Benzotrifluoride: Tracking a Chemical’s Real-World Impact
The Backstory: From Curiosity to Chemical Staple
Benzotrifluoride didn’t turn up in history books as some critical breakthrough on its own. This compound first caught the attention of chemists looking to find aromatic hydrocarbons that could carry some unusual properties: toughness against oxidation, mighty resistance in harsh environments, and a neat ability to facilitate reactions where traditional solvents just didn’t cut it. Early on, laboratories put it through its paces as both a solvent and a building block for more complex molecules. The more scientists worked with benzotrifluoride, the more they realized how it could handle jobs that knock other chemicals out of the running. With origins that trace back more than half a century, the story of this compound picks up steam as modern industries demand more specialized, high-performing materials.
Looking Closer: Physicochemical Features
Benzotrifluoride lives up to its reputation for stability. The molecule itself builds a stable trifluoromethyl group attached to a benzene ring, which changes how it behaves compared to old-fashioned toluene or benzene. This arrangement gives it a boiling point well above 100°C, thanks to those hefty fluorine atoms. Toss benzotrifluoride into water, and you’ll see instant separation—it hates mixing with water. The molecule shrugs off acids a lot better than regular aromatics, a key reason laboratories turn to it for demanding reactions. As a liquid at room temperature, it carries a sweet, sharp aroma that’s unmistakable once you’ve worked with it. Unlike some volatile organic compounds, benzotrifluoride ranks lower in flammability, and that trifluoromethyl branch helps it stick around without breaking down, even in the presence of light or oxygen.
Preparation and Operating Tricks
Chemists generally synthesize benzotrifluoride using a straightforward process: classical routes relied on chlorination and fluorination steps, starting from toluene or its derivatives. Those steps have gotten cleaner over time thanks to modern catalysts and improved heat management. The most common method involves treating benzotrichloride with hydrogen fluoride or another fluorinating agent. This reaction demands careful attention to pressure, temperature, and reagents that don’t blow up or corrode the equipment. Later, companies started refining conditions to boost yields and reduce waste. As far as technical labeling, the benzotrifluoride name gets swapped for synonyms like trifluorotoluene, α,α,α-trifluorotoluene, or even BTF among industry insiders. Regulatory labels push for clear hazard icons since the material comes with distinct warnings about inhalation and environmental exposure.
Changing Its Tune: Chemical Reactions and Derivatives
That trifluoromethyl side group doesn’t just sit tight on the benzene ring—it makes benzotrifluoride behave in distinctive ways in chemical reactions. The electron-withdrawing nature of those three fluorine atoms means the ring itself becomes less reactive in classic electrophilic substitutions. Still, chemists have figured out how to push reactions like halogenation, nitration, or sulfonation if they get conditions just right, like strong acids or specialized catalysts. On the flip side, the molecule wins big as a starting point for pharmaceuticals, agricultural chemicals, and specialty polymers. For instance, swap atoms on the benzene ring and you can open doors to new herbicides or active pharmaceutical ingredients designed to resist metabolic breakdown. The ability to branch off into new derivatives extends its reach into industries that rely on materials built to last.
Safety Out in the Real World
Handling benzotrifluoride means respecting its risks as much as its utility. In labs, teams follow protocols to minimize skin exposure, inhalation, and eye contact. Breathing in its vapor causes headaches or dizziness, so fume hoods aren’t a luxury—they’re non-negotiable. Despite lower flammability, storage still means keeping it away from heat sources, open flames, and oxidizers. Benzotrifluoride does not break down easily in the environment. Researchers have found that wildlife exposed to significant concentrations shows signs of toxicity, including changes in liver enzymes or behavioral patterns. Anyone moving drums of this liquid or working with kilo-scale batches runs monthly checks on gloves, goggles, and ventilation. Regulatory agencies raise the bar each year, sometimes updating workplace exposure limits as new data rolls in.
