Boric Acid: A Closer Look at an Old Workhorse with New Potential
Historic Routes and Today’s Relevance
Boric acid has stuck around for good reasons. Ancient folks in the Middle East dug out borate minerals for glass glazing, cleaning wounds, and keeping away creepy crawlies. Decades later, boric acid wound up in the hands of pharmacists and scientists. In labs and home cupboards, jars labeled “H₃BO₃” offer proof of its staying power. As industry changed gears after the industrial revolution, demand for boric compounds rose, especially in glassmaking, metallurgy, and agriculture. Even though it’s a simple white powder at first glance, boric acid keeps making itself useful. Over the years, governments began to demand tighter purity and labeling, and that led to today’s tightly regulated and clearly labeled containers.
Physical & Chemical Side of the Story
From a chemistry perspective, boric acid looks unassuming, but don’t let appearances fool you. Its crystals show transparency and softness, dissolving in warm water with ease but not exactly racing to break down in alcohol. Chemists can rattle off its melting point—about 170°C—and its mild acidity. It quietly buffers and stabilizes other compounds. It’s not very volatile and sticks around unless heated up. A lot of people miss that it doesn’t have the stinging burn of hydrochloric acid or sulfuric acid, but it can still shift the pH scale and slightly slow bacterial growth. Commercial products must hit tight purity marks for medical and technical applications, but in many industrial settings, lower grades with minor impurities get the job done.
Labeling and Specification Ground Rules
Walk into a modern chemical warehouse and snag a drum labeled “boracic acid”—just another of its many names—and you’ll see a fat stack of specifications. Labels cover chemical formula, weight, batch numbers, and hazard signs calling out skin irritation or environmental risk. Government rules push for full transparency, with warnings in bold print. Physical purity (at least 99.5 percent for high-grade stuff), moisture content, and traces of unwanted metals all end up in the documentation. Over the years, these efforts keep risks in check, ensuring everyone—from glassblowers to farmers—knows what they’re dealing with.
Making Boric Acid: No Mystery, But Plenty of Science
Making boric acid traces a pretty old path: run borate minerals like colemanite or borax through sulfuric or hydrochloric acid, stir, filter, and you end up with boric acid crystals. The process sounds easy, but there’s plenty of technical fine-tuning behind it. Temperature controls the size and purity of the crystals. Big companies keep everything in sealed systems, not just to avoid waste but to cut down dust that could cause breathing problems. There’s also a focus on recycling water and capturing any leftover acid to meet today’s pollution control standards. Small changes in the washing or drying step mean a lot to people who need either medical-grade purity or agricultural material by the ton.
The Chemistry: More Than Meets the Eye
Mix boric acid with sodium carbonate or sodium hydroxide and you create sodium tetraborate—better known as borax, a fixture in cleaning products and glassmaking. Heating alone pushes boric acid to lose water, creating metaboric or tetraboric acid, each with its own uses. In the world of organic chemistry, boric acid steps into more complex reactions, forming esters with alcohols or cross-linking agents for polymers. A story from my university lab: boric acid’s buffering helped pull off stubborn protein precipitations, saving hours of effort. It’s not the flashiest chemical, but its ability to nudge pH and form gentle bonds keeps it in the toolbox for a wide sweep of syntheses and product tweaks.
Names and Synonyms Across Different Fields
Plenty of terms appear on labels, depending on who’s buying—“boracic acid,” “orthoboric acid,” or just plain “H₃BO₃.” In agriculture, it may ride under a fertilizer code. In pharma circles, it pops up with purity stamps and medical warnings. People sometimes mix it up with borax or other borates, but the technical teams in industry know the score. Beyond that, trade names crop up regionally, some dating back to before chemical harmonization. For all the linguistic clutter, what’s inside the drum is the thing: a simple, reliable chemical with different names depending on who signs the purchase order.
