Bromomethane: Chemical Legacy, Challenges, and the Road Ahead
A Glance Back: The Journey of Bromomethane
Bromomethane’s story goes back deeper into chemical lore than many realize. A simple molecule, known for much of the last century as methyl bromide, it carved a unique space across agriculture, industry, and science. Early on, folks saw its knack for wiping out pests in stored grain and soil. Farmers leaned on it hard, finding no match for its power against nematodes, fungi, and insects. It looked simple—just a carbon, three hydrogens, and a bromine. But that simplicity masked a slippery set of consequences, both beneficial and troubling. Older agricultural records show how wide its reach once ran, turning up in warehouses, silos, even quarantine stations at busy ports. And with time those early wins started to turn bittersweet, as evidence built up about hidden dangers.
Recognizing the Product: What Sets Bromomethane Apart
Anyone who’s handled or read up on bromomethane knows its hallmarks: a clear, almost invisible liquid at room temperature, with a faintly sweet scent that doesn’t always tip you off to its power. It packs a punch as a fumigant, thanks to how quickly it vaporizes and spreads. That volatile nature made it both handy and tricky to control. Its manufacturers, recognizing both the promise and the risks, usually ship it in pressurized containers, with warnings splashed across the labels. Its trade names span from “Methyl Bromide” to less familiar codes known mostly to commercial users, but the power it wields speaks for itself: few compounds clear a warehouse of bugs and mold so efficiently.
Physical & Chemical Properties: A Balancing Act
This isn’t a heavy, sluggish fluid. Bromomethane boils at a chilly 3.6°C, turning to gas almost as soon as it sees an open space. That’s a boon in fumigation jobs where thorough penetration counts. Density checks in at about 1.73 g/cm³. It doesn’t play well with water, dissolving just a bit—enough to make clean-up a headache if spills happen. Chemically, bromomethane acts as a methylating agent. In lab work, I’ve watched it snap into reactions with nucleophiles, putting down methyl groups on everything from simple amines to complex pharmaceutical intermediates. Dangerous, yes, but precise in the right hands. Its chemical structure (CH3Br) and reactivity mean it keeps professionals on their toes. Cold storage limits its vapor, but you’ve got to respect how quickly it jumps phases at the warmth of daylight.
Preparation, Synthesis, and Reactions: Inside the Lab
Manufacturing bromomethane usually revolves around the reaction of methanol with hydrobromic acid or with bromine and phosphorus compounds. Sometimes in college labs, we’d talk through old-school syntheses with bromide salts, but modern production favors efficiency and scale. The chemical’s reactivity shows its teeth in methylation reactions—an asset in organic synthesis, pharmaceuticals, and agrochemical development. In those contexts, it shifts methyl groups like a pro, altering the chemical skeleton of bioactive molecules in a single step. But the same readiness to react makes it hazardous. Anyone dealing with it needs airtight controls: fume hoods hum, gloves thicken, and extra layers of chemical-resistant clothing come out. Waste disposal can’t be cavalier. The halogen power at its core pulls other molecules together—or tears them apart—meaning spill management and air filtration never take a back seat.
Names and Language Matter: Synonyms Across Industries
Bromomethane answers to more than one name. Old-timers and regulatory folks call it methyl bromide, a tag born of clarity but sometimes glossing over its legacy. Chemists stick with the International Union of Pure and Applied Chemistry—bromomethane, plain and precise. In trade circles, odd codes and shorthand nicknames float around, handed down in anecdote and scribble, but labels now demand clear, unambiguous identification. This shift in naming tracks a growing awareness—calling a chemical by its most precise name keeps everyone swimming in the same stream, especially as regulatory scrutiny tightens.
