Butyl Propionate: A Closer Look at a Modern Industrial Solvent
Historical Development
Butyl propionate didn’t leap into the chemical limelight overnight. Its journey grew from the broader quest for performance solvents that saw chemists tinkering for decades. During the boom of the mid-twentieth century, factories searched for alternatives to slow-drying, heavily toxic options. People wanted something less dangerous to handle, but also versatile enough to work well in coatings, inks, and cleaners. My years working alongside process engineers revealed the value of something reliable, free from the fuss of the old genera of harsh-smelling solvents. Butyl propionate took time to earn its reputation, mostly because early commercial blends struggled to strike a balance between evaporation rate and odor profile.
Product Overview
At a glance, butyl propionate looks and acts much like other mid-weight esters: clear, colorless, and packing a slightly fruity odor. My own time spent in formulation labs taught me quickly that this isn’t just another face in the ester crowd. The compound walks that fine line between efficiency and gentleness. This makes it a recurring player in industries that can’t afford to compromise finish quality but don’t want to risk overexposing workers to stronger, legacy chemicals.
Physical & Chemical Properties
Scientists checking the specs see a liquid with a density lower than water and a modest boiling point that helps it evaporate neither too fast nor too slow. Anyone who’s spilled a beaker across a stainless bench knows the telltale aroma streak, unmistakable but not overwhelming. With a flash point that managers won’t lose sleep over and a solvency suited to pigments, butyl propionate fits standard operating environments well. Its miscibility with many organics opens doors across paint and pigment production, creating reliable, even coatings. I’ve seen how this translates directly to easier cleanup and thinner application, providing smoother finishes without sacrificing coverage.
Technical Specifications & Labeling
Every time drums arrive on the loading dock, quality teams check specific gravity and purity. Trust comes from consistent distillation and verified labels, so labs pay close attention to supplier standards. I’ve stood over enough titration setups to respect a sample that sits true to its label. Reading an SDS will confirm most of what production teams already know: butyl propionate comes with its safety demands, but it beats out a host of far more volatile peers. Good labeling means fewer surprises at transfer and blending, saving both cost and time for everyone on the floor.
Preparation Method
The classic route sees butanol and propionic acid combined in a straightforward esterification reaction. Watching skilled technicians work, I’ve seen this process honed with the addition of a touch of acid catalyst and smart water removal, ensuring high yields in commercial reactors. Careful temperature control reduces by-products, and good vacuum distillation strips away any trace impurities. In an industry where bad batches cost real money and trust, operators don’t cut corners. Getting the preparation right improves not just yield, but worker trust, customer satisfaction, and environmental compliance.
Chemical Reactions & Modifications
Chemists like to push boundaries, and butyl propionate’s ester bond offers a straightforward target. Hydrolysis brings it back to butanol and propionic acid, a reversible step easily leveraged for recovery or test analysis. In real production, though, it’s the physical interactions—how it dissolves pigments, wets surfaces and acts in blends—that matter most. The molecule keeps stable under most industrial settings, so companies can store it without worrying about spontaneous changes that would degrade product performance.
Synonyms & Product Names
Old-timers may call it butyl propanoate, and regulatory filings use similar terms. Manufacturers stamp their own brands, but the chemistry remains unmistakable. This language dance often trips up new buyers, but the savvy know it's the CAS number that locks down what’s actually in the drum. I’ve seen confusion cost hours in production delays—underscoring the benefit of clear communication in procurement and operations.
Safety & Operational Standards
Over the years, regulations pushed companies to tighten control over solvent handling, and for good reason. Inhalation of vapors can bring headaches and dizziness, especially in poorly ventilated areas. Standard safety protocols like fume hoods and PPE form the front-line defense. Training helps too: reminding staff to avoid open flames and use grounded pumps. Facilities performing regular air monitoring avoid long-term exposure issues, and proper storage practices reduce fire risk. I’ve witnessed the difference a well-led safety culture makes—near-miss incidents drop, insurance headaches fade, and it’s easier to attract good talent.
Application Areas
Butyl propionate found a strong foothold in paints and coatings, prized for balancing speed of application with even drying. Printers enjoying smooth press runs appreciate its ability to thin inks without tempering pigment strength. Its gentle solvency helps chemists formulate cleaning solutions for electronic and automotive components. In these fields, I’ve noticed how small changes in solvent blend can make or break a batch—surface finish, application ease, and worker comfort all depend on details like this. Trends in green chemistry focus on solvents that offer performance without heavy residue or persistent environmental impact, making butyl propionate increasingly attractive.
