Butyl Benzoate: Science, Industry, and the Next Chapter
Historical Development
Butyl benzoate doesn’t grab headlines like plastics or pharmaceuticals, yet its story stretches well over a century. Chemists stumbled across benzoate esters as part of a much broader hunt to improve fragrances, preservatives, and solvents. The marriage of butanol and benzoic acid falls into a family of reactions—the classic esterification pushed forward by pioneers like Fischer and Speier in the late 19th century. Researchers took simple raw materials that, by themselves, didn’t offer much, and with some acid catalysis, built a whole menu of esters—each with its own scent, texture, and industrial uses. Over years, butyl benzoate found itself in the thick of perfumery labs, lacquer factories, and on the bench tops of scientists designing better pesticide carriers.
Product Overview
Hardly anyone outside of chemical circles recognizes butyl benzoate, but it quietly finds its way into dozens of products. Transparent, oily, and boasting a sweet, faintly fruity scent, this ester appeals because it dissolves things other compounds can’t touch, resists many forms of chemical attack, and doesn’t break down under reasonable heat or light. Factories depend on it as a solvent, formulators lean on it to deliver medications through skin, and even perfumers add it for a smooth, subtle background note. So, although consumers rarely see its name, they benefit from its reliability and the way it fits into so many chemical toolkits.
Physical & Chemical Properties
Butyl benzoate appears as a colorless liquid, heavier than water, and slips easily across glass or metal surfaces. Its boiling point sits beyond what most household stoves can reach, and vapor pressure stays low under typical storage conditions, making it less likely to evaporate or cause indoor air problems unless handled in bulk. One reason it pops up in specialty uses comes from its ability to dissolve resins, plastics, and cellulose, all without attacking the structure itself. I’ve watched solvents chew up certain plastics in seconds— butyl benzoate won’t do that.
Technical Specifications & Labeling
Every barrel, drum, and bottle of butyl benzoate attracts attention from both regulators and customers for purity and safety. Both the benzoic acid and the butanol source need to meet strict criteria, and the final compound must hit benchmarks for color, water content, and the absence of byproducts like benzyl alcohol or dibutyl ether. Labels identify the batch, purity percentage, and any stabilizers, with hazard pictograms to warn of both fire risk and the need for eye and skin protection. I’ve seen manufacturers rely on robust, transparent data sheets when shipping or receiving this product, knowing that attention to technical details is non-negotiable.
Preparation Method
Industries lean on classic acid-catalyzed esterification for making butyl benzoate. Chemists add benzoic acid and butanol together, often in the presence of sulfuric acid or similar catalyst, stirring and warming the solution until water distills off and the reaction shifts toward completion. Large-scale outfits recycle excess alcohol and neutralize remaining acid, using vacuum distillation to purify the ester. These production lines balance efficiency with safety, because any spill, especially in hot reactors, can create vapor risks or slippery surfaces.
Chemical Reactions & Modifications
Chemists value butyl benzoate’s stability: it doesn’t break down or interact with many ingredients under shelf conditions. Still, if pushed—through hydrolysis in strong acid or base—it can break back into its parent acid and alcohol. Scientists sometimes use it as a stepping stone, modifying the benzoic acid backbone to tack on new groups, chasing targeted pharmaceutical or agricultural properties. Its moderate reactivity means it fits well in recipes where more aggressive solvents fail or cause side reactions.
Synonyms & Product Names
While the label usually says butyl benzoate, researchers and vendors keep a few other names handy—benzoic acid butyl ester, n-butyl benzoate, and among European firms, Butyloxybenzoate. Knowing all these variations helps buyers avoid mix-ups and ensures proper hazard tagging, especially in multilingual corridors of international trade.
Safety & Operational Standards
Anybody who handles butyl benzoate in a lab or plant needs a sharp eye and reliable gear. Liquid spills create slip hazards. Bodily contact can cause irritation, especially with repeated exposure to unprotected skin—a quick rub and rinse won’t always keep problems at bay. Respirators come out when dealing with vapors, especially in poorly ventilated spaces or high-volume operations. Product storage policies require secure containers kept cool and away from open flames, since it burns if given the chance. Companies cross-check each batch with global safety standards, from Europe’s REACH to OSHA guidelines, to build a strong safety culture.