Keeping Workplaces Safer: Standards and Best Practices
Meeting safety standards with bromomethane ranks near the top of industrial priorities. Anyone working with this gas gets indoctrinated with stories about old mishaps and hard-learned lessons. In industry, you guard against leaks with regular checks, sealed systems, and active air monitoring. Good labeling means unmistakable warnings, hazard pictograms, and color codes set by national and international standards. Emergency training isn’t optional. Regulations like those from OSHA or the EPA aren’t just checkboxes—they shape day-to-day routines. I remember one facility drill where the alarms cut through our daily rhythm, reminding everyone just how little room for error exists. Respirators, gloves, eye protection—these aren’t formalities. Teams constantly re-check, document, and audit. The best plants go further, layering redundancy into every system, and refusing shortcuts even under time pressure.
Fields of Use: Value and Risk in Every Acre
For decades, agriculture relied on bromomethane to keep pests out of high-value crops. It didn’t take long for growers to see yield improvements, fewer post-harvest losses, and cleaner export shipments. As a pre-plant fumigant, it carved channels for roots by clearing out biological threats. Shipping companies used it to sterilize containers, quarantine inspectors depended on it, and even some medical manufacturers tapped into its disinfecting strength. Yet, the rise of resistant pests, and worries about environmental fallout, began to stir. Organic farmers and environmental advocates sounded alarms about residues, leaching, and bioaccumulation. Certification bodies, looking over every step, pushed for alternatives, pressuring industry to innovate. Despite restrictions, it still lingers in some specialty applications, mostly where no viable replacement provides the same level of risk management.
Research Momentum: Pushing the Boundaries
Modern research focuses both on reducing dependency and understanding long-term impacts. Universities dig into soil microbiome shifts, tracking how repeated bromomethane use thins populations of beneficial organisms. Atmospheric scientists measure its ability to break down ozone, linking agricultural practices to broader planetary health. Newer technologies aim for “green chemistry” approaches. I’ve seen projects in conference posters that harness targeted delivery—microencapsulation, precision soil injection, or combinations with biological pest controls—hoping to use less chemical and do less damage. Academics also plot the fate of breakdown products, hunting for ways to trap or neutralize the more stubborn residues. Funding bulges around projects that promise new answers, and policy groups often sit in on research meetings, demanding updates and real-world timelines.
Toxicity and Environmental Toll: A Growing Reckoning
Stories of acute exposure—nausea, respiratory pain, neurological symptoms—fill old toxicology reports and more recent case studies. Bromomethane crosses workplace boundaries, sometimes hitting adjacent communities if leaks run unchecked. Long-term health concerns still stir debate among experts. The molecule’s not just dangerous to people; it hits non-target species as well, rippling through soil life, water, and even the upper atmosphere. Decades of use left a measurable shadow, particularly in vulnerable habitats where fumigant drift poisoned beyond the field boundary. Environment-focused organizations keep pushing for wider bans. Some countries phased it out almost entirely, except for “critical use exemptions,” sparking heated arguments about food security versus environmental risk.
Looking Forward: Tighter Rules, Better Solutions, and Unfinished Work
No one denies bromomethane changed agriculture and storage practices for the better in its early days. But the present moment demands a reckoning: the risks became too glaring to ignore, and the alternatives—physical, biological, synthetic—won’t all fill the same shoes, yet. Regulatory bodies now take a closer look at every step: where and how the chemical is applied, what personal protections shield workers, and how residues are managed from field to dinner table. Some labs chase engineered enzymes that might break bromomethane into harmless fragments. Others test rotations of cover crops to build up biological resistance in soil, cutting chemical reliance at the root. Future prospects will depend on bridging hard science with on-the-ground work. Growers, regulators, scientists, and community groups each have a seat at this table, searching for answers that deliver both safe food and a safer planet. Progress—uneven, sometimes slow—flows from the ongoing tug-of-war between technological possibility and the hard limits set by health and nature. My hope, rooted in long hours spent discussing policy and new tools in drafty meeting halls, is that the next generation manages both the legacy and promise of chemicals like bromomethane with more wisdom than we did in the past.
What is Bromomethane used for?
The Many Faces of Bromomethane
Bromomethane, better known as methyl bromide, draws headlines every few years. Farmers know it as a strong soil fumigant, a gas released to kill off a list of stubborn pests, nematodes, weeds, and fungi. Crop growers across the world—from strawberry patches in California to tomato fields in Spain—have counted on its ability to knock out invisible threats before planting. Its speed and thoroughness let growers guarantee harvests that feed millions.
