Allylamine: Finding Meaning Beyond the Lab Bench
Looking Back at Allylamine’s Road
Allylamine turned up in the pages of chemical history as part of a surge in organic synthesis research in the early twentieth century. Chemists once faced a tough landscape, hunting new ways to shape simple hydrocarbons into substances that could change agriculture, medicine, and industry. By the 1940s, as research into amines picked up pace, allylamine earned a spot thanks to its unique nitrogens and double bonds. Early investigators recognized the promise sitting in this molecule’s modest structure. Today, its story stands as an example of practical curiosity paying off—layer by layer—just like Edison’s quest for a better filament or the agricultural push for nitrogen fixers. Studying its progress over generations, you start to understand why chemists often say research is built on a thousand small advances, each profound in its modesty.
Getting to Know the Molecule Up Close
Allylamine looks almost too simple to hide such utility: three carbons, a double bond on the tail, and a lone nitrogen at the front, written in shorthand as C3H7N. Clear in color but powerfully sharp to the nose, its watery liquid form signals reactivity from the moment the bottle opens. Chemists who meet allylamine remember its fishy odor, a side effect of its amine group, and how quickly it mixes with water thanks to hydrogen bonding. Flammability demands respect. Like many small amines, even light sources can set off the vapor. In any room working with it, exhaust hoods don’t run for show.
Specifications and Labels: More Than Red Tape
What makes a chemical safe to handle, transport, or sell isn’t just data sheets. It’s about knowing at a glance what this thing can do, what it can harm, and who could get hurt. Labels warn of its irritation potential, but the education has to go deeper than sticker symbols. Training helps prevent mishaps: from the splash that burns skin to the shortness of breath that creeps up if inhaled. A small error in measurement, forgotten gloves, or an absentminded moment can mean trouble. Regulations have improved transparency—hazard pictograms, boiling points, concentration limits—but ultimately, personal vigilance draws the line between safe use and regret.
Crafting Allylamine: The Preparation Route
In the lab or at scale, making allylamine isn’t just about mixing chemicals. Starting points often come from allyl chloride or allyl alcohol, each bringing its own quirks and safety lessons. The classic route mixes allyl chloride with ammonia, often under pressure, with side products complicating the separation. The resulting allylamine often needs to be distilled gently to prevent side reactions. Industrial routes focus on yield and purity, sometimes involving catalytic hydrogenation of acrylonitrile. Each leg of synthesis weighs energy, resources, and safety, echoing real-world decisions in any operation: do you save on time, or play it safer and slower? There’s no perfect route, just an ongoing judgment about what risks and benefits match your setting.
Chemical Reactions and Its Capacity for Change
The double bond and amine group let allylamine serve as a handy building block. This chemical stains its fingerprints all over the pharmaceutical industry—for instance, in antifungal drug manufacturing. It snaps into place in Michael additions, acts as a nucleophile, and forms salts with acids, all hallmarks that earn it a seat at many synthetic tables. Working in a bench-scale lab, you remember the satisfaction of coaxing allylamine to form bigger, more complex molecules. One wrong move—a little too much heat, a misunderstood catalyst—and the reaction heads off course, showing just how demanding precision can be even for such a small molecule.
Common Names, Different Worlds
Ask people across chemical industries, and you’ll hear everything from 3-aminoprop-1-ene to propenylamine. While synonyms might seem bureaucratic, they reflect varied uses and traditions. Sometimes the name highlights its structure, sometimes its role in a process or product. Good communication between chemists, regulators, and safety professionals relies on knowing when someone means the same thing, or something subtly different—a lesson learned the hard way if a mislabel leads to the wrong drum arriving at a plant gate.
Safety and Working Standards on the Ground
Every safe day with allylamine is a small victory over complacency. Exposure hurts the skin and can damage the eyes and lungs, so gloves and goggles are hardly optional. Some operations shift work to closed systems. Procedures put a premium on quick access to emergency showers and eyewash stations. Regular training keeps new hires and old hands alert to the invisible dangers—vapors linger, and chronic exposure adds up. In the real world, it’s often the culture of respect for what you handle, not only written standards, that teaches everyone the value of a solid safety baseline.
Where Chemistry Strikes the Real World: Applications
Allylamine never makes a splash directly with the public—its value comes in what it helps build. Pharmaceutical lines, like those making terbinafine for athlete’s foot, count on its reactivity. Water treatment chemicals, rubber accelerators, and specialty resins all get their edge by adding this simple amine along the way. Plenty of advances in polymer science come from clever substitutions around that double bond or nitrogen, tuning flexibility or adhesion drop by drop. If you’ve ever noticed a synthetic product with unusual durability or a medicine that took years off a clinical trial, there’s a fair chance molecules like allylamine were key players in the background.