Application Area
Few chemicals cross as many industry lines as butyl benzoate. Pesticide makers pick it for its carrier abilities, letting active ingredients ride into plants or insect coats where pure water-based sprays fall short. Perfumers craft fragrances with its faint, pleasant aroma and ability to blend oil and alcohol phases. Drug developers use it to ferry actives through skin in topical gels or creams—it won’t cause sharp stings or break formulations like harsher solvents. Paint and coating factories count on it for smoothness and steady evaporation, helping prevent clumping or cracking as finishes dry. Even polymer researchers work it into plasticizers, trading off flexibility and flow in materials, usually after plenty of trial-and-error in the lab.
Research & Development
Over decades, teams across continents kept experimenting with butyl benzoate, both as a product and as a raw material to build better molecules. Synthetic chemists keep testing tweaks on the benzoic core, hoping for breakthroughs in drug delivery, agricultural sprays, or safer solvents. Green chemistry labs chase renewable feedstocks and catalysts that shift production away from fossil fuels and cut waste streams down to the bone. I’ve seen these projects in action—scientists juggling yields, economic reality, and customer needs, trying to squeeze one more percentage point from every batch or one fewer allergen from the finished formula.
Toxicity Research
Decades of animal testing, cell experiments, and human monitoring offer a picture of manageable—but not zero—risk. Skin contact for short spells causes mild irritation in sensitive users, but doesn’t seem to provoke long-term or systemic injuries. Inhalation at moderate levels yields headaches or nausea before anything worse, but all guidelines echo the basics: keep exposure low, use proper gear, and clean up promptly. Environmental authorities track how it moves through wastewater and soil, checking for bioaccumulation or breakdown into more toxic pieces. So far, regulation accepts its uses in many consumer fields, provided that manufacturers and handlers keep exposure reasonable and waste pathways tight.
Future Prospects
Looking ahead, butyl benzoate’s future links tightly with the push toward greener manufacturing and safer chemicals. As regulatory frameworks evolve and more attention falls on worker health, researchers keep searching for tweaks—either in the molecule, the process, or end-use recipes—that can lift both efficiency and safety. Firms also dig into recycling options, seeking ways to recover spent solvent or redesign processes to leave less chemical residue behind. In my experience, the next breakthroughs will likely come from hybrid labs that merge green chemistry, industrial engineering, and digital controls for cleaner, tighter processes. As environmental pressures mount, every chemical—even the quiet performers like butyl benzoate—gets put under the magnifying glass, prompting smarter decisions and bolder innovation for decades to come.
What is Butyl Benzoate used for?
What’s in a Name?
Butyl benzoate sounds like something you’d find in an old chemistry set, but this chemical shows up in a lot more places than most folks expect. I ran across it years ago during a summer job at a factory, mixing fragrances for soaps. That was my first brush with it, but I learned quickly that it does a whole lot more than just add a dash of aroma.
Diving into Daily Use
Most people have no idea what goes into their colognes or lotions, let alone the stuff that keeps them running smoothly. Butyl benzoate acts as a solvent, so it helps dissolve substances that don’t blend well with water. Perfume makers rely on it because it works quietly in the background, softening intense scents and helping everything mix without turning your favorite scent into a clumpy mess.
Think about that next time a bottle of sunscreen slides over your skin without feeling greasy. There’s a decent chance butyl benzoate lent a hand there. It thins out sticky ingredients, making them easier to spread. I remember how my grandfather, who spent his afternoons fishing, would complain about sunscreen that felt thick and heavy. Lighter, smoother creams always won him over. The science behind that comfort often comes down to smart ingredient choices like butyl benzoate.
Not Just in Your Bathroom
Butyl benzoate pulls its weight outside of cosmetics, too. Tackling pests at home or on farms used to involve some rough chemicals, but butyl benzoate started popping up in pesticides and insecticides as well. It doesn’t score headlines, but this chemical helps dissolve the main actives, making sure the spray fits the job. That means stronger results with less need for raw power. Farmers and gardeners count on it, whether they realize it or not.