The chemical doesn’t stay in the fields. Exporters often use it to treat products headed overseas. Think of wood packaging, fresh produce, or even historic artifacts traveling by sea. Methyl bromide keeps bugs from hitching along for the ride and spreading across new regions.
Dangers Lurking Behind Convenience
Methyl bromide’s track record comes with some hidden costs. Breathing the gas even at low levels can irritate the lungs, eyes, and skin. Larger doses bring on tremors, nausea, and, in serious cases, neurological damage. Some communities close to fumigated fields have reported headaches and breathing trouble during fumigation season.
The biggest alarm, though, isn’t visible on a CT scan. Methyl bromide attacks the ozone layer—the invisible shield stopping dangerous ultraviolet rays from reaching the earth. In 1992, global leaders included it in the Montreal Protocol, aiming to protect the air above everyone’s heads. Countries agreed to phase out most uses, but a few critical spots, like quarantine treatments and some research, still get exemptions.
Looking for Better Answers
Breakthroughs don’t appear out of thin air. I’ve watched heated meetings between scientists, farmers, and regulators as everyone stakes a claim in the debate. Nobody wants another hole in the ozone, but walking away from reliable chemicals like methyl bromide never feels simple. In California, for instance, growers tried switching to alternatives like 1,3-dichloropropene or chloropicrin, only to face new health and environmental worries.
Some farmers shift their focus entirely—rotating crops more often, planting pest-resistant varieties, or adding organic compost to restore balance underground. These approaches often require patience and extra labor. Small family farms struggle to afford these changes; large operations need a push to break habits built over decades.
Calling for Smarter Regulations
Policymakers walk a tough path. Farmers pressure them to keep waivers going, but the health and environmental hazards loom over any decision. I’ve seen some regions help with funding for non-chemical solutions or support the move to organic methods as a carrot—offering up tax credits, grants, or technical help. As research churns along, the best shot comes from sharing knowledge as fast as possible. What works for a melon grower in Spain might help a Californian strawberry farmer save a season too.
The methyl bromide story leaves us with a lesson: balancing immediate needs with long-term safety isn’t easy, and rarely fits into neat boxes. Staying honest about risks, listening to all sides, and keeping the door open for new science moves things forward. That’s the only way to keep both food on the table and the sky above safe.
Is Bromomethane hazardous to health?
What Is Bromomethane and Where Does It Show Up?
Bromomethane shows up in places most people never think about—like farms, food storage, and fumigation sheds. Workers in agriculture use it as a fumigant to keep bugs out of soil before planting certain crops. In the past, people used bromomethane a lot more often, but its reputation has taken a serious hit over the years. Today, seeing the warning labels and protective gear at commercial farms makes you realize that this chemical isn’t just another tool in the shed.
Bromomethane’s Impact on Human Health
Coming into contact with bromomethane isn’t like touching water or dirt. The vapors from bromomethane enter the body fast, either through the air or by touching skin. Shortness of breath and coughing usually show up right away. As the dose climbs, headaches, dizziness, and nausea quickly follow. Spend more time with bromomethane, and the symptoms turn serious: tremors, convulsions, and trouble with your lungs, kidneys, or nervous system. Inhaling large amounts sometimes even turns deadly.
Back in the 1980s and 1990s, workplace safety wasn’t always strict, and a lot of folks didn’t think twice about new chemicals. Some workers never wore masks or gloves and learned the hard way that chemicals like bromomethane bring invisible dangers. The U.S. National Institute for Occupational Safety and Health now considers bromomethane a hazardous substance because it attacks the body silently and swiftly.
How Bromomethane Travels From Fields to Food
The Environmental Protection Agency tracks bromomethane because even small leftovers on food can cause concern. In the past, residues made their way onto strawberries, nuts, and grains. Even people not working on farms found traces in their kitchens. Many countries now limit bromomethane’s use, but old habits—or loopholes—sometimes let it leak into the food chain. Over time, low-level exposure builds up and risks stack higher for kids, pregnant women, and anyone with breathing problems.
