Bismuth Potassium Citrate: A Deep Dive into Its Journey, Qualities, and Future
Historical Development
Bismuth potassium citrate has an interesting backstory that stretches over several decades. It first popped up in laboratories that were searching for new compounds to treat stomach conditions. In the days before widespread antibiotics, bismuth salts took on the task of fighting gastrointestinal infections. Potassium citrate, known for its work in buffering acids, was paired with bismuth to make the compound easier on the digestive system and boost its performance. Several companies across Asia and Europe picked up the formula during the late 20th century, with a sharp increase in research output after the rise of Helicobacter pylori treatments. Generations of patients relied on bismuth-based therapies long before the term “triple therapy” entered medical textbooks, and that practical legacy continues to inform medical choices today.
Product Overview
This compound finds heavy use in the world of gastric health. It acts as both a shield and a weapon: shielding the stomach lining from damage while going after bacteria like H. pylori. Most forms on the market are fine, pale powders or white crystalline granules. Manufacturers offer it as bulk raw material for pharmaceutical formulations, or already included in tablets and suspensions. Instead of being toxic or causing high levels of irritation, bismuth potassium citrate provides a safe route to symptom relief for ulcers and chronic gastritis. In pharmacies across Asia and select regions in Europe, one can see its product names interspersed with other bismuth combinations, giving doctors and pharmacists choices tailored to the patient’s needs.
Physical & Chemical Properties
The crystal structure stands out under a microscope. Water solubility comes in low to moderate, so suspensions settle if not stirred. The color usually hovers near white or very pale yellow, which helps quality control spot impurities quickly. It doesn’t smell, nor does it easily absorb water from the air, so storage is pretty straightforward. Chemically, you’ve got three players: bismuth (a heavy, less-reactive metalloid), citrate from citric acid, and potassium. At common pH levels found in medications, the molecule stays stable. It holds up well under normal storage, resisting breakdown even after sitting months on a pharmacy shelf. Melting comes in at a high temperature, which is a plus for transport safety—very little risk of accidental melting in a non-lab setting.
Technical Specifications & Labeling
Pharmaceutical suppliers stick to a tight set of rules on this compound. Purity shoots for over 98% bismuth potassium citrate by dry weight, leaving little room for contamination by other metals or non-reactive salts. The label must spell things out: manufacturers list the active content, storage instructions (keep cool, avoid humidity), shelf life, and intended use. Pharmaceutical grades are checked for lead, arsenic, and other heavy metals that sometimes sneak into bismuth supplies from mining. Particle size matters, too—a coarse batch clumps differently, so suppliers standardize the grinding to keep mixing in the lab easy. Product codes, batch numbers, and exact suppliers show up on every drum or bottle, which helps trace any problems back to their source during a recall. Attention to labeling details lines up with the principle of keeping both healthcare providers and patients safe.
Preparation Method
Large-scale synthesis depends on the quality of bismuth nitrate and potassium citrate. Most factories run a wet-chemistry route, blending bismuth nitrate in solution with potassium citrate under controlled conditions. Workers add the reactants in specific ratios, then agitate the mixture at a set temperature for a few hours. The reaction produces insoluble bismuth potassium citrate, which is then filtered out, washed several times to clear out any leftover acid or nitrate, and dried in low-humidity ovens. Scale-up from lab to factory often reveals issues with clumping, so modern plants operate with continuous stirring equipment and closed systems to protect both the workers and the product. Quality checks tops off each batch—company chemists check for unreacted nitrate and test how well the final product resists decomposition under high humidity.
Chemical Reactions & Modifications
Bismuth potassium citrate rarely undergoes wild transformations in the body or in the pharmacy. The structure loves to stay put, barring strong acids or bases. Strong acid breaks it into free bismuth and citrate ions, which then take their own separate chemical journeys. In research labs, chemists experiment with tweaking the potassium part, swapping sodium in for certain medical applications. On a more advanced front, some researchers graft the molecule onto delivery polymers, hoping to steer the bismuth exactly where it’s needed. Such modifications sometimes increase absorption from the gut, raising hopes for lower doses and even stronger effects against persistent bacterial infections.
