Chloromethyl Pivalate: More Than Just a Name in an Industry Catalog
Unearthing the Past: Historical Footprints and Chemistry’s Course
Chloromethyl pivalate stubbornly resists celebrity status outside chemical manufacturing, but its origins paint a picture of how niche molecules often fill critical gaps. Chemists first started using pivalate esters between the 1960s and 1970s, during an era of rapid innovation in organic synthesis. Pivalic acid itself, with that distinctively bulky tert-butyl group, grew popular for its talent in protecting sensitive functions in reaction schemes. Adding a chloromethyl arm into this equation gave scientists a way to play mix-and-match with reagents that needed both reactivity and stability. The growth spurt in industrial synthesis during the late 20th century kept pushing new variations; chloromethyl pivalate became a regular player for anyone wanting a straightforward chlorination agent that wouldn’t collapse under mild processing.
Getting Down to Basics: What’s Chloromethyl Pivalate All About?
This compound isn’t flashy. It comes as a clear oily liquid, easy to pour and handle, with a scent that gives more away than you might think—sharp and slightly sweet, with an edge reminding everyone about its chemical punch. For chemists, the formula sticks in memory: C6H11ClO2, with a carbon backbone branching out. It works best in cold storage, since any warmth or moisture often shortens shelf life or leads to undesired side products. Unlike more hazardous halides, this one’s manageable, though gloves aren’t optional; the reactivity can catch fingers off guard.
Peeking Into the Details: Physical and Chemical Punch
Chloromethyl pivalate doesn’t give away its nature right away. The oily consistency comes with a tendency to creep up glass surfaces, betraying its low-to-medium viscosity. Water doesn’t mix in, and it floats on top, which signals the need for anhydrous conditions in most reactions. In the lab, it gives off fumes under heat, and contact with strong bases drives off the pivalic acid, leaving behind a stingy layer of residue. A small whiff can wake up anyone in the vicinity—a sure sign to review the ventilation system before opening a new bottle. Solubility runs higher in organic solvents like dichloromethane, illustrating why it fits neatly into workflows where water’s not welcome.
Technical Specs Aren’t Just for Nerds
Specifications matter most when people start comparing different batches from various suppliers. For researchers, purity usually falls in the 97–99 percent range, with color and odor as rough field checks. Density hovers just below that of water, which influences storage and dispensing. Boiling sits above 100°C, with decomposition setting in if someone pushes it. During the busiest times in my own lab work, I’ve found temperature and moisture control absolutely essential. Labels on shipping cans tell much of the story, with strong warnings about skin and eye contact, but also notes about ambient light protection, as exposure over time seems to yellow the liquid. Attention to detail when noting these specifications, especially the refractive index and possible contaminants, saves pain later on when side reactions confuse a synthesis.
How It’s Cooked Up: Synthesis That Demands Caution
Industrial preparation routes for chloromethyl pivalate need tight procedural discipline. The main path goes through reacting pivalic acid or pivaloyl chloride with chloromethanol or a chloromethylating agent under controlled heat. Acid scavengers like triethylamine often play a role, absorbing the released acid and smoothing out reaction bumps. The use of phosgene or thionyl chloride, both notorious for their hazards, underlines why many setups invest so fully in glove boxes or reliable fume extraction. Findings from operations stress the difference between theoretical yields and real-life outcomes—small leaks, contamination, or uneven cooling can quickly eat away at final mass. Filtering, washing, and drying finish the job, but every operator knows not to rush the last part, since residue moisture spells trouble during next-stage syntheses.
How It Changes and Reacts: Not for the Impatient
This molecule shows classic behavior for a reactive ester, blending the pivalate's bulk with a highly electrophilic chloromethyl group. In nucleophilic substitution reactions, it passes off the chlorine to good leaving groups—amines, thiols, even enolates—forming new bonds for pharmaceuticals or specialty polymers. Attempts to overheat the reaction often spark hydrolysis or decomposition, driving up the cost and cleanup. In my experience, meticulous addition rates and close monitoring of pH keep things under control, though automated systems have helped reduce variables. Derivatization and modification work best in inert atmospheres, and mistakes with glassware or solvents can trigger side chains or polymerization—messy, sticky, and tedious to separate.