Where It Works: Applications in the Field
Benzotrifluoride shows up in more industries than its obscure name suggests. Chemical manufacturers count on it as a high-stability solvent, especially when making compounds that fall apart under acidic or oxidative stress. The compound acts as a reliable intermediate for synthesizing modern agrochemicals—think weed killers with better soil lifespans, or insecticides designed to tackle stubborn pests without washing away at the first rain. In pharmaceuticals, some labs use BTF derivatives to design drugs that hang around a bit longer in the bloodstream, sidestepping rapid breakdown in the liver. Polymer makers like its contribution to specialty plastics and coatings that demand water and chemical resistance. Even electronics industries dabble in using benzotrifluoride for photoresists, adhesives, and advanced materials where trace contamination must stay low. Knowing where to use this material often separates average tech from high-performing designs in competitive sectors.
Pushing the Boundaries: R&D and Toxicology
Research teams tackle questions that haven’t been answered about benzotrifluoride’s behavior in living systems and in the environment. Toxicologists focus on understanding how the body handles exposure: the compound absorbs through skin, but the major danger comes from inhalation or accidental ingestion. Long-term animal studies track liver and kidney effects, sometimes linking exposure to subtle metabolic changes. Based on animal testing, regulatory organizations have issued strict guidelines for occupational exposure, although benzotrifluoride rates lower in acute toxicity compared to similar solvents. On the research front, scientists keep pressing to tweak the molecule or its derivatives for greener, safer performance without sacrificing industrial strength. Some universities run studies on photocatalytic breakdown, searching for cost-effective ways to clean up accidental spills or reduce environmental persistence. It’s a tricky balance—bringing these advances into mainstream production without driving up costs or introducing new risks.
Chasing Tomorrow: Prospects and Challenges
Looking ahead, benzotrifluoride faces pressure from multiple sides. Green chemistry puts a spotlight on how long chemicals linger in the environment and whether alternatives can do the same jobs with less hazard. Growing regulatory interest in PFAS and other persistent substances means ongoing review of how benzotrifluoride fits into sustainable manufacturing. At the same time, industries keep asking for stronger, more resilient materials and niche intermediates that only compounds like BTF can deliver. The solution often means investing in recycling methods, safer synthesis pathways, and breakdown technologies that close the loop on chemical use. For R&D teams, the push goes toward less toxic derivatives, biodegradable alternatives, and manufacturing processes that trim energy needs. In a world moving toward safer, cleaner solutions, benzotrifluoride wears the badge of a proven performer while offering a tough challenge for scientists committed to progress without compromise.
What is Benzotrifluoride used for?
More Than Just a Solvent
Benzotrifluoride shows up a lot in industries we count on, even if most people haven’t heard its name. You’ll find it in paints, adhesives, cleaning agents, and especially as a building block for making other chemicals. I once worked on a project involving specialty coatings, and we learned fast how often manufacturers depend on consistent, reliable solvents that can handle heat and harsh conditions. Benzotrifluoride brings both toughness and stability—qualities that turn basic compounds into durable products.
A Behind-the-Scenes Player in Pharmaceuticals
This chemical plays a role in making both over-the-counter drugs and newer medications. Take a look at the pharmaceutical sector, and benzotrifluoride keeps popping up, not as a medicine, but as a starting point for creating complex molecules. That’s important. New cancer treatments and antiviral drugs often get their start from building blocks like this one. Process chemists like to mention its chemical stability, which helps reduce unwanted reactions, making it easier to produce high-purity compounds that doctors count on.
Driving Safer and More Efficient Reactions
Working in research, I saw how often a safe alternative to older, harsher solvents became a game changer. Benzotrifluoride steps in for toluene and xylene, which come with baggage: flammability worries and stricter workplace regulation. With benzotrifluoride, manufacturers can meet safety rules without lowering output. This matters to people on the ground. It cuts risks for workers and sometimes trims clean-up costs.
Environmental Questions and Industry Responsibility
Benzotrifluoride doesn’t just disappear after industry uses it. Waste oversight matters, since improper handling can lead to slow breakdown and possible environmental buildup. European and North American agencies have flagged it for monitoring due to possible water and soil impacts. In my role at a chemical plant, I saw firsthand the pressure to reuse solvents and set up recovery systems. Investments in closed-loop operations, like distillation units and sealed tanks, stop leaks and save money in the long run.