Research, Discovery, and New Frontiers
Continued research into allylamine keeps uncovering fresh applications and cautionary tales. The hunger for greener synthesis methods pushes chemists toward lower waste streams and safer byproducts. Search engines light up every month with new papers on improved catalysts, efficiency gains, and novel routes to functionalized amine derivatives. Pharmaceutical chemists keep chasing ways to tweak the molecule, aiming for lower toxicity and fewer side effects. Every breakthrough gets measured against cost, reliability, and regulatory scrutiny, reminding everyone that creativity thrives best under real-world constraints.
Toxicity: Lessons in Handling Risk
Toxicity sits front and center in any conversation about allylamine. Researchers list clear dangers, from irritation and chemical burns during acute exposure to long-term organ impacts for workers lacking protection. Animal studies back up the need for strict limits. Conversations in the workplace about venting, spill control, and storage aren’t just checkbox exercises. They grow from lived experiences—each mishap, even if minor, shapes better habits. The ongoing challenge lies in making sure everyone, from interns to supervisors, understands that a fast-paced environment never forgives shortcuts. The best safety stories are the ones where nothing dramatic happens, despite years of routine handling.
Looking Ahead: The Future for Allylamine
Forecasting allylamine’s next chapter means watching broader trends. Industrial processes lean toward balance—minimal waste, maximum safety, and lower costs. Environmental rules around the globe push plant managers and chemical engineers to rethink old methods, making syntheses cleaner and more sustainable. Research continues peeling back more possibilities, especially as demand grows for high-value polymers, specialty chemicals, and new drug scaffolds that call for amine building blocks. As more companies put money into green chemistry, expect allylamine to show up in places and products few outside the field would ever suspect, all while scientists and safety pros keep striving for that elusive blend of productivity and protection.
What is Allylamine used for?
What Allylamine Brings to the Table
Allylamine often stays behind the curtain, but it plays a bigger part in everyday products than most folks realize. This organic compound, built from carbon, hydrogen, and nitrogen, packs a punch in multiple industries. Its most visible flag waves in the world of prescription medicine, where it steps up in the fight against fungal infections. Terbinafine, a drug widely prescribed for athlete's foot and similar problems, owes much of its effectiveness to allylamine at its core.
Making Tough Materials Stronger
Beyond medicine, allylamine shapes the backbone of some industrial products. Paints and coatings often begin with complex chemical reactions, and allylamine can help get those started. It acts as a building block for resins and polymers, making materials more durable and weather-resistant. This is not just chemistry for chemistry’s sake. Stronger coatings mean longer-lasting bridges, safer cars, and better-protected electronics.
Contributing to Everyday Health and Cleanliness
Not only limited to doctors’ offices and industrial plants, the reach of allylamine can slip into cleaning products as part of surface disinfectants or household sprays. Robust antifungal and antimicrobial properties—first noticed in clinical drug development—sparked the use of allylamine-derived compounds in formulations that keep everything from bathroom tiles to kitchen counters safer.
What Science Says about Handling and Safety
While it serves good purposes, working with allylamine brings its own set of challenges. Safety data from the Centers for Disease Control and Prevention highlights its potential dangers if inhaled or accidentally spilled onto skin. Short-term exposure can cause headache, dizziness, or even severe irritation. This isn’t a product most people want to buy at the hardware store themselves. Most of the time, it stays locked away at facilities set up with equipment designed to protect employees and the environment.
Walking the Fine Line: Environment and Regulation
As with many chemical building blocks, allylamine’s growing use calls for careful regulation. Countries like the United States and those in the European Union enforce strict guidelines on its handling, storage, and disposal. Rules come down hard for good reason: even small leaks can spell trouble for water supplies and wildlife. Environmental scientists continue to keep their eyes on how facilities manage allylamine-based waste, pushing for greener chemistry where possible.
Better Practices, Safer Products
Companies using allylamine bear the weight of consumer trust. One solution comes from investing in new manufacturing methods that capture and recycle excess material. Innovative researchers also develop substitutes that deliver similar benefits without as much risk. Some of these solutions use different amines or plant-based components, aiming not only to protect workers but also to shrink the environmental footprint.
Looking Ahead
Every time a fungal infection clears up or a structure stands strong against weather and time, allylamine’s influence can be felt. Scientists, manufacturers, and regulators share responsibility in balancing useful applications with health and safety. That trust, built on transparent research and tough standards, guides this small but important compound’s role in daily life.
