Aluminum Sulfate: Unearthing Its History, Uses, Science, and What Comes Next
Looking Back: The Story of Aluminum Sulfate
Aluminum sulfate didn’t earn its place in industry overnight. Turning the clock back to the dawn of large-scale municipal water projects, this chemical was already finding work as a valuable coagulant. Evidence from the late nineteenth century points to its adoption in the growing field of city water purification, with early plant operators discovering it made muddy, particle-filled river water clearer and less prone to foul taste or odor. This early role really set the stage for its global spread. In my own academic years, water treatment always featured aluminum compounds as foundational tools—every chemistry lab seemed to keep at least a jar or two. Over decades, paper makers figured out it could help paper fibers stick together, producing smoother, higher-quality sheets. Textile workers found value in its dye-fixation capabilities. Such repeated lessons from the past remind me that yesterday’s experiment often shapes tomorrow’s staple.
What Is Aluminum Sulfate and How Is It Recognized?
Sometimes labeled “alum,” this compound carries a reputation that extends far and wide. Commercial documents might call it papermakers’ alum or filter alum. I’ve found it in unassuming bags, crystalline and almost slippery between my fingers, not looking like much at all—but don’t let the looks fool you. Marketed powders range from pure white to an icy blue tinge, a clue about source minerals or minor impurities. Its molecular formula usually shows up as Al2(SO4)3, and even though synonyms pop up depending on the market, the core chemistry stays constant. The product labels can vary by region, which can puzzle buyers—what you get in one country might carry a whole set of alternative names elsewhere.
Looking Inside: Aluminum Sulfate’s Properties
Aluminum sulfate isn’t flashy, but its properties speak volumes about its utility. It’s one of those rare minerals that dissolves very easily in water, even in cool conditions. In my own work, I’ve seen how it reacts with chalky minerals to form a cloudy solution, a telltale sign when testing unknown powders. Its melting point sits comfortably above standard gasoline engine temperatures, so you won’t see it liquefy except in a blast furnace or kiln. Acidity plays a big role here: drop it into water and the solution quickly turns slightly acidic, a hint at why it makes water treatment work so well—charged particles come out of hiding, grab hold of dirt and bacteria, and pull them into removable clumps. This trick, used by engineers for years, helps safeguard what many take for granted—a glass of safe, clean drinking water.
Technical Specifications and Labeling in the Real World
Technical documents for aluminum sulfate can run long, but real-life use comes down to a handful of practical figures. I’ve sorted hundreds of vendor samples, and all the spec sheets, whether from a major supplier or a regional distributor, demand crystal clarity about a few things: active aluminum content, presence of heavy metals, and the degree of hydration. These specifications matter far more than fancy jargon. It’s not about mystifying statistics—it’s about making sure users don’t accidentally dose the wrong levels into a water system, dyebath, or papermaking process. Labels on bags and barrels highlight safety, warn about accidental dust inhalation, and often flag whether the batch passed current regulations, especially in the European Union or North America. Sometimes, small differences in hydration directly affect how users dissolve or measure it. Getting this wrong can mean uneven batches, lost time, or in the worst case, damaging a city’s water filters or a factory’s delicate machinery.
How Aluminum Sulfate Gets Made
The manufacture of aluminum sulfate doesn’t rely on secret sauce so much as on sturdy chemical processes. The core process, which I’ve seen replicated in plant after plant, involves reacting aluminum hydroxide or aluminum oxide with concentrated sulfuric acid. You watch as the solid material fizzes, giving off heat and making a thick, syrupy solution. Factories cool this hot mixture and then evaporate water to yield crystals. Some operations tap into natural bauxite ores, running these through the same acid reaction. Technological advances now make it possible to fine-tune purity at each stage. These improvements ensure fewer impurities—especially important for water treatment, where just a trace of iron or manganese can cause color or taste issues. The science sounds simple, but handling concentrated acid and high temperatures calls for sharp attention and steady hands, not just automation.
Aluminum Sulfate in Chemical Reactions and as a Base for Modifications
What really excites chemists and industrial users about aluminum sulfate is how it opens doors to other reactions. Add it to lime, and you’ll notice a white sludge—classic aluminum hydroxide, perfect for further processing or as a precursor for other useful chemicals. Mix it with alkalis, and you’ll see interesting byproducts forming, often used in pigment production and as fillers for rubbers and plastics. I learned early on that modifying aluminum sulfate’s composition, perhaps with iron or other transition metals, tunes it for specialized water treatment problems or rare dyes. Each reaction offers a specific advantage—one batch might be best for tropical aquarium water, another for stubborn industrial waste streams.
