Isoamyl Butyrate: More Than Just a Fruity Note in Chemistry’s Long Story
The Road from Discovery to Modern Use
The world often overlooks the stories behind the small molecules that power our daily comforts, and isoamyl butyrate deserves a spot in that conversation. This quirky ester, with its sweet scent that reminds some of juicy pears or overripe bananas, traces its roots back to early synthetic organic chemistry. The work of nineteenth-century chemists, striving to mimic the heady fragrances of fruits, sparked the first deliberate production of isoamyl butyrate through classic Fischer esterification. Their goal wasn’t always clear at the outset, but the result has rippled through food, flavor, and even chemical manufacturing ever since. I remember studying the practical aspects of ester synthesis, using isoamyl butyrate as a perfect model for teaching acid-catalyzed condensation reactions. College labs and textbooks treat it as familiar territory, but the journey from lab flask to full-scale flavor manufacture never happens by accident.
Sizing Up the Product
So, what’s inside that clear, colorless bottle labeled “isoamyl butyrate”? At its core, this compound brings together isoamyl alcohol and butyric acid into a molecular handshake that both pleases the nose and brings useful solvency. It pours as a slightly oily liquid, with a flash point that demands respect in warehouse settings. Chemically, it stands apart from its cousins in the ester family largely due to that waggish branched amyl group, which transforms a base chemical into a sought-after note in candies, beverages, and perfumes. The purity chase never ends—producers screen for trace acids and residual alcohol to avoid off-odors in finished products. Sometimes that pursuit of the “cleanest” aroma adds a layer of cost for processors but saves headaches for formulators seeking a consistent result batch after batch.
Physical Behavior and Chemical Quirks
Anyone handling isoamyl butyrate learns fast that its low water solubility and pleasant volatility shape both its promise and its risk. That volatility gives flavors a lift but also contributes to evaporation losses in open systems. It tolerates mild acids and bases but buckles under aggressive hydrolysis, breaking back down into the smell of rancid butter and cheap moonshine. More than one mistake in a research lab has underscored the importance of dry, inert storage—isoamyl butyrate loves to react if you leave it exposed for too long, especially somewhere hot and humid.
The Details Behind The Label
Every bottle tells a story—answers to labeling regulations, purity declarations, and batch traceability. Food industry demand for detailed technical information forces producers to document everything from density to refractive index. Regulations by agencies like FDA, EFSA, and their peers keep a close eye on any compound entering the supply chain, so isoamyl butyrate that goes into gums or sodas carries paperwork as thick as a season’s worth of receipts. Fine print matters: the content of each bottle needs to be precise, or else manufacturers face the wrath of both regulators and consumers with sharp noses.
Mixing Up Isoamyl Butyrate
Making isoamyl butyrate isn’t exactly rocket science, but making it well takes attention and know-how. Most pathways rely on the age-old Fischer method, marrying isoamyl alcohol with butyric acid using an acid catalyst under controlled heat. Sometimes modern alternatives—like enzymatic esterification—pop up for specialty applications, especially in “natural” flavor compounds. Side products can sneak in, especially if quality controls slip or reagents get reused past their prime. This process, repeated and refined over decades, keeps turning out a building block for flavors, research reagents, and perfumery.
Reactions and Tinkering Around the Edges
Isoamyl butyrate plays well in chemical modifications, which lets researchers push its uses a bit further than the basic ester link. Under the right lab tricks, it can be transesterified to form new compounds, or hydrolyzed to reclaim its component parts if someone needs to rework a batch. In the presence of strong acids or bases, it won’t last long, which is exactly the point for those who want to break down food flavorings during analysis or recycling. Some labs experiment with derivatization or isotope labeling, which feeds back into better analytical techniques and tighter quality controls.
Names and Faces Across Borders
Walk through a chemical storehouse and isoamyl butyrate wears different names: 3-methylbutyl butanoate, isopentyl butyrate, or even “pear oil.” These synonyms don’t just reflect local nomenclature—they signal to buyers and regulators what’s inside. In flavorist catalogs, it appears alongside dozens of similar compounds; in research circles, each naming convention tells a little about its origin and intended use. Keeping those names straight matters when different countries impose different expectations or restrict uses to certain sectors like confectionery or fine fragrances.
