Aluminum Trichloride: More Than Just a Laboratory Staple
Historical Development
People have worked with aluminum trichloride for quite a while, reaching back to the days when chemistry mostly meant hands-on experiment and plenty of patience. Through the late 19th and early 20th centuries, this compound threw its weight around in new directions, especially as the understanding of Lewis acids took shape. Rather than being an obscure laboratory curiosity, it powered a leap forward in synthesizing dyes and perfumes. The material owes much of its early fame to the Friedel–Crafts reaction. Without this crystalline yellow—or sometimes white—substance, key organic syntheses would have slowed to a crawl. Researchers found that harnessing the reactivity of aluminum trichloride meant unlocking whole new branches of industrial chemistry. Its place in the chemist’s toolkit grew as spillover from those discoveries rolled into commercial and academic labs alike.
Product Overview, Physical & Chemical Properties
The features of aluminum trichloride jump out immediately. Sitting as a yellowish-white solid at room temperature, it quickly releases fumes if the air’s even a bit humid. The stuff reacts with water in a way that grabs your attention—steam rushes out, and as hydrogen chloride gas escapes, no one forgets the acrid bite in their nose. Its melting point hovers around 190°C, but if left out, it doesn’t hang around solid for long, shuffling off into the air as it sublimes. You don’t want to let this substance escape its bottle, because once it reacts with moisture, you get a stinging cloud no one welcomes. Chemists know to use well-sealed glassware and keep it dry.
Technical Specifications & Labeling
Most bottles carry a sharp warning label. Anyone handling it pays attention to the purity, since trace moisture or impurities kick off side reactions nobody welcomes. Proper hazard labeling appears clear and unmistakable—skull and crossbones, corrosive pictograms, and those bold R- and S-phrases set the tone. In laboratory stockrooms, this bottle always sits in secondary containment, double-checked for leaks. This isn’t something you toss onto a shelf and forget about. Workers require gloves, eye protection, and training before popping open a seal. Some might feel tempted to treat it like table salt—it looks similar if you’re not thinking—but a whiff or a splash can do permanent damage if you aren’t careful.
Preparation Method
Chemists usually make aluminum trichloride by passing dry chlorine gas over hot aluminum metal. The reaction kicks off rapidly and, once started, keeps rolling until the aluminum turns into a cloud of energized yellow crystals. It’s an exothermic rush, and the setup needs to channel off fumes efficiently. Large-scale manufacturers set up sealed reactors loaded with aluminum chips and feed in streams of chlorine. The product collects as it cools, forming solid clumps or loose powder, ready to pack away in air-tight containers. Some laboratories might turn to the reaction between hydrogen chloride and aluminum oxide, but the classic method—aluminum and chlorine—remains the go-to for scale and purity.
Chemical Reactions & Modifications
Aluminum trichloride stands out in the world of Lewis acids. In organic chemistry, it pulls electrons from other molecules, priming them for all manner of substitutions and rearrangements. In Friedel–Crafts alkylation and acylation, it sits front and center, taking raw benzene and turning it into a massive library of aromatic compounds. This compound can flip between its solid, dimeric, and vapor forms, each one with its own quirks. Add water, and it hydrolyzes in a flash, giving off hydrogen chloride and forming aluminum hydroxide. Push it through more challenging reactions, and you see it serve as a catalyst in making pharmaceuticals, flavorings, and polymers. Over time, researchers found ways to tweak its reactivity: pairing with other halides, swapping solvents, changing temperatures. The compound opened up doors to customizing both its reactions and those of other chemicals sent through its sphere of influence.
Synonyms & Product Names
The chemistry world doesn’t always settle on just one name. Aluminum trichloride pops up in papers and catalogs as AlCl3, trichloroaluminum, and sometimes just “aluminum chloride”—though the “trichloride” version helps avoid any confusion with its lesser-known chloride cousins. Most science-minded people stick with AlCl3 for shorthand, but crossing over into industry, one starts to see trade names and proprietary blends, some tweaked with stabilizers or purity levels dialed for specific niche uses.