Is Allylamine hazardous to health?
Understanding Allylamine and Its Risks
I’ve spent years around chemicals both in the lab and in industrial settings. Allylamine stands out as one of those substances that draws a lot of concern because of how it behaves and how people might come into contact with it. It appears as a clear, colorless liquid and smells a bit like ammonia, which often tips folks off before they even see the label.
Touching or inhaling allylamine can irritate the nose, throat, and lungs almost right away. Getting it on your skin or in your eyes causes burning and swelling. I watched a coworker’s skin turn red and angry after just a small splash. OSHA put the limit at 2 parts per million in the air for an 8-hour workday, reflecting the fact that anyone working with this chemical needs serious protection.
Why Worry About Chronic Exposure?
Repeated exposure brings separate worries. Breathing in low levels repeatedly can lead to chronic bronchitis, a wheezy cough, and breathing trouble that just won’t quit. The National Institute for Occupational Safety and Health (NIOSH) calls out big risks for the heart as well. Animal research shows allylamine can lead to heart lesions, thickened arteries, and sometimes heart failure. Though most people won’t get near these doses, people in production plants can face higher odds if safety steps get skipped.
I remember walking through a plant that produced specialty plastics where allylamine was in use. It only took a small leak to force an evacuation—the supervisor drilled us on what could happen if the chemical got loose indoors: confusion, headache, or even loss of consciousness. Thankfully, everyone moved fast, but the whole event left a mark.
Beyond the Workplace
Allylamine seldom shows up outside of industrial sites, but downstream users—people who mix or transport chemicals, even staff at water treatment plants—can get exposed if safety equipment fails. Spills can become a real hazard quickly, especially since allylamine evaporates and can pump dangerous vapor into enclosed spaces. If rescue teams aren’t ready with self-contained breathing gear, they could wind up as victims too.
Communities near plants don’t face the same direct risk, since regulations keep emissions low. People further out sometimes worry about contaminated water, but allylamine breaks down fairly quickly in the environment, reducing longer-term risks. That’s been echoed in local monitoring reports and EPA summaries.
Protecting Health in Real Settings
Tough workplace rules, proper training, and up-to-date gear make all the difference. I’ve seen simple steps like splash goggles, chemical suits, and air monitors stop a bad day before it starts. Emergency showers placed near work areas limit damage when things go wrong. Regular medical checks for exposed workers catch health problems early—one small cough can be a signal to step back and get checked out.
No magic bullet exists for handling chemicals like allylamine, but relying on layers of protection helps keep people safe. Building a culture where people speak up about leaks or near-misses matters even more. In places that take these rules seriously, health risks shrink and work moves forward without unnecessary danger.
How should Allylamine be stored?
Allylamine Storage: Not Something to Take Lightly
Years of lab work teach you that overlooking proper chemical storage turns into a hazard fast. Allylamine is no harmless shelf-filler. Workers and researchers need to remember: this stuff adds risks beyond your typical solvent. It doesn’t smell pleasant—there’s a strong ammonia-like odor—and exposures can harm eyes, skin, and lungs. News stories about chemical leaks often have one thing in common: sloppy preparation. Forgetting basic safety steps can turn a simple task into an emergency.
Vapor, Fire, and Corrosion Risks
This compound evaporates quickly, and its vapors catch fire easily. If it leaks or spills, vapors can travel and ignite from a distant spark. Not just that—allylamine reacts with acids, some metals, and oxidizers, which can turn any small lab accident into a major headache. The CDC and NIOSH both flag it as a major inhalation risk and advise storage in tightly closed containers, far from sources of ignition.
What Works in Practice
Storing allylamine means two things: cool and well-ventilated. I’ve learned that keeping it below room temperature (preferably in a flammables refrigerator) helps limit those constant vapors. Room temperatures work for short-term storage in a safe space, but colder storage is better for longer stints.
Metal drums? Glass jars? I’ve seen folks try all sorts. In reality, only containers rated for amines make sense. Stainless steel and glass with Teflon-lined lids stand up best against corrosion. Forget about regular steel or low-quality plastics—allylamine chews them up over time.
Isolation and Organization
Fire codes don’t leave much room for cutting corners. Chemical storage lockers, equipped with self-closing doors and spill containment trays, offer a basic starting point. Separate allylamine from acids, oxidizers, and even food or personal items. I’ve seen even experienced teams forget this detail, which turns cross-contamination into a real threat.
Labels, fresh inventory logs, and easy-to-read safety data sheets should never gather dust. It’s tough to track down information during an emergency, so make it visible and accessible. That isn’t just best practice—it’s common sense.
