D-Mannitol: A Deep Dive into Its Journey, Properties, and Horizons
Historical Development
D-Mannitol has a story going back hundreds of years. Originally, folks discovered it in natural sources like seaweeds and certain plants, making it one of the oldest sugar alcohols in both Eastern and Western medicine. By the 19th century, chemists isolated and named the compound after the manna plant, drawn by its sweet taste and curious physical form. Industrial production started booming in the 20th century when ways to extract or synthesize mannitol became more efficient, reshaping how it’s used in pharma, food, and chemical fields. The compound appears in the history of intravenous therapies, and anyone working in healthcare will remember mannitol’s longstanding role in acute care settings, especially for treating brain swelling and kidney issues.
Product Overview
D-Mannitol is one of those ingredients that shows up in all sorts of industries. People come across it in chewable tablets, candies, gum, and some health foods. Its sweet profile means it can replace sugar without causing a spike in blood sugar. It’s found in everything from cough syrups to processed snacks. Many folks—myself included during pharmacy rotations—have seen D-mannitol prescribed as an osmotic diuretic, drawing fluid out of tissues. The powdery, crystalline texture makes it easy to use in mixes, and it doesn’t break down under most cooking or processing temperatures.
Physical & Chemical Properties
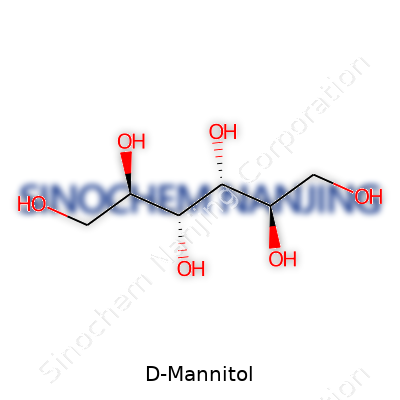
On the physical side, D-Mannitol forms a white crystalline powder or granules, doesn’t have a strong odor, and gives a cooling sensation on the tongue. It has a melting point around 165-168°C and dissolves in water, but not nearly as well in alcohol or most oils. As for the chemical makeup, the formula is C6H14O6, and as a hexitol, it holds six carbon atoms with plenty of hydroxyl groups—great for chemical modifications or reactions. The stability keeps it from reacting much under normal storage conditions, which explains why it lasts so long and is handy in many manufacturing processes.
Technical Specifications & Labeling
Manufacturers offer D-Mannitol with set criteria: high purity (often above 98%), fine particle size grading, and low levels of residual solvents or impurities. Labels must give clear ingredient lists, the source (natural or synthetic), and allergen declarations if required. Pharmaceutical and food packaging usually carry batch numbers, production dates, country of origin, and health warnings for those with rare hereditary sugar alcohol intolerance. Over the years, labeling standards have tightened, thanks to increased consumer scrutiny and tougher regulatory checks in both food and medicine.
Preparation Method
Historically, people extracted D-Mannitol from natural plant saps and seaweed, but today, most production relies on catalytic hydrogenation of fructose and glucose solutions, often sourced from starch hydrolysates. Some setups use microbial fermentation, harnessing the enzymatic power of fungi or bacteria, which appeals to companies chasing sustainable or non-GMO claims. Large industry reactors control temperature, pressure, and hydrogen flow to fine-tune the yield, and I’ve seen how a small slip in these controls can throw off purity or crystal form—a headache for downstream uses.
Chemical Reactions & Modifications
Truth is, D-Mannitol has plenty of chemistry under the hood. Chemists can oxidize it to produce mannose or use it to make certain specialty polymers. In synthetic organic chemistry, it steps in as a chiral auxiliary or a protecting group for aldehydes and ketones. Chemical reactions typically focus on the abundant hydroxyls, either for esterification, etherification, or even to prepare halogenated derivatives. Industrial setups limit these modifications due to cost, but academic labs keep exploring new routes for drug development or advanced materials, which could change the commercial outlook down the road.
Synonyms & Product Names
The world knows D-Mannitol by different names: manna sugar, mannite, osmitrol (in medical use), and E421 (as a food additive in the EU). For those working in pharmacy and food science, all these names turn up with regularity on product sheets, regulatory filings, or hospital dispensation lists. With more countries standardizing codes and additive names, tracking the stuff across markets grows easier, but old-school labels still create confusion for people switching between regions or industries.
