Butyl Salicylate: A Commentary on Its Past, Present, and Future
Tracing the Roots: How Butyl Salicylate Emerged
There’s something almost cinematic about the way chemicals like butyl salicylate find their foothold across industries. This compound dates back to a time when the world leaned heavily on plant-based remedies and chemists worked overtime to turn nature’s scents and healing agents into bottled products. Salicylic acid itself comes with a long-standing legacy as a building block in both pain relief and skincare. By linking it with butanol, chemists unlocked qualities that could cross over into fragrances, personal care, and specialized coatings. My interest in laboratory work made it clear pretty early that the leap from an old-fashioned willow bark extract to a modern ester like butyl salicylate hinges on practical chemistry and a solid sense for what people actually want from everyday products.
Product Overview: More than a Smell-good Additive
Mention butyl salicylate and perfumers think of a gentle, sweet aroma, while industrial chemists look to its solvency power and stability. As an ester, this compound manages to be both fragrant and functional—a rare balance. Its role in the beauty world as a fragrance fixative or masking agent comes from its pleasant, sweet scent that manages to take the edge off harsher base notes. Meanwhile, some labs see it as a lubricant additive or even an intermediate for more complex molecules. I’ve seen firsthand how versatile it gets, sliding between delicate tasks like scent harmonization and the gritty jobs inside chemical manufacturing plants.
Tangible Traits: Physical and Chemical Properties
You won’t get far in any chemical application without knowing its traits. Butyl salicylate, as a colorless to pale yellow oily liquid, doesn’t try to hide its presence. It’s got a decent boiling point and holds its shape in both hot and cold weather, making it a reliable option in formulas that demand stability. Its mild, pleasant scent also means a small amount can transform a product’s profile. Unlike heavier esters, it flows smoothly and blends well, with low viscosity that lets it spread thin and wide. The solubility factor—barely mixing with water but blending easily in alcohols and oils—gives it an edge in oil-based perfumes and skin creams. An old chemistry instructor used to say, “If you want something that won’t fight your carrier solvents, reach for an ester like this.” That advice holds up—flexibility in blending is gold in the formulation world.
Understanding Standards: Labeling and Technical Details
Product stewardship goes hand in hand with technical discipline. Real talk—no one wants to go through a lineup of chemicals at the warehouse and discover a missing or vague label. With butyl salicylate, labeling needs to capture the essential warning signs, the UN number for logistics, and correct hazard icons. Producers who cut corners on clear labeling invite headaches on the regulatory side. Sticking to clear labeling helps protect workers and keeps shipments moving. Over the years, regulatory pressure has only gone up, with more demand for traceability and batch tracking. The specifics on purity, boiling and flash points, and other hazard data can swing depending on the supply chain and synthesis quality, but top-tier suppliers support rigorous data sheets and barcodes to avoid confusion and unnecessary risks.
Preparation: The Core Chemistry Behind Butyl Salicylate
Heating up salicylic acid with butanol in the presence of a strong acid sets the stage for making butyl salicylate. This is textbook esterification, and I remember grinding through the glassware cleanup after long reflux runs during my time in the lab. Getting this step right matters—not just for high yield, but for stripping out leftover acid and washing out impurities. Big plants use stainless steel reactors, pressure controls, and always a nose for lingering butanol that hasn’t reacted. Lab-scale batches sometimes hit snags with side reactions if the process runs hot or goes too long, but industry sites have dialed in their flows to keep things moving smoothly. As with many esters, water forms during synthesis and gets removed to push the reaction beyond equilibrium, otherwise you risk low conversion or contaminated product.
What Really Happens: Common Chemical Reactions and Modifications
Talk to anyone spending serious time in synthesis, and they’ll tell you that every ester serves as both an endpoint and a possible starting point. Butyl salicylate hydrolyzes under strong acid or base back to salicylic acid and butanol. Anyone in coatings or surfactants knows that tweaking the alcohol or acid part of the molecule can encourage new traits—a process called transesterification. In pharma, researchers sometimes modify the salicylate part to tune how quickly the ester breaks down or how gently it delivers active ingredients. I recall a pharma R&D meeting where a small change in the ester fragment flipped the bioavailability profile, forcing the team to circle back and rethink everything. Tweaking small bits, like the butyl chain, offers an entry to new performance areas, whether adjusting volatility, solubility, or scent.