Safety & Operational Standards
Nobody should downplay the safety issues with aluminum trichloride. One careless moment, one snap of a damp glove, and you’re facing burns or worse. Direct contact stings and blisters skin, while accidental inhalation strips moisture from airways, burning as it goes. Regulatory agencies such as OSHA and the EU’s REACH register place this compound on their lists requiring strict exposure limits, personal protective equipment, and venting setups for any workspace. In my own experience working around AlCl3, there’s no room for shortcuts. The labs that handle it well use gloveboxes, strong hoods, and clear protocols. Waste gets neutralized, sealed, and tagged for specialist disposal. Routine audits and safety training matter just as much as the science—one slip-up changes lives.
Application Area
Aluminum trichloride earns its keep across more than just academic chemistry. It forms the backbone of big reactions for dyes, perfumes, and flavors. Many plastics—especially polystyrene—get their start with this compound as a catalyst, making it foundational for everyday items from insulation to packaging. It’s crucial in making ethylbenzene, used in styrene and synthetic rubbers. The pharmaceutical industry leans on its chemistry to build complex drugs and intermediates. Even water treatment relies on aluminum compounds derived from this reaction base. Early in my career, I saw huge production lines for detergents and cosmetics depend on careful use of AlCl3 to tweak fragrance and dye molecules. It’s invisible to most consumers, but factory engineers keep a sharp eye on supplies, quality, and waste treatment for this essential ingredient.
Research & Development
In R&D circles, aluminum trichloride stands at the crossroads of tradition and innovation. Chemists have wrung generations of reactions out of those yellow crystals but refuse to rest. Recent work chases alternatives that mimic its strong-acid action without the drawbacks—less toxicity, safer handling, wider green credentials. There’s real interest in moving toward catalytic cycles that recycle the reagent or use milder analogs based on iron or less volatile halides. Computational chemists model ways to harness its electron demands for new syntheses, while process engineers try to build reactors that keep any exposure to workers or the atmosphere near zero. Getting more productivity from smaller loads and making every gram count sits at the beating heart of modern industrial chemistry. Calls for “greener” routes—made safer and still cost-competitive—push R&D budgets toward new synthesis strategies every year.
Toxicity Research
Few in the chemical industry take toxicity research for granted. Repeated studies on exposure risk keep shaping workplace policy and medical advice. Skin burns, severe eye damage, lung scarring—these aren’t hypothetical risks. Decades of published research flag the damage from even short, intense exposure. No one wants to relive those lessons learned the hard way on factory floors or in poorly-ventilated labs. Further down the line, toxicologists study breakdown products and residues in wastewater. They look for chronic hazards and environmental fallout. Any evidence for long-term problems drives reform, as community advocates and regulatory agencies demand demonstrated safety, not just claims. The answer isn’t to avoid these chemicals altogether, but to keep raising the bar on protections.
Future Prospects
Looking ahead, the future of aluminum trichloride points toward automation, safer substitutes, and more closed-system chemistry. Robotics and remotely-operated reactors could take people further away from direct exposure. Market trends keep shifting toward stricter limits for environmental discharge, with every factory line and academic lab watching costs alongside compliance. Some chemical makers explore variants that give similar reactivity with reduced volatility and lower toxicity. You can spot real momentum behind academic–industry partnerships searching for “drop-in” replacements or new catalysis strategies. The push for green chemistry isn’t a passing trend—it’s only picking up speed, with AlCl3 sitting on the frontline as both problem and opportunity. For those of us close to industrial and research labs, the drive feels immediate. Better containment, smarter process design, robust worker training, and ongoing monitoring become the next natural steps.
What is Aluminum Trichloride used for?
What Exactly Does Aluminum Trichloride Do?
Aluminum trichloride doesn’t show up in everyday headlines, but take a good look at nearly any touchpoint in the chemical world, and you’ll find it working hard in the background. It plays a key role where the stakes are high: in everything from making the plastics that wrap food to the medicines taken for granted. For anyone who spends time around chemical plants or in a research lab, the sharp, earthy scent of “AlCl3” means business.
The Cornerstone of the Friedel-Crafts Reaction
Chemists have long leaned on aluminum trichloride for what’s called the Friedel-Crafts reaction. That’s a reaction used to stitch carbon atoms together to make all sorts of things, including dyes, prescription drugs, and the kinds of fragrances found in soaps and colognes. I remember my university days, learning how a handful of white powder (this very compound) could turn a bland starting material into something colorful or useful, given the right mix of heat and patience. Without this process, many of the chemicals that frame modern life would be a lot tougher to make.
