Caprolactam: Exploring Its Journey, Impact, and Next Steps
A Look Back: How Caprolactam Shaped Modern Industry
People sometimes overlook the quiet giants of chemistry, but caprolactam has earned its stripes over the years. Developed in the early twentieth century as folks scoured for alternatives to natural fibers, caprolactam paved the way for nylon 6—a fiber that made its way to everything from stockings to parachute cords. Before caprolactam came onto the scene, silk shortages during war times spurred scientists like Paul Schlack to get creative in Germany. By the late 1940s, the world started noticing nylon’s ability to stand up to daily wear, leading to booming demand. This one molecule became the backbone for countless textiles and engineering plastics, quietly transforming fashion, engineering, and even the automotive industry. Seeing caprolactam go from an obscure laboratory curiosity to a chemical mainstay strengthens the case for betting on science and ingenuity in times of crisis.
What Caprolactam Brings to the Table
Caprolactam sits at the root of nylon 6 production. This crystalline solid looks white and smells faintly medicinal. Heavy-duty machinery churns out millions of tons of it every year. Chemically, it's a cyclic amide with the formula C6H11NO—made up of a six-carbon ring and a nitrogen atom. Without this set of atoms arranged the right way, nylon 6 would never show its signature elasticity and heat resistance. Straight out of the bag, caprolactam feels oily, dissolving nicely in water but eager to polymerize when the right catalysts step in. While people often talk about polymers, everything starts with the simple structure of caprolactam, which cracks open and links up into chains during polymerization. One gram of caprolactam contains plenty of energy to feed a whole chain reaction, which is part of its unique draw.
Understanding the Chemistry: From Lab to Factory
Chemists discovered early on that if you treat cyclohexanone with hydroxylamine and run it through a set of reactions to form oxime, a dash of strong acid eventually triggers the Beckmann rearrangement—flipping the molecule inside out and creating caprolactam. Over time, researchers engineered this sequence for large-scale use. Newer methods still spring from that classic approach, but tweaks help trim byproducts and improve yields, whether by using cleaner catalysts or fine-tuning temperatures. Some facilities focus on recycling waste streams and converting lower-value feedstocks, while others add purification to strip out water and unwanted residues. Factories need caprolactam to meet strict benchmarks for color, purity, and moisture. Only the best batches get carried forward to make nylon 6 fibers or resins. Hard numbers like melting point, boiling point, and reactivity matter for engineers but also for folks concerned with safety and storage.
Putting a Name to the Product: Caprolactam’s Many Aliases
Scientists and manufacturers know caprolactam by a batch of names. Some labels like epsilon-caprolactam or 1-aza-cycloheptan-2-one show up in technical documents, especially in patents or journals. In ordinary conversation, people stick to just “caprolactam.” This compound often appears in shipping manifests or regulatory forms because of how much industry moves it around the globe. Over the decades, caprolactam kept its place in chemical textbooks and factory logs, earning a reputation for reliability.
On the Ground: Handling and Safety in Real Workplaces
Factories handling caprolactam run tight ships. People working with the raw material wear gloves and goggles, since dust or vapors can irritate eyes or skin. Facilities often use enclosed processes or localized ventilation, limiting spills or runaway reactions. Regulators demand companies publish detailed safety data sheets—making sure everyone from line workers to cleanup crews knows what they’re facing. Storage tanks need protection from moisture and acids, and spills get cleaned up quickly to avoid slips, heat buildup, or environmental leaks. Only trained staff handle high temperatures, and everyone sticks to routines that minimize accidental exposure. I’ve walked factory floors and seen how much time goes into training and drills, with safety managers double-checking every corner on a regular basis.