Safer Consumer Products Start With Better Building Blocks
Every day, people rely on safe packaging, medical devices, and home improvement supplies. Many of these goods start from advanced polymers and specialty chemicals made with solvents such as benzotrifluoride. The push for greener chemistry means more labs search for tweaks or replacements that keep performance high and impact low. Smaller startups experiment with biosourced or biodegradable alternatives, and larger players fund safer chemical research. The market rewards companies that pay attention to both safety and sustainability.
Finding a Balance
Benzotrifluoride reminds me that industry choices can ripple out much further than the plant gates. Factories that keep safety gear updated, invest in worker training, and rethink their waste recovery practices stand a better chance at long-term success. Public health and sustainability groups often push companies past what’s legally required, but that’s where real progress happens. The chemical itself is just a tool; the bigger question is how wisely we use it and what responsible stewardship looks like as science keeps pushing forward.
Is Benzotrifluoride hazardous to health?
A Familiar Story from the Lab
Many folks working in industrial or laboratory settings have handled benzotrifluoride. It often shows up as a solvent and intermediate in chemical processes. On the job, the label usually has the same warnings: avoid breathing vapors, keep away from flames, use proper protection. Many of us follow those precautions because we trust the science and the real-world experience behind those recommendations.
Health Risks in the Real World
I remember the first time a drum of benzotrifluoride arrived at work—my supervisor pointed out the faint, sweet odor. It seemed less harsh than other solvents I’ve worked with, like toluene or xylene, but looks can be deceiving. Studies show benzotrifluoride can irritate the skin, eyes, and respiratory tract. Short-term exposure often gives headaches or dizziness, similar to many organic solvents. Spraying it without a fume hood once left me lightheaded for hours. That’s not a memory I want to repeat.
Chronic effects draw more concern. There’s not much long-term data on human cases, but reports indicate possible liver and kidney stress after long exposure in lab animals. European regulators list it as a substance requiring careful handling; the Environmental Protection Agency recommends limiting exposure. If you spill a drop or two, the odor stands as a warning, but the colorless, volatile nature means it can go unnoticed and accumulate—a silent hazard waiting for a breach in protocol.
Why It Matters Beyond the Lab
This chemical isn’t just a lab problem. It plays a role in making pharmaceuticals, pesticides, and dyes. Any place workers use it regularly faces the same repeat exposure risk. Sometimes, regulators lag behind what’s happening in factories or waste facilities. In some cases, warehouses with poor ventilation or outdated safety practices grow ripe for accidents.
Supporting the Warnings with Facts
The American Conference of Governmental Industrial Hygienists (ACGIH) lists a low threshold limit value, suggesting caution against overexposure. The National Institute for Occupational Safety and Health (NIOSH) recognizes the potential for health effects after both short and long periods of contact. European Classification labels it as an environmental hazard. That’s a clear sign that containment and ventilation should rank as high as actual chemical use in daily procedures.
One important point: water solubility stays low, so it won’t spread easily in wastewater. But its persistence means it lingers in the environment. Some studies flag it as a probable contributor to long-term contamination in soils near production sites.
Practical Steps Toward Safer Use
No one wants a job-related health scare. For anyone working with benzotrifluoride, gloves, eye protection, and well-maintained ventilation make a big difference. Engineering controls like local exhaust systems help keep air quality in check. Good spill response plans and frequent air monitoring build confidence and stop harm before it starts.
Training and open communication also matter. Everyone working with more than a few liters should know the emergency plan and the reasons behind strict safety steps. New workers and seasoned hands both benefit from reminders and retraining. Labeling helps, but nothing beats a culture where taking safety seriously is a shared goal.
Looking Forward
Safer alternatives may exist, or processes could shift to minimize benzotrifluoride use where possible. Industry leaders and regulators should stay in contact to keep standards updated as more data becomes available. Preventing exposures today keeps workers, the environment, and communities safer for years to come.