Safety & Operational Standards
Safety with D-Mannitol starts with clear workplace guidelines, such as dust control, personal protective equipment, and spill management to avoid respiratory irritation. Storage away from moisture limits caking and keeps it safe for months—sometimes years—if sealed tight. Food and drug quality mannitol must obey global codes like USP, EP, JP, or national food safety acts. Auditors in food and pharma won’t let shipments in without proof of compliance with thresholds for heavy metals, microbial count, or residual solvents. Training and automated controls cut down the risk of mistakes, and near misses—having seen one too many—show why solid operational routines matter more than anything written on paper.
Application Area
Use cases for D-Mannitol stretch further than most expect. In the food world, it turns up in sugar-free gum, low-calorie chocolates, and diabetic snacks as a bulking agent. Bakeries that want to avoid sticky pastries often turn to mannitol since it resists browning and keeps moisture low. Pharmacies and hospitals rely on intravenous mannitol drips to treat brain injury, increase urine output, and fight glaucoma. Labs use it for culturing bacteria or customizing drug delivery vehicles in research. Construction materials and specialty coatings, surprisingly, also draw from mannitol for niche plastics or adhesives. Every industry finds a different way to solve persistent problems with this unassuming polyol.
Research & Development
Active research looks well beyond food or medicine. Teams explore nano-D-mannitol as a carrier for targeted drug delivery and develop new polymers that could cut environmental impact. Researchers tackle tricky stability questions in combination medicines, especially for sensitive drugs that break down when mixed with other excipients. There’s also work on greener production methods—think fermentation with engineered microbes, which could bring prices down and sidestep reliance on fossil-fuel-derived starting materials. I’ve watched university-industry partnerships try to turn research wins into commercial processes, but scaling these ideas sometimes runs into old bottlenecks like supply chain interruptions or unpredictable yields.
Toxicity Research
Through decades of studies, D-Mannitol comes across as safe for most people in moderate quantities, passing through the digestive system largely unmetabolized. High doses can cause laxative effects, and there are case reports of rare allergic reactions in sensitive individuals. Hospitals keep an eye out for kidney overload or electrolyte imbalances when administering high-dose IV mannitol. Animal studies focus on chronic exposure and mutagenicity, with results showing low toxicity profiles. Regulatory bodies like the FDA and EFSA set maximum daily intakes based on human and animal data, helping prevent misuse. Transparency around side effects remains a challenge, as people with digestive disorders face higher risk and can’t always find clear warnings on supplement labels.
Future Prospects
The landscape for D-Mannitol keeps shifting. Interest grows in using it for advanced medical treatments, including targeted brain therapies where mannitol can open the blood-brain barrier, letting medication slip through more easily. Demand increases in health-conscious consumers looking for low-calorie, tooth-friendly sweeteners without aftertaste. Chemical companies invest in greener manufacturing, and researchers chase better synthetic methods that cut waste. Regulatory attention ramps up on labeling accuracy, ensuring vulnerable groups—such as those with hereditary metabolic diseases—get the information they need before consuming products. As more discoveries come out of university labs, chances grow for innovative applications in areas like bioplastics, specialty coatings, and even climate-friendly farming aids. Open communication between industry, scientists, and consumers will shape how D-mannitol moves into the spotlight as an old staple finding new relevance.
What is D-Mannitol used for?
How D-Mannitol Finds Its Way Into Medicine Cabinets
D-Mannitol sits on pharmacy shelves for reasons most people don’t notice. This sugar alcohol goes beyond sweetening sugar-free gum. In hospitals, D-Mannitol serves as a tried-and-true diuretic, meaning it helps the body flush out extra water. Many of us have crossed paths with D-Mannitol in some form, especially if we or someone we know has spent time in the hospital for brain swelling or kidney trouble. Nurses and doctors turn to it during emergencies when lowering pressure inside the skull or eyes. Think of injuries, strokes, or serious infections where every second can matter. Its ability to pull water out of tissues and into urine makes it indispensable there.
Careful Solutions for Kidney Patients
Dialysis patients sometimes need a helping hand. D-Mannitol can enter the picture to help protect the kidneys during tough times, especially in acute kidney failure or before surgery when surgeons risk clogging the kidneys with old blood. It’s been part of the toolkit for decades and remains useful in specific situations, but not without debate among kidney experts. Overuse can tip someone from hydrated to dangerously dehydrated. So, getting the dosage right isn’t just guesswork—it’s a skill that takes judgment and training.