Industrial Backstage
Back in my college lab days, I’d catch the faint, sweet scent of benzoate during long days in the research wing. Textile manufacturing, paints, and even the polish at the shoemaker’s bench all give butyl benzoate a job. It smooths finishing touches, helps dyes penetrate fibers, and polishes surfaces so colors come out sharp instead of streaky.
Even in plastics and resins, butyl benzoate steps in to keep things flexible. Manufacturers blend it with PVC or nitrocellulose, balancing products for the real world—think flexible cables or shiny coatings on furniture. Engineering plastics can’t cut corners. If your phone case bends without snapping or wallpaper resists cracking, ingredients like butyl benzoate deliver the goods without much fanfare.
Safety Steps
Long-term trust in a chemical depends on safety. Butyl benzoate doesn’t stir up the same fears as some other old-school industrial additives, but smart companies monitor exposure and keep a close eye on research. Reports suggest low toxicity, especially at the tiny amounts found in finished goods. Still, allergic reactions can turn up, just like they do with almost any synthetic compound. Learning from my early misadventures with skin rashes, I always recommend checking labels and paying attention to how your body reacts to anything new.
Looking Ahead with Open Eyes
There’s always room for smarter solutions and closer examination. Researchers keep tabs on how butyl benzoate interacts with the planet and with us. Sustainable sourcing and clean production should sit at the top of future priorities. If safer or greener alternatives step forward, the industry ought to keep an open mind. But for now, this behind-the-scenes helper supports everything from comfy lotions to critical crops, reminding us that tiny details often keep the world running smoothly.
Is Butyl Benzoate safe for skin contact?
What Butyl Benzoate Is, and Where You Find It
Butyl benzoate pops up in personal care lines and fragrance mixes. It works as a solvent, helping perfumes blend together smoothly, and turns up as an ingredient in some creams and ointments. Anyone reading labels in health and beauty aisles has probably seen it listed. I always look twice when a chemical pops up in something that goes on skin, since allergic reactions and irritation can sneak up on anyone.
How the Body Handles Butyl Benzoate
Dermatology studies show butyl benzoate doesn’t sink deep into the skin. Most of it stays near the surface. A 2017 review published in
Contact Dermatitisshowed that butyl benzoate, used in medication creams, generally stays put and gets broken down by the top layers, limiting buildup inside the body. For routine use in small amounts, it hasn’t been flagged as a risk by big review panels like the Cosmetic Ingredient Review (CIR). No government warning sticker shows up for it on shelves in the U.S. or through European consumer agencies.
Real-World Reactions
Most folks don’t notice any difference after putting a lotion with butyl benzoate on their skin. For perspective, people working in labs handling the pure compound every day have a much higher chance of any reaction compared to someone using a cologne or hand cream a few times a week. Medical records reviewed in occupational safety journals point out that rare skin reactions or mild discomfort mostly happen with spills of the raw liquid or extra-long exposure.
Skin patch tests checked on adults with sensitive skin show mild or no irritation in most cases. That fits what dermatologists see: for healthy skin, occasional contact in the tiny amounts found in finished products rarely sets off problems. Still, those with eczema or a known fragrance allergy should be watchful. From my own experience living with sensitive skin, patch testing new products helps catch any personal triggers before they become a larger problem.
Allergies and Sensitivities
Anyone can react to an ingredient, no matter how “safe” it looks in broad studies. There are people who react to butyl benzoate, especially if they react to similar esters or fragrance blends. As with most cosmetic ingredients, irritation risks rise if the skin’s already broken or inflamed. Manufacturers often run “repeat insult patch tests,” exposing volunteers over time to check for delayed reactions. The published data suggest issues from butyl benzoate stay pretty rare, especially when compared to far more aggressive solvents or harsh preservatives.
Ways to Reduce Any Risk
It helps to stick with products from trusted brands that list every ingredient. If anyone has ever had a reaction to benzoates or strong perfumes before, looking for “fragrance-free” or “for sensitive skin” claims can save hassle. I recommend testing a new cream on a small part of your forearm for a day or two before slathering it everywhere. Be aware of any new redness, itching, or bumps.