What You Call It Depends on Where You Stand
Navigating the world of aluminum sulfate means getting comfortable with aliases. You might be looking for “alum” and find it sold under a local name, or you might need to check if you’re buying “filter alum” or “paper alum”—the product may be almost identical, or subtly different in purity and particle size. In my own travels and professional circles, confusion pops up when someone orders from a foreign supplier and receives product under a less familiar label. This isn’t just a matter of semantics—mislabeling or misunderstanding can mean failed quality checks or regulatory problems downstream. It’s a real reminder that language shapes the practical side of global trade in chemicals.
Staying Safe and Running a Tight Operation
Any industry vet will tell you: safety rules aren’t just legal hurdles but hard-learned lessons from years of factory and fieldwork. Aluminum sulfate comes with specific handling requirements—ignore the dust masks and you might end up coughing through a work shift, or worse, with irritated eyes all day. Regulations push for low levels of impurities, partly to protect human and ecological health. Operators stick to clear signage and training, keeping even seasoned factory workers on their toes. Accidental spills get managed fast to avoid polluting the local water table. Over the years, international standards have tightened, holding importers to account and forcing companies to prove that their product won’t introduce lead, arsenic, or other contaminants into drinking water or consumer goods.
Key Applications: Not Just a Water Cleaner
Talk to an industry chemist, and the first use that comes to mind will probably be water treatment. Walk into any medium-sized municipal plant and you’ll see bags or silos dedicated to coagulant dosing—a daily dance that keeps water drinkable and clear. But that’s only part of the story. The paper industry swears by it, using it to give finished sheets that clean, even look. Textile plants depend on it as a mordant, helping dyes grip fabric fibers more effectively than relying on organic chemicals. I’ve seen landscapers and farmers apply it to acidic soils, aiming to balance conditions after years of nitrogen-heavy fertilizer use. Dyes, pigments, even certain food manufacturing processes make room for this humble salt. Its versatility means that even as our industries change, aluminum sulfate continues to pull its weight.
Labs and New Directions: What Research Uncovers
Modern research throws fresh light on an old compound. Universities study ways to make its production more efficient and less polluting, cutting down acid consumption or using recycled bauxite. Environmental scientists dig into how aluminum compounds move through rivers and soils, trying to make sure that large-scale use won’t tip delicate ecosystems out of balance. Engineers test whether blending it with other coagulants will speed up water purification or let smaller plants treat more water at lower cost. I remember one research project focusing on wastewater from electronics factories—the team found aluminum sulfate could pull out stubborn metallic residues, improving both safety and recovery rates. This kind of applied R&D has a way of bending old uses in new directions, helping industries adapt to tougher pollution rules.
Diving Deeper Into Toxicity and Risk
No chemical can escape scrutiny, and aluminum sulfate is no exception. Toxicity research stretches back decades, driven by fears about aluminum ingestion and chronic exposure. Epidemiological studies from the 1970s and 80s probed possible links between high aluminum intake and neurological disorders, though the science still leaves room for interpretation. More recent analysis zeroes in on occupational hazards, such as inhaled dust in closed processing areas—something I’ve experienced firsthand despite protective gear. Regulators generally allow its use in potable water as long as residual concentrations fall well below established safety thresholds. Scientists agree that acute toxicity is low, but long-term ecological exposure worries persist, especially near discharge outlets. Better monitoring, stricter purity guidelines, and incremental changes in handling protocols all form part of the collective effort to keep this old staple both useful and as safe as possible.
Outlook and Future Prospects: Where Next?
Peering ahead, aluminum sulfate faces new challenges and opportunities alike. Green chemistry movements and tightening regulatory landscapes push suppliers to innovate, seeking cleaner process routes and less energy-intensive manufacturing steps. Municipal water systems look for ways to minimize residuals and chase more selective coagulants, while keeping costs in check. Academic groups model how climate shifts could change seasonal water chemistry, altering how facilities use coagulants year-round. In regions facing the hardest water or the trickiest contamination profiles, newly engineered blends and dose automation are likely to further optimize results. Incremental improvements in purity, packaging, and process integration suggest that aluminum sulfate will keep earning its spot in the industrial toolkit, even as researchers eye alternatives and the chemical world marches forward.