Rules for Safe Handling
Dealing with isoamyl butyrate means working with common-sense precautions: splash goggles, gloves, good ventilation, and thoughtful storage, all count. It isn’t especially noxious at low concentrations—after all, we eat it all the time in candy and soft drinks—but concentrated vapor can irritate airways or trigger headaches in poorly-ventilated labs. Flammability counts as an ever-present risk, with lower explosive limits not so far above typical operating concentrations. I’ve seen warehouse managers drill their crews on spill containment, and flavorists will vouch for the wisdom of capped bottles, clean benches, and clear labeling. Most accidents trace back to simple oversight, not malice or incompetence.
Where Isoamyl Butyrate Finds its Place
Its strongest influence lands on the flavor and fragrance industries, even though it never makes kitchen headlines. Food makers lean hard on it for fruit flavors that punch above their natural weight—juicy pears, tropical blends, and certain soft drinks all benefit from a dash of this ester. Outside candy and beverages, perfumers value its crisp, uplifting character to evoke freshness in a wide sweep of consumer goods. There’s a role too in solvent applications, especially for specialty coatings or cleaning formulations, though competition from cheaper substitutes keeps this a side-line rather than the main show.
Scientific and Industrial Curiosity
Academic research around isoamyl butyrate stretches beyond flavor science—covering synthesis pathways, green chemistry, and analytical modeling. Green production gets attention: enzymatic routes, renewable feedstocks, and lower-waste techniques reflect a move away from petroleum-based chemistry. The analytical community picks apart trace levels in food matrices, aiming for ever-lower detection limits and a better understanding of its breakdown during storage. This work doesn’t always make headlines, but it quietly powers more reliable labeling and tighter safety standards.
Health, Hazards, and Open Questions
Toxicity studies give isoamyl butyrate a favorable profile in low doses, which fits its high-volume use in food and scent products. Lab animals and cell line tests haven’t turned up carcinogenic or strongly mutagenic effects at levels typically encountered by consumers. That said, the science keeps an eye on long-term metabolic impacts of heavy flavor use, especially in processed foods where esters stack up in complex mixtures. As with most chemicals, the occupational hazard falls most on those who handle it day-to-day: skin or eye contact means irritation, and inhaled vapors become a problem in unventilated rooms. Regulatory agencies have responded by tightening workplace exposure limits, but gaps linger in worldwide harmonization, especially outside North America and Europe.
The Road Ahead: Demand, Disruption, and Green Chemistry
Isoamyl butyrate’s future tracks the growing scramble for natural flavorings, plant-derived fragrances, and “clean label” ingredients. As food and beverage giants chase sustainability targets, questions grow about sourcing isoamyl alcohol from fermentation rather than refinery side-streams. Similarly, butyric acid production shows signs of moving into bio-based territory, which cuts the carbon footprint and aligns with climate commitments. Researchers keep digging for more efficient, less wasteful ways to stitch these molecules together. The consumer tide keeps pushing upwards—snack food demand, fruity sparkling drinks, and specialty confections drive an appetite for clean, strong, consistent flavors. So while isoamyl butyrate might seem like just another cog in the flavor wheel, it’s one that bears watching as chemistry’s priorities shift toward both sensory delight and environmental responsibility.
What is Isoamyl Butyrate used for?
Isoamyl Butyrate: A Sweet Detail in Everyday Life
The name isoamyl butyrate doesn’t come up at most kitchen tables, but this compound pops up more than many realize. If you’ve ever bitten into a candy and tasted a pear or banana note, you’ve encountered it. Isoamyl butyrate packs a punch as a flavoring in food and drink. I’ve seen it labeled as a “flavoring agent” on a variety of sweet snacks — it’s there lending fruitiness to everything from bubblegum to soda.
In the Flavor Industry
Food technologists have leaned on isoamyl butyrate to craft flavors that mimic nature. It shines in pear, apple, and sometimes pineapple profiles. My younger days experimenting with baking revealed how adding a touch of “banana essence” can shift a recipe. That same idea drives industrial use. This compound helps manufacturers skip the headaches and costs of extracting flavor from fruit, which can get expensive and unreliable, especially off-harvest.