What are the physical and chemical properties of Benzotrifluoride?
Getting Familiar with Benzotrifluoride
Benzotrifluoride brings to mind a clear, colorless liquid you might spot in labs and factories. Its chemical formula, C7H5F3, stands out because each molecule tacks three fluorine atoms onto a benzene ring through a single carbon. This structure leads to a unique blend of properties that make it more than just another chemical in the warehouse.
What’s Special About the Physical Side?
Pick up a container of benzotrifluoride and you notice a light, slightly sweet smell. At room temperature, it stays liquid, with a boiling point around 102 °C and a freezing point down near -30 °C. I’ve seen it pour easily between glassware, since its viscosity is low. Its specific gravity lands at about 1.25, heavier than water, so it forms a separate layer if mixed. If there’s a small spill, it’ll evaporate faster than something like motor oil, but not nearly as quickly as acetone. That speed of evaporation helps workers notice its presence before it pools and becomes a hazard. It's also not soluble in water, so it tends to float or stay apart, which people in the field use to help clean up or contain it.
Chemical Foundations That Matter
Benzotrifluoride keeps its cool better than quite a few other organic solvents. It’s naturally stable because those fluorine atoms pull electrons tight, making the ring less eager to react. This matches up with lab work where stability keeps things safe and predictable. It resists mild acids and bases, so it doesn’t break down easily in storage. Still, it can light up in the right conditions. Its flash point comes in at about 32 °C, so storage away from open flames and high heat makes good common sense.
Health and Environmental Considerations
Working with benzotrifluoride, you learn not to mess around—breathing the vapors or letting your skin soak it up doesn’t do your body any favors. Short exposure sometimes brings headaches or dizziness, maybe skin irritation. Long-term effects aren’t fully mapped out, but the chemical’s persistence in soil and water means it can linger. Unlike many solvents, it doesn’t break apart easily in the environment. In my own experience, even small traces stick around after spills unless cleaned up quickly and thoroughly. That’s a risk for both workers and wildlife. Good ventilation, gloves, and eye protection always stay within reach while using it.
Industry Uses and What Drives Them
Benzotrifluoride shows up all over the industrial map. Paints and coatings companies lean on it as a solvent; chemists use it as a starting point for making pharmaceuticals and pesticides. What’s striking is how the trifluoromethyl group molds the reactivity, letting scientists coax out molecules that few other starting materials can match. Automotive and electronics firms appreciate its tough, resistant nature in specialty fluids. While the demand grows, so does responsibility to handle it safely—no room for shortcuts.
Sensible Solutions for Safer Use
Some facilities now rely on recovery and recycling systems, cutting down the release of benzotrifluoride into waste streams. That technology, paired with training and strong safety habits, lowers risk on the production line and in downstream use. Regulatory guidelines also shape how this chemical travels and gets used, from shipping to disposal. There’s room for improvement in safeguards, like real-time detection tools and secondary containment, especially in crowded workplaces. Addressing the persistence means not just keeping exposure down but also looking for greener alternatives where production volume keeps rising.
How should Benzotrifluoride be stored and handled?
The Need for Caution
Benzotrifluoride finds its way into countless labs and industrial spaces. From my experience in a mid-size chemistry lab, anything with “trifluoride” in it rings alarms for good reason. It looks harmless enough in a flask, but behind clear liquid and a faint sweet odor, real hazards lurk. Let’s not take shortcuts with this one—sloppy storage or mishandling almost always equals risk.
Understanding the Hazards
Benzotrifluoride evaporates quickly at room temperature. Vapors can catch fire with a single spark. Splash a few drops on your skin and irritation follows. Breathe in enough vapors, and you’ll feel dizzy or short of breath after just a few minutes. In the lab, I’ve seen someone brush off a splash as no big deal until a rash formed an hour later. Nobody should want a label as the person who closed a safety shower room for the day.