Research & Development
Research teams carry out formulation studies, searching for ways to cut hazardous emissions without losing drying performance. Laboratory tests track compatibility with film-formers, resins, and colorants. I’ve seen teams devote months to studying evaporation curves under changing humidity—and then toss it all aside when a new flame-retardant regulation comes out. The real value comes from cross-department collaboration: chemists, application engineers, and end users working to tweak formulas so that operators feel the change in day-to-day work. New blends focus on reducing odor and boosting green content, trends sure to continue as rules tighten worldwide.
Toxicity Research
Toxicologists take a close look at how butyl propionate behaves in biological systems. Short-term inhalation usually causes mild irritation, not enough to rule out its use, but enough to require thoughtful ventilation and personal protection. Environmental studies dig into degradation, looking for any sign of persistent risk. Disposal practices concentrate on keeping effluents below critical thresholds. Places with strong occupational monitoring see fewer complaints of chronic symptoms—testament to the importance of robust health surveillance and open reporting channels.
Future Prospects
The next years look bright for this solvent, especially as manufacturers seek to balance tradition with innovation. Pushes toward waterborne coatings pressure companies to find add-ins like butyl propionate that enhance leveling without excessive VOC emissions. As more governments introduce stricter air quality controls, demand will swing towards safer, biodegradable chemicals. Emerging research looks at renewable feedstocks for ester production, hinting at a world where butyl propionate starts with plant-based butanol or propionic acid. If regulatory frameworks support this, suppliers will move quickly, eager to capture premium markets. I’ve watched reformulation cycles turn on changes like this, sometimes taking months, but always driven by the real-world need to combine high performance with sustainability and health. There’s little doubt the story of butyl propionate remains unfinished—its role will keep evolving with the chemistry of tomorrow.
What is Butyl Propionate used for?
More Than Just a Chemical Name
You see names like butyl propionate in ingredient lists and safety data sheets. Most people scroll right past it without thinking. I did the same until I managed shipments for a paint company. Seeing barrels stamped with the name, I realized this wasn't just technical lingo—it touched almost everything from art supplies to auto shops.
Making Paint Work
Pick up a can of paint, especially quick-drying types for cars or furniture, and chances are it owes something to butyl propionate. This liquid keeps paints from drying inside the can. High-quality spray finishes wouldn’t come out smooth or even without help from solvents like this one. With low odor and not as flammable as many others, butyl propionate stands out in busy factories where safety and performance matter daily.
Helping Cleaners and Fragrances
Open a strong cleaning product in a public bathroom or kitchen and notice how it doesn’t burn your nose the way old-school chemicals did. Manufacturers switched to butyl propionate to get grease-cutting power but leave harsh smells behind. It dissolves oily messes fast and leaves glass shining, all without sticking around in the air. Even fragrances in some personal care products blend more easily thanks to this lesser-known solvent.
Sneaking into Printing and Inks
Printers—from small presses to massive machines—take liquid color and make it dry in seconds. Here, butyl propionate gives ink both its flow and snap. The compound keeps ink wet just until it hits the paper, then helps it set quickly without blurring. This matters for magazines, books, boxes—anything mass-produced. In my own time working at a local newspaper, I saw the difference its absence made—smears, smudges, wasted runs.
Safer, Yes—But Still Needs Respect
Even with its advantages, butyl propionate needs attention. Safety agencies require special handling to prevent spills or accidental inhalation. The chemical is less toxic than many predecessors, but constant exposure has risks. Factory workers, truck drivers, custodians—anyone using it should get basic training and gloves. Over the years, making these standards part of my routine meant fewer accidents, and everyone got home safe.
Finding Smarter Substitutes
Many companies have moved away from heavier, more hazardous solvents. Some switch to water-based systems or blend in renewable chemicals, but not all jobs suit these swaps. Short-chain esters like butyl propionate fill a tricky gap for now, where performance and safety walk a fine line. As regulations grow tighter and green chemistry keeps pushing forward, expect even better options ahead—but for the moment, this compound keeps thousands of businesses ticking.
Why the Details Matter
Butyl propionate isn’t some corporate concoction with no real-world impact. Every time a painter lays down a clean coat or a janitor wipes a streak-free window, the role of this solvent comes through. Making smart, informed decisions about chemicals like these keeps products safe, jobs running, and communities protected. If you’re in a workshop, a lab, or even just buying new furniture, knowing these names helps you ask the right questions.
Is Butyl Propionate safe to handle?