More Than Just Chemistry
Take a walk through any major factory making aluminum metal, and you’ll see aluminum trichloride at work again. During the production of aluminum, workers use it to help refine the ore. Here, efficiency isn’t just about saving a buck, either — it’s about reducing waste and lowering the burden on the environment. Years in manufacturing have shown me that even a small step forward in metal refining makes a huge ripple all the way down the supply chain, affecting both energy use and environmental stewardship.
Catalyst for Everyday Essentials
Move past heavy industry and you’ll find aluminum trichloride showing up in the production of antiperspirants. The antiperspirant products on store shelves rely on aluminum compounds to stop sweat, keeping folks feeling clean and confident. I’ve spoken with product developers who point to consistency, reliability, and purity as critical factors. Aluminum compounds, created with processes involving aluminum trichloride, tick all those boxes.
Risks and How People Handle Them
Job safety remains a priority with any chemical, and this one calls for respect. Aluminum trichloride reacts strongly with water and can release hydrochloric acid fumes. Nobody in a lab will forget what happens if it’s handled without eye protection or if someone gets careless. Regulations require clear lab training, good ventilation, and the right protective gear. Experience tells me that those guardrails reduce accidents. Still, enforcement needs attention, particularly in places where corners get cut under pressure.
Moving Toward Greener Chemistry
Right now, researchers and industry leaders are looking toward safer, cleaner catalysts to fill some of the roles aluminum trichloride handles. Some processes try to recycle or recover it, so less waste finds its way into the environment. Others aim to swap it out for less reactive chemicals in certain applications, chipping away at the health risks and keeping things sustainable. It’s a complicated puzzle, but progress shows up as new safety procedures and innovations make their way into standard practice.
Everyday Impacts
Most consumers never see aluminum trichloride. Yet its reach stretches into medicine cabinets, kitchen drawers, and even the cars parked outside. Industry insiders see room for improvement in both its usage and in the training people receive around it. Lessons learned from both successes and mishaps push the field forward, making products safer while looking out for the people and the planet behind every process.
Is Aluminum Trichloride hazardous or toxic?
What Happens When You Work with Aluminum Trichloride
Aluminum trichloride draws attention in chemistry labs and industries. White, easily fuming, and quick to react with water or air, this compound possesses properties that raise safety flags for both small-scale and industrial users. After years in research settings and training new lab workers, I learned quickly to treat aluminum trichloride with plenty of respect. Young lab techs sometimes underestimate the risks, thinking “clear and simple” means “safe and easy.” That’s a mistake.
Direct Health Concerns
Exposing skin or eyes to even small amounts of aluminum trichloride causes serious pain and damage. This stuff reacts aggressively with water, even the moisture in your skin. Any spill or splash makes burning, itching, and persistent chemical irritation likely. Inhaling the fumes brings on coughing fits and choking; longer exposures may leave people wheezing for days. In heavy doses, animal studies pin this compound as a potential lung corrosive.
Contact with body moisture rapidly turns aluminum trichloride into hydrochloric acid and aluminum hydroxide. These products drive up the toxic risk. Burns from the acid take weeks to heal. Worst cases documented involve blindness and permanent scarring. Even handling the container calls for full gloves, goggles or a face shield, and work inside a fume hood.
Risks in Everyday Environments
Outside the lab, most folks never cross paths with aluminum trichloride itself. It’s too unstable to sit in many households or workplaces. Instead, its biggest role sits in large-scale chemical industries, especially making aluminum metal and certain drugs. Still, accidents from transportation or plant leaks impact air and water quality nearby. In my experience living near a chemical corridor years ago, runoff after spills brought acrid smells drifting for blocks. That air forced residents inside for hours from just a small leak.
Environmental Impact
Spilling aluminum trichloride into water threatens aquatic life fast, since it dumps hydrochloric acid as it breaks down. Fish, insects, and plants die off quickly. This chemical does not linger for months, but its immediate impact wipes out stream communities before dissipating. Runoff also leaches aluminum salts into soil, creating new trouble for crops and wild habitats.