Nicknames and Known Aliases: Decoding Chemical Language
Like many lab staples, chloromethyl pivalate goes by a small army of synonyms. Some will call it pivalic acid chloromethyl ester, while others reach for the less catchy 2-chloromethyl-2,2-dimethylpropanoate. The abbreviation “CMP” gets tossed around, usually in notes or inventory systems, but it’s rarely used on labels outside large-volume operations. Looking up CAS numbers before orders or literature searches trims the confusion; misunderstandings over synonyms have stalled more than a few research projects over the decades.
Working With It: Safety and Responsibility, No Shortcuts
Even seasoned chemists show respect for chloromethyl pivalate. It irritates skin upon contact, and the fumes irritate eyes and lungs. In poorly ventilated spaces, it builds up quickly, leading to headaches or worse. Standard PPE isn’t negotiable: gloves, goggles, fully buttoned coats, and access to emergency showers matter as much as the compound’s purity. Disposal routines involve specialized waste containers to avoid accidental release into drains or open air. Emergency teams drill for spills or exposure since reactivity with water and bases poses real hazards in mixed-use labs. Layers of procedure, checklists, and well-marked storage keep everyone safer.
Where It Gets Used: Beyond the Chemistry Classroom
Chloromethyl pivalate isn’t a household term, but its reach touches pharmaceuticals, agrochemicals, and specialty materials. Medicinal chemists use it for building blocks in active drug molecules, where its ability to deliver a pivalate-anchored group in a controlled manner opens doors to stable intermediates. Crop protection specialists lean on it for tailored pesticides and herbicides, seeking molecules that break down on schedule in the environment. Performance polymer manufacturing taps its reactivity too, aiming for materials that resist heat or oxidation. The hands-on reality—one I’ve dealt with myself—is tweaking every step in the workflow for optimal yield alongside manageable safety measures.
Deep Dive: Research, Risks, and What’s Next
Academic groups around the world keep charting new ways to harness chloromethyl pivalate. Recent papers have focused on tweaking reactivity or pairing it with milder reagents to dial down hazardous byproducts. Some investigation heads into greener alternatives, searching for routes that skip over the risky gas-phase intermediates of the past. Toxicity research has revealed moderate acute effects; animal models show respiratory irritation and skin reactions at low thresholds, so risk management strategies remain central. Regulatory standards typically demand detailed documentation for production, transport, and use—far more than simple “handle with care” labels.
The Road Ahead: Adaptation and a Push for Safer Chemistry
Future development moves in two directions: tackling safety and boosting the molecule’s versatility. Teams keep looking for alternative synthesis that sidestep notorious reagents, with some small-scale pilots experimenting with enzymatic or photochemical routes. Digital process monitoring, as seen in advanced manufacturing facilities, helps reduce accidents by catching deviations early. In my view, regulatory guidance and real-world operational lessons shape chloromethyl pivalate’s future; safer processes will help maintain its place in modern synthesis while attention to emerging health data and environmental effects shapes how and where it gets used. As the culture around industrial and academic chemistry grows ever more conscious of safety and green practices, expect to see innovation not just in the lab but in the entire support infrastructure around these specialized, essential compounds.
What is Chloromethyl Pivalate used for?
Chemical Building Blocks and Daily Life
In the noisy world of specialty chemicals, a compound like chloromethyl pivalate does not grab front-page attention, but its importance quietly ripples through labs and industries. Over the years, my work with organic synthesis has introduced me to many chemicals, and chloromethyl pivalate always stood out for its subtle yet significant contribution to drug development and advanced materials. At its core, it serves as a crucial building block, offering chemists a way to add functional groups to molecules. This trick powers a cascade of transformations in pharmaceutical research and fine chemical production.
The Backbone of Synthesis in Pharmaceuticals
Drug discovery thrives on innovation in synthetic chemistry. Many blockbuster medications trace part of their journey to obscure intermediates like chloromethyl pivalate. Through its reactive chloromethyl group, it links to other molecules, opening doors for the synthesis of esterified drugs, antiviral compounds, and more complex structures. In practice, I’ve seen teams streamline projects that once took months by reaching for this single intermediate, letting creativity and speed reshape pipelines in ways few chemicals can match. Pharmacology teams lean on it for its reliability and the specificity it brings to tough synthesis routes.