A Careful Look at Butyl Propionate
Butyl propionate doesn’t show up on most folk’s radar unless they work in a lab or on an industrial site. It lives in the world of solvents, coatings, and the chemical recipes behind paints, fragrances, and cleaning products. If you’re unlucky enough to spend time around it, safety questions tend to pop up right away. Having handled my share of chemical jars and soaked rags during a college job in an auto shop, I learned firsthand that familiar chemicals don’t always play nice—especially when used without care.
What Happens on Contact?
On a basic level, butyl propionate is a clear liquid with a mild odor that reminds me a bit of apples, though nobody recommends sniffing it. Touching the liquid on your skin can cause dryness or mild irritation. Prolonged contact makes things worse—peeling skin, redness, even tiny chemical burns in bad cases. Eyes sting sharply if a splash lands there. Vapors in a poorly ventilated room don’t do the lungs any favors, either, setting off coughing or lightheadedness if you breathe too much in.
Research from the National Institute for Occupational Safety and Health shows that exposure limits exist for good reason. Even moderate exposure over a shift leads to headaches, sore throats, or worse. A lot of cases crop up where someone ignores gloves. Once, a coworker wiped butyl propionate off a bench with a bare hand, brushed her face, and got red streaks an hour later. Lab safety sheets aren’t being dramatic; stories like these remind me that our skin isn’t as tough as we think.
Fire Risks and Spills
Butyl propionate lights up with a single spark and burns with a nearly invisible flame. Once, we had a spill where a cleaning rag sat too close to a space heater. Smoldering, not obvious at first, until the strong, sweet smell turned into an acrid warning. Firefighters arriving later said we got off lucky; the blast radius could’ve sent glass flying. The lesson: store this liquid away from heat or static, always recap bottles, and never trust a casual clean-up with old habits.
Handling It Right
Gloves, goggles, and fresh air—the trio keeps you out of trouble. Companies that invest in proper ventilation save themselves sick days and liability, something my old shop skimped on until a routine inspection forced their hand. Even short exposure in a poorly-ventilated space lets vapors build up, making everyone feel foggy or nauseous by lunch.
Plenty of guides show how to use spill kits and fire extinguishers with the right class rating. Workers must know exits and keep safety data right on a clipboard, not buried in a drawer. Emergency showers belong close by, never at the far end of a hallway. Training should come before you ever twist open the cap. So much harm comes from simple shortcuts—saving thirty seconds, risking weeks of irritation or worse.
Room for Improvement
If a workplace depends on butyl propionate, a safer alternative deserves real consideration. Green chemistry keeps churning out new options every year, cutting down fire hazards and health risks without losing performance. Until that day comes, honest training and strict habits matter most. I’ve seen what carelessness looks like, and it’s never worth the cost.
What are the main physical and chemical properties of Butyl Propionate?
What Butyl Propionate Really Brings to the Table
Butyl propionate holds its own among solvents with a clear, colorless liquid form and a fruity odor—not unlike many esters from the same family. Chemical formulas and numbers may seem intimidating, but here it’s pretty straightforward: butyl propionate combines the building blocks of butanol and propionic acid. This combination shapes both its physical feel and how it actually behaves in the world.
How Butyl Propionate Behaves Physically
Pick up a bottle of butyl propionate, and you'll find a low-viscosity liquid. This stuff pours easily, more like water than syrup. It boils at about 145°C and freezes somewhere around -78°C. People handling it in the field notice the quick evaporation, even at room temperature, which makes it valuable for applications where fast drying really matters. Its specific gravity hangs just under one, meaning it floats on water, and it sports a vapor pressure of roughly 3-4 mmHg at 20°C, so it’ll give off vapor you can often smell in the air. Many who work with solvents know to keep it capped tight – otherwise, it slips away fast.
Chemical Properties That Stand Out
Putting butyl propionate to the test in the lab or industry, you’ll see it doesn't stay idle: it mixes smoothly with a long list of organic solvents—alcohols, ketones, ethers—but won’t cut it with water. This helps explain why it shows up in paints and inks so often. Butyl propionate reacts like a typical ester, meaning acids and bases can break it down, churning out butanol and propionic acid along the way. Under normal conditions, it stays pretty stable, not just sitting in the container itching to react. When exposed to heat or strong oxidizers, that story flips, highlighting the need for careful handling in busy workplaces.
Real-World Importance and Industry Caution
My years in chemical distribution brought plenty of chances to see butyl propionate put to work. It keeps popping up because it pairs low toxicity (for a solvent) with pleasant smell and solid dissolving power. In manufacturing paint, coatings, and cleaning products, operators value how it makes products easier to apply—nobody wants sticky streaks or a bottle that separates overnight.