From Car Tires to Medical Devices: The Broad Reach of Caprolactam
If you pull apart the components in everyday goods, caprolactam pops up everywhere. Nylon 6, made from this chemical, lines the carpets in homes and offices, weaves into fishing lines, and builds tough under-the-hood parts for cars. Engineers prefer nylon 6 when a product needs to be both strong and lightweight, like electrical casings or gears inside machines. Even the medical field has found roles for nylon derived from caprolactam—think surgical sutures that hold strong but flex with healing tissue. The consistent qualities and price of caprolactam-based plastics let companies turn piles of powder or pellets into everything from cable ties to food packaging. When shopping for camping gear or repair kits, nylon 6 gear holds up under rough use year after year. No matter the application, caprolactam proves that behind every product sits a backbone built on sound chemistry.
Toward a Safer, Smarter Future: Health, Environment, and Research
No story about industrial chemicals feels honest without facing up to their downsides. Plenty of studies track caprolactam’s effects on people and the planet. Most research finds that modest exposure over short periods rarely causes lasting harm, but repeated contact or inhalation brings risks. Eyes, skin, and lungs react to dust or fumes, and countries set tight exposure limits for workplace air. Researchers in Europe, Asia, and North America spend years checking toxicity and monitoring local water and soil for any buildup, especially since water-soluble residues can leak from wastewater if not handled right. Newer designs for factories scramble to close loops—cutting waste, recycling process water, or capturing vapors before they drift into surrounding communities. People want manufacturing to get cleaner and safer. If the industry stalls, activists and lawmakers still push for answers, so steady pressure means safety practices never stand still.
Driving Innovation: Fresh Ideas Breathe Life into Caprolactam
The chemistry world keeps evolving, and caprolactam hasn't slipped into the shadows yet. Scientists continue hunting for greener catalysts, better ways to tap into bio-based starting materials, and new approaches that cut CO2 footprints. Some research teams design tweaks in the core structure, forging “modified nylons” with built-in resistance to fire or better compatibility with recycling systems. Others see promise in caprolactam for 3D printing filaments or advanced composites in wind turbines and EVs. Moving from fossil feedstocks to renewable sources could slash dependence on oil, though tough hurdles still wait. As customers care more about environmental labeling and transparent sourcing, caprolactam production needs to keep pace—not just in keeping costs low, but in tracking emissions and taking responsibility for waste.
What Comes Next for Caprolactam?
Industrial chemistry never stands still. More regulation on emissions, pressure for circular economies, and demand for sustainable design all push caprolactam manufacturers to rethink their playbooks. The best hope for balancing progress and responsibility lives in closer partnerships between companies, regulators, and researchers—sharing data, investing in new tech, and training future chemists to handle challenges head-on. By tracing the story of caprolactam across decades, it's clear this molecule will keep shaping industries. Down the line, success won’t hinge on pumping out more product at any cost but on finding smarter ways to serve practical needs without sidestepping safety or the environment.
What is caprolactam used for?
What Is Caprolactam?
Caprolactam might not be a household name, but plenty of household items rely on it. It’s the main ingredient in producing nylon 6, a strong, dependable plastic. Think of nylon 6 as the staple that holds a huge range of products together: clothing, carpets, fishing nets, tire cords, and even automotive parts. With global demand for tough, versatile materials always on the rise, caprolactam isn't fading from significance any time soon.
Why Nylon 6 Matters
Nylon 6 has carved out a reputation for being both lightweight and durable. The roots stretch all the way back to caprolactam. Factories polymerize caprolactam to make millions of tons of nylon 6 each year. Out of all the uses, synthetic fibers for textiles and carpets take up the largest share. Wander through any clothing store—sportswear, leggings, windbreakers—countless items show off what caprolactam can achieve.
Carpet manufacturers also lean on caprolactam’s byproduct. Nylon carpets offer stain-resistance, vibrant colors, and last through years of heavy foot traffic. In factories, nylon 6 comes in handy for producing industrial yarns, ropes, and even fishing nets used all over the world.
Beyond Clothes: Caprolactam in Industrial Materials
The story doesn't stop at fabric. Car parts, garden tools, and electrical components all owe something to caprolactam. Its chemical properties give manufacturers flexibility to mold, stretch, and shape nylon 6 into whatever the job demands. Air intake manifolds, oil pans, gearwheels, and cable ties all become possible with caprolactam. It has helped the auto industry shave weight off vehicles, cut costs, and make cars more fuel-efficient.