Synonyms & Product Names
Anyone shopping for this compound in academic literature or commercial catalogs faces a mess of names. Besides “bismuth potassium citrate,” the compound shows up as “tripotassium dicitrato bismuthate,” and sometimes under code names used in clinical studies from the ‘90s. Trade names differ by region—one finds names like De-Nol, Gastrodenol, or Helizid attached to similar formulations intended for ulcer treatment. When reading research or safety documents, checking synonyms avoids confusion, since different countries use different naming conventions based on historical patents and clinical development quirks.
Safety & Operational Standards
Current drug safety laws require both manufacturers and healthcare workers to handle this compound with respect for both its benefits and risks. Miners and processors involved in bismuth supply chains use personal protection to avoid long-term exposure to the fine dust. In the final pharmaceutical product, most patients experience little trouble, but rare cases of bismuth-related side effects—renal issues, chronic encephalopathy—must be spotted early. The safety paperwork for factories, hospitals, and clinics lines up with international guidelines: gloves and dust masks in bulk handling, locked cabinets for finished goods in the pharmacy, and prompt disposal of any unused product. Medical staff track patients for any hint of cumulative bismuth exposure, especially in long-term use. These routines have come from painful lessons a century ago, when heavy metals in medicine escaped into the wider world with less oversight.
Application Area
Doctors lean on bismuth potassium citrate most as a weapon against stomach troubles. It forms the backbone of quadruple-therapy regimens against Helicobacter pylori, often teamed up with antibiotics like metronidazole and tetracycline. The combination works two ways: the bismuth starves bacteria of the metals they need, and the citrate smooths the journey by stopping stomach acid from getting in the way. Beyond infection, the compound shows results in easing pain from gastritis and peptic ulcers—the lining gets a protective coat that helps healing. Some countries have kept it in their official treatment guidelines, while others rely more heavily on antibiotics or proton pump inhibitors. Folk remedies and off-label uses pop up in rural clinics, showing the reach of bismuth potassium citrate into both formal and informal healthcare settings. It has even emerged as a candidate for treating some protozoal infections in early lab studies, hinting at broader application.
Research & Development
The last ten years expanded researchers’ curiosity about how this compound works down to the atomic level. Teaming up with physicists, chemists have mapped binding sites on both stomach bacteria and stomach cells, chasing ways to make the drug gentler and more powerful. Multi-center clinical trials in China and Europe showed improved eradication rates for H. pylori when bismuth potassium citrate formed part of quadruple therapy, even in regions with high antibiotic resistance. Antibacterial resistance trends remain a hot topic at medical conferences, with speakers urging for renewed focus on non-antibiotic agents like bismuth compounds. Startups now run pilot tests with new versions: some release bismuth more slowly, others pair with probiotics to rebuild gut flora after treatment. Funding continues to flow from both government research councils and public-private partnerships. Universities publish results around toxicity, new delivery forms, and combination therapies, feeding a pipeline of next-generation medicines that keep bismuth potassium citrate in play.
Toxicity Research
Most toxicity studies set out to define what’s safe for both patients and workers. Short-term dosing in both animal studies and live clinical trials shows low accumulations of bismuth in liver and brain tissues. But long-term or chronic use, particularly at higher-than-recommended doses, draws some red flags. Teams in Europe documented a handful of bismuth-induced encephalopathy cases, usually in patients who received large doses over many months without proper monitoring. For most people, the kidneys flush out excess bismuth and potassium fairly quickly. Still, researchers keep a sharp eye on patients with kidney problems or advanced age, since these groups process heavy metals more slowly. Toxicologists today work with trace-level detectors that spot bismuth ions in urine and blood, helping draw sharp lines between safe use and risk zones. These advances slice out most danger for everyday users, but no one drops their guard when large batches roll out of the factory.
Future Prospects
If the research community’s excitement offers any clues, bismuth potassium citrate isn’t fading out of the medical landscape anytime soon. With antibiotic resistance building at a worrying pace worldwide, therapies that don’t rely on broad-spectrum antimicrobials gain special attention. The unique way bismuth targets bacteria without harming most healthy cells sets it apart as a foundation for new stomach medicines. As more is learned about microbiome balance, researchers look for ways to pair this compound with gut-friendly agents, hoping to heal without wiping out the body’s good bacteria. There’s also fresh energy going toward easier and more precise dosing forms—think fast-dissolve tablets or targeted delayed-release capsules. Continuous updates to international safety standards and lab testing protocols will likely follow, improving purity and cutting contamination risk. In some corners of the research world, teams explore the potential for using bismuth-based compounds in cancer therapy and as selective drugs for other challenging infections. The need for safer, well-proven alternatives gives bismuth potassium citrate a steady role in both pharmacies and research labs as the search for better health solutions evolves.