Why the Ban Didn’t Erase the Problem
Bromomethane’s damaging side isn’t news to scientists or public health groups. The Montreal Protocol, signed in 1987, called for most uses of bromomethane to end because of its effect on the Earth’s ozone layer. Some exceptions stick around since replacing bromomethane isn’t always easy. Crops like strawberries and sweet potatoes fight tough pests, and growers push for exemptions. Many farmers keep using it under “critical use” waivers, so the threat hasn’t fully disappeared.
Where Do We Go From Here?
Awareness brings change. More farmers have switched to safer alternatives, like steam or pest-resistant crops. Still, switching isn’t cheap, and some say the alternatives don’t always deliver the same results. More research has pointed the way to integrated pest management—growing more diverse crops and rotating fields to reduce the need for chemicals. People in cities can help by paying attention to labels and supporting organic and low-residue produce, though not everyone can afford to buy this way.
With health and science on our side, keeping bromomethane out of homes and diets isn’t impossible. Wearing protective gear, following safety guidelines, and pushing for better alternatives can make a difference. Everyone who handles or eats food deserves clean air, safe water, and protection from hidden hazards like bromomethane.
How should Bromomethane be stored?
Understanding the Risks
Bromomethane doesn’t get much daily attention outside specialist circles, but for anyone handling chemicals in agriculture, fumigation, or industrial labs, it holds a notorious reputation for good reason. It brings both danger and usefulness in equal measure. Plenty of accidents have traced their roots to mishandling or bad storage, so this isn’t just about rules—it’s about protecting your health, your co-workers, and your neighborhood.
The Trouble with Bromomethane
Exposure to bromomethane causes trouble fast. It’s colorless and gives off vapors you can’t smell until concentrations start hurting your nose. Breathing it in for a little while can lead to headaches, dizziness, and in worse cases, lung and nerve damage. Long exposure edges toward chronic health trouble and even cancer. The chemical doesn’t break down quickly when it escapes storage, so the environment also pays the price. Years ago, regulators started clamping down on its widespread use thanks to the way it eats away at the ozone layer. Even though its usage has dropped, improper handling still lingers as a threat.
Choosing the Right Storage Setup
Proper storage always starts with temperature and moisture control. Storing bromomethane means using a cool, dry, and well-ventilated spot. Where I’ve seen things go wrong, heat is often the culprit. Leave a metal drum in the sun, and pressure inside climbs. You risk breaches, leaks, or a burst container. I’ve watched colleagues move drums out from poorly designed chemical rooms after someone left a thermostat unchecked. So, keeping things below 25°C or 77°F makes sense, and it’s worth putting alarms on thermostats or using digital monitoring, especially in big facilities.
Humidity causes another headache. Moisture creeps in around worn seals or a cracked container, setting off chemical breakdown and the release of toxic gases. A dry chemical room with dehumidifiers offers a layer of insurance. Simple steps like checking for standing water, fixing ceiling leaks fast, or keeping containers off the floor (using pallets or metal grates) go a long way.
Container Material and Leak Prevention
Containers demand extra scrutiny. Bromomethane eats through certain plastics over time, so always lean on heavy-duty steel or cylinders built for pressurized gases. I’ve seen new hires tempted to transfer leftovers into “temporary” plastic jugs—a recipe for leaks, even explosions. Use original containers with tight-sealing, corrosion-resistant valves and clearly printed hazard labels. Double-check for any corrosion, warping, or degraded gaskets each time you move or open a drum. A damaged seal spells trouble in days, not weeks.
Ventilation and Monitoring
Airflow matters almost as much as locking the door. You don’t want a build-up of invisible, toxic vapor. Vent hoods, extraction fans, and gas detectors should all run in areas used for storage. Install meters that trigger alarms if concentrations spike. It’s not just a box-ticking exercise—I saw an old storeroom fine before a fan died. A few days later, levels had crept up, putting everyone in the building at risk. Simple spot checks with hand-held sensors during weekly walk-throughs can save a lot of pain later.