Last year, I went shopping for a power tool. The casing felt oddly lightweight but rock solid. That’s the magic of nylon 6. I cringed at the thought of dropping it on my concrete garage floor, yet it’s survived plenty of hard tumbles. Over time, I realized how much faith we put in these tough plastics. The average consumer may not recognize the chemical behind the product, but they depend on its strength and reliability every day.
Environmental Concerns
With such wide use, it’s not all upside. Manufacturing caprolactam needs energy and produces nitrous oxide, which poses environmental risks. Some regions with large chemical plants have struggled with water and air pollution. Tackling these issues isn’t just about policy or industry—there’s a role for buyers too.
I’ve talked to textile workers who now look for suppliers recycling old nylon 6 into new fibers. This method, called “closed-loop recycling,” makes a significant difference in cutting waste. Factories in Europe and Asia have started tapping into renewable energy for their processes, lowering caprolactam’s carbon footprint.
What Needs to Happen Next
The market isn’t slowing down. With everything from running shoes to electrical housing relying on nylon 6, demand keeps rising. Companies can keep investing in better pollution controls and improved recycling. Designers have started reimagining products for longer life or easier disassembly, and some offer take-back programs for old carpets and clothing. As individuals, each purchase makes a difference, especially when we support recycled or sustainably produced nylon products. Caprolactam’s reach is vast, but the future can be cleaner—and still tough as ever.
Is caprolactam hazardous to health?
Understanding Caprolactam and Everyday Exposure
Caprolactam finds its way into daily life more than most folks realize. It’s a building block for making nylon—used in clothes, carpets, and countless household products. Factories pump out tons of it every year, and workers around these plants handle it often. For most people, the biggest risk comes from breathing in fumes near manufacturing sites or living close to chemical plants. It doesn’t tend to linger long outdoors, since sunlight and water break it down, but where production ramps up, exposure goes up right along with it.
Real-World Health Facts
Breathing in caprolactam dust or fumes can mess with your lungs and eyes. Workers say their throats feel tight, noses get stuffy, and eyes sting when they breathe it in for a stretch. The U.S. Occupational Safety and Health Administration has set limits on how much is safe in workplace air — about one part per million over eight hours. That isn’t just a cautionary number: hospital visits pop up whenever people breathe in too much, and not just in one-off cases. Nausea, headaches, and sore throats show up pretty quickly if safety gear slips, or ventilation stalls.
Now and then, factories mess up. Leaks release higher doses, and folks nearby get exposed. It doesn’t take a chemist to see that consistent chest pain or headaches in an industrial community mean something’s getting in the air that shouldn’t. Even so, short spurts of exposure rarely trigger lasting harm; the trick is not letting small lapses stack up into big health problems.
The science on cancer or major chronic issues from caprolactam hasn’t nailed down a clear link for most people. The U.S. Environmental Protection Agency doesn’t label it as a cancer-causing agent. That might sound reassuring, but a lack of evidence doesn’t mean proof of safety—for people breathing it often, the stakes feel different.
Trace Chemicals, Larger Impact
Some people are already vulnerable: kids, the elderly, or anyone with sensitive lungs. It only takes visiting a hospital to see how easily something mild sends asthma patients into coughing fits. Factories can’t just shrug off those statistics; their emissions affect these groups differently. Caprolactam isn’t the only chemical that floats through a fence line, but it’s one that can be controlled.
Personal Experiences and Community Worries
My neighbor once worked in a fiber plant, and every day he came home, his collar smelled sharp, like plastic left too close to a heater. He never wore masks at first—nobody talked about them. Years later, when stricter safety rules hit, some older workers joked about “soft lungs.” Few thought much about exposure until friends started calling in sick more often. Trust in plant managers didn’t come easy after that.
Health agencies say washing hands and changing out of work clothes helps, and from what I’ve seen with my neighbor’s family, the small habits matter. When he started keeping work shoes outside and clothes in a separate hamper, sniffles and coughs at home faded.