Everyday Uses That Hide in Plain Sight
Most people eating sugar-free treats don’t realize those foods sometimes use D-Mannitol. Ice cream, chocolate, cough syrup, chewable vitamins, and even toothpaste may rely on it. The reason? It’s sweet without spiking blood sugar, so people with diabetes get a pleasant taste and fewer surprises at glucose checks. Dentists like it, too, because bacteria don’t break it down as easily, cutting down on cavity risk. If you have eaten a mint that cooled your mouth but didn’t taste cloying, D-Mannitol may well have played a part.
The Gut: Friend or Foe?
There’s a catch with sugar alcohols, including D-Mannitol. Eat or drink too much, and cramping and diarrhea follow. Pharmacies print warnings on products for a reason. Kids and adults with delicate stomachs often notice effects sooner than others. For people with irritable bowel syndrome, just a small serving can turn into hours of discomfort. So, anyone new to these products should start slow and pay attention to what their body says.
Safety, Science, and the Need for Vigilance
D-Mannitol has beaten the test of time because doctors and chemists keep asking questions about safety and effectiveness. Most people tolerate it just fine in low doses, and the U.S. Food and Drug Administration lists it as generally recognized as safe. For doctors, knowing the signs of overuse or allergic reactions means the difference between a successful treatment and a setback. Public health experts look out for hidden sugars and encourage food labeling so people understand what’s in the foods they eat.
Better Education and Smarter Choices
Teaching medical students, dietitians, and patients the facts behind D-Mannitol helps prevent mistakes. Clear labeling, honest guidance at the doctor’s office, and practical choices at the grocery store make a difference. Solutions start with informed people—a lesson every healthcare system can stand to remember as new ingredients find their way into medicine and onto our plates.
References: FDA: GRAS Notice Inventory for Mannitol; National Kidney Foundation: Mannitol Use in Renal Patients; Journal of Critical Care: Mannitol in Neurosurgery; Mayo Clinic: Sugar Alcohols in Food Products.
Is D-Mannitol safe for human consumption?
What is D-Mannitol?
D-Mannitol often pops up on the back of snack packaging or as a white, sweet powder in lots of foods and pharmaceuticals. It’s a type of sugar alcohol, like xylitol or sorbitol, and manufacturers use it as a sweetener, stabilizer, or to add a cooling sensation in minty products. Doctors and pharmacists know it well as a diuretic or to reduce pressure in the brain for certain medical cases.
How the Body Handles D-Mannitol
The body doesn’t process D-Mannitol in the same way as glucose or other sugars. Most of it passes through the intestines without much change, which means it produces fewer calories than regular sugar. The kidneys help filter it out, and it usually comes out in the urine. For most healthy adults, this process doesn’t cause many issues. But anyone with kidney problems should probably check with their doctor before eating large amounts on a regular basis.
Research on Safety
Plenty of scientific studies support the safety of D-Mannitol in moderate amounts. The U.S. Food and Drug Administration (FDA) classifies it as “generally recognized as safe” (GRAS). So do the European Food Safety Authority and other major regulators. Decades of studies show little risk for most people when they eat mannitol in small to moderate quantities.
Some people have spent years handling drugs and dietetic products, and D-Mannitol comes up again and again as an ingredient. No horror stories stand out in the medical literature about this additive causing lasting harm to ordinary folks who are not allergic or who don't have certain bowel diseases.
Common Side Effects
Anyone who’s eaten a sugar-free candy binge knows the warning sign: “Excess consumption may cause a laxative effect.” Sugar alcohols push more water into the colon, making you head for the bathroom if you take in too much. The same goes for D-Mannitol. Most people can handle around 10-20 grams a day without any issue, but go above that, and gas, bloating, and diarrhea show up. Kids, folks with irritable bowel syndrome, or anyone with sensitive digestion should keep an eye on the serving size.
Allergies and Special Populations
Allergies to D-Mannitol turn up only rarely. For those with hereditary fructose intolerance, even small amounts can spell trouble. People on certain medications or with chronic kidney disease should double-check with their physician if mannitol’s in their diet or prescribed for medical reasons. In the hospital, IV mannitol sometimes gets used for brain swelling or to help kidneys flush out toxins. Those uses sit under tight medical supervision.
