Allyl Methacrylate: A Chemical Key in Today’s Industry and Research
The Historical Path of Allyl Methacrylate
Allyl methacrylate has been around the chemistry world for nearly a century, tracing back to early work on acrylic esters. Researchers looking for new ways to strengthen resins and coatings stumbled on a group of monomers with promising potential, and among them stood allyl methacrylate. Its journey started in the laboratories of the mid-1900s, during a period when thermoset plastics needed better cross-linking efficiency. Chemists began paying close attention to the molecular backbone: two functional groups in one molecule—an allyl group and a methacrylic group—allowed it to stand out as a dual-purpose actor in radical polymerization. This property boosted its reputation not just as a participant in making polymers but as a building block for tearing down and constructing more sophisticated materials. Over time, allyl methacrylate moved from specialty labs into mainstream industrial settings, marking its importance as companies and universities looked for ways to refine paints, adhesives, and advanced composites. Without these early investments in acrylic and allylic chemistry, many modern plastics and specialty materials would still be out of reach.
What Allyl Methacrylate Brings to the Table
This compound isn’t just another raw material on a long list of chemical intermediates. With a clear, pungent liquid appearance, allyl methacrylate offers unique properties that go beyond traditional methacrylates. Its dual-reactive structure catches attention in formulating robust networks for cross-linking polymers. That’s why it has become a go-to additive for improving impact resistance and toughness in both thermosets and thermoplastics. Nearly every major synthetic resin producer has explored using allyl methacrylate at some stage, especially when they seek greater flexibility or higher heat resistance. Its ability to branch polymers and create complex cross-linked architectures sets it apart. In practice, this means final products—from dental materials to lightweight panels—can endure repeated stress that other materials cannot.
Physical and Chemical Traits Worth Noting
A pungent odor accompanies allyl methacrylate, reminding experienced chemists that it’s a volatile, flammable liquid. Boiling at roughly 142°C and with a density close to 0.98 g/cm³, this monomer evaporates easily, which prompts tight handling protocols. Chemically, the presence of both allyl and methacrylic unsaturated bonds means it’s reactive towards free radical initiators, allowing it to blend smoothly in co-polymerization processes. It dissolves well in most organic solvents, adding flexibility to formulation work. Yet, its volatility and tendency to polymerize—sometimes even during storage—calls for stabilization, often achieved with inhibitors or cold storage. Water barely dissolves it, which suits many composite fabrication techniques where separating phases is important. Anyone using or storing allyl methacrylate learns not to overlook its strong odor, fast evaporation, and reactivity, both in routine work and in research.
Technical Specs and Labeling Challenges
Labeling allyl methacrylate isn’t just about placing a sticker on a bottle. Regulations demand clear hazard and precautionary statements due to its flammability, skin and eye irritation risks, and environmental hazards. In my own lab days, I learned the hard way that even strong secondary containment sometimes cannot mask the acrid scent if a cap isn’t tight. Modern standards require specific attention to batch purity, inhibitor content to suppress unwanted polymerization, and clear indication of storage conditions. Tech specs often focus on purity measured by gas chromatography, stabilizer levels, and obvious color or phase changes. Shipping it calls for UN-approved containers, and anyone receiving a drum or bottle needs up-to-date safety documentation. Small lapses in handling or mislabeling have big consequences, particularly for workplace safety and downstream product reliability.
Preparation on the Production Line
Making allyl methacrylate involves a classic esterification between methacrylic acid and allyl alcohol, usually catalyzed by acids and conducted under strictly controlled conditions. In my time observing pilot plant runs, I saw how precise temperature and pH adjustments make a huge difference not only in yield but also in the color and stability of the final product. Reaction by-products like water must be stripped off, with distillation taking care of both purification and solvent recovery. After main distillation, the product is stabilized and stored in tight, light-resistant vessels. Quality checks come thick and fast at each stage—one off-kilter batch can halt polymer lines downstream. Scale-up from lab glassware to industrial reactors brings its own headaches, especially with heat exchange and volatile vapor containment, which makes robust engineering controls a must.