Food safety is a constant concern with any ingredient. According to reviews by health agencies, isoamyl butyrate is considered safe in the small amounts found in food and beverages. These agencies track exposure closely so consumers can enjoy flavor without worry.
There’s More Than Sweets
Beyond kitchen shelves, isoamyl butyrate has a strong presence in fragrances. Perfumers use it for its fruity character, balancing out floral and citrus notes. It has popped up in shampoos, soaps, and lotions. Perfume creation is a bit like cooking; building a memorable scent relies on finding the right building blocks. My personal experience with essential oils taught me the difference even a few drops make — and that’s how fragrance companies often use this ester.
It’s also tapped for its mild solvent properties. In industrial settings, that means paint removers, cleaning products, and sometimes printing inks. The fruity odor even helps mask harsh chemical smells in certain household items. As a solvent, isoamyl butyrate can make difficult sticky jobs easier — though always used with proper care.
Safety and Environmental Impact
The health and environment aspects matter more today than ever before. Isoamyl butyrate’s volatility means it evaporates quickly, which could lead to air quality concerns if used in large amounts or poorly ventilated spaces. Workers in factories rely on good ventilation and protective gear. Having spent time in a food lab, I’ve seen firsthand how small measures, like well-placed exhaust fans or rotating staff duties, protect against accidental overexposure.
Disposal also draws attention. As a synthetic compound, isoamyl butyrate shouldn’t go down drains unchecked. Waste management guidelines call for proper collection and treatment. Most industries take steps to reduce spills and leaks. Some smaller businesses have partnered with waste processing companies to handle leftover chemicals safely.
New Directions and Alternatives
Interest in natural and sustainable ingredients has soared. Some researchers look at bio-based production of isoamyl butyrate, starting from plant material instead of oil-based chemicals. Enzyme technology shows promise here. If these efforts scale, more food and fragrance brands could claim a smaller environmental footprint.
This push reminds me of changes in the dairy industry — as demand for "plant-based" rose, companies adjusted to meet expectations. Isoamyl butyrate’s future might look similar, driven by consumers who want both flavor and responsibility in a product. Businesses tracking trends and investing in cleaner methods can stay ahead while keeping trust strong.
Is Isoamyl Butyrate safe for human consumption?
What Is Isoamyl Butyrate?
Isoamyl butyrate pops up in all kinds of foods and drinks. This chemical may sound technical, but it's just an ester put together from isoamyl alcohol and butyric acid. Bakers and beverage companies like it because it smells fruity, a little bit like banana or pineapple, and works wonders in flavoring candies, soft drinks, and baked goods. You’ll even find it in perfumes and cosmetics because that light tropical note is tough to match.
Evidence on Safety
The question of safety makes sense, especially since no one wants mystery chemicals slipping into their snacks. The U.S. Food and Drug Administration (FDA) calls isoamyl butyrate "Generally Recognized as Safe" (GRAS) for use in food when used in small amounts. The European Food Safety Authority classifies it as safe too. That means scientists have studied it for years and haven’t found evidence of harm at levels commonly used in food.
Researchers often test flavor additives like isoamyl butyrate on animals and human cells, looking for any sign they might cause long-term issues. Isoamyl butyrate doesn’t build up in the body. After digestion, enzymes break it down into the basic building blocks everyone eats daily: alcohol and fatty acids. There is barely any literature showing toxic effects in animals, even when scientists push the doses much higher than you’d ever find in a piece of candy or a muffin.
Allergy and Sensitivity Questions
Some people have a sensitive stomach or find themselves allergic to almost anything new. Isoamyl butyrate hasn’t shown up on common allergen lists. Over the years, there haven’t been clusters of people getting sick from foods that use it for flavor. Still, just about any ingredient can set off a reaction in rare cases, especially in people with strong histories of chemical sensitivity. Anyone who worries might want to stick to foods made from scratch to keep an eye on every ingredient.
Natural vs. Synthetic Flavors
The flavor industry loves to debate natural and synthetic additives. Isoamyl butyrate sometimes shows up as ‘natural flavor’ if someone makes it from plant sources. Chemists produce most of the supply in factories, though, since it’s identical in structure whether you squeeze it out of a fruit or build it in a lab. Safety testing treats both forms the same—what matters is the molecule, not the origin. People looking for clean labels might still want to check how companies describe it on their ingredient lists.