Getting Storage Right
You won’t find this chemical sitting on an open bench in any workplace that puts safety first. Metal drums or tightly sealed glass bottles work best, but only inside rooms built for flammable chemicals. Storage areas should maintain steady, cool temps—heat invites pressure buildup, and pressure means containers might burst. I always watch for heavy sunlight sneaking through windows, especially in older buildings. One summer at our campus, we found out the hard way: a forgotten bottle warmed to nearly 40°C behind a broken shade. The cap bulged and hissed. Now, every bottle gets double-checked before leaving it even overnight.
Don’t Overlook Ventilation
Strong air flow works better than any mask. If vapors escape, good ventilation whisks them away before anyone soaks up a lungful. Fume hoods aren’t optional. I remember trying to get a simple distillation done, only to trip a sensor in a hood with poor airflow. The alarm blared until maintenance fixed the filter. I learned that day: no shortcut saves time if you can’t breathe or see straight.
Everyday Handling Steps
Goggles and gloves are your friends. Nitrile or neoprene work best; latex doesn’t last long with organic solvents. Pour slowly—splashing can mean ruined clothes, injured skin, or worse. Absorb any spills with sand or commercial pads before they dry. Open flames, static discharge, and even sparks from failing equipment have started fires. A colleague once knocked over a beaker and barely avoided a flare-up because we kept flame detectors working and an extinguisher within reach. Don’t let unfamiliarity tempt you into carelessness. Routine drills help keep everyone sharp. In our lab, we run through spill response every month; nobody rolls their eyes when real emergencies happen.
Thinking Ahead
Safety records improve when teams invest in basic awareness. Put up simple signs about hazards. Train even the most seasoned workers regularly. Encourage reporting on near-misses—some of the most important lessons come from accidents that almost happened. Proactive safety costs less time and money than cleanup and medical bills. If storage feels like an afterthought, it’s probably time to revisit policy. For me, knowing how to handle Benzotrifluoride safely means confidence in my workday—and that’s something everybody deserves.
What safety measures are necessary when working with Benzotrifluoride?
Getting Straight to the Risks
Benzotrifluoride looks pretty tame at first glance, but looks rarely tell the whole story. This chemical doesn’t mess around—swallowing it or breathing too much of its vapor sets off rapid heartbeats, headaches, and even confusion. Skin contact goes beyond just irritation; some folks develop chemical burns or reactions that show up days later. No job is worth that panic attack you get from respiratory distress or a trip to the ER with burns up your arms.
Personal Choices Can Make a Big Difference
Every lab tech and production worker knows the feeling of "just this once" when it comes to safety gear. Slipping on gloves or goggles might sound like nagging, but I’ve seen a colleague miss weeks of work over a careless splash. Nitrile gloves, not latex, give the best shot at keeping chemical exposure low. Eye protection should seal well—goggles, not glasses. Clothing ought to cover as much skin as possible; long sleeves, closed shoes, the works. It makes me wince thinking about a friend who ignored advice, only to learn about contact dermatitis the hard way.
Ventilation Strategies People Overlook
One time, a coworker shrugged off the fume hood, convinced a cracked window would do the trick. It didn’t. Fume hoods don’t just check a box—they cut vapor buildup and keep breathing air safe. Good ventilation matters even for short tasks, especially since benzotrifluoride vapors hang around. Relying on luck or open office windows isn’t enough. Proper airflow, routinely checked for leaks and clogs, keeps everyone safer and stops a small mistake from turning into a big medical bill.
Storing with Respect
Storing chemicals might feel like background work, but emergencies start with a missing cap or careless shelving. Benzotrifluoride belongs on its own shelf, away from heat or open flames. It catches fire faster than most expect. Workers sometimes pile everything together out of convenience, but I saw a costly warehouse fire caused by just such a shortcut. Flammable storage cabinets, clear labeling, and tight-fitting caps go a long way. Never underestimate the peace of mind from double-checking the label or confirming the cap is tight before heading home.
