Raising the Bar
Set routines save labs and people. Training keeps everyone sharp: regular safety drills, policy refreshers, and honest conversations about near-misses all help. If you spot a crack in a bottle or a sticky cap, flag it fast and replace it. This acid has earned real respect. If more workplaces raised their standards, stories of burns and fumes would show up less often. I always tell newer folks: skip shortcuts, and you finish every shift healthy.
Can dichloroacetic acid be used for cancer treatment?
Why Dichloroacetic Acid Got Attention
For years, cancer has pushed people toward unconventional approaches. Dichloroacetic acid, or DCA, has stirred hope in corners of the medical world. Reports of DCA shrinking tumors in lab animals made waves. Early cell studies suggested DCA could help target how cancer cells create energy, which sounded like the Holy Grail to folks grasping at any real improvement in outlook.
Understanding the Science
Cancer cells tend to switch their energy production to a process called glycolysis, even with plenty of oxygen around. This switch, known as the “Warburg effect,” allows rapid growth. DCA, a simple chemical, flips a switch inside those cells, nudging their powerhouses, the mitochondria, back into action. When this happens, it’s thought the cancer cell becomes less aggressive or might even die.
Holding Out for Reliable Evidence
I remember people passing around stories featuring DCA as a do-it-yourself cure. Searching the Internet turns up desperate talk on forums, showing just how hungry people are for options that dodge chemotherapy’s side effects. Yet, university labs performing animal studies don’t translate into treatments you can count on in real life.
Several early-stage clinical trials looked at DCA in people with advanced brain cancers and other tough diagnoses. Researchers ran into hurdles. DCA can cause nerve damage and mess with liver function. Some patients in these studies got side effects like numb fingers and toes or feeling shaky on their feet. The science community hasn’t gathered enough long-term, controlled data in larger groups to show clear survival benefits or a broad safety profile.
Who Stands to Gain—and What’s Stopping Progress
Desperation lays traps. Cancer patients, feeling let down by standard care’s limits, chase unproven fixes. Over-the-counter DCA, sold on unregulated websites, skips the check-ups that keep buyers from real harm. No two online batches match. The risks can pile up with other medications or existing health problems. Family members ask whether to trust testimonials or scientific process; the answer never really gets easier when time feels short.
Doctors have the training and experience to weigh new research, but prescribing DCA lies outside current cancer care. Medical boards and regulatory agencies ask for proof—proof that outweighs known risks, proof that stands the test of controlled, peer-reviewed trials. Stories about DCA working for a neighbor spread fast; retractions or studies showing mixed results rarely reach the same ears. This gap keeps lives in limbo.
More Than a Magic Bullet
Living with cancer often means sorting hope from hype. DCA reminds us how complicated this dance becomes. Medical science marches step by step, not in big jumps, because patients deserve treatments that help more than they harm. Real breakthroughs take time. Teams at big cancer centers won’t write off new angles like DCA—but new angles require tough, expensive proof.
Better oversight could stop dangerous shortcuts and provide clear guidelines. Expanding access to clinical trials and transparent reporting on outcomes, good and bad, may help families decide their next move without falling victim to empty promises. For now, DCA highlights both the drive to conquer cancer and the cost of chasing shortcuts without enough evidence in hand.
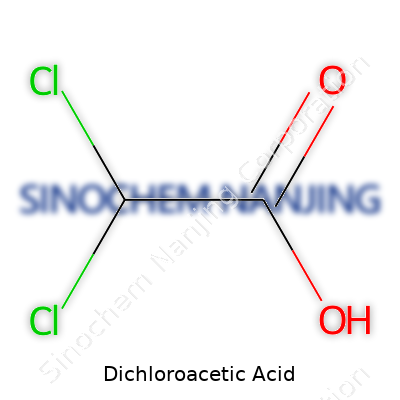
| Names | |
| Preferred IUPAC name | 2,2-Dichloroacetic acid |
| Other names |
Dichloroethanoic acid Dichloroacetic acid DCAA Bichloroacetic acid Acetic acid, dichloro- UN 1764 |
| Pronunciation | /daɪˌklɔːroʊəˈsiːtɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 79-43-6 |
| Beilstein Reference | 1209221 |
| ChEBI | CHEBI:28441 |
| ChEMBL | CHEMBL15737 |
| ChemSpider | 6199 |
| DrugBank | DB03431 |
| ECHA InfoCard | 03a1d5d2-3453-4065-b64d-5d27edcbc8fd |
| EC Number | 200-871-4 |
| Gmelin Reference | 1648 |
| KEGG | C01758 |
| MeSH | Dichloroacetic Acid |
| PubChem CID | 3039 |
| RTECS number | AJ9625000 |
| UNII | 1CB5X020QU |
| UN number | UN1764 |
| CompTox Dashboard (EPA) | DTXSID2020102 |
| Properties | |
| Chemical formula | C2H2Cl2O2 |
| Molar mass | 128.944 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 1.57 g/cm³ |
| Solubility in water | Very soluble |
| log P | 1.48 |
| Vapor pressure | 14 mmHg (20 °C) |
| Acidity (pKa) | 1.35 |
| Basicity (pKb) | 1.36 |
| Magnetic susceptibility (χ) | -29.7×10^-6 cm³/mol |
| Refractive index (nD) | 1.424 |
| Viscosity | 1.25 mPa·s (25 °C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 165.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -447.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -711.5 kJ/mol |
| Pharmacology | |
| ATC code | D08AX01 |
| Hazards | |
| Main hazards | Corrosive, harmful if swallowed or inhaled, causes burns to skin and eyes, may cause respiratory irritation, suspected carcinogen. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H351 |
| Precautionary statements | P260, P264, P270, P271, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P403+P233, P405, P501 |
| Flash point | > 98 °C |
| Autoignition temperature | > 440 °C |
| Lethal dose or concentration | LD50 oral rat 1217 mg/kg |
| LD50 (median dose) | 1,210 mg/kg (rat, oral) |
| NIOSH | AJ8750000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 ppm (5 mg/m³) |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Chloroacetic acid Trichloroacetic acid Monochloroacetic acid Dichloroacetate Acetic acid |