Chemical Behavior and Main Modifications
Allyl methacrylate’s real magic comes from its double bond setup. During free radical polymerization, each double bond offers a reactive site, allowing it to act as a cross-linker. Chemists have used this to toughen up standard resins, making tougher adhesives and coatings as a result. Sometimes it reacts with epoxides or amines for further functionalization—this opens doors for fine-tuning hydrophobicity or thermal resistance. Many labs also look at partial hydrolysis or acid-catalyzed reactions as routes to turn it into new acrylic intermediates. Yet, its tendency to self-polymerize poses shelf-life headaches unless inhibitors stay present at correct concentrations.
Names and Nomenclature: Avoiding Mix-ups
Confusion often springs up with chemical names. Allyl methacrylate also turns up in paperwork as 2-Propenoic acid, 2-methyl-, allyl ester or simply AMA in some product guides or patents. Supply chains juggling international shipments need everyone on the same page about these synonyms to avoid costly errors. In research, precise naming streamlines literature searches and ordering, while in manufacturing, matching product codes avoids mixing up chemicals with radically different properties. Even a simple slip on a shipping manifest can lead to process shutdowns or safety breaches—something any laboratory or plant manager wants to avoid.
Safety Culture and Operational Realities
Handling allyl methacrylate calls for discipline and trust in standard operating procedures. Flammability ranks high on the list—open flames or stray sparks present constant risks. Even personal experience reinforces this point; a single spill on stainless steel benchtops can stink up a whole wing and, left unchecked, evaporate within hours. Emergency showers, eyewash stations, and proper gloves form the core of daily life for chemists using this substance. At scale, negative-pressure hoods and vapor sensors make a clear difference in day-to-day safety. Regular training ensures staff never get complacent, and detailed records for storage and waste disposal help keep regulatory flags at bay. For workers new to this chemical, strong induction programs focus on odor recognition, evacuation routes, and what to do if clothing gets splashed. In operations where allyl methacrylate stays in use for years, these practices shift from being “compliance” to lifelines that protect everyone onsite.
Where it Finds a Home: Industry and Research
Few chemicals boast a bigger footprint across so many industries. In plastics production, allyl methacrylate helps build strong, flexible networks in acrylic resins, often serving roles in lightweight automotive panels, wind turbine blades, or aerospace composites. Electronics makers lean on its cross-linking power to build durable encapsulants and rigid circuit boards. Adhesive and sealant developers tap its unique structure to create products that handle extreme temperatures and stresses. Dentistry has adopted it for stronger, longer-lasting fillings and prosthetic components. Out in the field, protective coatings with this monomer have pushed construction and infrastructure projects to new durability standards, especially for concrete and steel. Technical textiles and membrane producers blend it into performance fabrics and filter media, seeking resilience and precision. Each application emerged from the groundwork laid by chemists and engineers generations ago, always searching for ways to stretch material limits.
Ongoing Research and Fresh Discoveries
Crowds of researchers worldwide continue probing how allyl methacrylate might push materials science deeper into the future. Academic labs want to expand the bandwidth of properties it gives to co-polymers. For example, recent work explores greener synthesis using less harsh catalysts and more efficient separation methods to cut waste. Startups in sustainable packaging give it a shot as a base for biodegradable composites or more easily recycled plastics. A few teams work at the intersection of materials and biomedical science, aiming to lower residual monomer content in dental and medical devices while maintaining mechanical toughness. Even high-performance 3D printing benefits: allyl methacrylate lends ways to craft tough lattices and intricate parts with strength-to-weight ratios that rival metals. Each project builds on decades’ worth of accumulated know-how—failures and successes alike feeding a constant feedback loop between basic research and industrial application.
Diving into Toxicity and Human Health
Researchers and safety officers keep a close watch on the toxicity front. Standard testing points to eye and skin irritation as primary hazards, and workers exposed without protection risk dermatitis or respiratory discomfort. Animal studies and limited human data suggest that long-term exposure, especially to high airborne concentrations, can challenge respiratory or immune responses. These insights shape workplace exposure limits and propel investments in ventilation and process containment. In my own circle, strict adherence to personal protective equipment and regular monitoring have become non-negotiable expectations. The push for alternatives with lower toxicity continues, though the unique profile of allyl methacrylate means outright replacement remains tricky for critical applications. Companies invest in process automation and enclosed systems, aiming to cut direct worker exposure and improve downstream material purity. Regulatory agencies in the US and EU routinely update hazard classifications, pushing companies toward innovation in formulation and packaging to keep risks low.