Staying Safe: Operational Habits and Standards Matter
Too many people outside industry take chemical safety for granted. Boric acid can cause skin irritation or temporary eye issues, and it’s risky to inhale the powder over long periods. Employers hand out gloves, goggles, and much stronger masks than anyone would use for sweeping a dusty porch. Safety guides in chemical plants stick close to OSHA or EU standards, checking for spills and keeping the workspace clean. Medical and home uses rely on strict dosing, since too much boric acid causes poisoning—an actual risk rarely flagged in do-it-yourself blogs. In my own experience, one cause of concern came from seeing old pest control recipes floating online with no caution note. Safety boils down to education and following well-marked operational rules, plus clear disposal methods to avoid environmental damage.
Applications: Not Just Roach Traps and Eye Washes
Most people first meet boric acid as an ingredient in bug powders or old-fashioned eyewashes. That’s just the tip of the iceberg. Glassmakers use it to tweak the thermal and chemical durability of certain glasses, an unsung reason everyday glass cookware survives stove-to-oven treatment. Metallurgists rely on boric acid flux to keep metal surfaces free of oxidation. In agriculture, small but crucial doses supply essential boron for healthy plant growth, especially in fruit and root crops. Textile workers lean on its fire-retardant qualities, soaking fabric so it will smolder instead of flare up. Nuclear power plants draw a different benefit: boric acid helps slow neutrons, so it lands in reactor cooling circuits. In much smaller doses, pharmacies sell it to soothe skin irritations and treat fungal infections, though new restrictions have dialed back its over-the-counter use. Its power comes from not overpromising—steady performance in each role, not miracle claims.
Research, R&D, and the Edges of Modern Chemistry
Researchers keep finding ways to stretch boric acid’s abilities. In medicine, labs explore its antifungal and potentially antiviral effects, although regulatory barriers remain steep. Materials scientists have tinkered with boric acid to create boron-rich ceramics and flame-retardant plastics. Nanotechnologists are busy embedding it in new sensors, coatings, and treating boron nitride nanotubes with it to control growth. Some food packaging researchers look at it for antimicrobial barriers, but safety hurdles and tight oversight slow those projects. R&D isn’t always headline-grabbing, but steady work on improved synthesis and recycling aims to cut costs and environmental strain. From my side of industry, I see environmental teams pushing for better ways to recover boric acid from process waste so nothing goes down the drain unless rendered harmless.
Digging Into Toxicity and Safety Data
Boric acid wears a badge as “low-to-moderate” toxicity, which means caution and common sense. Acute effects crop up at high doses: nausea, skin and eye irritation, possible reproductive effects if someone ignores recommended exposure limits. Prefilled bottles and premixed products cut accidental risks, but bulk users must stick to strict handling rules. Long-term studies trace links to developmental toxicity, which led to recent European moves to restrict its use in personal care products and home remedies. Chemical manufacturers monitor atmospheric dust in plants and enforce exposure limits well below what triggers health concerns. Disposal stays tricky, since even low toxicity can harm aquatic organisms—so environmental guidelines now limit wastewater boron content. Relying on solid research, safety sheets evolve, and so should old habits.
Where’s It All Heading?
Looking forward, boric acid shows signs of renewed interest. The push for greener flame retardants spotlights boric acid’s gentle touch compared to older, nastier chemicals. Glass and ceramics industries expect demand to rise as new battery technologies require specialized borates. In agriculture, microdosing techniques balance the scales to avoid both deficiency and excess boron—a big deal for global food security. Medical researchers keep testing formulations for wound care in high-infection settings. Environmental hurdles won’t just go away: proper use, improved recovery, and strict disposal must become standard. As technology brings new challenges, boric acid stands like an example of how old substances, used carefully and thoughtfully, still carve out room in our future. Right now, its story hangs on whether industry and regulators work together—turning a simple mineral salt into a tool for tomorrow’s problems.
What is boric acid used for?
A Quiet Workhorse at Home
Boric acid turns up in the back of cabinets and under kitchen sinks. That white powder might look plain, but in the real world, it earns its keep. My grandmother relied on boric acid to deal with roaches. Sprinkling it behind appliances and in cabinet corners helped us stay ahead of bugs in a humid, Southern climate. It doesn’t poison pests outright; instead, it damages their exoskeletons and dehydrates them. There’s no slick advertising campaign, but people keep using it because it works and doesn’t smell nasty.