What is Bismuth Potassium Citrate used for?
Looking at How Bismuth Potassium Citrate Helps Tackle Stomach Troubles
Not every stomach pain comes from stress or spicy food. Behind many digestive complaints sits a pesky bacteria called Helicobacter pylori. Decades ago, doctors noticed that ulcers showed up even in folks who ate a clean diet. Scientists discovered H. pylori lodged deep in the stomach lining, causing irritation and upset for millions.
Bismuth potassium citrate caught the eye of researchers after they saw how bismuth compounds soothed stomachs. This compound might not steal headlines, but it takes up a central spot in multiple-drug treatments targeting stubborn H. pylori infections. You will often see it packaged as part of a therapy alongside antibiotics and acid reducers, like the famous quadruple therapy. Folks in China and other parts of Asia became quite familiar with this combo long before it spread worldwide.
The Science: More Than Just Soothing
People sometimes call bismuth salts “old-fashioned.” Yet, lab work keeps showing that bismuth potassium citrate creates a tough environment for H. pylori. Studies from journals such as Alimentary Pharmacology & Therapeutics and Gut confirm that bismuth cuts off vital functions in the bacteria, blocks enzymes it needs, and even helps reduce the bug’s ability to stick to the stomach lining.
Real-world experience backs up these experiments. In my neighborhood pharmacy, people with recurring ulcers often ask for detailed explanations of their medications. After years of chatting with patients and reading the fine print on prescriptions, it’s plain to see that their stomach issues often disappear when bismuth sits alongside antibiotics.
Why Safety and Proper Use Matter
Every drug—even minerals—can go sideways if people misuse it. Too much bismuth, taken for too long, brings side effects. Anyone who has given or received quadruple therapy knows about the black tongue or dark stools. It scares folks who haven’t heard about it before. Beyond that, rare complications—like nervous system problems—make headlines once in a while. That’s why using these medicines with doctor oversight really matters.
As more people grow resistant to basic antibiotics, bismuth potassium citrate serves as a defense line. Meta-analyses, including one published by the World Journal of Gastroenterology in 2022, show that regimens with bismuth deliver a better cure rate—especially for people who already tried and failed with other treatments.
Building Better Access and Smarter Treatment Plans
The real challenge shows up in countries with tight prescription laws or medicine shortages. Folks with ulcers but limited means can’t always get the more advanced regimens. Health authorities and doctors need to look at policies, supply lines, and patient education around these compounds.
With digestive conditions on the rise, boosting doctor awareness and patient trust could go a long way. Encouraging transparent information about possible side effects and clear instructions helps people stick to their treatment plan. In my own work with a community health collective, I’ve seen that clear talk from healthcare workers pays off—people finish their medications, flare-ups drop, and life gets better for families.
Facing the Next Challenge
As antibiotic resistance keeps building, solutions like bismuth potassium citrate play a bigger role. It’s not a miracle fix, but with smart use, solid science, and upfront conversations between doctors and patients, this compound continues to earn its spot—and saves countless stomachs from years of pain.
How should I take Bismuth Potassium Citrate?
Understanding the Role of Bismuth Potassium Citrate in Treatment
Bismuth potassium citrate shows up from time to time in clinics for one big reason: it fights Helicobacter pylori, the stubborn bacteria behind many stomach ulcers and some forms of gastritis. Stomach pain and bloating have sent a lot of folks to their doctor, and sometimes the culprit points straight to this bug. Doctors discovered through decades of research that bismuth-based medicine, especially in combination with other antibiotics, brings good results for getting rid of H. pylori. If you’ve picked up your prescription and are staring at the bottle, it’s natural to wonder about the right way to take it.
Finding the Right Timing and Dose
Doctors often suggest taking bismuth potassium citrate two or four times a day, usually before meals. This helps the medicine coat the lining of your stomach, blocking bacteria from attaching or causing damage. Meals can screw with how much medicine gets absorbed, so leaving that hour before food helps maximize the benefits. That routine might feel strict, but sticking to it helps reduce the chance for the bug to rebound.