Looking Ahead: Future Prospects and Innovation
Allyl methacrylate stands as both a cornerstone of classic chemistry and a springboard for new frontiers. As green chemistry takes root across the globe, manufacturers hunt for energy-saving synthesis routes, safer stabilizers, and recycling-friendly resins. Research into bio-based feedstocks for producing allyl methacrylate could shift reliance away from fossil sources, fundamentally changing its supply chain and carbon footprint. Advanced composites—especially those aimed at the transportation and renewable energy sectors—look to this monomer for further breakthroughs in performance and longevity. Nanomaterials and smart coatings draw interest for its compatibility in multi-functional layers. The race among labs and startups now focuses on lowering environmental impact without sacrificing mechanical advantages. Regulatory pushback on workplace exposure only intensifies the need for innovation geared toward safety, traceability, and zero-waste manufacturing. Every day, the chemical and its community of researchers, engineers, and operators rewrite what is possible, drawing on experience and imagination to solve urgent challenges in society and industry.
What is Allyl Methacrylate used for?
What Makes Allyl Methacrylate Special?
Working in labs over the years, I've seen many chemicals come and go, but allyl methacrylate keeps showing up in unexpected places. Its real draw is the combination of an allyl group and a methacrylate group in the same molecule. That simple structure leads to huge versatility for creating new things that simply last longer and work harder.
Doctors, engineers, and manufacturers rely on tough materials. The products all around us—from tire rubber to dental fillings—often begin their lives with chemistry like allyl methacrylate. Once mixed in, the finished items often hold up better, shrug off heat and wear, and stay strong year after year.
Stronger Plastics, Reliable Rubbers
The most direct role for allyl methacrylate shows up in plastics and rubbers. Industrial makers add tiny amounts when making acrylic sheets, pipes, or coatings. This tweaks the final material—making it tougher, stretching without breaking, and taking years of sunlight or vibrations without cracking. It comes down to crosslinking; the chemical bonds that join up different parts of the plastic, stopping the plastic chains from sliding past each other.
Think of tough bowling balls, rugged car parts, or weather-resistant window frames. All of them benefit from the tiny amounts of allyl methacrylate working at a molecular level. Without it, these objects wouldn’t handle daily punishment nearly as well.
Making Teeth Repairs Last Longer
In dentistry, there’s no patience for brittle fillings. Allyl methacrylate helps dental plastics set better and hold strong against years of chewing, brushing, coffee, and pizza. My dentist once told me about the challenge of repairing cracked fillings that just didn’t last. Modern composite resins with allyl methacrylate survive that test better. Clinical studies back it up; these resins resist wear, avoid shrinking, and let dentists shape repairs that blend right in.
Electronics and Coatings
On factory floors, circuit makers rely on stable coatings for their printed boards. Allyl methacrylate steps in to toughen these coatings, so they hold up under the heat of soldering and the steady current running through tiny chips. Companies making LEDs, touchscreen gadgets, or solar panels look for materials that resist cracking and peeling. Allyl methacrylate delivers that, without adding unwanted color or blocking signals.
Paint companies use it, too, for specialized finishes. Cars, boats, outdoor equipment—all benefit from paints that shrug off rain, UV rays, and harsh chemicals. The chemistry stops flaking and fading, meaning products don’t need fresh coats as quickly.
Safe Use and Future Solutions
Handling allyl methacrylate in the lab reminds me that safety never takes a break. The liquid itself can irritate skin and eyes, and fumes ask for good ventilation. Industry always pushes to lower risks and manage waste well. Regulatory agencies keep tabs on workplace limits and set rules to prevent spills.