Regulations and Worker Safety
Manufacturers keep aluminum trichloride behind strict regulations. The Occupational Safety and Health Administration designates exposure limits and calls for special training. Stores and shippers put the chemical in double-sealed containers with hazard labels. High school teachers give clear demonstrations in video form; universities supervise any lab use closely. For people who build chemical facilities or drive tankers, handling spills means carrying neutralizing compounds and knowing first aid.
Practical Solutions
Substitute materials play a role in cutting risk. Some companies shift to less volatile compounds where possible or redesign processes for closed-system containment. Training fresh workers about the chemical’s dangers makes a difference. I’ve seen companies run full-scale spill drills, mixing practical steps with real information on past accidents. Quick action with neutralizing agents and rapid evacuation drills turn a dangerous release into a controlled cleanup.
Aluminum trichloride holds important uses in chemistry and industry, but its health and safety hazards demand full attention. With careful planning, education, and safer alternatives, the risks shrink for everyone involved.
How should Aluminum Trichloride be stored?
Looking Out for Your Team and Facility
Aluminum trichloride doesn’t win any popularity contests in a stockroom. During my years working in laboratories, I saw firsthand the havoc it can cause if someone grew careless. This isn’t the type of chemical you just toss in the corner after your shift. Its tendency to react with water and moist air means a single oversight can fill a room with toxic fumes or spark a corrosive mess nobody wants to clean up.
Keeping Moisture Away
I often tell new technicians the most important lesson: keep it bone dry. Aluminum trichloride grabs onto water at the slightest chance, releasing hydrochloric acid gas. That gas burns your lungs and eyes. I still remember a day a bottle cap wasn’t tightened, and the whiff that hit me nearly sent me running. Glass or plastic containers with tight-fitting lids keep leaks and accidents in check. I’ve always checked for cracks or old seals before stowing containers. No shortcuts turned into an unspoken rule in our chemical cabinets.
Controlling Temperature Makes a Difference
Temperature swings invite trouble. Leaving aluminum trichloride near pipes or heaters creates extra risk. I’d never put it anywhere above room temperature. Too warm, and it can build pressure inside its bottle or even start reacting with vapor in the air. Our storage space sat well ventilated, far from direct sunlight, and the thermostat hovered around 20°C. That kept everyone safer, not just the chemists with their name on the logbook.
Choosing the Right Spot
I can’t stress the importance of isolation. Storing aluminum trichloride in a cabinet with water-based solutions or acids spells disaster. The thought of an accidental spill or a bottle getting knocked from a shelf would keep me up at night. Designated corrosive cabinets, ideally lined with non-reactive materials, are the way to go. I made a point to separate it from any possible reactants, especially anything that sat open or released vapor.
Protecting Your Health—And the Law
Safety rules for chemicals like this come from real spills and real injuries. Accidents land people in hospitals, and most countries carry strict storage guidelines for good reason. Eye wash stations, gloves, and good ventilation aren’t just recommendations—these practices stop trips to the emergency room. Keeping a clear logbook of every transfer or storage location helped our lab pass every inspection and made emergencies easier to handle.
Solutions Worth Considering
Substituting less hazardous compounds isn’t always possible in industry, but regular staff training on those worst-case scenarios can be the difference between a bad day and a disaster. Proper signage—bright, readable, and not just a faded sticker—lets everyone know what they’re dealing with. If your facility goes through a lot of aluminum trichloride, investing in sealed transfer systems and personal gas monitors can catch small problems before they become big ones. A routine audit of storage practices, not just paperwork, brings peace of mind and helps spot issues early.
What is the chemical formula of Aluminum Trichloride?
Everyday Chemistry: More Than Letters and Numbers
Plenty of folks have glanced at a bottle in a lab or read a product label and seen something like AlCl3 staring back. That combination—one aluminum atom and three chlorine atoms—builds the foundation for aluminum trichloride. Learning the formula is more than school trivia. You find it woven through real work, from water treatment to making medicines to shaping products that fill kitchen shelves.