Making It Safer
Companies hold a responsibility to guard people’s health around caprolactam. Improved ventilation makes a measurable difference. Daily air quality checks catch leaks before they spiral. Personal protective equipment, including masks with proper filters, lines up with best medical advice. Regular health checkups catch red flags faster, instead of waiting on someone to land in a clinic.
Calls from the community push regulators to adopt tougher safety checks. Bigger barriers or greener production practices won’t replace vigilance, but they ease the risk. Where factories keep open lines with neighbors and share results from air monitors, trust grows—and so does safety.
How is caprolactam produced?
The Building Block for More Than Fabric
Caprolactam turns up almost everywhere. Nylon fibers owe their toughness to it, so do engineering plastics sitting under car hoods. If you zipped up a jacket or walked across a carpet today, you touched the end of a process that no one calls glamorous but shapes how we live. From a writer’s chair, this story matters, because we take these chemical journeys for granted, even though they shape global supply chains and raise environmental questions that touch hometown rivers and distant farmland alike.
What It Takes to Make Caprolactam
Industry makes caprolactam from cyclohexanone, a liquid distilled from crude oil. Most producers start with benzene, a simple molecule from petroleum refineries. Factories transform benzene into cyclohexane by hydrogenation. Then, they oxidize it to cyclohexanone, blend it with ammonia, and create cyclohexanone oxime. The real magic kicks in with a process called Beckmann rearrangement: treating that oxime with sulfuric acid to shuffle atoms, freeing caprolactam from the stew. Afterward, chemists neutralize the mixture and separate the product from whatever else came along for the ride. Each step involves high heat, specific pressures, and some very sharp engineering.
I’ve spoken with people who live near chemical plants and heard their worries about what lands in their water from these operations. The old methods created a lot of ammonium sulfate, a fertilizer that piles up at plant gates. Some regions can absorb all the extra nitrogen; others can’t safely use that much, so they end up with mountains of waste. The industry leans on technical progress to address these piles. Over the past decade, leading companies retooled their setups, swapping older batch tanks for continuous flow systems that squeeze out more product, recover excess chemicals, and reduce leftover fertilizer. They aim to waste less and pollute less, since communities now demand cleaner manufacturing.
Why This Process Stays in the Spotlight
It’s tempting to shrug off chemicals as someone else’s business, but the way caprolactam gets made ripples far beyond factory gates. Worldwide, over four million tons are produced each year. In China, huge plants anchor industrial parks and feed global exports. Small changes in demand for clothing or electronics can shake up whole supply chains. Price shocks a decade ago forced carpet makers, automakers, and even sportswear brands to scramble, showing how fragile modern supply can become when one chemical source falters.
The environmental impact travels with the product. Ammonium waste, nitrogen runoff, and air emissions trouble communities from Texas to Zhejiang. Companies respond by investing in closed-loop water recycling, adopting catalysts that work with lower temperatures, and shifting away from sulfuric acid when possible. These aren’t just engineering tweaks. They bring real changes for local residents who want safer air and water, and for workers who care about safer conditions. Some European firms have even started exploring biobased routes from renewable feedstocks—early days, but progress brings hope.
Looking Down the Road
Caprolactam’s chemical path might seem technical or distant, but it’s deeply tied to daily life. Engineering advances shaved waste, improved energy use, and cleaned up old processes, but every new plant or expansion brings fresh challenges. Old habits die hard, and markets still reward the cheapest output. Solutions will demand that producers balance profit with social responsibility. That mix—upgrading technology, investing in safer methods, and taking local concerns seriously—looks like the surest way forward in an industry too important to ignore.
What is the main application of caprolactam?
A Look at Caprolactam in Everyday Life
Caprolactam doesn’t show up on a supermarket shelf. Most people have never even heard its name. Funny thing is, everyone bumps into products made thanks to this one ingredient almost every day. It's the key ingredient that lets us have things like tough carpeting that makes it through years of family traffic, and the seat covers in buses and trains that don’t tear after months of use. Caprolactam’s main claim to fame boils down to a single material: nylon 6. This is a synthetic fiber and plastic that keeps popping up across industries. Jackets that hold up against sharp wind, fishing nets that don’t rot out at sea, and sturdy liners for tires all rely on this unique man-made material.