Looking ahead, researchers focus on making the production process cleaner, aiming for greener chemistry with less waste. Recyclable materials using allyl methacrylate are starting to show promise. Europe has pushed for tighter controls, and big companies follow suit to keep both workers and the environment safer while keeping the benefits.
Allyl methacrylate keeps earning its spot not because of flashy headlines, but thanks to real-world reliability. From daily commutes to doctor visits, its chemistry helps products stay tough, safe, and ready for whatever life brings.
What are the safety precautions when handling Allyl Methacrylate?
Understanding a Tricky Chemical
Allyl methacrylate gives off a sharp, pungent odor before you even open the drum. Over the years, I’ve learned to respect these clues. This liquid burns if it touches your skin, stings the eyes, and can leave you coughing from just its fumes. In the labs I’ve worked at, seasoned techs always wore goggles and nitrile gloves. That sharp smell isn’t a suggestion. It’s telling you to gear up.
The Hazards Up Close
Direct contact will irritate skin and eyes. Even brief exposure triggers red, itchy skin or sore throat. The vapors can knock you back, especially in tight spaces with poor ventilation. In my early days, I once saw someone splash a drop on their forearm—immediate redness, a trip to the eyewash just in case. OSHA lists it as harmful by inhalation, skin absorption, and ingestion. Long-term, there’s a risk of allergic skin reaction. Grabbing a respirator and washing your hands after every use is not overkill. It’s just common sense.
Working Safe Means Working Smart
Prioritizing personal protective equipment (PPE) can make all the difference. Chemical-resistant gloves, splash goggles, and lab coats shield you from accidental spills and splashes. I’ve moved from using latex to nitrile gloves after watching latex break down too fast in practice. When pouring large amounts or cleaning up spills, a face shield adds extra peace of mind. Proper lab work means never standing over an open bottle, never handling it without gloves, and storing it below eye level to limit spill risk.
Ventilation keeps vapors from building up. In every reputable workplace I have visited, all work with allyl methacrylate happens inside a fume hood. Airflow pulls fumes away from breathing zones and disperses them safely. The days of working in a closed room without a hood belong in the past. Fire safety deserves attention too. This stuff ignites easily, so flames and sparks have no place nearby. Check your fire extinguishers—they need to be compatible with chemical fires, not just paper or wood.
Storage and Cleanup Aren’t Afterthoughts
Storing allyl methacrylate properly prevents big headaches later. Tightly seal containers and keep them in a cool, dry room away from direct sunlight and incompatible chemicals like oxidizers and acids. I once saw a drum leak because the cap wasn’t sealed; fixing that became a whole afternoon of cleanup and paperwork. If a spill happens, soak it up with absorbent materials, ventilate the room, and dispose of waste as hazardous. Rags and gloves go in a special bin, never with everyday trash.
Training: Everyone Plays a Role
Supervisors set the rules, but everyone on the team keeps each other safe. I’ve watched new techs pick up safe habits by watching mentors model each step. Regular training updates keep everyone alert. If you see unsafe handling or broken gear, calling it out saves trouble. No one outgrows asking questions or double-checking instructions. Stay sharp, and incidents stay rare.
Finding Better Ways
Some teams have switched to less hazardous alternatives when possible, but not every process allows it. Engineers keep pushing for better ventilation systems. Regular safety reviews often reveal small tweaks—relocating a container, changing gloves more often, or labeling storage cabinets more clearly. These simple fixes save injuries and help your lab or workplace stay out of the headlines.
Sources:- OSHA: Chemical Hazard Information for Allyl Methacrylate
- CDC/NIOSH: Pocket Guide to Chemical Hazards
- Personal experience with laboratory safety protocols
What is the chemical formula of Allyl Methacrylate?
Chemistry in Everyday Life: Where Allyl Methacrylate Fits In
Allyl methacrylate, written chemically as C7H10O2, plays a role in manufacturing that often goes unnoticed outside of labs and factories. For many years, I worked with polymers in an industry lab, and allyl methacrylate regularly came up when engineers wanted to toughen up plastics or rubbery materials without making them brittle. The formula itself—C7H10O2—means you get seven carbon atoms, ten hydrogens, and two oxygens, giving a hint of what this molecule brings to a chemical reaction.