Emergency Readiness: Small Habits Become Lifesavers
I once witnessed a spill where the worker froze, not sure which extinguisher to grab or where the nearest eyewash was. Practicing spill responses isn’t just for new hires. Regular drills, clear instructions, and easy access to eyewash stations can shave seconds off response time. Knowing exactly where to find the emergency shower or the right fire extinguisher stops panic from costing someone their eyesight or worse. A spill kit should always sit close by—with sorbents rated for organic solvents, not just “universal” products that can fall short.
Training and Clear Communication Matter
It’s tempting to treat safety briefings as formalities, something to tune out or gloss over. That attitude invites disaster. I’ve seen teams that swap tips before each shift head home healthy for years, while others who skip the talk rack up injuries. New workers need reminders just as much as veterans. Open communication—sharing near-misses and lessons learned—raises the bar and fosters a culture where looking out for each other isn’t just lip service.
Moving Forward Together
Staying healthy on the job—especially around chemicals like benzotrifluoride—calls for respect, vigilance, and teamwork. PPE, ventilation, careful storage, emergency preparation, and honest talk keep accidents from escalating. Safety works best when everyone buys in and shares the responsibility, creating an environment where workers can focus on the job without fearing the hidden dangers of what’s in the bottle.
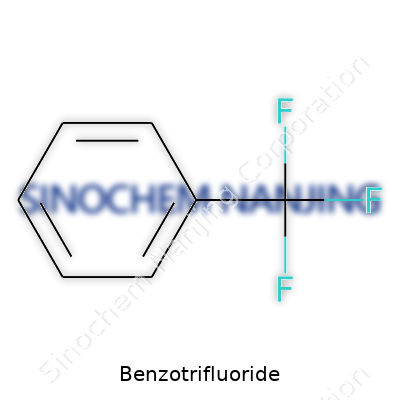
| Names | |
| Preferred IUPAC name | 1-Phenyl-1,1,1-trifluoromethane |
| Other names |
Benzotrifluoride Trifluoromethylbenzene α,α,α-Trifluorotoluene PhCF3 1,1,1-Trifluorotoluene |
| Pronunciation | /ˌbɛn.zoʊ.trɪˈfluː.ə.raɪd/ |
| Identifiers | |
| CAS Number | 98-08-8 |
| Beilstein Reference | 1631189 |
| ChEBI | CHEBI:142733 |
| ChEMBL | CHEMBL14238 |
| ChemSpider | 10615 |
| DrugBank | DB14018 |
| ECHA InfoCard | 100.008.634 |
| EC Number | 206-398-3 |
| Gmelin Reference | 2952 |
| KEGG | C02514 |
| MeSH | D000594 |
| PubChem CID | 7535 |
| RTECS number | CU1400000 |
| UNII | 4H1K03RO41 |
| UN number | UN2321 |
| Properties | |
| Chemical formula | C7H5F3 |
| Molar mass | 170.12 g/mol |
| Appearance | Clear, colorless liquid |
| Odor | Aromatic |
| Density | 1.353 g/cm³ |
| Solubility in water | 0.13 g/100 mL (20 °C) |
| log P | 2.9 |
| Vapor pressure | 4.2 mmHg (20 °C) |
| Acidity (pKa) | 43 |
| Basicity (pKb) | 12.76 |
| Magnetic susceptibility (χ) | -60.5e-6 cm³/mol |
| Refractive index (nD) | 1.333 |
| Viscosity | 0.86 mPa·s (at 25 °C) |
| Dipole moment | 2.81 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 248.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -211.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3523 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P261, P271, P280, P301+P312, P304+P340, P305+P351+P338, P312, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 50°C (122°F) |
| Autoignition temperature | 565°C |
| Explosive limits | Upper: 7.0%, Lower: 1.3% |
| Lethal dose or concentration | LD50 oral rat 6810 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 6810 mg/kg |
| NIOSH | WA8400000 |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 40 ppm |
| Related compounds | |
| Related compounds |
Trifluorotoluene Fluorobenzene Bromobenzene Chlorobenzene Nitrobenzene Toluene Benzene |