What are the main uses of Aluminum Sulfate?
Water Purification
Safe drinking water is easy to take for granted, but behind the scenes, a lot of chemistry makes it possible. Aluminum sulfate plays a heavy role in cleaning up water. In municipal water plants, it’s poured in to help clump together dirt and particles in raw water. People call this “flocculation.” The process lets big bits settle out, so the water that reaches the tap turns out clear and safe. According to the World Health Organization, access to safe drinking water has been a driving force in public health for decades, and aluminum sulfate stands out as a key ingredient. Without it, turning rivers or reservoirs into tap water would be a tough job.
Pulp and Paper Industry
Paper wouldn’t make it from wood pulp to notebook without quite a bit of chemistry, either. The paper industry has leaned on aluminum sulfate for over a century. Manufacturers add it to help fibers bond and improve stiffness. It also fixes dyes on the paper, producing clearer prints. In my experience, touring a paper plant makes it pretty clear: production lines move fast, and consistency matters. Without some kind of chemical aid, the sheets would turn out blotchy or weak. The U.S. Geological Survey has noted that paper making remains one of the top consumers of aluminum sulfate worldwide. That’s a direct link between chemistry and everyday items on our desks.
Wastewater Treatment
Trying to clean wastewater is a bigger mess than most people realize, and city sewage often carries a stew of phosphates and pollutants. Aluminum sulfate helps remove these unwanted chemicals. Pour it into the system, and it binds with phosphates, letting them settle to the bottom. This fights back against algal blooms, which have shut down beaches and lakes in some communities. The Environmental Protection Agency tracks dozens of water clean-up projects that rely on aluminum sulfate to meet environmental rules year after year. These improvements ripple out, giving towns cleaner rivers and safer recreation.
Other Everyday Uses
People might not see aluminum sulfate mentioned on products, but it crops up in some everyday spots. Gardeners sometimes use it to make hydrangea flowers blue, lowering soil pH with a sprinkle of this compound. Textile makers rely on it as a mordant, fixing dye so that clothes keep their color through wash after wash. In construction, it features in fire-retardant formulations for wood. Companies even put it in animal feed, aiming to cut down ammonia smells in chicken barns.
Moving Toward Better Solutions
Using aluminum sulfate brings real benefits, but it’s smart to ask about downsides. Some researchers worry about links between aluminum exposure and health issues, though current safety standards keep levels well below danger. Waste from aluminum sulfate can build up, making disposal a challenge for water utilities and factories. Scientists experiment with alternatives, like polyaluminum chloride or greener filtration options, aiming to lower chemical footprints. While new methods keep showing up in journals, the old standard sticks around mainly because it balances cost, reliability, and impact. My work in community outreach for water systems taught me that future solutions need to fit not just the science, but budgets and local needs.
Is Aluminum Sulfate safe for drinking water treatment?
Understanding the Use in Water Treatment Plants
Open just about any report on municipal drinking water, and you’ll probably bump into the words “aluminum sulfate.” For decades, this compound, usually called alum, has played a huge role in cleaning up raw water. In water treatment, workers add alum to bind together dirt, particles, and other stuff that clouds up the water. Big clumps form and drop out, letting the plant filter out what people don’t want to drink. The result: clearer, cleaner water.
What’s Actually Getting Into the Water?
Plenty of studies support alum’s usefulness, but what about safety? Some folks worry about aluminum traces left behind in tap water. That concern deserves a closer look. After alum does its thing, water passes through several stages of filtering and settling. So, most of the aluminum settles out and gets removed. Most US and EU tap water winds up with less than 0.1 milligrams per liter, far below limits set by authorities. The World Health Organization suggests up to 0.2 mg/l doesn’t pose a health risk.
I remember chatting with a plant operator after a city switched to a different source—he told me how operators logged alum dosages precisely, always watching to prevent overdosing. They know high alum levels can gunk up pipes and sometimes impact taste. Over many years, the evidence from these steps shows why health authorities feel comfortable approving its use.
Are There Real Health Risks?