Despite those pluses, there’s a flip side in safety. Butyl propionate is flammable, which means storage calls for more than a casual shelf spot in a hot warehouse. Proper ventilation and sensible protective gear keep people clear of overexposure. Short-term whiffs might just annoy with headaches or dizziness, but long-term exposure deserves respect and professional attention. That's why companies often keep extensive safety training for team members who handle these types of chemicals.
Pursuing Safer and Smarter Usage
With supply chain changes in recent years, companies watch costs while weighing health and environmental rules. Tightening up workplace ventilation and swapping in greener alternatives if the job allows can reduce headaches—both literally and for compliance teams. Regulatory agencies track esters including butyl propionate, urging checks on how much gets released into the air. Responsible manufacturers invest in containment and recovery systems to hold down emissions and keep workplaces safe without tossing aside performance. On a personal note, seeing companies step up with better controls made teams more confident and kept risk in check.
How should Butyl Propionate be stored and transported?
Understanding the Real Risks
Dealing with chemicals such as Butyl Propionate takes more than a technical manual and a quick glance at a safety sheet. Its fruity odor hides a volatile side, and missing details can cost real money or even safety. One slip—too much heat, spark, or a poorly sealed cap—can set off headaches that spill far beyond the warehouse.
Temperature: No Room for Guesswork
Leaving drums in sun-baked yards or next to a heat vent doesn’t just risk ruined batches. At warmer temperatures, Butyl Propionate churns out flammable vapors. That’s the kind of hazard that can turn minor negligence into a headline. Workers deserve peace of mind, and so do nearby neighbors. Storing drums in cool, shaded, and well-ventilated buildings cuts risk. HVAC, away from direct sun, and regular temperature checks keep storage honest. Those steps never feel like overkill after an accident.
Moisture and Contamination: Little Things Build Up
I’ve seen chemists cringe when water finds its way inside sealed pails. Even one careless lid can lead to ruined raw material. Moisture brings unwanted reactions, which might degrade Butyl Propionate or generate acids and pressure inside containers. Simple routines—tightly sealed caps, dry storage areas, pallets off the concrete—block small leaks from turning into big problems. Taking the time to label everything sharply avoids mix-ups. “Close enough” never satisfied anyone, especially with chemicals.
Sparks, Static, and Open Flames: Keep Distance
Forklift batteries, frayed extension cords, or a casual cigarette near the loading dock have all sparked headaches in places I’ve worked. Take the warnings on flammable liquids seriously. Keep Butyl Propionate far from open flames. Use spark-proof tools, ground metal drums, and avoid static build-up during transfer. Safety showers, eye wash stations, and fire extinguishers aren’t just for show. Workers should always know their location and how to use them.
Moving Butyl Propionate: Not a Routine Haul
Transporting Butyl Propionate needs drivers trained beyond a run-of-the-mill delivery. Tanks and drums must survive bumps in the road, temperature swings, and the chance of a rollover. Secure containers tightly, protect from impacts, and use certified hazardous material carriers. Keep the paperwork right and placards clear—regulations never forgive ignorance. Direct routes and time limits shave risk. Busy terminals raise odds of delays, so skip the shortcuts.
People and Training Matter Most
Accidents almost always point to skipped steps or rushed jobs. No checklist can replace experienced hands. Ongoing training keeps new hires from making rookie mistakes. Managers walking the site spot problems better than office-bound memos ever could. Questions deserve answers, and nobody should fear raising a concern about a leaking drum or missing label.
Room for Smarter Solutions
Real-time temperature loggers and leak detectors give early warnings before a problem explodes. Bulk tank monitoring, automatic shutoff valves, or double-walled storage pay off over the years. Insurers notice, too. Spend now, save later.
Bottom Line
Take Butyl Propionate seriously. It’s not about worrying over every drop—just giving people the tools, space, and time to do the job right. Health, safety, and business depend on it.
What is the CAS number of Butyl Propionate?
The Nuances of Chemical Identification
Butyl Propionate has earned its spot in labs and factories for a reason. It belongs to a class of chemicals known as esters, and it finds its way into several industries, from paints to perfumes. What ties this chemical to regulations and smooth operations? Its CAS number: 590-01-2. That string of digits might look like just another code, but in chemical work, it can make the difference between success and costly mistakes.
Why a CAS Number Matters More Than You Think
Every working chemist has run into a jumble of names for a single compound. Sometimes, one chemical gets half a dozen different nicknames, trade names, or translations from around the world. This makes hunting down the exact substance a risky business. A mislabel in a recipe or shipping label could easily lead to the wrong chemical arriving at the lab bench or production line. The CAS number works as a sort of chemical fingerprint—unique and universal. Butyl Propionate’s 590-01-2 tells anyone worldwide, no matter the language, exactly which compound sits in front of them.