Best Habits for Handling Allylamine
Accidents don’t wait for a quiet day. Workers should always use splash-proof goggles, chemical-resistant gloves, and lab coats when handling allylamine. Keep a reliable spill kit within reach and test the fume hood regularly. Storage alone can’t prevent every problem, but regular checks and clear rules keep surprises to a minimum.
Knowledge Saves More Than Regulations
In research settings, rushed chemistry or ignored storage advice can have a domino effect. Learning from the incidents reported by the Chemical Safety Board convinced me that safety isn’t optional. Training sessions on storage situations aren’t paperwork exercises—they’re about making sure everyone goes home unscathed.
Getting the storage right for allylamine means a lot more than pleasing inspectors. It prevents injury, property damage, and lots of paperwork later on. Keeping procedures clear and simple gives peace of mind to everyone around—because in the end, people matter far more than the chemicals.
What are the safety precautions when handling Allylamine?
Understanding What’s at Stake
Allylamine isn’t a chemical you want to take lightly. It packs quite a punch—its pungent smell tells your nose plenty before you even look at the safety sheet. Folks use it in the lab for making medicines, polymers, pesticides, and even fuel additives. Getting careless around it brings real risks. Your skin, lungs, and eyes feel the brunt if things slip up.
Personal Protective Equipment: No Shortcuts
Rubber gloves are a must—nitrile works better than latex since allylamine likes to chew through thinner barriers. A solid pair of goggles forms your best defense for your eyes. Full-face shields take that a step further. In one factory job, I saw a co-worker get a splash on his cheek, and although he rinsed right away, he still needed medical attention. Face protection isn’t negotiable.
A simple mask won’t cut it. Respirators with organic vapor cartridges can mean the difference between a tough day and a trip to the ER. Allylamine’s fumes go straight for the throat and lungs, causing sore throats and even trouble breathing. Those fumes linger, so good ventilation matters just as much as gear.
Workspaces: Better Safe than Sorry
Forget using allylamine on a cluttered bench with a fan in the corner. Fume hoods earn their keep here. Air gets pulled away from your face, drawing the vapors out before they can build up in the room. Closed systems and proper seals protect everyone—not just the one pouring out the bottle.
Never eat, drink, or snack at the bench. Hand washing takes just a minute but saves a world of trouble. Even with gloves, traces can stick to surfaces.
Know the Signs of Exposure
A spill on your hands can cause redness, pain, and burns that grow worse as time goes on. It isn’t just skin deep: breathing allylamine vapors might leave you dizzy, coughing, or feeling pressure in your chest. Long-term exposure leads to lasting breathing problems and possibly heart issues. Get any contact checked by a health professional, no matter how small it seems.
Storage and Clean-Up: No Corners Cut
Allylamine catches fire easily. Store it cool and dry in well-marked containers, nowhere near sources of ignition. If you spill, soak it up with sand or vermiculite. Those fancy paper towels only make things worse by spreading it around and creating flammable waste. Contaminated gear needs proper handling—not tossing it into the regular trash.
Training and Teamwork
Regular safety drills make all the difference. Teams who practice together spot risks and catch mistakes fast. People remember what works because they rely on each other—not just on rules written on the wall. In some companies, updated laminated instructions hung near workstations helped even experienced folks double-check each step. Building a safety culture can start with one worker speaking up if they see something off.
Fixes and Improvements
Basic gear upgrades change outcomes overnight. Swapping gloves or goggles costs little but pays back every shift. Automated dispensers or robots for handling nasty chemicals have cut accident rates sharply in some places. Clear labeling and regular training won’t solve everything, but they make mistakes far less likely. Real safety means never assuming “it’ll be fine.” Companies who invest here protect lives and business, plain and simple.
What are the common applications of Allylamine in industry?
Why Industry Values Allylamine
In the chemical world, it’s easy to skip over small molecules like allylamine and pay all the attention to finished products. But look closer, and these building blocks power a surprising number of essential industries. Allylamine stands out for its ability to kick off larger chemical chains, serving as a starting point for products most people use and depend on every day.
Pushing Forward with Pharmaceuticals
Pharmaceutical companies count on allylamine to make antifungal drugs. Terbinafine, a well-known medication for treating fungal infections of skin and nails, comes directly from chemical pathways launched by allylamine. The way these molecules react lets pharma researchers design medications that attack infections right at the source. This matters a lot for people who struggle with persistent fungal conditions, which can be stubborn and resistant to older treatments.