People notice headlines about a possible connection between aluminum exposure and diseases like Alzheimer’s. This worry tends to linger. The science here gets complicated. While researchers still explore potential long-term effects of high aluminum exposure, the consensus is that the level coming from treated drinking water is minuscule compared to other sources. Antacids, some foods, cookware—those contribute more aluminum than a glass of tap water.
For young kids or people with kidney problems, some extra caution makes sense. Both the Centers for Disease Control and Prevention and the US Environmental Protection Agency keep tabs on this, revising advice as new research appears. After following several high-profile reviews, regulators worldwide agree: keeping residual aluminum low in tap water means the benefit of clean, safe water far outweighs any possible risk.
What About Alternatives?
Polymers and ferric chloride offer substitutes that some regions use to cut back on aluminum-based treatments. Each one brings its own package of costs, safety checks, and effectiveness. Water utilities sometimes switch processes based on changing source water or technology. Where budgets tighten or routine supplies dry up, switching coagulants isn’t always practical. For now, alum stands out for how well it works and its long safety record.
How Can Utilities Keep Building Trust?
Nobody wants surprises in their drinking water. Keeping customers informed matters more than ever. Publicly available test results and clear language from municipal water teams help cut through confusion and build trust. Local officials should not only follow safety guidelines but also engage communities when questions pop up. Real accountability makes a difference—if something changes, people want the facts and reassurance that people are watching out for their well-being.
Good drinking water saves lives. Alum, when monitored responsibly, continues to serve millions without causing harm. Still, keeping science, transparency, and healthy skepticism at the core of public health policy keeps water safer for everyone.
What is the chemical formula of Aluminum Sulfate?
Why Formula Knowledge Matters in Daily Life
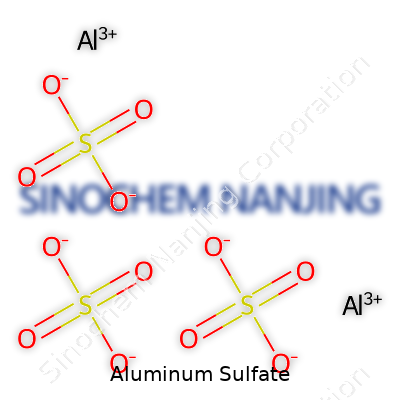
At first glance, “aluminum sulfate” sounds like something tucked away in a high school chemistry textbook or scribbled on a lab worksheet. That’s only part of the story. In reality, understanding the chemical makeup of common compounds shows up practically everywhere—in water treatment, gardening, even simple household fixes. The chemical formula for aluminum sulfate is Al₂(SO₄)₃. “Al two, S-O four three” may roll off the tongue of a chemistry teacher, but it represents a connection between science and real-world problem-solving.
Touching Lives: Where You See Aluminum Sulfate
Tap water owes its clarity to plenty of behind-the-scenes science. Water treatment plants often use aluminum sulfate. This compound pulls tiny particles together so they can be filtered out, cleaning up the water that comes through our faucets. Without a formula like Al₂(SO₄)₃ at play, whole cities might struggle with murky water and unsafe supplies.
Home gardeners know another side to aluminum sulfate. Acid-loving plants, like hydrangeas or blueberries, like a specific soil pH. By sprinkling just the right amount, it’s possible to make blue blooms pop in the yard or coax a slower harvest into something tastier. The same chemical that clears water helps folks enjoy better soil in their backyard.
Breaking Down the Composition
The formula Al₂(SO₄)₃ holds meaning once broken down. Each molecule contains two aluminum atoms and three sulfate groups. The sulfate group itself has one sulfur atom and four oxygens. Every bit of this counts—right down to the subscript numbers setting off predictable chemical reactions. Misreading a single part can lead to plant damage, stubborn stains, or even corroded pipes.
Safety and Environment
With everyday uses, safety becomes important. In large doses, aluminum sulfate can irritate skin or eyes, and mishandling it can hurt aquatic life if stormwater runs off containing extra chemicals. Reports from the Environmental Protection Agency note that following label instructions protects both health and nature. People should always store the material securely and measure carefully, especially around kids or pets.
Proper disposal also matters. Washing large quantities directly into drains can mess with local ecosystems, throwing off the natural balance in streams or lakes. Most community guidelines recommend returning extra material to a hazardous waste facility if there’s any doubt.