Specialty Polymers and Agrochemicals
Those who have worked in polymer labs know the struggle to control the design of advanced plastics. In this field, chloromethyl pivalate becomes a go-to starting point, helping add branching points to chains or fine-tune properties for coatings, electronics, and adhesives. The agricultural sphere turns to the same chemical to anchor molecules in crop protection agents, where effectiveness and safety matter in every blend. Here, its role may never become common knowledge to end users, but the impact filters down to better-performing products and safer outcomes for growers and consumers alike.
Balancing Safety and Sustainability
No story about chemicals is complete without looking at handling risks and environmental impact. Chloromethyl pivalate carries hazards—skin irritation, inhalation dangers, and persistence in waste streams all deserve careful attention. Laboratories and chemical plants must use well-ventilated workspaces, protective gear, and rigorous waste disposal plans. As a researcher, I learned early that cutting corners only leads to harm. Regulatory agencies and environmental scientists work together now, seeking safer alternatives and greener pathways without slowing innovation in industries that depend on such intermediates.
Pushing Toward Better Practices
Experience has taught me the value of sharing knowledge across sectors. Chemists swap lessons on cleaner reactions, companies invest in waste treatment, and researchers search for routes with less hazardous byproducts. The push for green chemistry is not just academic: real companies, facing real regulations and scrutiny from customers, have begun shifting to less toxic reagents where possible. The chemical supply chain grows stronger every time projects replace legacy methods with cleaner, smarter alternatives—without sacrificing the benefits that tools like chloromethyl pivalate bring to pharma, materials science, and agriculture.
Looking Forward
So, chloromethyl pivalate summarizes a theme in chemistry: simple compounds powering big improvements in technology, medicine, and industry. Its presence behind the scenes keeps research moving and innovation alive. Real progress comes from balancing the innovation this chemical brings with responsibility for health and the environment, making each cycle of synthesis just a little bit better.
What is the chemical formula and structure of Chloromethyl Pivalate?
Chemical Formula of Chloromethyl Pivalate
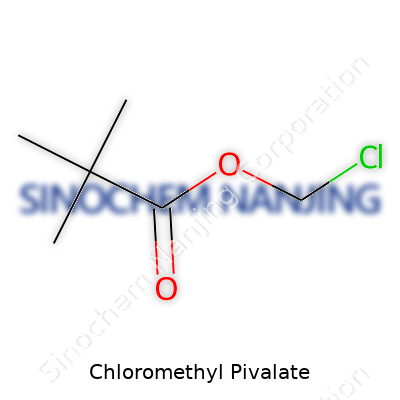
Chloromethyl Pivalate shows up with a straightforward formula: C6H11ClO2. Every molecule carries six carbons, eleven hydrogens, one chlorine, and two oxygens. The structure links a pivaloyl group to a chloromethyl group, forming a compact ester. Pivaloyl comes from pivalic acid, known formally as trimethylacetic acid. The backbone remains pretty easy to spot: a central carbon holds three methyl groups and connects through a carbonyl to an oxygen, which reaches out to the chloromethyl group.
Structure at the Atomic Level
The skeleton stands on five carbons making up the pivaloyl side: three methyls (–CH3) branching from a single carbon, itself sitting one carbon away from a double-bonded oxygen (the carbonyl). This setup forms the pivalic acid part. From there, it bonds with the chloromethyl group (–CH2Cl) through an ester linkage — basically, an oxygen joining the two.
Laid out by chemists, the usual structural formula will look like this:
(CH3)3C–COO–CH2Cl
So, if someone sketched it, they’d see three methyl groups fixed around a carbon, attached to a carbonyl, then through an oxygen to the chloromethyl piece. Recognizing this shape matters — familiarity with structural quirks lets scientists predict reactivity or how the molecule breaks down.
Real-World Uses and Safety Matters
Chemists turn to compounds like Chloromethyl Pivalate for building blocks. The molecule’s reactive chloromethyl corner makes for easy swaps, where another group can replace the chlorine through nucleophilic substitution. Pivaloyl parts, with all their bulk, steer reactivity and protect parts of new molecules during stepwise reactions. Synthesis labs reach for this stuff when building more complex pharmaceuticals, flavors, or fine chemicals.