For workplaces where the chemical gets handled in large quantities, gloves and good ventilation keep risks way lower. Public health officials agree that careful handling and labeling go a long way toward keeping most users safe.
Why It Matters
Staying aware of ingredients helps maintain healthy skin in the long run. Advocacy groups and regulatory agencies still keep an eye on studies around long-term use and vulnerable groups, like babies or pregnant women. Anyone with a chronic skin condition, or new to a product, should check with a dermatologist if something doesn’t feel right. Ingredient safety isn’t a one-size-fits-all answer — but knowing where butyl benzoate stands in current research gives people more control over what they put on their skin.
What are the storage and handling requirements for Butyl Benzoate?
Why Proper Conditions Matter
Anyone who spends time in a lab knows certain chemicals stick in your memory, not just for their use, but for how much trouble they can cause if handled poorly. Butyl benzoate stands out as one of those substances. It shows up in industrial products, cosmetics, and even acts as a solvent in pesticide formulations, so it moves through plenty of hands and facilities. Safe and smart stewardship goes far beyond ticking the regulatory boxes – it boils down to keeping people, property, and the environment out of harm’s way.
Main Storage Factors for Butyl Benzoate
Butyl benzoate carries flammable properties and can irritate skin and eyes. Anyone working with it should store it in a cool place. Heat or direct sunlight prompts decomposition, which does more than just waste the contents; it can send vapors into the air, and those bring fire hazards or even health problems by inhalation. I once visited a warehouse on a summer afternoon where sunlight streaming through the windows sent the thermometer soaring – the room smelled sharp and sweet, and workers mentioned headaches and eye irritation. A shaded spot with active ventilation would’ve avoided the risk and the complaints.
Closed containers make a world of difference. Butyl benzoate’s volatility means even small spills contribute to vapor buildup or sticky residue. Tight-sealing lids help more than most people realize – I watched a technician swap a cracked cap for a fresh one, and that single move put an end to the faint chemical whiff we’d noticed for days.
Keep water far from the storage area. Moisture can break down the chemical. Mix it in with the wrong compounds, and you can trigger byproducts that cause even more problems. Dry conditions take out a lot of guesswork about product purity or industrial reliability.
Handling: Paying Attention to Everyday Risks
Moving or measuring butyl benzoate means gloves, goggles, and lab coats are non-negotiable. The substance can irritate skin and cause real discomfort. I once slipped off my gloves for just a second to answer a phone call and noticed irritation where the liquid splashed. Proper protection turns what could be a medical report into just another routine task.
Spill control is not just about mop-up. Even small leaks require materials like absorbent pads or sand, followed by a thorough cleaning. Leaving chemical traces on a bench or into a floor drain risks long-term exposure downstream. Workers are safer when spill response materials stay close by, and training kicks in for even the smallest cleanups.
Labeling and Communication
Every drum, carboy, or dropper bottle holding butyl benzoate needs clear, durable labels. Faded pen scribbles on tape set up confusion, especially during shift changes or emergency checks. Accurate chemical identifiers and hazard warnings cut down on human error. In my experience, sharp labeling makes the difference between a near-miss and a well-handled job.
Safe Storage Means Responsible Use
Storage and handling routines keep chemicals where they belong and protect the people who use them. Ventilated spaces reduce invisible risks, and clear hazard communication nips confusion in the bud. With these habits in place, butyl benzoate supports manufacturing and research, not just today, but for the long haul.
Is Butyl Benzoate soluble in water?
Why Solubility Matters
Working in a chemistry lab, I’ve handled all sorts of liquids and powders. One thing that always stands out: the way a chemical mixes with water can change everything about how you use it. Some substances blend right in, while others just float around or sink, splitting away like oil on a rainy sidewalk. Solubility doesn’t just matter for the sake of curiosity. It shapes how industries clean spills, how perfumes are made, and how certain drugs reach their targets.
What’s the Deal With Butyl Benzoate?