What’s In a Name: Synonyms and Product Aliases
Butyl salicylate won’t always turn up under the same name, so anyone who works with procurement or regulatory codebooks needs to watch out for variants like “n-butyl salicylate”, “salicylic acid butyl ester”, or even EINECS and CAS registry numbers. One summer at a fragrance plant, half a day got burned chasing down a shipment labeled in French under an old trade name, which taught me just how tangled things can get in cross-border supply chains. Checking CAS number 205-487-5 clears up doubts, whatever language or supplier tag shows up.
Staying Safe: Standards in Handling and Transport
Protective eyewear, gloves, and good ventilation RULE in any setup where butyl salicylate gets poured, heated, or stored. Regulators classify it with low acute toxicity, though it can cause skin or eye irritation after repeat exposure. Roll up the sleeves in a pilot plant, and you’ll notice workers appreciate tight bottle caps and dedicated ventilation hoods during operational runs. Spills on the floor create a slip hazard, so the right handling protocols keep everyone on solid footing. Rules also demand careful waste disposal, and more facilities recycle solvent streams instead of dumping. Painting a picture, regulatory inspections in the EU and US make sure firms run regular risk assessments and keep the raw material under proper lock and key.
Fields of Use: Where Butyl Salicylate Finds a Home
Butyl salicylate spends more time behind the scenes than folks realize. It fills fragrance bottles as a fixative, tones down harsh scents in detergents, and plays a quiet role in certain adhesives, coatings, and topical skin care blends. During a tour of a natural cosmetics startup, I watched the chemist praise it for its mildness and how reliably it blended with plant oils. The paint and coatings crowd value it for leveling properties and delivery of active agents, showing just how each industry leans on specific features. Regulatory backlash against certain phthalates and parabens has opened the door for alt-esters like this one, letting product designers balance cost, logistics, and consumer safety.
Rising to the Challenge: Research and Innovation
Labs pour their budgets into pushing butyl salicylate further—seeking new roles outside legacy fragrance and coating niches. Pharmaceutics have peaked at slow-release pain relief using its backbone, while environmental scientists run tests on degradation rates to judge how “green” it really is. Tackling odor masking more efficiently, reducing allergenicity, and speeding up biodegradation—these are on the research wish list. Some green chemists push for conversion from bio-based feedstocks, aiming to shrink the carbon footprint while retaining high enough purity for sensitive products. Each incremental step lands us closer to a compound that ticks more boxes on both performance and sustainability.
Facing Toxicity Risks: Research and Awareness
Butyl salicylate isn’t exempt from health and safety scrutiny. There’s always a push to map out exactly how it behaves inside living tissues and the wider environment. Current toxicology reviews in Europe rate it as having “low concern” for acute toxicity, but persistent or high-dose exposure could still trigger discomfort, sensitization, or chronic effects. Regulators check for skin and eye irritation before approving new uses, which pushes companies to run patch tests and guarantee consumer safety. Recent animal studies suggest low bioaccumulation, but researchers keep a close eye on any long-term consequences, as regulatory groups won’t hesitate to clamp down if new risks emerge. Understanding these hazards helps keep formulation choices honest and clear; it prevents the corporate temptation to swap in a risky compound just because it sounds “nicer” on a label.
What Comes Next: Future Prospects and Trends
The market for butyl salicylate stands on the edge of change, driven by shifting tastes, regulatory tweaks, and technical progress. Synthetic fragrances and green chemistry both drive demand, with more R&D energy aimed at upgrading stability, lowering toxicity footprints, and stretching the value chain. In the next five or ten years, plant-based and upcycled versions will likely find their way into newer product lines. Emerging fields like controlled delivery for pharmaceuticals, advanced materials for art restoration, and special coatings for electronics might put their spin on the old ester. Industry, regulators, and researchers all play a role here, balancing consumer demands with higher safety standards and more transparent supply chain practices. For folks in the trenches—whether in labs, procurement, or production—flexibility and honesty about raw material sourcing and product safety will help steer the story of butyl salicylate into new territory.
What is Butyl Salicylate used for?
From Perfume Bottles to Cleaning Sprays
Butyl salicylate slips into daily routines almost unnoticed. For most people, the name probably sounds clinical, but its scent reminds me of the light sweetness you catch after spraying a body mist or using a new floor cleaner. The reason: butyl salicylate has been prized for its fresh, slightly herbal aroma since it first started showing up in perfumes and cosmetics more than a century ago.