Training and Emergency Planning
Proper chemical handling never works without solid training. Everyone on site needs to know the evacuation route, how to grab a respirator, and what to watch for during routine inspections. Spill kits and fire extinguishers should stay close, not buried three rooms away behind a locked cabinet. Regular drills, hazard refreshers, and easy-to-read signage keep awareness high. In places I’ve worked, asking someone to point out the nearest gas shutoff or eyewash station works better than any poster on the wall.
Making Safety a Daily Habit
Safe storage boils down to discipline and clear routines. Don’t just file away the safety data sheet—read it before the next shipment comes in. Double-check expiry dates, repair leaks right away, and write down every incident, no matter how small. Only certified professionals should move or dispose of leftovers. In the end, small oversights add up, but so do daily habits. Taking storage seriously keeps people safe, property intact, and the environment one step farther from the edge.
What are the safety precautions when handling Bromomethane?
Why Bromomethane Needs Respect
Bromomethane brings back memories of my short stint working with pest-control crews in citrus groves. Farmers use this stuff as a fumigant, but only after donning gear that looks more suited for a chemical spill than an orchard. The workers always double-checked their gloves and respirators before even opening a canister. One whiff of this chemical can put someone in the hospital with breathing trouble, dizziness, and much worse. The dangers are not hidden—OSHA and the CDC keep warnings right on the label for a reason. Long before regulations caught up, old-timers learned the hard way that carelessness left lasting damage.
Personal Protective Equipment: The Real Difference Maker
Rubber gloves, safety goggles, gloves lined with barrier cream, and a serious respirator—these are not optional if you ever find yourself around bromomethane. Regular latex or garden gloves won’t cut it; vapors seep right through. Splash-proof safety goggles protect against accidental sprays, and a lab coat, not just a regular shirt, shields sleeves from liquid exposure. When we worked near the tanks, the full-face mask made conversation tough, but everyone breathed easier knowing their lungs stayed clear of the gas. If skin contact happens, hand-washing with cold water—not warm, which opens up pores—goes a long way to limit absorption.
Safe Handling Starts Before the First Pour
A big lesson from my field days: never handle bromomethane in tight spaces. The gas collects near the floor, and a minor spill in a closed room quickly causes a lethal situation. Ventilation is critical. Big fans, fume hoods or all the doors and windows wide open—anything that keeps fresh air moving. We also used portable leak detectors, not just our noses. The vapor is colorless and sneaky, so relying on your senses is risky. Every drum and cylinder got tagged with clear hazard markings, and old containers got hauled away immediately to prevent confusion or cross-contamination.
Nutrition, Training, and Getting Out Fast
The best training covered warning signs of exposure: coughing, blurry vision, confusion, or a sudden headache. If anyone felt off, the supervisors insisted on a pause. No one stuck around to act tough. Medical kits sat close by, and clean water stations stood ready for fast rinsing. Eating or drinking anywhere near where bromomethane gets used is off-limits, since the vapors settle on food and water. Washing hands before breaks became second nature after just a few shifts.
Thinking About the Bigger Picture
Bromomethane is a tough chemical, but not everyone involved always receives top-notch education. Newer workers from small outfits sometimes hear about its risks only after their first headache or nagging cough. Frequent, blunt safety meetings need to be as normal as checking tire pressure before a long trip. Supervisors and managers who walk the talk—double-checking PPE, refusing shortcuts—show younger hands what matters. Pushing for alternatives might take longer, but less dangerous options are out there. Some cities and countries dropped bromomethane altogether after rising cancer and nerve damage cases. At the end of the day, health outlives any season’s harvest or fumigation deadline.
A Few Key Takeaways
- Wear gloves, goggles, and a tight-sealing respirator every time.
- Work only with proper ventilation.
- Keep exposure brief and wash up before eating or drinking.
- Label containers clearly and train all handlers, not just supervisors.