Butyl benzoate pops up in a few different worlds. Some folks know it as a solvent for dyes, others from its role in flavorings or fragrances. It even lands in treatments against lice and scabies. Each of these uses means someone somewhere needs to know: Will it dissolve in water, or separate out?
Rolling Up the Sleeves: Mixing Butyl Benzoate and Water
During my time prepping solutions, I poured butyl benzoate into water on several occasions. The result—never a complete mix. Instead, a layer forms, showing butyl benzoate doesn’t just blend with water. Science backs this up: with a solubility of under 0.05 grams per 100 milliliters at room temperature, it barely budges. Compare that to table salt, which vanishes by the tablespoon in a glass of water.
There’s a simple reason behind this behavior. Butyl benzoate is a larger, oil-like molecule, made mostly of carbon and hydrogen chains. Water, with its strict “like dissolves like” principle, frowns at large non-polar molecules. So, butyl benzoate stays mostly to itself, refusing to break up and mingle.
Why This Turns Into a Real-life Hurdle
Mixing butyl benzoate in water doesn’t just fall short in the lab. The challenge ripples into real-world uses. Consider pharmaceutical compounding. If a tiny dose of medicine must dissolve evenly, water-insoluble substances like butyl benzoate make the job tricky. You end up shaking bottles, adding surfactants, or searching for new ways to mix. In pest control, low water solubility shapes how the compound spreads and how long it lingers.
Environmental health raises questions here too. Water-soluble chemicals break down faster in rivers and lakes, giving nature a better shot at scrubbing them out. Butyl benzoate’s resistance means it tends to stick around in soils or fat stores, leading to different kinds of risks if dumped or spilled in the wrong place.
Possible Solutions for Poor Solubility
Folks in research and industry often try creative fixes. They’ll use cosolvents, stirring in alcohol, acetone, or similar agents to coax butyl benzoate into mixing. Some chemists build emulsions—tiny droplets carried in water with help from surfactants, much like mayonnaise. Encapsulation, another trick, packs tiny bits of the chemical into water-friendly shells. These approaches keep production lines moving, but none work perfectly in every situation.
Keeping safety in mind, people storing or transporting butyl benzoate should guard against leaks, knowing water won’t wash it away so quickly. Handling needs smart containment, good ventilation, and the right training to prevent any accidental exposure.
Final Thoughts
After seeing both spills and successful product batches in person, one thing holds true: no one should underestimate how a simple feature like water solubility changes the story of a chemical. Understanding butyl benzoate's limits clears a path for better decisions, safer workplaces, and cleaner environments.
What is the chemical formula of Butyl Benzoate?
Understanding Butyl Benzoate
Butyl benzoate comes with a simple formula: C12H16O2. Sometimes, a list of letters and numbers on a label gets ignored, but in the case of butyl benzoate, that formula carries a lot of meaning. This molecule forms when butanol, a basic type of alcohol, hooks up with benzoic acid, a preservative you might find in things like pickles or certain sodas. By combining the two, you get an ester—specifically, butyl benzoate—which feels slick on the skin and doesn’t evaporate as quickly as straight alcohol.
A Closer Look at Its Structure
Let’s break the formula down. C12H16O2 means there are 12 carbons, 16 hydrogens, and a couple of oxygens connected just so. The carbon "skeleton" includes a piece called a benzene ring, which creates stability and adds to its pleasant smell. The butyl part, coming from the alcohol, stretches the molecule out and brings some extra flexibility. This layout lets the compound move easily between oil and water phases, which is handy in making creams, lotions, and even some food additives.
Why the Formula Matters in Real Life
From my time in the chemical industry, knowing a formula like C12H16O2 gives an edge. It helps you predict how a material behaves in a formula, whether you’re trying to make a product feel silky or work as a mild solvent. Butyl benzoate dissolves a wide range of fragrances and may soften plastics, giving manufacturers a route to tweak products without hardcore industrial-grade chemicals.
The FDA already lists benzoic acid and its esters as safe at controlled levels. In this case, not much butyl benzoate is needed to make a big difference when used in topical products and flavors. It rarely causes an allergy, and its mild scent fits in all sorts of perfumes. That safe track record comes from a structure that the body can break down without much fuss. C12H16O2 looks complex to the average reader, but to a chemist, it’s a straightforward molecule with reliable behavior profile.