Staying Informed
Checking up on food ingredients has turned into something of a hobby for lots of people. Groups like the FDA, EFSA, and World Health Organization review new science regularly. So far, their reviews on isoamyl butyrate haven’t turned up red flags. Researchers would likely spot trouble if anything major emerged, thanks to food monitoring programs and ongoing toxicology reviews. Keep in mind, moderation remains key. Flavors and additives work best in small amounts—nobody benefits from guzzling them by the cupful, even if current science shows they’re safe at the levels used in food.
Solutions for Cautious Consumers
Anyone remaining unsure about isoamyl butyrate can look up foods made without flavor additives. Buying whole foods or products from brands that skip synthetic flavors guarantees simple ingredient lists. If concern continues, write to companies and ask how they source and use flavorings. Sometimes just knowing who made an ingredient and how it winds up in your snack can make all the difference.
What does Isoamyl Butyrate smell like?
Sniffing Out the Memories
One whiff of isoamyl butyrate takes the mind somewhere sunny, likely into a candy shop or an orchard just past harvest. This compound doesn’t hit the nose like ordinary laboratory fare. It brings sweetness, a feeling of ripe fruit, and—if you pay close attention—a trace of something almost fizzy. Think of banana-flavored taffy, juicy pears sitting in syrup, fruit punch cooling at a birthday party. Isoamyl butyrate carries that unmistakable “fake banana” charm that hardly feels artificial once you’ve made the association.
The Science of Sweet Aromas
Isoamyl butyrate falls under a family called esters. These compounds come together when acids and alcohols meet, and they’re partly responsible for the way fruit smells as it ripens. Nature knows what it’s doing; our brains are wired to notice and remember scents like these. Isoamyl butyrate appears in trace amounts in real fruit, but what ends up in candy and soft drinks usually comes from a lab. People have strong feelings about the “banana” note in gum or jellybeans—some love it for its nostalgic hit, others call it too bold or even fake. The scent feels deeply familiar because the compound helps flavors pop in everything from bubble gum to rainbow sherbet.
Safety, Trust, and Taste Buds
Food companies started using isoamyl butyrate to mimic fruitiness over a century ago. The process took off because real bananas and pears can’t always deliver a punchy, consistent aroma. Agencies like the FDA and EFSA have weighed in: at the levels used in food and cosmetics, this compound counts as safe. That trust comes from long studies and reviews involving toxicologists and flavor chemists, not just a taste test. The European Food Safety Authority, in fact, includes it in their list of flavorings “generally recognized as safe.” People might never know exactly what goes into that “banana” smell, but no one wants to bite into a mystery.
Common Concerns and Fresh Solutions
Many folks ask whether scents like these are too artificial. The answer often sits in the gray area. Artificial flavors fill a real need. Farmers can’t harvest perfectly ripe bananas year-round, and food makers need every batch of soda or chewy candy to taste the same. Still, shoppers these days want labels that feel transparent. Talking honestly about what goes into flavors and providing more info—even tracing the source of each component—helps build trust. Food scientists keep hunting down new ways to blend nature and science: some labs now use fermentation with yeast to create isoamyl butyrate, which falls closer to a “natural” label than formulas from petrochemicals.
Smell and Connection
Everyone carries a library of scents—childhood sweets, festival treats, fresh fruit just off the tree. Isoamyl butyrate holds a spot in that mental catalog for so many people. For me, walking past a taffy stand or unwrapping a piece of banana-flavored gum pulls up a flood of memories, all tied to this single compound. The right smell brings us back to birthdays, summer fairs, and sticky fingers coated in sugar. Sharing why something makes us feel at home adds meaning, even if the source started life in a beaker instead of a field.
How is Isoamyl Butyrate produced?
The Chemistry Behind Isoamyl Butyrate
Isoamyl butyrate gives off that enjoyable, fruity scent you might associate with pears or bananas. It belongs to the family of esters, a group that’s found all over nature and helps define the flavors and aromas in food and beverages. The story of how this compound comes together starts with a reaction between isoamyl alcohol and butyric acid. In the presence of an acid catalyst, they combine to form isoamyl butyrate and water. The reaction runs in big glass vessels or sturdy stainless steel tanks, heating the mixture for hours to ensure the best possible yield.