Caring For Feet and Eyes
Foot fungus creeps up in summer, and boric acid comes out again. A little in your socks or shoes or mixed with water for a soak can help fight off athlete’s foot. Doctors sometimes suggest boric acid suppositories for yeast infections, especially when more common medications fall short. The tablets are available in pharmacies, though you’ll want to follow dosing guidelines. Eyes drops with dilute boric acid also help clean out minor irritations and calm burning. Always check with a healthcare pro, but boric acid keeps showing up as a backup when store-bought stuff fails.
Tough Enough for Industrial Work
Manufacturers keep boric acid in steady rotation. Glass used in kitchen cookware and laboratory beakers wouldn’t stand up to high temperatures without boric acid in the recipe. It strengthens the material, keeping glassware from cracking just because it meets a hot flame. Ceramics and enamels—think brightly colored plates—call for it too. The boron in boric acid helps the finish stay smooth and glossy after firing. In welding, boric acid enters the picture as a flux, keeping oxidation at bay so joints come out clean. Fireproofing companies mix it into insulation because it slows down flames, giving families a chance to get out in an emergency.
Gardens and Crops
Boron makes a difference for plants. Boric acid acts as a micronutrient, necessary in tiny amounts for cell growth and seed development. Farmers and home gardeners sometimes turn to boric acid as an additive if soil tests show a shortage. But there’s a catch: the line between “needed” and “toxic” remains razor thin. I’ve seen fruit trees recover after a careful dose, and I’ve seen leaves burn from a little too much. Exact dosing matters, and over-application risks turning soil inhospitable.
Weighing Safety
People ask if boric acid is safe, and the answer is: respect it. Kids or pets who eat boric acid can get sick, and using too much in the home raises risks. The FDA doesn’t clear boric acid for food use, and some countries stopped letting manufacturers sell it as a household antiseptic. But handled with care, boric acid offers benefits for households and industries alike. It pays to read the label, keep it out of reach, and try old advice with new caution.
Thinking About the Alternatives
Some folks look for newer, “greener” products, but many modern bug sprays rely on boric acid’s basic chemistry. There’s always talk about phasing out any chemical with potential household hazards. At the same time, boric acid’s record shows low resistance, no aggressive fumes, and reliability across many jobs. In a world full of complicated chemicals, sometimes what works well gets quietly overlooked.
Is boric acid safe for humans and pets?
What Boric Acid Actually Is and Where It Shows Up
Boric acid pops up in a lot of everyday products. Ant baits, some laundry cleaners, roach powders, and certain eye washes use it. It comes from naturally occurring boron, often found in minerals, volcanic lakes, or even a bag from the hardware store. Over a hundred years ago, people counted on it for all kinds of home remedies and cleaning jobs. That doesn’t mean old-school equals safe for everyone, especially when pets and small kids live in the house.
Science on Human Safety: Facts and Limits
Touching boric acid doesn’t usually create trouble for healthy adults. The real trouble starts with eating it or breathing in its powder. Swallowing a significant amount—even as low as a teaspoon for kids—can lead to nausea, vomiting, diarrhea, headaches, and worse. Some reports show that just two to five grams can cause severe poisoning for toddlers. Medical journals tell of boric acid once being used for diaper rash until it caused fatal poisonings in babies. Skin contact in small amounts usually doesn’t trigger problems, but long-term exposure, or applying it to wounds, risks absorption into the body.
How Pets React: Risks for Cats and Dogs
Dogs and cats love to investigate anything new on the floor, especially powder or pellets. Pets who eat boric acid can have drooling, vomiting, tremors, and in severe cases, seizures. The American Society for the Prevention of Cruelty to Animals (ASPCA) lists boric acid as a toxin for animals. If pets like to chew on things or lick the floor, even a light dusting of boric acid around baseboards poses a danger.
Safer Alternatives Exist
Plenty of homes want to avoid strong chemicals, so boric acid might seem like a milder choice for bug problems. Still, there are options that lower the risk to children and pets. Gel baits kept inside tamper-proof stations cut down on loose powder. Diatomaceous earth—made from fossilized plankton—can kill insects without adding much danger to mammals, though it shouldn’t be inhaled. Sticky traps or simple housekeeping habits often go farther than powders.