Why Industry Bets on Nylon 6
Caprolactam makes nylon 6 possible through a process called polymerization. The outcome is a fiber that’s just plain tough. You’ll spot its strength in everything from climbing ropes to airbags. The real kicker is how well nylon 6 holds up under stress. Engineers and designers grab for it when they need something that stands up to friction, tension, and even chemicals. Factories that produce car parts choose nylon 6 for gear wheels and bushings. Medical suppliers rely on it for strong, flexible fibers in surgical sutures. Fashion brands use it in hosiery and activewear, all because the fiber holds its color and keeps its shape after wash after wash.
Caprolactam and the Global Supply Chain
Numbers tell the story here. Almost 90% of caprolactam production feeds directly into nylon 6. Major producers churn out millions of metric tons a year just to keep up with the growing demand for high-strength synthetic fibers and resins. Asia, and China especially, turn out more caprolactam than anywhere else, powering up global supply chains in textiles and auto parts. When shipping delays or cost spikes hit the market for caprolactam, prices for nylon-based goods follow. That’s tough for folks farther down the supply chain, from clothing brands to car manufacturers. Consumers may not see caprolactam on a clothing tag, but they feel the pinch if material prices jump.
Rethinking Caprolactam’s Environmental Footprint
Like most chemicals tied to heavy industry, caprolactam comes with baggage. Factories use plenty of energy and give off nitrous oxide, a greenhouse gas far stronger than carbon dioxide. A lot gets invested these days in making that process greener. Companies work to capture those gases and recycle water and raw ingredients. Some top firms started pulling from bio-based sources and even recycling old nylon back into the caprolactam chain. This effort can slash both emissions and waste, though it’s still catching on compared to traditional methods. These changes won’t show results overnight. Step by step, though, they point toward a world where strong synthetic fibers come with less of a price for the planet.
Finding Smarter Solutions for the Future
The world’s not backing away from synthetic materials any time soon. Caprolactam will keep driving the nylon 6 market for decades. Bringing real expertise from science and manufacturing together offers the best shot at building a more eco-friendly—and still affordable—market for caprolactam and its products. Smarter reuse of waste, tighter environmental controls, rewarding greener producers, and focusing on transparency can set up positive change that everyone, from giant factories to home sewers, can get behind.
What are the storage and handling requirements for caprolactam?
Understanding Caprolactam’s Nature
Caprolactam hits the shelves as small, white crystals or flakes with a faint, sometimes fishy odor. Most folks know it as the starting ingredient for nylon 6, but not everyone talks about what it takes to keep it stored and handled safely. If you’ve spent any time in a warehouse or plant, you understand that a big bag of chemicals is only as safe as the system around it. Caprolactam reacts quickly to moisture and dust goes airborne with the slightest breeze. Getting this wrong doesn’t just mean a lower-quality finished product—it means real risks for the people working with it every day.
Humidity and Temperature Aren’t Afterthoughts
Moisture works fast. Caprolactam absorbs water right out of the air, clumping together or turning sticky. Once clumped, it becomes tough to process and loses much of its chemical punch. Staff can keep this from happening with sealed, moisture-proof containers, usually lined barrels or sturdy multi-layer bags. Breathable packaging is just asking for trouble.
Climate-controlled storage plays a huge role. Caprolactam likes it cool—below 30 degrees Celsius, ideally. High heat speeds up degradation and sparks off self-polymerization. Where I’ve worked, a simple spike in summer temperatures led to material sticking and fouling the hopper in just a couple of days. Manufacturers sometimes suggest keeping the stuff around 20-25 degrees, and a well-ventilated storage area prevents any unwanted fumes from building up if the product breaks down. In places where air conditioning means high costs, rotating stock frequently and installing basic insulation makes a big difference.