Manufacturers rely on this ingredient to help blend fragrances together and mellow out harsher notes. A whiff of an old bottle of cologne sits on my shelf—well past its prime, but still holding onto a faint, recognizable warmth—and that’s butyl salicylate working behind the scenes. It doesn’t just sweeten up a formula; it helps keep fragrances stable so they last longer unopened.
Personal Care and Fragrance Science
Fragrance chemists pay attention to how ingredients interact with skin. The molecule behind butyl salicylate balances well with natural oils, improving not only scent but how subtle and even the application feels. Skin irritation and allergic reactions always stay top-of-mind for makers. In the United States and Europe, regulatory agencies have given it a nod as safe at low concentrations, following reviews of available research. This clearance comes after examining both real-world consumer use and laboratory tests, since health comes first.
I remember watching my partner struggle to find a lotion that felt light but lingered pleasantly. Her skin is sensitive—many complex perfumes left a rash, but unscented varieties didn’t interest her. Looking up ingredients lists, I noticed that milder blends often included butyl salicylate. Dermatologists and consumer advocates suggest checking ingredient panels because even though it’s considered safe, it can trigger problems for anyone with a salicylate allergy.
Beyond Perfume: In Your Cleaning Closet
Go down the cleaning aisle and you’ll spot this compound again. Cleaning products—from piney sprays to furniture polish—use butyl salicylate for more than just a pleasant smell. Chemists add it so harsh solvents smell less like a laboratory and more like home. I once helped my neighbor clean vintage speakers with a product that offset the musty scent with a crisp floral twist. He laughed, saying it barely smelled like a chemical at all. Factories that manufacture cleaning agents use butyl salicylate to bridge the gap between function and comfort, making the experience better for people who spend time scrubbing, dusting, or freshening up a space.
Environmental Concerns and Safer Choices
Science circles talk a lot now about exposure to synthetic chemicals and their impact on the environment. Butyl salicylate breaks down in soil and water, but chronic exposure raises questions. Studies tracking aquatic life and wastewater note that while the compound doesn’t accumulate easily, high concentrations can stress sensitive species. Some green product makers have started searching for alternatives that leave an even lighter touch, often turning to plant-based extracts or essential oils for similar white floral notes.
Plenty of folks read labels more carefully these days, whether motivated by allergies, chemical sensitivity, or concerns about long-term impacts. If you want to avoid butyl salicylate, you’ll need to pay attention—especially to fine fragrances and multi-purpose cleaners.
Spotting Opportunity in Transparency
Demand for cleaner ingredients keeps shaping industry trends. Perfumers and cleaning brands try to stay ahead by disclosing ingredient lists and funding safety research. That transparency helps consumers and health professionals make better choices. I’ve seen firsthand how friends in the beauty industry respond to questions about lesser-known ingredients—every conversation raises awareness, and in the long run, those questions push for safer products across the board.
Is Butyl Salicylate safe for skin?
Understanding Butyl Salicylate
Butyl Salicylate shows up in personal care products like perfumes, creams, and sunscreen. This colorless liquid has been widely used to carry fragrance and boost how products feel on skin. Many people have never paused to wonder about its safety; only the long ingredient lists catch the eye before being quickly ignored.
Why People Care About Ingredients
Many of us have dealt with skin trouble. Rashes, redness, or breakouts can completely shift your day. As someone with sensitive skin, I’ve learned the hard way to pay close attention to what goes in these bottles. Even ingredients with long scientific names deserve a closer look, especially if they end up on our faces or hands.
What Science Says About Butyl Salicylate
Butyl Salicylate does not have a history filled with red flags. Research from the European Chemicals Agency points to low toxicity levels, showing only mild irritation potential in animal studies, but no links to long-term harm like cancer or reproductive issues. The Cosmetic Ingredient Review Expert Panel, a group funded by the cosmetic industry but composed of independent scientists, reviewed this compound and found it safe for use in today’s typical concentrations.
One thing stands out: Butyl Salicylate belongs to the salicylate family, where aspirin also belongs. Those with “salicylate sensitivity” or aspirin allergies could react after exposure. These cases seem rare. Most people will brush, spray, or lather up products containing this ingredient and never feel a thing. Allergic types, or parents shopping for kids with unpredictable skin, might want to read labels a bit closer.