Responsible Use and Storage
If you decide to keep boric acid, store it high up in a locked cabinet, just as you would prescription drugs or toxic cleaners. Always wash hands well after handling it. Read product labels for warnings and follow them to the letter. Never sprinkle boric acid where kids or pets might crawl, sleep, or play. The National Pesticide Information Center and poison control centers can help answer urgent safety questions.
What Doctors and Veterinary Experts Say
Both the Centers for Disease Control and Prevention (CDC) and the Environmental Protection Agency (EPA) classify boric acid as a low-risk pesticide if used properly, but both agree that real problems arise when doses are not carefully controlled. Veterinarians see poisonings most often in curious pets or those living in homes using boric acid carelessly. If a pet eats any, or a child does, call a poison center or doctor right away.
Take Practical Steps for Safety
Most families and pet owners want peace of mind at home. Double-check what you actually need boric acid for, and compare with safer methods. Never assume a “old remedy” can’t cause harm to kids or animals who experience the world much closer to the floor. Prevention beats any cure, especially where a few dollars and a call to the vet may save a lot of heartbreak.
How do you use boric acid for pest control?
No Tricks, Just Real Experience
Folks at home—me included—get pretty tired of fighting off roaches, ants, and other little squatters. Boric acid is one of those things you hear about from neighbors or the corner hardware shop. You probably know it as that white powder in a plastic bottle, usually tucked near the cleaning supplies. I remember watching an uncle sprinkle it behind his fridge and in the cracks beneath the sink. He swore it knocked out cockroach problems without fail.
Where Science Backs Up Folk Wisdom
Boric acid is a mineral derived from boron and water. Decades of research document its effectiveness against a bunch of crawling insects—roaches, ants, silverfish, termites. Here’s why: boric acid acts as both a stomach poison and a desiccant. Insects walk through the powder, it clings to their legs, and they digest it as they clean themselves. It also scrapes up their waxy exoskeletons, leading to dehydration. Researchers from Cornell and Penn State confirm that given a choice, roaches often bumble right through the stuff before you notice results.
Real-World Use: Not Complicated, But Requires Care
Anyone who wants results starts with a clean area. Sweep up crumbs, take out the garbage, and keep things dry. You want boric acid to be the main thing drawing pests' attention. I’ve used a little squeeze bottle to puff a light dusting under appliances, along the back edge of cabinets, even inside wall voids if I can get access. Too much powder and you end up with unsightly piles; bugs just walk around them.
For ants, mix a small pinch with sugar or honey on a bit of cardboard. They carry the bait back to the colony, which attacks the source, not just the stragglers. Experts at the National Pesticide Information Center highlight that boric acid works slow but sure. Patience brings results in a week or two. You won’t see an instant kill; you’ll see fewer bugs with each passing day.
Keeping People and Pets Safe
Just because the powder is common doesn’t mean you treat it like flour. The EPA lists boric acid as low-risk for humans and pets provided you handle it smart. Always keep it away from food, kids, and animal dishes. Wash your hands after using it. I store the bottle up high with my paint thinner and ant traps.
If I have pets or kids visiting, I opt for targeted placements in cracks, not open shelves or floors. Less is more. The idea isn’t to coat the whole house—focus on the dark, undisturbed corners where pests hang out. Vacuum the extra powder after a couple of weeks, and repeat if needed.
Using Old Tools, Thinking Fresh
Some people go for chemical sprays or special traps, and sometimes these work just fine. Boric acid appeals because it’s affordable, doesn’t stink, and doesn’t startle the immune system like many modern sprays. It’s a tool, not a miracle solution, and seasoned pest pros will tell you: combining common-sense hygiene, sealing up cracks, and strategic use of safe powders keeps home invasions down to a dull roar.
Where can I buy boric acid?
Boric Acid’s Everyday Value
Boric acid keeps popping up in more places than most people realize. I first got curious about it when my grandmother mentioned using it to keep ants away from her kitchen. That advice led me down a rabbit hole: not only does boric acid help with pests, it also turns up in laundry, homemade cleaning mixes, foot soaks, and even as an ingredient in some health products. Its many uses keep it in demand, but tracking it down can get tricky depending on where you live and what you plan to do with it.