- Push for safer alternatives if possible, looking for both personal and environmental safety.
What is the chemical formula and molecular weight of Bromomethane?
Chemical Formula and Molecular Weight
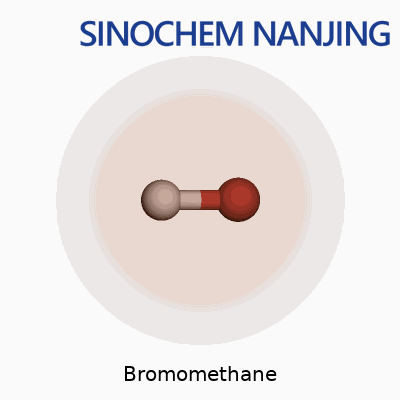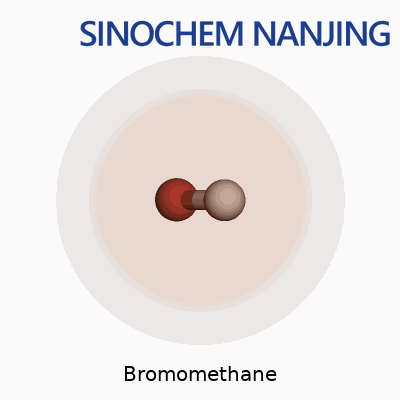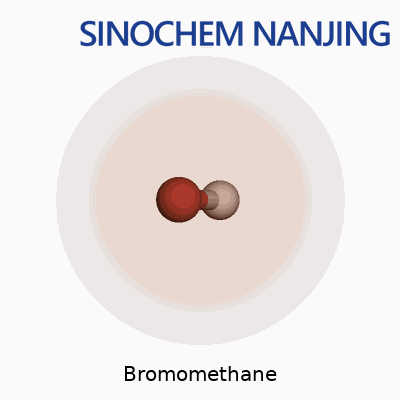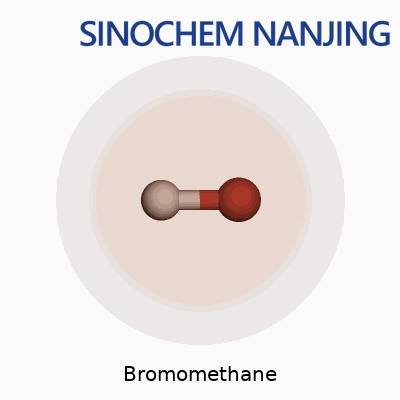
Bromomethane carries the formula CH3Br. Its molecular weight clocks in at 94.94 g/mol. It looks simple, just one carbon, three hydrogens, and one big heavy bromine atom latched on, but that combination gives it a set of properties and risks that need discussion.
Everyday Encounters With Bromomethane
This compound pops up more than most folks realize. For a long time, farmers counted on bromomethane (often called methyl bromide) to kill bugs and unwanted plants in the soil. It handled the job well, clearing fields for crops like strawberries, tomatoes, and peppers. It goes to work as a fumigant, turning from liquid to gas and working into the soil to attack pests where other solutions fail. The chemical’s volatility makes it potent, but also hard to control if not watched closely.
Human Health and Environmental Impact
Bromomethane might seem handy, but it packs some serious downsides. Breathing in too much can cause headaches, nausea, or even worse symptoms. It can damage lungs, kidneys, and the nervous system if exposure goes on for a while. Safety rules demand that people using it wear protective equipment and work in well-ventilated environments to keep the risks low. Beyond that, there’s another problem that stretches way past the field or workplace.
Methyl bromide rises into the atmosphere after use. This gas chews away at the ozone layer, making it harder for Earth to block out harmful ultraviolet rays. An increase in UV rays means extra cases of skin cancer, hurting people who may have never worked near this chemical themselves. Science journals and global studies confirm how noticeable the effect has proven over the decades. For this reason, global agreements like the Montreal Protocol took aim at methyl bromide, driving down its use and searching out safer options.