Possible Solutions to Risks
People can reduce any risks by reading labels and watching portion sizes, especially for kids. Manufacturers could make warnings clear, highlighting potential laxative effects once a certain amount shows up in a serving. Healthcare providers should ask about sugar alcohols during nutrition counseling, particularly for those with gut conditions. Education helps a lot—when parents and kids know what to expect, they make better decisions about treats and snacks.
Why Paying Attention Matters
D-Mannitol works well and is safe for most people. Problems mostly come from eating more than reasonable servings, just like with other sugar alcohols. The real solution sits in informed, moderate use and open conversations between health professionals and people about individual health concerns. Trust and transparency build up safety, and everyone walks away better off.
What are the side effects of D-Mannitol?
What Doctors and Patients Notice Most
D-Mannitol gets used for a range of medical reasons. Hospitals often turn to it for brain swelling after a head injury or for dropping pressure inside the eye during a crisis. Sometimes you’ll spot it in cough syrups and chewable tablets at your neighborhood pharmacy. But even a sugar alcohol with this many uses brings its own set of side effects.
Common Physical Discomforts
Most folks notice D-Mannitol’s impact on the gut first. Stomach cramps, bloating, or flatulence—these might catch you even at small amounts. For anyone with irritable bowel syndrome, taking this compound can lead to unpredictable bathroom trips, sometimes with sudden urgency. Diarrhea comes up pretty often, especially if the dosage rises. This isn’t just annoying; it can throw off your fluid balance or put you at risk for dehydration, which hits older adults hard.
This loss of fluids sometimes goes hand-in-hand with changes in blood chemistry. Folks who need high doses—often given through an IV—run the risk of losing electrolytes, especially potassium and sodium. These minerals keep your nerves and muscles working right. Drop them too low, and you might face weakness, cramps, irregular heart rhythms, or fatigue that seems to drag on. Hospitals usually watch these levels during treatment, but anyone self-medicating without close supervision could run into trouble fast.
Rare but Serious Dangers
Some patients see their kidneys take a hit. D-Mannitol draws water into the urine, pushing the kidneys to work overtime. In rare cases, this overload stresses the kidneys or, in patients with weak kidneys, tips them into failure. Research in the New England Journal of Medicine and JAMA supports this risk, especially for folks who already deal with kidney disease. Monitoring urine output and kidney blood tests matters a lot if you’re getting this sugar alcohol by IV.
There’s another angle to consider: heart patients. Too much volume shift in the blood can trigger or worsen heart failure, especially in those with existing cardiac conditions. Mild confusion, headaches, or dizziness can pop up with rapid changes in body fluids or pressure—hardly what you want in a hospital recovery room. Some allergic reactions, including rashes or swelling, get reported but stay pretty rare.
Sugar Substitute or Medical Tool?
Plenty of food makers use D-Mannitol to sweeten sugar-free gum or diet candy. Side effects show up here too, just less severe. For anyone eating a lot in one sitting, cramps and diarrhea can ruin the day. It works as a laxative, but getting caught off guard in a staff meeting brings little comfort. Labels often warn about this effect for a reason.
Many people don’t realize that even so-called safe sugar substitutes can spark uncomfortable reactions. Diet foods target folks with diabetes or those counting calories, yet digestive upset can make daily life tricky. Education matters. Understanding labels, starting any new supplement slowly, and telling your doctor about every medication or sweetener you use keeps you out of most trouble.
What Helps Keep Patients Safe
Doctors rely on monitoring. Blood work and kidney function checks catch big problems early. Clear instructions for patients carrying home products with D-Mannitol help, especially among diabetes patients or those with a chronic digestive ailment. The FDA knows about the laxative effect at higher doses, so food guidelines call for clear warnings. If you notice anything out of the ordinary—dizzy spells, muscle twitches, swelling, or bad belly pain—a quick check-in with a doctor beats waiting for things to get worse.
People hear about side effects and sometimes panic, but with clear information and good medical care, most get through treatment without major issues. In the end, knowing your body and keeping health professionals in the loop helps everyone steer around the rough patches.
How should D-Mannitol be stored?