Safety deserves a sober look. Chloromethyl groups can get feisty: many carry a risk of alkylating hands, eyes, or lungs of anyone exposed too casually. It takes training and the right gear — fume hoods, goggles, gloves — closer to the bench. Proper storage matters just as much. Moisture messes with the integrity of pivalate esters, with hydrolysis breaking them down over time, releasing corrosive acids. Locked chemical cabinets, good labeling, and careful logging protect both people and projects.
Looking at Solutions: Sustainable Handling and Research
While Chloromethyl Pivalate brings clear value in synthesis, its side effects demand action. Academic and industrial labs should adopt greener protocols, swapping in less hazardous reagents when possible. Updates to lab training can build mindfulness: reading MSDS sheets, handling spills, and preparing first aid responses. Waste handling stays critical, too, keeping byproducts and leftover chemicals out of the environment.
Open dialogue between chemists and safety officers can make a difference — feedback from the front lines shows what works and what falls short. Chemical suppliers now offer more detailed storage and disposal recommendations, stemming from studies about long-term health impacts. This shift doesn’t just keep people safe; it earns public trust and keeps science moving forward.
Further Knowledge and Scientific Growth
Digging into the formula and structure of Chloromethyl Pivalate isn’t about memorizing details. It’s about building a kind of fluency. With the right understanding, chemists and students spot patterns faster, design safer experiments, and react quickly if something stops working. Sharing this knowledge — through hands-on workshops and practical guides — lifts the whole field, letting everyone work smarter rather than just harder.
What are the storage and handling precautions for Chloromethyl Pivalate?
Understanding the Risks
Chloromethyl pivalate doesn’t show up in the headlines, but it hides serious hazards. Folks who run chemical labs or work in industrial settings understand that some reagents demand respect, and this one belongs on the list. It carries flammable, corrosive, and toxic threats that call for careful treatment every step from shipping to disposal.
Real-World Storage Strategies
Forget about shoving this chemical onto a random shelf. The right call is a cool, dry, and well-ventilated area, away from sunlight or heat sources. Locked flammable storage cabinets sit at the center of good practice. Moisture breaks down chloromethyl pivalate, so every container needs a tight seal. Water or even basic humidity speeds up decomposition, filling the space with toxic and corrosive fumes. Most experienced lab workers mark the labels and check regularly for leaks or cracks—small steps, but they save trouble down the line.
Compatibility deserves attention, too. Acids, bases, oxidizers and anything reactive need to live far from this material. Over the years, I’ve seen folks get comfortable, stacking incompatible bottles too close out of habit. One small spill can start a chain reaction, so separation stays non-negotiable. It pays to use secondary containment trays—those plastic pans under the bottles catch drips and give an extra line of defense.
Protecting People on the Job
A smart safety plan means everyone suiting up properly before they even touch the container. Standard goggles do not cut it: chemical splash goggles plus a face shield handle the job. Gloves, ideally nitrile or neoprene, protect from accidental splashes. For bigger tasks, lab coats with tight cuffs or even chemical aprons go a long way. I learned early on that quick access to an eyewash station and safety shower can save someone from lasting harm. Don’t skip routine checks—better to replace a broken bottle or worn gloves than to pay for a mistake.
Ventilation takes priority. Even quick handling or opening a bottle can release enough vapors to cause issues. Fume hoods, local exhaust systems, or even portable units stand between workers and breathing problems. I’ve spent long hours reminding colleagues to check the hood airflow before pouring—nobody gets used to that sharp, irritating smell that sometimes signals trouble.
Emergency Procedures and Environmental Risks
Spills do not wait for convenient hours. Fast response starts with planning. Spill kits should stand ready, with compatible absorbents, neutralizers, and heavy-duty disposal bags. Some labs tape up emergency instructions near the storage area. In case of a spill, isolating the area, ventilating, and cleaning up without delay lower the odds of escalation.
Disposal brings up environmental concerns. Simply throwing waste down the drain puts workers, communities, and aquatic life at risk. Best practice calls for collecting every bit—spent reagent, rags, gloves—and shipping it to licensed hazardous waste facilities. Documentation keeps things transparent and traceable, which matters during audits or accidental releases.