Beyond the Lab: Why E-E-A-T Principles Count
Experience builds trust. My curiosity about aluminum sulfate started with watching my dad treat a patchy lawn one spring. He explained the importance of not just picking any white powder from a hardware shelf, but making sure the label matched what the soil needed. That lesson has stuck—science and safety work best when handled with steady hands and clear facts.Alongside personal stories sits the need for reliable information. Trustworthy sources—including EPA reports and university extension guides—lay out how much to use and what cautions to take. Attention to these details makes home projects safer and results better.
Practical Solutions Going Forward
Reading labels, using the compound only for its intended purpose, and checking local guidelines all pay off. Homeowners, farmers, even science students get the best results when they follow expert advice and handle chemicals with respect. For those uncertain about what to do with leftovers, calling the local waste authority offers a safe route. Science isn’t just in books—it’s in every measured scoop and healthy plant, and it always starts with understanding the formula.
How should Aluminum Sulfate be stored and handled?
Understanding Why Storage Matters
Aluminum sulfate pops up in everyday spots—wastewater plants, paper mills, even some water filters. This white, powdery or chunky stuff reacts in contact with water and acids, so keeping it in the right place means fewer problems for workers and fewer headaches for facility managers.
Keep It Dry, Keep It Cool
Moisture and aluminum sulfate do not mix well. A humid storeroom will clump this chemical and, if things get too wet, set off an exothermic reaction. Sweat-heavy summer days in an unventilated shed spell trouble. Shelves made from polyethylene or similar materials won’t rust or react. Metal racks, especially iron, suffer corrosion. No one wants to pay for new shelving each year.
A buddy of mine working in a pool supply warehouse once learned this the hard way. They stored a batch in an old steel container. Within months, chunks welded themselves to the bottom—cleanup took weeks. Shelving made from plastic makes life easier every time someone needs to scoop out a bag for a treatment.
Separate from Incompatibles
Storage near acids, chlorine solutions, or even some organic chemicals risks either spoilage or a burst of fumes. I saw a small facility keep its aluminum sulfate in the same area as hydrochloric acid. Fumes made the air harsh on the lungs. The safest routine places incompatible chemicals with their own kind, away from main walkways or break areas.
The best storerooms go a step further—signs remind workers what’s stored inside and what to do if something spills. Employees who can read the room from twenty feet away have one less thing to remember during a busy shift.
Good Bags, Tight Containers
Aluminum sulfate shipped in strong, double-layered bags or sealed drums prevents spillage and limits moisture. Spills might seem minor, but inhaling the dust or touching it with bare hands can irritate the skin, eyes, or lungs. People often make the mistake of using old sacks that rip easily. Tough, intact packaging saves costs that would otherwise go to lost product or injury claims.
Smaller facilities often pour product into open bins for convenience. This shortcut cuts corners. I’ve watched folks rush to open bins and send up clouds of white dust, filling the air with fine particles. Locking lids and resealing containers every time keep dust down and reduce accidental exposure. A good habit like this helps maintain a clean workspace and keeps everyone healthier.
Protect Yourself Every Time
Gloves, goggles, and masks aren’t just for the lab. Direct contact with aluminum sulfate causes itching and rashes for some, and losing your eyesight in a splash accident could happen in seconds. I’ve never met anyone who said, “I wish I’d skipped my gloves that day.” Safety data sheets sometimes sit ignored on shelves, but a short reminder at the start of a shift—about where these supplies sit—can save a worker from a trip to the clinic.
Don’t Cut Corners with Waste
Used containers and sweepings shouldn’t end up in the regular trash. Two years ago, a small maintenance team dumped the remains of a spill down a storm drain. A few days later, fish turned up dead downstream. Check for local regulations on hazardous waste removal. Work with recognized chemical disposal outfits—they keep you covered and avoid environmental fines.
What are the potential health hazards of Aluminum Sulfate exposure?
What Happens With Direct Contact
My first memory of working around chemicals sticks in my mind—a big warning label on an old, dusty drum of aluminum sulfate in the school custodian’s closet. I didn’t know then, but even a spilled pinch could sting the skin. Most folks using it now wear gloves for good reason. Direct skin contact can leave rashes or burns, especially for those with sensitive skin. If it gets in your eyes, you’re talking about intense irritation, sometimes damage that needs real medical care. No heavy science needed to see why personal protection matters here.