Pharmacists usually recommend swallowing the tablet with plenty of water. Chewing or breaking tablets might lower how effective the treatment is, since the drug is meant to reach the stomach in its pretty intact state. If you miss a dose, take it as soon as you remember. But if it’s almost time for the next tablet, don’t double up—too much medicine leads to side effects like dark stools, or sometimes kidney trouble in people already at risk.
Managing Common Side Effects and Staying Safe
People have reported dark stools so often with bismuth potassium citrate that it no longer surprises medical staff. The medicine carries bismuth, a heavy metal, and the color change doesn’t spell trouble for most. What deserves attention? If you see ringing in your ears, rashes, or confusion, it’s worth calling the doctor right away. Rare allergic responses or problems with long-term use can happen, but short regimens, typically around 10 to 14 days, keep things safer for most.
I remember a few patients who wanted to stop early because black-colored bowel movements caught them off guard. A quick chat and some reassurance helped them stick with the plan and finished the treatment. Trust builds with clear, honest discussions about what to expect. Taking care not to use antacids containing aluminum, magnesium, or calcium close to your medicine also matters since these can mess with how the medicine works.
Why Completing the Full Course Matters
It might seem tempting to drop the medicine after a couple days of feeling better. Helicobacter pylori likes to hang around, even when symptoms fade. Stopping short of the planned course lets the bacteria gather strength and dodge future treatments. Doctors and pharmacists always drum this in for good reason: full completion gives the best shot at long-term cure and a settled stomach.
Most people easily tolerate bismuth potassium citrate, especially as part of the standard "quadruple therapy" for H. pylori, which research continues to back up. The rate of bacterial resistance looks lower than with single-drug regimens, making bismuth a valuable tool, especially in regions where the bug has learned to dodge other medicines.
Pay attention to the instructions from the pharmacy and reach out for clarification when things feel confusing. Your gut health deserves that level of care, especially when one simple step can keep more serious problems away.
What are the side effects of Bismuth Potassium Citrate?
What People Experience After Taking Bismuth Potassium Citrate
Bismuth potassium citrate, a common ingredient in drugs for stomach issues including ulcers and Helicobacter pylori infection, gets overlooked in everyday conversations about side effects. As someone who has spent time in both pharmacies and clinics, I can say patients usually care less about chemical formulas than about real-world outcomes. They ask what to watch out for and how it could mess with their day.
Typical Side Effects—What Patients Tell Us
The first question anyone asks is: Will this make me feel worse? The most talked-about side effect is a darkening of the tongue and stool. It looks alarming, but it’s rarely harmful. Dark stools can send people running to the ER worried about bleeding, only to find it’s the medicine and not a new health crisis. In my experience, clear communication up front stops a lot of panic and wasted hospital visits. Manufacturers and pharmacists should put this message front and center—people need straightforward warnings, not tiny footnotes.
Some users report mild stomach upset, nausea, or constipation. Less often, patients mention headaches or ringing in the ears. The rare complaints about joint pain or skin rashes carry extra weight, since allergic responses can snowball—they aren’t the sort where most folks can just sleep it off. Medical staff need to pay attention to rashes, swelling, or trouble breathing and act quickly if patients report them.
Why the Risks Matter More for Certain Groups
Older adults or those with kidney issues find their risks go up. The body simply doesn't clear bismuth as fast with age or poor kidney function—so taking too much for too long can lead to bismuth buildup. This isn’t theoretical. I’ve seen patients develop confusion, muscle twitching, and in severe cases, trouble walking, which all point to bismuth toxicity. Regular users—especially elderly folks—need more than a basic information sheet. A pharmacist should talk with them, run through other medicines, and check for symptoms at every refill.
Practical Steps to Stay Safe
The easy way to lower risks—stick to the recommended dose and timeline. People think, “If a little helps, more must work better.” This logic gets folks into trouble fast. If symptoms don’t improve or side effects show up, checking in with a doctor beats taking extra pills. Open conversations help patients flag early warning signs, and it keeps them off the internet looking for bad advice.
For those already on other medications—like blood thinners or diuretics—there’s a chance for drug interactions. Doctors need to know about everything a patient takes, not just this one medicine. In my experience, people underestimate what counts as “medicine,” skipping over supplements and vitamins. Direct questions in the clinic help catch interactions before they cause problems.
What We Should Do Better
There’s no room for half-truths in healthcare, especially with things as routine as acid relief. To keep people safe, professionals must use plain language, point out both the odd but harmless and the rare but serious side effects, and help connect the dots between how long a medicine is taken and how the body reacts. Giving people permission to ask “Is this normal?” makes a difference. Nobody should be afraid to speak up or feel brushed off.