Where It’s Sold
Most big-box hardware stores like Home Depot or Lowe’s carry small tubs or packets of boric acid, usually shelved near pest solutions. Grocery stores used to stock it with laundry boosters, and a few still do if you check in the cleaning aisle. Pharmacies have it behind the counter, especially if you ask the pharmacist directly. Online, boric acid sells through well-known retailers like Amazon and Walmart, along with specialty science shops. I’ve ordered it through Amazon to save time, but smaller hardware stores often keep a few boxes if you want to support local business.
Understanding What You’re Buying
Some folks chase boric acid without really checking the label. Not all boric acid products are the same—one box suggests cleaning tile grout, another prepares foot soak solutions, a third comes out in powder form in an ant trap kit. Quality varies, and not every use calls for the highest grade. Industrial-grade boric acid often costs less, but medical and food uses call for pharmaceutical-grade, which tends to cost more and requires more scrutiny.
Reviewing ingredient lists helps you avoid products containing unnecessary fillers or additives. I always check for a manufacturer or distributor with a clear track record—at least some basic information from the seller, and ideally some customer feedback. The FDA oversees medical boric acid, so pharmacy-sold products deliver accurate dosing through prefilled capsules. Bulk tubs from hardware retailers need more attention to instructions—improper use might mean health risks.
Health, Safety, and Honest Advice
Boric acid has a practical reputation, but it’s not a cure-all. It can irritate skin or eyes and quickly becomes dangerous if swallowed, especially for kids or pets. I keep mine on a high shelf, labeled clearly, and never mix it in the same area as food prep. Internet health trends sometimes promote homemade remedies, like suppositories. Science supports some uses against yeast infections, but dosage and purity matter here—medical advice beats TikTok tips.
Quality boric acid products have their place, but too many people take risks mixing their own capsules without talking to a doctor. The FDA has warned about improper or contaminated at-home treatments, which can lead to more harm than good. Pharmacies offering pre-measured, quality-controlled options make better sense for health uses than scooping powder from a hardware store box.
Getting It Right
Stores—online and brick-and-mortar—won’t always ask about your intended use, but that leaves responsibility with the buyer. Knowing where boric acid comes from, checking labels, and staying aware of risks turn a simple purchase into safe, smart household management. Keeping informed about changes in regulations helps too, since local policies about sales can shift.
If in doubt, ask your pharmacist or a store expert. Simple advice like this matters, whether you’re running pests off the property, cleaning tiles, or following a medical recommendation. Treat boric acid as a useful tool, not a magic fix, and you’ll get the benefits without the headaches.
What are the possible side effects or dangers of boric acid?
Boric Acid: Uses and Where You Find It
Boric acid lives in a lot of households, mostly under the kitchen sink or medicine cabinet. Some folks use it as a cleaning powder, an insect killer, or even as a home remedy for yeast infections. In the pharmacy, boric acid shows up in suppositories or eyewashes. Its many uses make it easy to forget that it’s a chemical with real risks attached.
Experiences With Boric Acid Exposure
I remember as a kid, seeing adults sprinkle white powder in the corners to stop cockroaches. Of course, no one explained that touching or breathing in that fine dust could cause problems. Years later, working in healthcare, I’ve seen patients suffer from accidental ingestion or improper use. The most common stories come from people who mixed up boric acid powder with baking powder in the kitchen or left it somewhere a curious child or pet might find it.
Health Risks to Watch For
Boric acid isn’t something to treat lightly. Swallowing even small amounts can cause nausea, vomiting, stomach pain, and diarrhea. People often ignore the risk because the symptoms start mild, like a stomach bug, but larger amounts can create bigger issues. More severe cases can lead to headaches, confusion, rash, or seizures. Death from ingestion isn’t common in adults, but it’s a real risk for children and pets.