Tales from the Field: Safety and Quality Assurance
Working on a university project in organic chemistry, I watched a mix-up unfold because someone read the trade name "butyl propionate" instead of the CAS number. It turned out the supplier had sent a different ester altogether. The experiment failed, wasting days of work, and also raising big safety questions. No one can feel confident around chemicals unless they know what they’re dealing with, both for health and for the result in the flask. Companies that ignore or fudge CAS numbers usually face fines, ruined batches, or, in the worst cases, injuries.
Over the years, I’ve learned that regulatory bodies worldwide—including OSHA in the U.S. and ECHA in the EU—lean on CAS numbers to keep substances catalogued, tracked, and understood. This system means ingredients lists, shipping manifests, and safety data sheets speak the same language, from the docs in the office to the folks on the factory floor.
The Real-Life Role of CAS 590-01-2 in Production
Manufacturers working on coatings, adhesives, or flavorings rely on precise sourcing. Ordering Butyl Propionate boils down to one simple phrase: “Send me CAS 590-01-2.” This eliminates confusion, even when new suppliers enter the picture. For engineers and lab managers, this small act ensures that paints dry with the right finish, that fragrances keep their note, and that no one breathes in fumes from a lookalike compound. Purity levels and toxicity concerns all get relayed back to that number in global chemical databases.
Keeps the Wheels Turning
Anyone looking to improve safety and quality in chemical supply chains should push for strict use of CAS numbers, not just for paperwork but at every stage of handling and labeling. Training new hires to check this detail will keep dangerous errors and financial hits at bay. I’ve seen that small investment in education pay off with fewer recalls and more consistent products landing on shelves. It’s easy to treat the CAS number as an afterthought, but it’s actually the foundation that keeps chemical handling running smoothly.
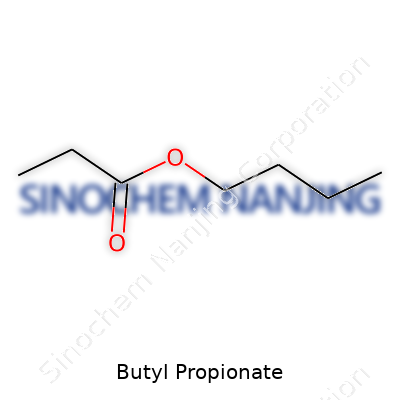
| Names | |
| Preferred IUPAC name | Butyl propanoate |
| Other names |
Propionic acid butyl ester Butyl propanoate n-Butyl propionate 1-Propanoic acid, 1-butyl ester Butyl n-propionate |
| Pronunciation | /ˈbjuːtɪl proʊˈpiːəneɪt/ |
| Identifiers | |
| CAS Number | 590-01-2 |
| Beilstein Reference | 1071403 |
| ChEBI | CHEBI:8772 |
| ChEMBL | CHEMBL3158728 |
| ChemSpider | 7075 |
| DrugBank | DB14596 |
| ECHA InfoCard | ECHA InfoCard: 100.011.597 |
| EC Number | 203-690-9 |
| Gmelin Reference | 6887 |
| KEGG | C13807 |
| MeSH | D017380 |
| PubChem CID | 8028 |
| RTECS number | UJ8750000 |
| UNII | J4R0332W4F |
| UN number | UN2348 |
| CompTox Dashboard (EPA) | 7QO6N6P52T |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | 130.18 g/mol |
| Appearance | Colorless liquid |
| Odor | Fruity |
| Density | 0.872 g/cm3 |
| Solubility in water | 0.6 g/L (20 °C) |
| log P | 0.97 |
| Vapor pressure | 0.6 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | pKb: 13.19 |
| Magnetic susceptibility (χ) | -7.31×10⁻⁶ |
| Refractive index (nD) | 1.399 |
| Viscosity | 0.64 mPa·s (at 25°C) |
| Dipole moment | 1.78 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 395.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -471.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3585.1 kJ/mol |
| Pharmacology | |
| ATC code | No ATC code |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H336 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| Flash point | 31 °C (closed cup) |
| Autoignition temperature | 285°C |
| Explosive limits | Explosive limits: 1.1% to 8.3% |
| Lethal dose or concentration | LD50 (oral, rat): 10,100 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 9500 mg/kg |
| NIOSH | WIY |
| PEL (Permissible) | PEL: 150 ppm |
| REL (Recommended) | 250 mg/m³ |
| IDLH (Immediate danger) | IDHL: 1500 ppm |
| Related compounds | |
| Related compounds |
Propyl butyrate Butyl acetate Propyl acetate |