Knowledge, Training, and Teamwork
Learning how to handle chloromethyl pivalate safely takes more than a glance at the label. Regular training and clear, honest communication help everyone spot risks and step in before accidents grow. People remember lessons from hands-on drills and real-life stories much longer than PowerPoint slides. Bringing veterans and new hires together to walk through emergency plans keeps these practices alive, not buried in a safety manual.
Safety culture grows from shared respect and the understanding that small oversights cause big problems. Taking these steps turns potential hazards into manageable risks—a lesson every workplace learns, sometimes the hard way.
Is Chloromethyl Pivalate hazardous or toxic?
Understanding the Risks of Chloromethyl Pivalate
Most folks outside the chemical industry probably haven’t come across chloromethyl pivalate. It sounds technical, maybe even a bit intimidating. This compound lands in a group of chemicals used for specialized manufacturing, sometimes popping up during pharmaceutical synthesis or within lab research. The topic of its safety is anything but abstract or distant—it has real effects, especially for people handling it daily.
Personal Experience in the Lab
Years ago, while working in a university chemistry department, I handled a range of chlorinated organics. Many had warning signs that meant serious business. Even with gloves and goggles, the idea of breathing in fumes or splashing the skin brought genuine concern. Safety data for chemicals often reads dry, but talking to colleagues hammered home the potential for burns, breathing problems, and worse.
What the Data Tells Us
Chloromethyl pivalate shares some traits with other chlorinated compounds that have a strong track record for toxicity. Reports on its effects paint a clear picture: direct skin or eye contact can lead to irritation or more serious injury. Fumes have the potential to damage the respiratory tract. There’s a link between similar chloromethyl compounds and cancer risks, traced back to how certain chemicals interact with DNA or break down in the body.
The European Chemicals Agency and other regulators classify many chlorinated organics as hazardous, and chloromethyl pivalate fits right in. Regulations point out a risk for acute toxicity—you touch or inhale too much, and health effects show up quickly. Chronic exposure carries a cloud of unknowns, which can be even more troubling. It’s not uncommon for safety data sheets to require full protective gear and fume hoods, signaling that this is not a compound you want to treat casually.
Why This Matters Beyond the Lab
Hazardous chemicals aren’t just an issue for chemists and factory workers. Accidents can happen during transport or disposal. Mishandled waste may seep into the environment, reaching soil and water. I’ve seen what a broken container in a loading bay looks like, and the scramble to contain the spill shows that risks are never theoretical. These events draw on firefighters and emergency responders, raising the stakes even higher.
Communities near chemical plants want transparency, real-time updates, and prevention strategies. Stories of leaks or unsafe working conditions fuel distrust, and with good reason. Whether folks are dealing with industrial practices or caring for families close to a plant, understanding chemicals like chloromethyl pivalate becomes a personal stake.
Improving Safety and Awareness
Better labeling, strict adherence to storage and handling rules, and regular training for workers can lower the risk. Companies account for this by improving ventilation in production spaces, investing in early warning sensors, and encouraging workers to speak up about concerns. Extending this commitment to the supply chain ensures safety doesn’t stop at the factory door.
Continued research and transparency matter. Health agencies and scientists who publish detailed findings on these compounds serve a vital role. Decision-makers in government and business can support safer alternatives when possible and push for regular updates to safety guidelines.
Each step toward accountability and clear communication helps make workplaces and communities safer. For the everyday person—and the worker on the factory floor—the stakes are high and worth every ounce of vigilance.
What is the purity specification of Chloromethyl Pivalate available for purchase?
Understanding the Stakes in Purity
Chloromethyl pivalate has a niche spot in organic synthesis, especially where selective blocking and deprotection steps must go off without a hitch. Any chemist working in pharmaceutical labs, crop science, or fine-chemical manufacturing knows: the purity of every intermediate can decide whether a batch moves forward or hits the waste drum. There’s a simple reason many buyers chase the highest available purity. A batch with unknown or sub-par purity can ruin a downstream reaction and skew analytical results, wasting time and raw material. Most suppliers offer chloromethyl pivalate at purities above 97%, with some products even touching 99%. The difference between 97% and 99% sounds minor on a spreadsheet, but that last percentage point can mean fewer worrying side products and more reliable yields. I’ve seen situations in the lab where switching to a higher purity source solved lingering problems that nobody could nail down.