The powder dries out the skin and can cause burns or rashes if left too long, especially in kids. Those with sensitive skin or eczema can notice irritation almost immediately after exposure. Breathing in boric acid dust in an unventilated space can irritate your nose, throat, and lungs. Eyes exposed to boric acid—directly or through residue left on your hands—feel a burning sting and can begin to water and redden fast.
Concerns With Medical Use
Many people swear by boric acid suppositories for yeast infections, especially when standard medicines fail. Doctors do prescribe it, but only in strict doses. Overdoing it isn’t just wasteful. It raises a real risk of vaginal burning or a worsened infection if used alongside broken skin or open sores. Pregnant women should avoid it entirely, since studies have linked boric acid to birth defects. The risk reaches beyond adults, too. Accidental ingestion by infants or young children can quickly spiral into a medical emergency.
Why Regulation and Education Matter
The Environmental Protection Agency and Food and Drug Administration both limit how boric acid gets labeled, sold, and used. Still, confusion crops up every year. Some people trust old advice or vague online sources that overlook the risks. One sore spot comes from lack of clear labeling. Products sometimes mention “borax” or “insecticide powder” without calling out boric acid as the danger inside. Pharmacies in some countries sell it freely, and not everyone understands the line between topical use and ingestion.
Safer Practices and Alternatives
Keeping boric acid out of reach makes a difference. Lock cabinets and store powders away from flours or other white powders in the kitchen. Gloves help avoid skin contact, and washing hands after use limits accidental transfer. For pest control, traps or baits offer lower risks to people and pets. When it comes to medical treatments, checking with a healthcare provider helps avoid accidental overdose or inappropriate use.
Boric acid solves real problems, but people deserve clear facts and smart guidance before using it at home. Wherever chemicals meet daily life, respect and awareness make all the difference.
| Names | |
| Preferred IUPAC name | trioxoborane |
| Other names |
Boracic Acid Orthoboric Acid Hydrogen Borate Acidum Boricum Boricum Acidum |
| Pronunciation | /ˈbɔːrɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 10043-35-3 |
| Beilstein Reference | Beilstein Reference: 3903787 |
| ChEBI | CHEBI:33118 |
| ChEMBL | CHEMBL1406 |
| ChemSpider | 12155 |
| DrugBank | DB00765 |
| ECHA InfoCard | 100.033.001 |
| EC Number | 233-139-2 |
| Gmelin Reference | Gmelin Reference: **1044** |
| KEGG | C01336 |
| MeSH | D001911 |
| PubChem CID | 6328 |
| RTECS number | ED4550000 |
| UNII | 91MBZ8H3QO |
| UN number | UN3077 |
| Properties | |
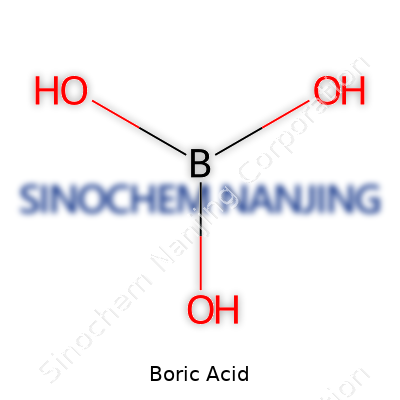
| Chemical formula | H3BO3 |
| Molar mass | 61.83 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.44 g/cm³ |
| Solubility in water | Soluble in water |
| log P | 0.17 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 9.24 |
| Basicity (pKb) | 9.27 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.340 |
| Viscosity | 1 cP |
| Dipole moment | 4.48 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 105.8 J·K⁻¹·mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1094.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1094 kJ/mol |
| Pharmacology | |
| ATC code | S02AA03 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may damage fertility or the unborn child |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H360FD: May damage fertility. May damage the unborn child. |
| Precautionary statements | P264, P270, P305+P351+P338, P301+P312, P337+P313 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Lethal dose or concentration | LD50 Oral Rat: 2,660 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2660 mg/kg (oral, rat) |
| NIOSH | PC1400000 |
| PEL (Permissible) | 10 mg/m³ |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | No IDLH established |
| Related compounds | |
| Related compounds |
Boric oxide Borax Sodium perborate Tetrafluoroboric acid |