Concentration and Exposure Matter
It’s not just which ingredients sit in the bottle; it’s how much. Regulatory bodies across North America and Europe only allow Butyl Salicylate at certain levels, keeping total exposure far below the amounts ever shown to hurt lab animals. Strong scientific oversight and frequent product testing help make sure companies stay in line. No ingredient is universally safe, though. If something doesn’t work for your body, all the safety reviews in the world don’t make a difference.
Balancing Benefits and Precautions
People keep turning to Butyl Salicylate because it boosts fragrance staying power and makes sunscreen spread more evenly. For most folks, these side benefits outweigh the small risk. I’ve used products with this ingredient, both on myself and my family, without issue. I always watch for any redness or strange feeling after applying a new product, and patch testing can help catch problems early. Contact a dermatologist for help with unexplained breakouts—many doctors can help sort out whether a product or ingredient causes trouble.
What to Do If Concerns Remain
Some shoppers want more control. Everyone deserves clear answers when it comes to what lands on their skin. Look for brands that publish full safety assessments. Explore alternatives without synthetic perfumes or lesser-known chemicals if skin tends to overreact. Don’t hesitate to call customer support lines and insist on specific information, especially if you manage allergies or sensitive skin.
Personal care should build trust. Science-driven oversight and open conversation about ingredients let consumers make informed, safe choices—no fancy language or complicated steps required.
What are the side effects of Butyl Salicylate?
Understanding Butyl Salicylate and Its Usage
Butyl salicylate plays a role in modern manufacturing, mostly showing up in fragrances, personal care products, and some cleaning items. People come across this compound more often than they realize; it hides in the ingredient lists with little fanfare. The main attraction comes from its ability to keep products smelling better for longer. Yet, even a common additive deserves a closer look—especially on what it brings along aside from the pleasant scent.
Short-Term Reactions People Might Notice
Working in environments with regular chemical exposure, I've seen colleagues develop random reactions that seem minor at first. Butyl salicylate gets absorbed by the skin and, for some, that means redness, itching, or even a mild rash. This isn’t imaginary. Research like the findings published by PubChem and the International Journal of Toxicology points out that mild irritation crops up in a portion of people, especially with repeated use. In rare cases, someone with particularly sensitive skin or an underlying allergy could face hives or swelling after using a scented lotion or soap.
It’s not just the skin, either. People using sprays or concentrated solutions indoors sometimes report headaches or dizziness—usually after working in stuffy rooms. Breathing in strong chemical odors day after day takes a toll. Salicylates as a group even carry a reputation for causing mild respiratory discomfort, and butyl salicylate fits this pattern.
Longer-Term and Internal Concerns
No one expects a fragrance ingredient to wander through the bloodstream, but certain chemicals do just that. Butyl salicylate can be absorbed in small amounts through the skin. The main worry centers on those already sensitive to salicylates—a category that includes folks who avoid aspirin for medical reasons. Large enough amounts, especially with regular exposure, could upset the stomach or cause ringing in the ears, similar to a mild overdose of salicylate medication. This risk stays low for most people using consumer products, but it’s not zero.
Long-term exposure without protection, like in factory settings, deserves attention. A few workplace studies (see CDC chemical safety bulletins) highlight employees who handled high volumes showing mild chronic effects—recurring headaches, mild upper airway irritation, or stomach discomfort. Workers tell stories about not wearing gloves or skipping proper ventilation only to feel fatigued or develop a “chemical taste” by the end of a shift.
Reducing Risks and Staying Informed
Transparency from manufacturers and accessible safety data play a big role in protecting people. Anyone handling raw butyl salicylate, especially in higher concentrations, benefits from gloves, goggles, and a workspace with good airflow. At home, reading ingredient labels on everyday products gives a bit of power back—choose options free from unneeded chemicals. Some skin reactions clear up as soon as people identify and cut out the irritant. For those with a known salicylate sensitivity, consulting an allergist before using a new personal care item can help avoid nasty surprises.
Better safety education holds value not just for industry folks but for the end user. In the rush toward scented, longer-lasting products, side effects deserve serious attention. Listening to your own body, trusting common sense, and asking questions whenever doubt creeps in—the basics go a long way in staying safe.
Is Butyl Salicylate the same as salicylic acid?
Looking at the Ingredients
It’s easy to spot salicylic acid on labels—skincare fans see it everywhere. Butyl salicylate appears less familiar. Flip over a sunscreen, fragrance, or cleansing oil and it sometimes sneaks in alongside more recognizable actives.