Understanding Why Storage Pays Off
Think about a dusty old pantry shelf. If someone puts sugar there, jars open, moisture creeping in, it clumps and spoils fast. D-Mannitol reacts the same way in careless conditions. As a sugar alcohol used in everything from food to medicine, its quality matters a lot. If it picks up moisture or sits in the wrong temperature, you risk wasting money and, more importantly, you might be delivering a product that doesn’t meet its promise.
Keeping It Dry Actually Matters
Ever noticed how powdered drink mix cakes up in damp weather? D-Mannitol pulls in water just like that, which means humidity becomes the enemy. Moisture encourages the material to clump, break down, or even grow unwanted microbes. In my own pharmacy days, I learned early that a failed batch usually traced back to sloppy handling or careless storage. Pharmacists and food manufacturers alike know humidity above 60% creates a nightmare for anything that behaves like a sugar.
Simple solutions go a long way. Use tightly sealed containers, preferably made of high-density plastic or glass. Store buckets or jars on shelves, not on the floor where contamination hits hardest. A dry storage room with a humidity meter cuts spoilage—and headaches. For larger operations, investing in a dehumidifier pays for itself many times over.
Room Temperature Works—But Not Too Hot or Cold
A lot of people make the mistake of tossing all sweeteners and additives in a fridge “just in case.” No need. D-Mannitol keeps well at regular room temperature (15°C to 25°C), the kind you’d want in a comfortable workspace. Temperatures higher than that, especially in stuffy warehouses in summer, can speed up degradation. I once saw a shipment ruined because someone stored it right by a heat vent. The clumps and yellowing told the story. Cold, on the other hand, can cause condensation when you pull the container out and open it up, which defeats the point of refrigeration.
Protecting from Light and Air
Sunlight does no favors for most chemicals or food additives. D-Mannitol holds up better than some ingredients, but it’s still smart to limit light exposure. Cabinets work well, or even tinted containers if you don’t have a dark spot to keep it. Keep containers closed tightly after every use, since oxygen and airborne contaminants speed up spoilage. Warehousing best practice always meant labeling every container with the open date, so if anything changed in color or texture you could spot problems quickly.
Risks of Cutting Corners
Quality drift rarely happens overnight, but ignoring basic steps brings trouble. I’ve seen manufacturers scramble over a rejected batch, only to trace it to material stored next to steam pipes, or bins left open after a busy day. Bad storage not only risks failed batches but also regulatory trouble, especially with food or pharmaceutical ingredients.
Taking Responsibility: Straightforward Steps
For anyone handling D-Mannitol, storage is part of the job—not an afterthought. Pick up a humidity gauge for the storeroom. Use proper bins and keep them off the floor. Add a checklist for your team so nobody forgets to close a container or check expiration dates. By making these habits part of the routine, you save money, avoid stress, and make sure every bag or bottle delivers what it promises.
Simple stewardship always makes a big difference, especially with something as widely used and sensitive as D-Mannitol.
Can D-Mannitol be used by people with diabetes?
A Sweetener That Doesn’t Fit All
D-Mannitol shows up on ingredient lists in a surprising number of sugar-free gums, candies, and even some medications. People see “sugar alcohol” and often think it’s a guilt-free swap for table sugar, especially for folks trying to keep blood sugar levels in check. But that assumption doesn’t always hold up. From my own time reading labels as a health-conscious grocery shopper, I noticed D-Mannitol advertised as non-glycemic or “diabetes-friendly." That’s only part of the story.
Blood Sugar Impact and Digestive Effects
D-Mannitol keeps its promises for the most part: the body absorbs it slowly, and it doesn’t spike blood glucose the way sucrose or glucose does. For people with diabetes, this sounds ideal. Research published by the American Diabetes Association points out that sugar alcohols like mannitol produce much smaller increases in blood sugar and insulin compared to regular sugar. So on paper, this looks good.
But another fact jumps out if you ask dietitians or look into real-world effects. D-Mannitol draws water into the gut. Eat more than 10 or 15 grams in one sitting, and you might feel the familiar gurgle that signals cramping or diarrhea. This isn’t just discomfort. Chronic digestive trouble can play havoc with day-to-day routines, sometimes disturbing sleep or hydration for people who already juggle complex self-care routines. That personal disruption is real, and numbers on a nutrition label don't tell the whole story.