Environmental and Safety Angles
Butyl benzoate’s chemical bones mean it breaks down in the environment better than some synthetic materials. That keeps streams and soil from piling up with non-biodegradable nasties. Companies watching their sustainability scorecards appreciate ingredients that don’t linger where they shouldn’t. In factories, it turns out butyl benzoate doesn’t ignite easily and has a low vapor pressure, which means fewer headaches about air quality or accidental flame.
Better Ingredient Choices
In my experience, choosing ingredients like butyl benzoate comes from weighing performance, safety, and environmental effort. Producers use this ester in personal care, not just for label-filling, but because its chemical stability means a product keeps working long after it leaves the plant. Using a molecule like C12H16O2 doesn’t fix every product challenge, but it’s a leap ahead of many heavier, more persistent chemicals.
Better transparency about what’s inside skin care, flavorings, and specialty applications builds trust. For folks working with these products or using them at home, knowing a compound’s formula isn’t about memorizing numbers; it’s about understanding what’s going on in the bottle and why manufacturers lean on certain choices.
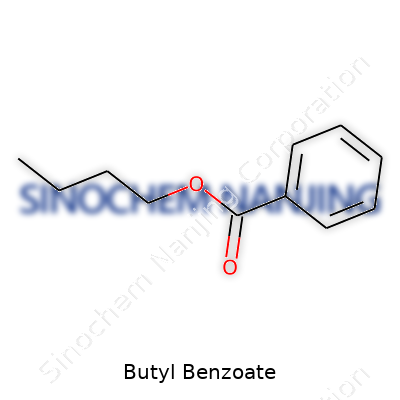
| Names | |
| Preferred IUPAC name | Butyl benzenecarboxylate |
| Other names |
Benzoic acid butyl ester Butyl benzenecarboxylate Butyl ester of benzoic acid n-Butyl benzoate Butyl benzoic acid ester |
| Pronunciation | /ˈbjuːtɪl ˈbɛn.zoʊ.eɪt/ |
| Identifiers | |
| CAS Number | 136-60-7 |
| 3D model (JSmol) | `/graphics/jsmol/jmol.php?model=C1CCC(CC1)COC(=O)C2=CC=CC=C2` |
| Beilstein Reference | 1908717 |
| ChEBI | CHEBI:3178 |
| ChEMBL | CHEMBL140211 |
| ChemSpider | 11087 |
| DrugBank | DB06710 |
| ECHA InfoCard | 100.007.363 |
| EC Number | 203-542-8 |
| Gmelin Reference | Gmelin Reference: **104594** |
| KEGG | C10465 |
| MeSH | D001432 |
| PubChem CID | 7772 |
| RTECS number | CD1925000 |
| UNII | 08A4M39HJJ |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C12H16O2 |
| Molar mass | 194.24 g/mol |
| Appearance | Colorless liquid |
| Odor | Faint, balsamic |
| Density | 1.01 g/cm³ |
| Solubility in water | insoluble |
| log P | 3.9 |
| Vapor pressure | 0.01 mmHg (25°C) |
| Acidity (pKa) | 13.3 |
| Basicity (pKb) | Product Butyl Benzoate has a pKb of approximately 15.0 |
| Magnetic susceptibility (χ) | χ = -7.02×10⁻⁶ |
| Refractive index (nD) | 1.499-1.503 |
| Viscosity | 15 mPa·s (20 °C) |
| Dipole moment | 3.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 355.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -332.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5886.6 kJ/mol |
| Pharmacology | |
| ATC code | D04AA16 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-2-NFPA704 |
| Flash point | 140°C |
| Autoignition temperature | 610°C |
| Lethal dose or concentration | LD50 (oral, rat): 6,600 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Butyl Benzoate: "6.6 g/kg (oral, rat) |
| NIOSH | BQ9625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Butyl Benzoate: Not established |
| REL (Recommended) | 25 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Benzoic acid Benzyl benzoate Propyl benzoate Methyl benzoate Ethyl benzoate |