Water Treatment: Clean Water, Clear Minds
Engineers turn to allylamine derivatives to make chemicals called ion exchange resins. These specialized resins pull unwanted minerals and metals from drinking water and keep heavy industries running efficiently. Safe, reliable water isn’t a small issue—without processes like this, homes and hospitals would have a much harder time delivering clean water. It’s one thing to install a filter at a sink; it’s another to build a city’s water system around materials that last and stand up to tough conditions.
Rubber and Plastics: The Hidden Link
Makers of synthetic rubber and plastics use allylamine as a key starting point. For example, it helps build polymers that end up as coatings for wires and pipes, or even in the tires on family cars. The chemical structure of allylamine lets companies create materials with the flexibility and strength that other techniques just can’t match. This isn’t just a convenience; strong, light plastics keep vehicles safer and more efficient, and durable rubber keeps roadways quieter and longer-lasting.
Agrochemicals and Specialty Chemicals
Allylamine also plays a big part in agriculture. Manufacturers use it to produce pesticides and herbicides that protect crops from insects and weeds. It’s not just about higher yields; it’s about making sure those who farm the land can afford to keep going year after year, with crops that remain marketable and safe for the table. In research and development labs, chemists put allylamine into specialty compounds that improve paint adhesion and drive advances in adhesives and coatings. These materials show up everywhere from house paint to high-end electronics.
Looking Ahead: Responsibility and Innovation
Like many useful chemicals, allylamine comes with safety concerns. Its vapors irritate eyes and skin, and workers need real training and good protective gear in factories. Companies that handle allylamine regularly push for higher standards, aiming to reduce accidental releases and protect their teams. Newer technologies in chemical manufacturing focus on limiting waste and recycling solvents—steps that keep plants efficient and cut down on environmental risks.
Solutions for the Long Run
People working in industrial settings, public health, and environmental safety have learned a lot from decades of experience with allylamine. By investing in better ventilation, personal protective equipment, and workplace education, industries can lower risks. Cleaner production methods, stronger chemical containers, and near-real-time air monitoring keep progress moving forward. It takes commitment from both leadership and teams on the ground, but the payoff is clear: safer factories, reliable products, and a better environment for everyone.
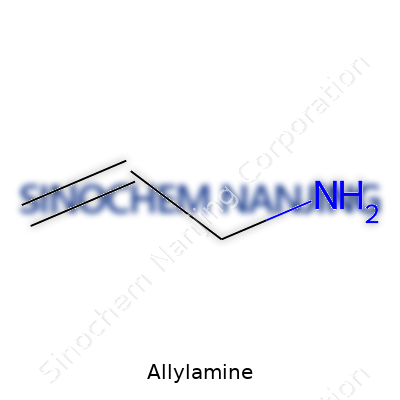
| Names | |
| Preferred IUPAC name | Prop-2-en-1-amine |
| Other names |
aminopropene 3-aminopropene propenylamine allylamin |
| Pronunciation | /ˈæl.ɪ.lə.miːn/ |
| Identifiers | |
| CAS Number | 107-11-9 |
| Beilstein Reference | 1718739 |
| ChEBI | CHEBI:29016 |
| ChEMBL | CHEMBL1408 |
| ChemSpider | 8187 |
| DrugBank | DB00797 |
| ECHA InfoCard | 100.003.738 |
| EC Number | 203-699-2 |
| Gmelin Reference | 6355 |
| KEGG | C06591 |
| MeSH | D017355 |
| PubChem CID | 7847 |
| RTECS number | AS1050000 |
| UNII | 4QD397987E |
| UN number | UN1605 |
| Properties | |
| Chemical formula | C3H7N |
| Molar mass | 57.10 g/mol |
| Appearance | Colorless to yellow liquid with an ammonia-like odor. |
| Odor | Ammonia-like |
| Density | 0.76 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | 0.64 |
| Vapor pressure | 233 mmHg (20°C) |
| Acidity (pKa) | 9.5 |
| Basicity (pKb) | 3.16 |
| Magnetic susceptibility (χ) | -14×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.414 |
| Viscosity | 1.0 mPa·s (at 20 °C) |
| Dipole moment | 1.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 252.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -20.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4422.7 kJ/mol |
| Pharmacology | |
| ATC code | D01AE06 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H314, H331 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P311, P321, P330, P337+P313, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Autoignition temperature | ### 327°C |
| Explosive limits | 3.0–40% |
| Lethal dose or concentration | LD50 rabbit oral 106 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 107 mg/kg |
| NIOSH | NIOSH: SD6475000 |
| PEL (Permissible) | 2 ppm |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | 40 ppm |
| Related compounds | |
| Related compounds |
Allylammonium N-Methylallylamine Methallylamine Allyl group Ammonia |