Based on decades spent reading ingredient lists as both a science lover and frequent buyer, it’s clear these two aren’t identical. Both start with a similar chemical backbone, but the way brands use them couldn’t be more different. Salicylic acid acts as a beta hydroxy acid, celebrated for unclogging pores and exfoliating skin. My first introduction came through an acne face wash—tingling, drying, but effective.
Chemistry Matters
Salicylic acid contains an aromatic ring with a carboxylic acid and a hydroxyl group. These chemical parts allow it to shed dead skin and help manage blackheads.
Butyl salicylate changes the structure by adding a butyl group via an ester link. This tweak transforms its behavior. Instead of aggressively clearing out skin or pores, butyl salicylate becomes a “fragrance fixative” — something that helps scents last longer and blend more smoothly. You won’t find any exfoliating power here. I notice it mainly in products designed to smell pleasant.
Safety and Regulation
Regulators keep a close watch on both chemicals. Salicylic acid counts as a drug in some places, especially when levels go over 2%. Butyl salicylate falls under a different set of rules. It can cause allergic reactions if used in high concentrations, but it’s not seen as an irritant like its acidic cousin.
Organizations like the US FDA or EU’s SCCS set limits based on current evidence and reported side effects. The Cosmetic Ingredient Review sees butyl salicylate as safe in typical concentrations. Salicylic acid gets its clearance at low levels—enough to clean up skin, not cause burns.
Why Confusion Happens
People sometimes mix them up because both feature “salicylate” in the name. This happens a lot in online discussions, especially when shopping for new skincare products. Understandably so—the beauty world keeps introducing fresh-sounding molecules every month. Salicylates make up a big family, used in sunscreens, pain-relievers, and fragrances. Both these ingredients can coexist in one formula, working totally separate jobs.
What Shoppers Deserve to Know
It’s important to know the difference before picking a product. If clearing up acne stands as the goal, salicylic acid remains the only proven bet among the two. If relaxing with a nice-smelling lotion or sunscreen matters more, spotting butyl salicylate in the ingredients just means added fragrance staying power.
Labels don’t always make things obvious. I recommend looking for the full ingredient list before buying anything that promises benefits for skin health. When in doubt, asking a dermatologist or trusted pharmacist works wonders.
Clear Demand for Better Education
There’s a lot of power in teaching shoppers about chemistry in everyday products. With so many beauty fads swirling online, reliable information cuts through hype. Forums, social channels, and brand websites need to keep explanations honest and free from bold claims. Regulators, brands, and skin health professionals can all step up to explain why certain molecules appear in our routines—and what risks or benefits stand behind them.
Separating marketing from science gives people a real shot at making smarter choices.
Can Butyl Salicylate cause allergic reactions?
Getting Real About Butyl Salicylate and the Skin
Most people walk through daily routines without thinking too much about every ingredient in their favorite products. Butyl salicylate—a chemical often tucked away in ingredient lists—pops up in perfumes, sunscreens, and even skincare. It brings a floral fragrance and acts as a solvent. Not many pause to consider how their skin reacts until something feels off. That itchiness isn’t just in their imagination.
Allergy Risk: Why It Matters
Back in my own college days, standing in a drugstore aisle, I believed anything at the local pharmacy was safe. A few rashes later, I started reading labels. Many like me never connect scented lotions or sprays to red, irritated skin. Yet, reports from dermatologists and studies from places like the American Contact Dermatitis Society paint a different picture. Allergic contact dermatitis linked to preservatives and fragrant molecules, including some salicylates, keeps climbing. Even stuff that smells great on a test strip can mean trouble on real skin.
Who’s At Risk?
Some folks shrug off minor rashes, but others end up with real discomfort. Sensitized skin—already sensitive or damaged—reacts more quickly. Kids, people with eczema, and those spending hours in the sun start seeing patterns. The trickiest part? Reactions don’t show up overnight. They might build up over days. Suddenly, what felt like a tiny inconvenience turns into full-blown redness and swelling.
Research pieces, like the data cataloged by the European Chemicals Agency, confirm butyl salicylate can cause allergic reactions in rare cases. The incidence rates sit lower than for the big offenders like parabens or formaldehyde-releasing preservatives. Still, “rare” matters if you happen to be the one affected.
Why Listen?