Why AlCl3 Pops Up in Real Life
Aluminum trichloride’s role steps beyond theory. Sitting at the intersection of industry and science, it acts as a strong Lewis acid. That lets it pull double duty: sparking up key reactions for making dyes, detergents, and even perfumes. Chemists reach for it while making medicines—plenty of pain relievers owe their jumpstart to an AlCl3-driven process. This isn’t about rare, obscure knowledge. It’s about the daily gears that turn the world’s engines.
Back in college, I watched a friend fumble with drawing the structural formula on a test. Half the class tripped up the same way. That moment drilled home how skipping the basics turns a small detail into a big roadblock. Even outside classrooms, plant engineers and lab techs use this formula for everything from figuring out how much to mix in a batch, to troubleshooting fouled-up reactions.
From Research to Safety: What People Miss About AlCl3
The formula packs a message about handling and risk. AlCl3 doesn’t love water. Toss it into a damp beaker, and you'll see fumes and heat. Most accident reports involving this compound link to underestimating its reactivity. Picking up the facts—what’s inside the bottle, what can go wrong—brings a clearer sense of responsibility. OSHA’s guidelines stress that anyone working with AlCl3 keeps water away and wears solid protective gear. Missteps rarely forgive.
Understanding formulas helps make chemistry safer. The EPA and other agencies track how aluminum compounds interact with air and water. The formula’s simplicity lets regulators model pollution and set far better safety guidelines for storage or transport. Without clarity, accidents or leaks get harder to contain and trace.
How to Build Chemical Know-How
Killing confusion around chemical formulas starts early. Teaching kids the rules of valence—the basics of combining elements—sparks bigger questions: “How does this behave in water?” or “Why does it matter for recycling?” Sliding a lesson about AlCl3 into a science fair project or a classroom demo can close the gap between symbols and real-world impact.
Industry often leans on digital tools today, mixing up compounds with software guidance and automation. Still, people check formulas by hand, especially if something goes sideways in the plant. Back when I shadowed a chemical engineer, she caught a shipping mistake because she remembered what AlCl3 should look like, not just as a formula but as a pale yellow powder, sometimes forming “chunks” if exposed to air. Details like that stem from a grounded understanding of what the shorthand really means.
Building Toward Solutions That Stick
Part of building trust in chemistry comes from openness. Companies and research teams can publish clearer, easier-to-access data sheets. Material labels and safety training should link formulas with pictures, warnings, or quick facts. Demonstrations and videos help shine a light on how each formula, like AlCl3, can affect everything from the cost of a product to its effect on the planet. In the end, seeing beyond the formula—tying it to real risks and real benefits—encourages a culture where everyone, not just scientists, can ask better questions and make safer choices.
What are the safety precautions when handling Aluminum Trichloride?
Understanding the Risks
Aluminum trichloride sits among reagents that can make or break a day in the lab. It comes as a yellowish solid that quickly reacts with moisture, stubbornly releasing hydrogen chloride gas wherever it finds a tiny drop of water, including the air around us. This isn’t a gentle fume; it stings the nose and eyes, and even a low dose can make breathing tough. Direct skin contact with the solid or solutions leads to nasty burns—no small irritation, but real injuries that take time to heal. These facts alone raise the stakes for anyone moving, mixing, or storing this chemical.
PPE Isn’t Optional—It’s Non-Negotiable
In my own time maintaining university equipment, I saw what happens when gloves or goggles stay on the shelf. Cases of chemical splash injuries don’t make for easy stories. To work safely, start with the basics: splash goggles with side shields and a reliable lab coat. Choose gloves made from neoprene or nitrile—latex doesn’t stand up to this chemical’s hunger for destruction. Protect skin fully; regular cotton sleeves and lightweight aprons buckle under the corrosive weight of this stuff. Always finish with closed shoes—no sandals, no excuses. Every forgotten piece of equipment becomes a gap in your armor.
Keep Air Clean and Reactions Contained
There are no shortcuts to good ventilation. Fume hoods matter because they catch the sharp, acidic vapor that forms the moment the container cracks open. Years ago, I watched a careless transfer outside a hood fill a whole room with a thick, choking cloud. There’s no heroism in toughing it out—get the reaction under control inside the fume hood, always. Store all containers in a dry spot, far from sinks, emergency eyewash stations, or benchtops known for spills and splashes. Any hint of a leak, and the room will know in minutes.