Is Bismuth Potassium Citrate safe during pregnancy or breastfeeding?
What Is Bismuth Potassium Citrate Used For?
Bismuth potassium citrate appears in some medications for managing digestive problems such as heartburn, indigestion, and Helicobacter pylori infections. Sitting at the pharmacy shelf, those bottles look innocent enough, promising quick relief from upset stomachs or ulcers. During pregnancy or breastfeeding, though, choices about medicine impact more than just personal comfort — they can shape the baby’s well-being too. That fact deserves serious attention from parents, not just clinicians.
What Do Doctors Say About Safety?
Doctors often take a cautious route with treatments in pregnancy and breastfeeding. The FDA no longer uses pregnancy categories on labels, but data around bismuth potassium citrate remains thin. Compared to common options like antacids or H2 blockers, bismuth-based medications have not gone through enough controlled tests in pregnant or nursing women. A lack of evidence does not automatically mean danger, yet it does signal uncertainty. I’ve talked with pharmacists and OB-GYNs who often suggest safer, well-studied alternatives such as calcium carbonate or ranitidine (except for recent recalls of ranitidine over impurity concerns). Their message: stick with medicines backed by experience and research whenever possible.
Pitfalls of Using Bismuth Compounds
Bismuth, the main active metal in this medicine, sometimes builds up in the body, especially with long or repeated use. In rare cases, this buildup causes brain or kidney issues. A pregnant woman’s body changes how drugs get absorbed and cleared. Impact on the baby’s development or on milk for a nursing child has not been well studied, and that’s a real blind spot. Historic reports of bismuth toxicity in kids raise further concern, even if the risks seem rare today.
What Trusted Sources Advise
Reputable organizations like the American College of Obstetricians and Gynecologists (ACOG) and LactMed, a database for medications in breastfeeding, tend to err on the side of safety with unknowns. They recommend leaning on medications with strong safety profiles. Treatments for acid reflux or stomach pain that do not use bismuth potassium citrate show better safety records in pregnancy. Consultation with healthcare providers remains essential before taking anything over the counter.
Better Options Exist
Hearing stories from friends and patients, antacids such as Tums, Maalox, or even low-dose omeprazole brought needed relief during pregnancy without increased risk to their babies. The fact that these choices have clear data behind them helps everyone feel more confident. Bismuth potassium citrate rarely shows up as the first recommendation from doctors. Insurance coverage, affordability, and easy access to information all nudge parents in the direction of established therapies.
Simple Steps for Relief and Safety
A healthy diet, smaller meals, and less caffeine or spicy food can go a long way in cutting down on digestive trouble during pregnancy and breastfeeding. If medication is still needed, relying on options reviewed and recommended by medical organizations brings peace of mind. Sharing any medication use, including over-the-counter options, at every prenatal or pediatric appointment never goes to waste. Nothing replaces that conversation with a healthcare professional who knows both the parent and the latest research.
Resources for Parents Seeking Answers
Every parent has a right to clear, science-backed answers. Talking to a trusted healthcare team, checking FDA resources, and using evidence-based databases such as LactMed keep decision-making grounded in the safest path forward for both mother and child.
Can Bismuth Potassium Citrate interact with other medications?
Why People Use Bismuth Potassium Citrate
The stomach doesn’t always cooperate. For years, doctors have relied on bismuth compounds to help people struggling with ulcers and persistent belly pain. Bismuth potassium citrate, found in some combinations for Helicobacter pylori infection, brings relief when dealing with stubborn bacteria. Not everyone’s day job involves thinking about medications, but once a gut infection crops up, suddenly the labels on your pills matter more than what’s on your sandwich.
The Heart of The Issue: Mixing Medicines
In high school, they never taught us about drug interactions. Only after spending time with a family member on multiple medications did I recognize how even simple over-the-counter products can trip you up. Bismuth potassium citrate, for all its needed work inside the stomach, can tangle with other pills.
Doctors and pharmacists commonly warn about certain combinations. For example, speaking from experience, managing heartburn with antacids and then adding bismuth on top can throw off absorption. Antacids, particularly those packed with aluminum or magnesium, can block the body from taking in bismuth as it should. That’s not speculation; researchers tracked these slowdowns in clinical studies. It’s tough enough treating an ulcer without undoing your own efforts with another tablet.