Ignoring small symptoms gives the false sense that everything’s fine. But continued exposure only piles on. That’s exactly how a simple rash drifted into chronic problems for me. I once learned the hard way with a favorite aftershave. What worked for a while started turning my jawline raw. My dermatologist broke it down: repeated contact lets even mild allergens train the immune system to attack them.
Safer Choices in Daily Life
Finding another product often feels overwhelming. Transparency helps. Brands providing full disclosure—and clear, readable lists—stand out. People living with allergies benefit from patch testing. My own route meant carrying a small notepad of “problem” ingredients. That simple note saved time and pain. New apps scan ingredient lists, making the hunt less frustrating.
A few basic steps go a long way. Try a new lotion on a small area. Give it a couple of days. Itchy? That spot tells you what’s coming if you commit to daily use. For people dealing with repeated irritation, asking a dermatologist about ingredient testing opens up real answers.
Better Awareness, Better Results
The rise of “clean” beauty pushes companies to rethink what goes onto shelves. Recognizing that butyl salicylate poses a potential allergy risk—however small—keeps both companies and customers alert. Choices in the self-care aisle shouldn’t mean rolling the dice on comfort. My experience taught me: reading the fine print and having an honest conversation with my doctor paid off more than any promise on a label.
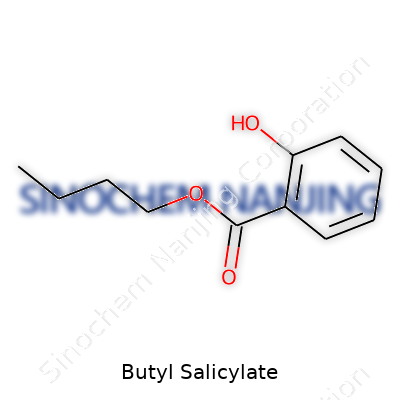
| Names | |
| Preferred IUPAC name | Butyl 2-hydroxybenzoate |
| Other names |
Butyl 2-hydroxybenzoate Salicylic acid butyl ester Benzoic acid, 2-hydroxy-, butyl ester |
| Pronunciation | /ˈbjuː.tɪl səˈlɪ.sɪ.leɪt/ |
| Identifiers | |
| CAS Number | 205-664-1 |
| 3D model (JSmol) | `load chemical.molcoord "O=C(OCCCC)C1=CC=CC=C1O"` |
| Beilstein Reference | Beilstein 1208731 |
| ChEBI | CHEBI:34660 |
| ChEMBL | CHEMBL147121 |
| ChemSpider | 12145 |
| DrugBank | DB14692 |
| ECHA InfoCard | ECHA InfoCard: 1000006-593 |
| EC Number | 205-427-8 |
| Gmelin Reference | 72186 |
| KEGG | C17238 |
| MeSH | D017377 |
| PubChem CID | 7137 |
| RTECS number | WZ0450000 |
| UNII | 3TI5LT896A |
| UN number | UN1986 |
| CompTox Dashboard (EPA) | DTXSID7020182 |
| Properties | |
| Chemical formula | C11H14O3 |
| Molar mass | 208.25 g/mol |
| Appearance | Colorless liquid |
| Odor | ester-like; sweet |
| Density | 1.055 g/cm3 |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.0015 mmHg (25°C) |
| Acidity (pKa) | 13.6 |
| Basicity (pKb) | 13.18 |
| Magnetic susceptibility (χ) | -66.0e-6 cm³/mol |
| Refractive index (nD) | 1.502 |
| Viscosity | 7.05 mPa·s (25 °C) |
| Dipole moment | 3.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 415.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -602.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7346.7 kJ/mol |
| Pharmacology | |
| ATC code | N02BA15 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause an allergic skin reaction. Toxic to aquatic life with long lasting effects. |
| Precautionary statements | Precautionary statements: **"If medical advice is needed, have product container or label at hand. Keep out of reach of children. Read label before use."** |
| Flash point | 122°C |
| Autoignition temperature | 400°C |
| Lethal dose or concentration | LD50 oral rat 16,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral Rat 4,890 mg/kg |
| NIOSH | NA3476000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Butyl Salicylate: Not established |
| REL (Recommended) | 5000 mg/L |
| Related compounds | |
| Related compounds |
Methyl salicylate Ethyl salicylate Propyl salicylate Amyl salicylate Salicylic acid |