Emergency Equipment: Use It or Regret It
I was once present during a spill where a rapid response stopped small panic turning into big regret. Keep well-marked eyewash stations and safety showers close to working and storage areas—no one wants a frantic search in the burning moment of exposure. Neutralizing agents, such as sodium bicarbonate, belong within arm’s reach. Water should never meet dry aluminum trichloride directly; controlled neutralization with something safer protects both people and plumbing.
Solid Storage and Team Training
Label every bottle clearly, and don’t trust memory alone. Never recycle containers for other chemicals, and don’t let containers pile up unorganized. I’ve seen confusion here lead to close calls. The chemical sits fine in a glass or polyethylene container with a snug, undamaged seal. Every member of the team should get straightforward training, straight talk about real risks, not just handed sheets of procedures. Even veterans slip up—the best routine is regular reminders and honest stories of what happens when routine breaks down.
Disposal Means Thinking Ahead
After tasks are finished, waste gets the same respect as fresh reagent. Don’t tip leftovers down the drain—even traces will rip through pipes and turn into toxic vapor. Gather all cleanup material and remnants for hazardous waste pickup. If you share a lab, triple check disposal protocols. Responsible handling doesn’t just keep your workspace safe—it protects colleagues further down the line whom you may never meet.
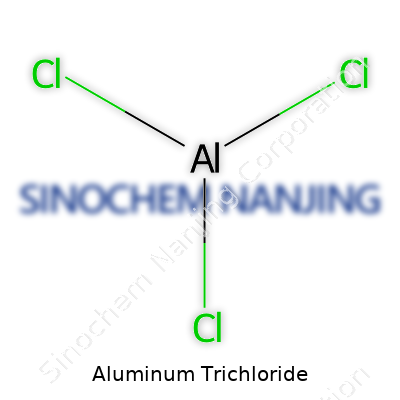
| Names | |
| Preferred IUPAC name | trichloroalumane |
| Other names |
Aluminum chloride Aluminium chloride Trichloroaluminum AlCl3 |
| Pronunciation | /əˌluː.mɪ.nəm traɪˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 7446-70-0 |
| Beilstein Reference | 3587261 |
| ChEBI | CHEBI:30189 |
| ChEMBL | CHEMBL1200897 |
| ChemSpider | 21527 |
| DrugBank | DB14784 |
| ECHA InfoCard | ECHA InfoCard: 100.028.807 |
| EC Number | 231-208-1 |
| Gmelin Reference | 844 |
| KEGG | C07065 |
| MeSH | D000693 |
| PubChem CID | 24012 |
| RTECS number | BO0875000 |
| UNII | F5P3YP1VG1 |
| UN number | UN1726 |
| Properties | |
| Chemical formula | AlCl3 |
| Molar mass | 133.34 g/mol |
| Appearance | White or yellowish solid |
| Odor | Pungent |
| Density | 2.44 g/cm³ |
| Solubility in water | reacts with water |
| log P | -1.7 |
| Vapor pressure | 1 mmHg (100°C) |
| Acidity (pKa) | -5 |
| Basicity (pKb) | -4 |
| Magnetic susceptibility (χ) | +38.5·10^-6 |
| Refractive index (nD) | 1.398 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 109.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -704.2 kJ/mol |
| Pharmacology | |
| ATC code | A01AB03 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS05, GHS07, GHS08 |
| Signal word | Danger |
| Hazard statements | H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P234, P260, P264, P280, P301+P330+P331, P303+P361+P353, P305+P351+P338, P310, P304+P340, P308+P311, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-W |
| Autoignition temperature | 130°C (266°F) |
| Lethal dose or concentration | LD50 oral rat 380 mg/kg |
| LD50 (median dose) | LC50 inhalation rat 20,000 mg/m³ 2 h |
| NIOSH | GM4725000 |
| PEL (Permissible) | 2 mg/m3 |
| REL (Recommended) | 25 mg/m3 |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Gallium trichloride Indium trichloride Aluminum tribromide Aluminum trifluoride Aluminum triiodide |