How Purity Gets Measured
Lab specifications usually give chloromethyl pivalate’s purity as a percentage, often paired with results from gas chromatography (GC) or high-performance liquid chromatography (HPLC). Alongside these, some certificates show results for water content (using Karl Fischer titration), acid value, and sometimes identification by infrared spectroscopy. A top-shelf supplier lists impurities clearly instead of hiding them behind technical jargon or generic marketing language. I tend to trust companies willing to show raw data, especially data on isomer content or residual solvents. Mislabeling or sloppy purification has landed more than one research group in hot water, whether by failed reactions or regulatory headaches when unknown impurities crop up during analysis.
Purity’s Impact on Safety and Compliance
Let’s not forget the safety angle. Chloromethyl pivalate belongs to a group of chemicals that can release toxic fumes. Low-purity material means a higher chance for unexpected reactions or volatile byproducts. For people with their hands on the pipettes, those unknowns can turn a simple weighing exercise into something riskier. Labs following ISO or GMP rules look for a certificate of analysis (COA) that goes beyond just the minimum. Investment in purity testing and transparent reporting builds trust and shortens the paperwork nightmare if a regulator comes calling.
Responsible Sourcing: Avoiding Unwanted Surprises
Many labs source chemicals through distributors that provide repackaged compounds. Sometimes, lower-cost options hide gaps in quality control, with poorly cleaned containers or lax storage ruining purity before the bottle even arrives. Scrutinizing the purity spec means looking at the supply chain. Reputable sources keep a chain of custody and lot-to-lot consistency. Companies cutting corners show it in missing documentation, vague answers to technical questions, or shipped material that doesn’t match what’s promised on paper. I’ve learned to check every COA and push for batch-specific data, especially before scaling up any synthesis.
Pushing for Better Purity Standards
Buyers who demand higher purity specifications help set better industry norms. Tighter specs reduce batch failures, minimize unknown variables, and make scale-up less stressful. Working with partners who are open about testing and batch records makes a supplier easy to recommend. In my own experience, insisting on open communication and accountability from suppliers is worth every extra email or phone call. The small details in purity specs, and in how they’re measured, do more for workflow, safety, and final product quality than most realize until something goes wrong.
| Names | |
| Preferred IUPAC name | chloromethyl 2,2-dimethylpropanoate |
| Other names |
Pivalic acid, chloromethyl ester Chloromethyl neopentanoate Chloromethyl trimethylacetate Trimethylacetic acid, chloromethyl ester |
| Pronunciation | /ˌklɔːrəˈmɛθɪl pɪˈveɪleɪt/ |
| Identifiers | |
| CAS Number | 4640-01-1 |
| Beilstein Reference | 1361003 |
| ChEBI | CHEBI:139468 |
| ChEMBL | CHEMBL21805 |
| ChemSpider | 16634388 |
| DrugBank | DB08259 |
| ECHA InfoCard | '03a9206a-7a55-40a2-96d7-42ec05d34d85' |
| EC Number | 205-703-7 |
| Gmelin Reference | 80519 |
| KEGG | C14321 |
| MeSH | D015245 |
| PubChem CID | 11449212 |
| RTECS number | MO2625000 |
| UNII | X586072T5B |
| UN number | UN3271 |
| CompTox Dashboard (EPA) | DTXSID2050546 |
| Properties | |
| Chemical formula | C6H11ClO2 |
| Molar mass | 164.63 g/mol |
| Appearance | Colorless to pale yellow transparent liquid |
| Odor | Pungent |
| Density | 1.048 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 1.7 |
| Vapor pressure | 0.6 mmHg (20 °C) |
| Acidity (pKa) | Estimated pKa ≈ 16 |
| Basicity (pKb) | 15.96 |
| Magnetic susceptibility (χ) | -6.44 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.418 |
| Viscosity | 0.93 mPa.s (20°C) |
| Dipole moment | 2.34 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 383.3 J·mol⁻¹·K⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H315, H319, H331, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-1-W |
| Flash point | 85 °C |
| Autoignition temperature | 450°C |
| Lethal dose or concentration | LD50 oral rat 1600 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 940 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | ≥98% |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Pivalic acid Pivaloyl chloride Methyl pivalate Chloromethyl acetate Chloromethyl benzoate |