Why Process Control Matters
The ratio of raw materials and the precise control over temperature and timing decide how clean the product turns out. Poorly handled reactions mean leftover alcohol, acid, or odd-smelling byproducts. Factories use continuous stirring and monitor acidity, chasing that sweet spot where product comes out pure and simple. Filtering and distilling further helps, separating the isoamyl butyrate from everything else. From experience, I’ve seen how neglect in any of these areas leads to wasted time and product loss—something far more common than most assume in smaller set-ups.
Sourcing and Sustainability Concerns
Butyric acid sometimes comes from natural fermentation: think of how cheese gets its bite. Isoamyl alcohol can come from fermentation or as a byproduct of the petrochemical industry. Many consumers prefer ingredients from plants or natural fermentation. The flavor industry has seen a big shift in the last decade, with more companies moving away from petroleum-based chemicals. Green chemistry approaches make a difference. Enzyme-catalyzed synthesis cuts back on hazardous waste and energy use. Fermentation-based production taps into renewable raw materials, resulting in fewer byproduct headaches and less environmental harm.
Quality and Human Safety
Every batch destined for food or perfume has to clear tests for purity. High levels of leftover butyric acid won’t just mess with the odor—they can trigger skin or eye irritation. Too much isoamyl alcohol risks unpleasant notes and allergic responses. The industry uses gas chromatography and other sharp analytical tools to verify that every drop fits the required standards. These checks don’t just tick regulatory boxes; they keep trust with chefs, perfumers, and consumers who want safe and reliable ingredients on the label.
Health and Environmental Considerations
Isoamyl butyrate itself breaks down easily in the environment. It evaporates into the air and gets chewed up by sunlight and microbes. Wastewater from manufacturing does need attention, as traces of acid or alcohol can harm aquatic life. The most responsible producers reuse solvents and treat effluent, reducing the overall impact. My work with small-scale producers showed that even modest investment in waste capture and solvent recycling pays off both in local environmental quality and business costs.
Pushing for Greener and Safer Production
Innovation keeps changing this industry. More companies explore bio-catalytic routes and low-waste processes. Sharing best practices, investing in process upgrades, and keeping an honest line to the public all build a stronger sector. For businesses, cutting corners often backfires in quality or safety issues. For consumers, supporting brands that value sustainable practices matters. In today’s world, clean chemistry and clear labeling set the standard for how ingredients, such as isoamyl butyrate, ought to be produced.
Can Isoamyl Butyrate be used in cosmetics or perfumes?
The Appeal of Isoamyl Butyrate
Isoamyl butyrate brings a fresh, fruity smell that sits somewhere between banana and pear. This isn’t some obscure compound only chemists appreciate. You can sniff it out in fruity drinks or candy. But many in the beauty industry want to know: Does isoamyl butyrate belong in bottles of perfume or in the colorful world of cosmetics?
A Breath of Freshness for Fragrance
Pick up a bottle of niche perfume or a mainstream body spray and read the ingredients — it’s rare to see every aromatic compound spelled out, especially when it comes to those naturally found in fruit. Isoamyl butyrate’s scent profile pairs nicely with blends that chase a light, tropical feel. Testing on skin reveals how it mingles with other notes: it brings sparkle at the top, then fades into a subtle sweetness. Synthetic perfumery seeks these kinds of building blocks for affordable and safe scents because harvesting enough fruit extracts to supply a whole market proves unreliable and costly.
Safety Sits at the Core
Any ingredient used on skin spends time under the spotlight. Bodies like the European Chemicals Agency and the International Fragrance Association keep this sector from running wild, analyzing old studies and demanding new data for chemicals like isoamyl butyrate. Current research suggests it rarely triggers skin irritation or allergic reactions when properly diluted. The body naturally encounters esters like this in fruit and in daily food flavorings, and the doses involved in fragrances sit at the low end compared to what’s eaten. Still, cosmetic chemists never just throw something into a bottle because it smells nice — they run patch tests, stability checks, and observe how it wears over time.