Kidney Concerns Get Overlooked
There’s another angle few product ads mention. D-Mannitol passes through the kidneys mostly unchanged. In hospital settings, doctors sometimes use this molecule to provoke urine production. So if someone already struggles with kidney conditions—which people with diabetes face at higher rates—managing D-Mannitol intake becomes a real health consideration. With kidney disease, the body can build up molecules like mannitol, which may worsen swelling or overload delicate filters. My own family’s experiences with diabetic nephropathy taught me that every ingredient matters, especially as complications add up over time.
Dosing and Daily Life
Serving size rarely matches snacking habits. People might grab a handful of sugar-free candy at a movie or chew several pieces of gum over an afternoon. That adds up quickly. Each gram doesn’t seem like much, but cumulative effects sneak up—causing unpredictable blood sugar shifts if labels are misleading or portion sizes grow. Healthcare providers often urge patients to experiment cautiously and track reactions in real time with glucose meters, especially after meals including new sweeteners.
Health Professional Advice and Label Transparency
Reliable health advice always emphasizes checking with a doctor or registered dietitian before changing dietary patterns, especially in diabetes care. D-Mannitol isn’t off-limits, but no two people respond exactly alike. Some tolerate small amounts easily, while others experience gut issues or subtle blood glucose changes. More transparent packaging and standardized labeling would help people gauge their own responses. Stronger regulation of “sugar-free” labels supports informed choices, especially among people who rely on details to stay healthy.
What Works: Individual Experimentation and Science-Based Guidance
Using D-Mannitol safely depends on learning from small experiments, careful monitoring, and ongoing communication with medical professionals. While it creates options for variety and sweet taste, its side effects, kidney clearance, and impact on daily routines need as much attention as its sweetness. Building habits around those facts helps people with diabetes create diets that suit their bodies, supported by honest conversations with trusted clinicians.
| Names | |
| Preferred IUPAC name | (2R,3R,4R,5R)-hexane-1,2,3,4,5,6-hexol |
| Other names |
Mannitol Osmitrol D-Mannite Manna sugar |
| Pronunciation | /diːˈmæn.ɪ.tɒl/ |
| Identifiers | |
| CAS Number | 69-65-8 |
| Beilstein Reference | 1724228 |
| ChEBI | CHEBI:16899 |
| ChEMBL | CHEMBL921 |
| ChemSpider | 7279 |
| DrugBank | DB00742 |
| ECHA InfoCard | 100.043.244 |
| EC Number | EC 200-711-8 |
| Gmelin Reference | 82277 |
| KEGG | C00392 |
| MeSH | D002044 |
| PubChem CID | 6251 |
| RTECS number | OP2060000 |
| UNII | F5TD010360 |
| UN number | UN 1327 |
| CompTox Dashboard (EPA) | urn:ietf:wg:oauth:2.0:oob#DTXSID2028480 |
| Properties | |
| Chemical formula | C6H14O6 |
| Molar mass | 182.17 g/mol |
| Appearance | white crystalline powder |
| Odor | Odorless |
| Density | 1.52 g/cm3 |
| Solubility in water | 22 g/100 mL (25 °C) |
| log P | -3.1 |
| Vapor pressure | <0.1 hPa (20 °C) |
| Acidity (pKa) | 14.16 |
| Basicity (pKb) | pKb: 10.62 |
| Magnetic susceptibility (χ) | -7.2e-6 |
| Refractive index (nD) | 1.333 |
| Viscosity | Viscosity: 3.7 cP (10% w/w solution, 25 °C) |
| Dipole moment | 1.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1177 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3334 kJ/mol |
| Pharmacology | |
| ATC code | A06AD16 |
| Hazards | |
| GHS labelling | GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264, P270, P301+P312, P330 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Flash point | 185 °C |
| Autoignition temperature | 430 °C |
| Explosive limits | Upper: 24% ; Lower: 2.2% |
| Lethal dose or concentration | LD50 Oral Rat 13,500 mg/kg |
| LD50 (median dose) | LD50 (median dose): 13 g/kg (oral, rat) |
| NIOSH | MW8200000 |
| PEL (Permissible) | PEL = Not established |
| REL (Recommended) | 100 g |
| Related compounds | |
| Related compounds |
D-Mannose D-Sorbitol Iditol Dulcitol Ribitol |