Protecting Workers on the Front Line
Unloading and moving bulk quantities isn’t just a physical task. Caprolactam dust can easily irritate skin, eyes, and airways. Workers deal with it best using personal protective equipment—always gloves, safety glasses, and dust masks. Over the years, I’ve seen people get a little casual about protocols, yet the cost for them comes in the form of rashes or respiratory symptoms. Sites committed to regular safety training keep minor accidents down. A simple rule: never eat, drink, or smoke anywhere near the storage or handling areas. Washing up right after a shift might sound old-fashioned, but it makes all the difference.
Main Hazards and Real-Life Incidents
Caprolactam gets pretty flammable at high temperatures, giving off toxic fumes—including ammonia—if caught in a fire. This isn’t just textbook knowledge—it’s played out in more than one smoky warehouse, turning a small problem into a fire department event. Fire extinguishers with foam or dry chemicals need to stand ready, and any spill needs fast containment. It helps to keep floor drains covered, so material doesn’t sneak into the water system.
Tackling Problems With Training and Controls
An efficient plant gives its storage teams the knowledge and tools they need. Regular training sessions mean everyone stays sharp. Signage in easy-to-read language about emergency action plans and hazard controls keeps procedures clear. Chemical management software and routine inspections keep tabs on shelf life and prevent old material from sitting too long. As far as solutions go, nothing beats investing in reliable packaging and staff knowledge. Together, these steps keep caprolactam in the barrel—right where it belongs.
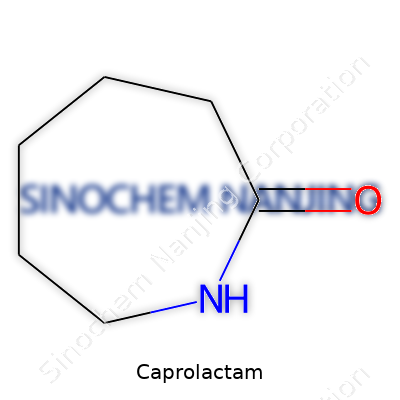
| Names | |
| Preferred IUPAC name | azepan-2-one |
| Pronunciation | /ˌkæprəˈlæktæm/ |
| Identifiers | |
| CAS Number | 105-60-2 |
| 3D model (JSmol) | `3D model (JSmol)` string for **Caprolactam**: ``` C1CCNC(=O)C1 ``` |
| Beilstein Reference | 1710491 |
| ChEBI | CHEBI:18127 |
| ChEMBL | CHEMBL71539 |
| ChemSpider | 5481 |
| DrugBank | DB02996 |
| ECHA InfoCard | 06c4166a-e973-4b07-b663-97f733cb16cf |
| EC Number | 203-313-2 |
| Gmelin Reference | 85374 |
| KEGG | C01745 |
| MeSH | D002199 |
| PubChem CID | 7064 |
| RTECS number | GV9825000 |
| UNII | F07G78CVXW |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C6H11NO |
| Molar mass | 113.16 g/mol |
| Appearance | White crystalline solid |
| Odor | characteristic |
| Density | 1.012 g/cm³ |
| Solubility in water | Very soluble |
| log P | -0.26 |
| Vapor pressure | 0.005 mmHg (25°C) |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | -1.7 |
| Magnetic susceptibility (χ) | -9.9e-6 |
| Refractive index (nD) | 1.484 |
| Viscosity | 0.968 mPa·s (at 80°C) |
| Dipole moment | 4.49 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -588.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3227 kJ/mol |
| Pharmacology | |
| ATC code | J01XX15 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P333+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2 |
| Flash point | 113°C |
| Autoignition temperature | 630°C |
| Explosive limits | Explosive limits: 1.7–9.8% |
| Lethal dose or concentration | LD50 (oral, rat): 1870 mg/kg |
| LD50 (median dose) | 1,210 mg/kg (rat, oral) |
| NIOSH | RJ2100000 |
| PEL (Permissible) | 5 ppm |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | 40 mg/m3 |