Masters of Mixing: Formulation Know-How
Formulators like to keep their ingredient lists both effective and understandable. Adding isoamyl butyrate means weighing fruitiness against how the compound might react with emulsifiers, preservatives, or colorants already in the formula. Not every fragrance ingredient stays stable under sunlight or in contact with other common cosmetic materials. I’ve watched perfumers experiment in labs with a drop here or there, tweaking until the result is safe, stable, and appealing. There’s an art to balancing the scent’s lift without veering into artificial or overpowering territory.
Environmental and Ethical Questions
Part of the conversation around any cosmetic ingredient now circles back to how it gets made. Isoamyl butyrate can come from renewable sources by fermenting plant-based alcohols. Synthetically, it’s made in labs, but processes have improved, reducing waste and stepping up purity. Consumers want transparency. They read labels, research where ingredients come from, and ask about possible allergens or residues, pushing the industry to favor green chemistry approaches and full disclosure.
Looking Ahead: Solutions and Trends
For brands that crave a signature fruity note without heavy florals, isoamyl butyrate offers a clean, sweet aroma that supports the trend for playful and personalized fragrance experiences. Eco-conscious production and comprehensive safety reviews build trust, making this ingredient one that both formulators and end-users can appreciate. Blending nature and science gives us products that are safe, fun, and honest about what they deliver.
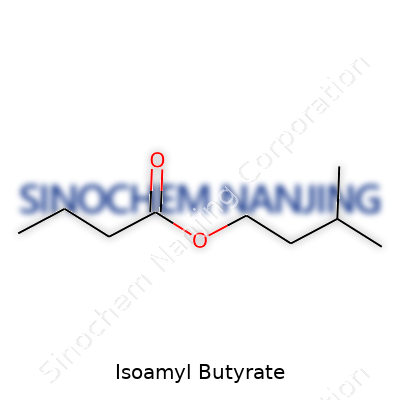
| Names | |
| Preferred IUPAC name | 3-methylbutyl butanoate |
| Other names |
Isoamyl butanoate Isopentyl butyrate |
| Pronunciation | /ˌaɪ.soʊˈæm.ɪl ˈbjuː.tɪ.reɪt/ |
| Identifiers | |
| CAS Number | 2035-66-7 |
| 3D model (JSmol) | `Isoamyl Butyrate: "C1COC(C)CC(=O)OCC(C)C"` |
| Beilstein Reference | 1368737 |
| ChEBI | CHEBI:8871 |
| ChEMBL | CHEBI:88570 |
| ChemSpider | 7711 |
| DrugBank | DB14156 |
| ECHA InfoCard | 03e3c6e7-6cf4-462e-ba85-dc75c1225d7a |
| EC Number | 203-729-6 |
| Gmelin Reference | 7958 |
| KEGG | C12543 |
| MeSH | D02.241.081.700.107.676 |
| PubChem CID | 31253 |
| RTECS number | EN2060000 |
| UNII | 1IS1VHN1NC |
| UN number | UN3272 |
| CompTox Dashboard (EPA) | DTXSID2021734 |
| Properties | |
| Chemical formula | C9H18O2 |
| Molar mass | 130.19 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | fruity, banana-like |
| Density | 0.867 g/cm³ |
| Solubility in water | insoluble |
| log P | 2.82 |
| Vapor pressure | 0.2 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 16 (for the ester alpha-hydrogen) |
| Magnetic susceptibility (χ) | -8.68×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4100 |
| Viscosity | 2.393 mPa·s |
| Dipole moment | 2.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 354.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -516.05 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4462.8 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements: P210, P233, P280, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | NFPA 704: 1-2-0 |
| Flash point | 71 °C |
| Autoignition temperature | 410 °C |
| Explosive limits | Explosive limits: 0.87–6.4% |
| Lethal dose or concentration | LD50 (oral, rat): 16,600 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Isoamyl Butyrate: "16 g/kg (oral, rat) |
| NIOSH | NIOSH: EC9275000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 mg/L |
| Related compounds | |
| Related compounds |
Isoamyl acetate Butyl butyrate Ethyl butyrate Pentyl butyrate Isoamyl alcohol Butyric acid |