Bismuth and Antibiotics
Ulcer treatment often involves more than one drug. If you’re using antibiotics like tetracycline or doxycycline, adding bismuth potassium citrate can slow the antibiotics down. I once watched a close friend get prescribed both, and her pharmacist pointed out that spacing them out over several hours helped. Pharmacological evidence backs this up—bismuth binds to the antibiotics in the gut and carries them out before they do their job. People sometimes wonder why their treatments stall; this is one unsung culprit.
Blood Thinners and Aspirin
I’ve known folks hesitating to share medication lists with doctors, not realizing bismuth combinations might irritate the lining of the stomach. Mixing this compound with aspirin or blood thinners, like warfarin, increases the risk of bleeding. The American Gastroenterological Association advises keeping a close eye on stools, as black or tarry stools can signal a bleeding stomach.
Kidney Worries
People managing kidney problems walk a careful line. Poor kidney function limits the body’s ability to clear bismuth. I remember stories from nephrologists describing cases where bismuth built up and started causing neurological symptoms, like confusion and tremors. That’s not headline news, but it’s well-documented and a real risk for anyone with chronic kidney disease.
What Really Helps
Too many people depend on “Dr. Google,” piecing together advice from forums and friends. From long hours spent volunteering at community clinics, I noticed, time and again, the biggest shift comes when folks list their medications before starting a new one. Pharmacists can spot dangerous overlaps. Sticking to clear schedules—spacing out doses—can help the good drugs work as expected.
The FDA, alongside leading health bodies, keeps a running list of drug interactions. It’s worth five minutes at the pharmacy counter or on the phone with your doctor before picking up something new. For those who help neighbors or care for family, making one extra call or jotting down a medication list makes the stomach, and the whole body, a lot safer.
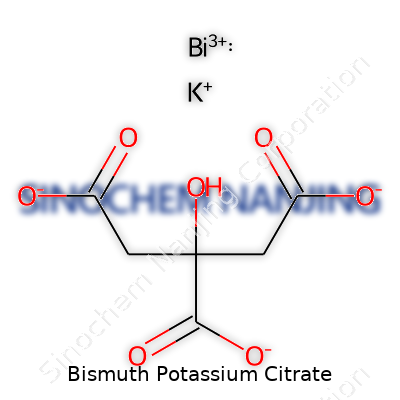
| Names | |
| Preferred IUPAC name | potassium;2-hydroxypropane-1,2,3-tricarboxylate;bismuth(3+) |
| Other names |
Tripotassium dicitratobismuthate Bismuth tripotassium dicitrate Potassium bismuth citrate Colloidal bismuth subcitrate CBS |
| Pronunciation | /ˈbɪz.məθ pəˈtæsiəm ˈsɪtreɪt/ |
| Identifiers | |
| CAS Number | [57644-54-9] |
| Beilstein Reference | 3581319 |
| ChEBI | CHEBI:91249 |
| ChEMBL | CHEMBL2106109 |
| ChemSpider | 171137 |
| DrugBank | DB09227 |
| ECHA InfoCard | 101000069078 |
| EC Number | 262-993-8 |
| Gmelin Reference | Gm.13255 |
| KEGG | C18907 |
| MeSH | D019473 |
| PubChem CID | 187631 |
| RTECS number | GG4050000 |
| UNII | J439C7P4G6 |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID0077072 |
| Properties | |
| Chemical formula | K6Bi2(C6H4O7)4 |
| Molar mass | 766.5 g/mol |
| Appearance | White or almost white crystalline powder |
| Odor | Odorless |
| Density | 2.55 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -3.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | Approximately 3.1 |
| Basicity (pKb) | 5.8 |
| Magnetic susceptibility (χ) | −92.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.94 |
| Viscosity | Viscous liquid |
| Dipole moment | 13.8 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 489.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | A02BX05 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | oral, human, prescription |
| Signal word | Warning |
| Hazard statements | Hazard statements: "H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Lethal dose or concentration | LD50 oral rat > 5,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 2200 mg/kg |
| NIOSH | WT1158000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 180 mg |
| Related compounds | |
| Related compounds |
Bismuth Subcitrate Colloidal Bismuth Subcitrate Bismuth Subgallate Bismuth Subsalicylate Ranitidine Bismuth Citrate |