Inhalation Risks in Unventilated Spaces
I once met a pool technician who described the “chalk dust” from aluminum sulfate as something that got everywhere—even in his nose. Breathing in the powder or mist, even a few times, tickles the throat and nose, makes it tough to catch a deep breath, and often sets off coughing spells. People with asthma or a history of allergies can have a much rougher time. Over long periods, constant exposure may lead to ongoing respiratory problems. According to the Centers for Disease Control and Prevention, repeated inhalation sometimes even causes lung and airway problems.
Drinking Water Concerns
Aluminum sulfate shows up in water treatment plants all over the planet. Here’s the catch—while it's meant to settle out impurities, too much in the final drinking water stirs up concerns about aluminum build-up in the body. Some studies suggest a possible connection between high aluminum levels in water and neurological conditions. While definite links to diseases such as Alzheimer’s remain debated, health organizations continue to advise close monitoring of water supplies. So it’s not about panic, but about steady, careful oversight.
Workplace Exposure & Chronic Issues
Anyone working with this chemical day after day faces extra risks. I’ve seen old-school municipal workers cough and rub their eyes after dusty shifts. Chronic exposure, primarily through inhalation, sometimes brings persistent headaches, nausea, and even changes in bone health. The National Institute for Occupational Safety and Health marks aluminum sulfate as a substance requiring proper ventilation and routine air quality checks. Without consistent monitoring and safety habits, workers can end up with long-term health problems that stack up over the years.
Community & Environmental Impact
In communities near factories or facilities storing large amounts of aluminum sulfate, accidental releases can contaminate local water bodies and soil. I’ve lived in a town where a chemical spill led to warnings not to fish or swim for weeks. High concentrations can poison aquatic life and disrupt entire ecosystems. For residents, regular water quality testing and fast response plans from city agencies can reduce this risk. People expect real accountability, not just apologies, if something goes sideways.
Practical Ways Forward
Basic safety starts with protective gear: gloves, goggles, and masks go a long way. Companies need to teach proper handling and enforce good ventilation in workspaces. For households, keeping cleaning chemicals and pool supplies, including aluminum sulfate, locked away from kids beats any warning label. Regulators owe the public clear limits for drinking water and strong oversight of industrial practices. Science keeps moving, so water quality checks and health guidelines will need to keep up.
| Names | |
| Preferred IUPAC name | dialuminium trisulfate |
| Other names |
Alum Dialuminum sulfate Aluminium sulphate Al2(SO4)3 |
| Pronunciation | /əˈluː.mɪ.nəm ˈsʌl.feɪt/ |
| Identifiers | |
| CAS Number | 10043-01-3 |
| 3D model (JSmol) | Aluminum Sulfate (JSmol 3D model string): `Al2.3S.O12` |
| Beilstein Reference | 1902300 |
| ChEBI | CHEBI:29974 |
| ChEMBL | CHEMBL1200888 |
| ChemSpider | 28734 |
| DrugBank | DB11110 |
| ECHA InfoCard | ECHA InfoCard: 027-003-00-2 |
| EC Number | 10043-01-3 |
| Gmelin Reference | 71738 |
| KEGG | C14547 |
| MeSH | D012073 |
| PubChem CID | 24850 |
| RTECS number | WS4250000 |
| UNII | C3H89G223N |
| UN number | UN3077 |
| Properties | |
| Chemical formula | Al2(SO4)3 |
| Molar mass | 342.15 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.71 g/cm³ |
| Solubility in water | soluble |
| log P | -2.9 |
| Vapor pressure | negligible |
| Acidity (pKa) | 2.9 |
| Basicity (pKb) | 2.9 |
| Magnetic susceptibility (χ) | 'Magnetic susceptibility (χ): -52.0×10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.47 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 342.3 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | −3441.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3442 kJ/mol |
| Pharmacology | |
| ATC code | A12AB01 |
| Hazards | |
| Main hazards | Irritating to eyes, skin, and respiratory tract; may cause gastrointestinal discomfort if ingested. |
| GHS labelling | GHS07, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H318: Causes serious eye damage. |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| Lethal dose or concentration | LD50 oral rat 6207 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 6207 mg/kg |
| PEL (Permissible) | 2 mg/m3 |
| REL (Recommended) | 10 mg/L |
| Related compounds | |
| Related compounds |
Alum Iron(III) sulfate Sodium aluminate Aluminum chloride Ammonium alum Potassium alum |