Alternatives and Paths Forward
Replacing bromomethane hasn’t been easy. Nothing quite matches its killing power on pests without bringing other problems to the table. Some growers switched to different fumigants, but these substitutes often carry their own risks. Others embraced crop rotations or solarization—covering the ground with plastic and letting sunlight do the work. These take more time and patience, but they chip away at the dangers tied to synthetic chemicals.
On the chemistry side, labs look for new solutions that split the difference. Biopesticides and smarter, localized treatments target pests more directly and let the rest of the ecosystem bounce back. Governments, university extensions, and farmers have to work together to make these transitions affordable. It’s tempting to keep running business as usual, but a growing body of evidence means more folks recognize the need for change. Life experience tells me that regulations only go so far; progress really takes off when new options line up with what folks need to feed their families and safeguard the health of their communities.
Personal Reflection
Knowing the chemical size of bromomethane helps, but it becomes more meaningful seeing how it shows up in daily life, both in the fields and in the sky above. Every element in its formula brings benefits and costs. Science hands us the facts, but society decides how much risk feels acceptable and where to draw the line. Folks grow up learning respect for chemicals after witnessing firsthand how a shortcut in one season can set the stage for bigger problems down the road. As new materials emerge, staying curious and adaptable gives everyone a better shot at farming well and living healthy, not just this season but for decades yet to come.

| Names | |
| Preferred IUPAC name | Bromomethane |
| Other names |
Methyl bromide Brommethan Monobromomethane Methly bromide Bromoform MeBr |
| Pronunciation | /ˌbrəʊ.moʊˈmiː.θeɪn/ |
| Identifiers | |
| CAS Number | 74-83-9 |
| Beilstein Reference | 3587152 |
| ChEBI | CHEBI:3136 |
| ChEMBL | CHEMBL17564 |
| ChemSpider | 5356 |
| DrugBank | DB06770 |
| ECHA InfoCard | DTXSID2020150 |
| EC Number | 200-813-2 |
| Gmelin Reference | Gmelin Reference: 1351 |
| KEGG | C01381 |
| MeSH | D001942 |
| PubChem CID | 6327 |
| RTECS number | PA4900000 |
| UNII | 39G5A2UM3M |
| UN number | 1062 |
| CompTox Dashboard (EPA) | DJ3UP0199Y |
| Properties | |
| Chemical formula | CH3Br |
| Molar mass | 94.94 g/mol |
| Appearance | Colorless gas or liquid with chloroform-like odor |
| Odor | Sweetish odor |
| Density | 1.77 g/cm³ |
| Solubility in water | 13.4 g/100 mL (20 °C) |
| log P | 1.24 |
| Vapor pressure | 1430 mmHg (at 20 °C) |
| Acidity (pKa) | 15.7 |
| Basicity (pKb) | Basicity (pKb) = -4 |
| Magnetic susceptibility (χ) | -62.3·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.422 |
| Viscosity | 0.326 cP (25 °C) |
| Dipole moment | 1.80 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 245.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -35.25 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -654.0 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | T60.2 |
| Hazards | |
| Main hazards | Toxic if inhaled, skin and eye irritant, may cause genetic defects, suspected of causing cancer, harmful to the ozone layer |
| GHS labelling | GHS02, GHS04, GHS06 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H331, H315, H319, H335, H340, H350, H373, H400 |
| Precautionary statements | P210, P260, P264, P271, P280, P284, P301+P310, P304+P340, P320, P308+P311, P370+P376, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-4-0◆ |
| Autoignition temperature | 537 °C |
| Explosive limits | 10-15% |
| Lethal dose or concentration | LDLo (oral, human): 14 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Bromomethane: Rat oral 214 mg/kg |
| NIOSH | NIOSH: PA4900000 |
| PEL (Permissible) | 5 ppm |
| REL (Recommended) | 0.05 ppm |
| IDLH (Immediate danger) | **1600 ppm** |
| Related compounds | |
| Related compounds |
Methanol Methanol-d4 Methyl iodide Methyl chloride Methylene bromide |