What the Formula Means for Industry
Each atom in allyl methacrylate's formula does more than add up to a total. There’s a methacrylate group that wants to join in polymer chains; the allyl group gives a branching site. This combination creates flexibility and durability in end products. Many plastic parts in cars, electronics, or appliances depend on chemical links formed by monomers like this one. If any of these atoms were missing, the finished plastic could end up cracking or breaking under stress.
Real-World Impact: Beyond the Lab
In practice, allyl methacrylate steps in to give certain plastics better heat resistance and strength. For example, companies use it to help paints stick to surfaces or make adhesives last longer. Its ability to form three bonds sets off crosslinking, which transforms runny liquid resins into solid materials. I’ve watched team members in coatings development swear by this monomer for getting tough, glossy layers in demanding environments—from airplane parts to high-end furniture. Without that C7H10O2 backbone, those same coatings might wear off much faster.
Safety and Environmental Considerations
The story does not end with performance. Like many strong-smelling chemicals, allyl methacrylate poses health risks. Direct contact might cause skin reactions, and breathing its fumes can harm the lungs. Responsible companies train workers on safe handling techniques—wearing gloves, using proper ventilation, and measuring air quality. This helps lower risks and protects long-term health, which I see as non-negotiable in any chemical workplace.
Questions about environmental impact also come up. Once released, organic monomers like these can stick around. Responsible disposal and tight controls cut back on spills and emissions. Certification systems, like ISO or REACH in Europe, encourage transparency and safety. If every lab and factory takes chemical stewardship seriously, we can use materials like allyl methacrylate without tipping harm onto people or the environment.
The Way Forward: Smarter Chemistry With Purpose
Chemical formulas like C7H10O2 are just codes unless people put them to work. Engineers and chemists use allyl methacrylate for good reason. The goal should always be to find just enough of these molecules to serve real needs—never wasting, always thinking ahead. In my own work, pushing for leaner, cleaner processes not only saved money but also built more trust with workers and customers. Chemical safety and environmental care can go hand in hand with industrial progress if everyone takes ownership, one molecule at a time.
How should Allyl Methacrylate be stored?
Understanding the Risks
Allyl methacrylate is a chemical with sharp odors and a high level of reactivity. I’ve stood in a lab where someone cracked open a half-forgotten drum only to find the inside sticky, yellowed, and warm—never a good sign. This chemical doesn’t play nice with oxygen, heat, or light. Even an absentminded mistake with its storage can result in disaster. It’s more than a matter of compliance; it’s about real safety.
What Makes Allyl Methacrylate Tricky
Allyl methacrylate likes to polymerize on its own. It doesn’t need much provocation—ambient sunlight, left long enough, kicks off a chain reaction turning clear liquid into a gelatinous mass. Uncontrolled polymerization sometimes even causes pressure build-up. There’s also the issue of vapor; this stuff stings the nose and can irritate your lungs and eyes if you’re sloppy with venting. Direct skin contact burns fast, so handling with care isn’t negotiable in any setting.
Lessons in Practical Storage
The best labs I’ve worked in use a few simple principles: keep allyl methacrylate in cool, dark spots, in tightly sealed containers, away from anything that throws off heat or sparks. I remember packing it into metal drums stored in ventilated cabinets, far from direct sunlight. If a refrigerator sits in the chemical room, it’s no accident—controlling the temperature matters for integrity and safety alike.
Using containers made of compatible materials helps prevent reactions. Stainless steel or glass containers with solid seals cut down the risk of leaks or exposure to oxygen and moisture. Expired containers, cracked lids, or questionable seals—these spell trouble. It’s worth it to inspect storage drums monthly and to swap out anything showing wear. I’ve seen chemicals brought out from storage that looked fine on the outside but had partially polymerized inside. One sharp-eyed tech saves a whole lab from potential headaches just by being thorough.
Keep an Eye on Stabilizers
Allyl methacrylate shipped with stabilizers lasts longer. Removing these stabilizers—or accidentally letting them degrade—increases the chance of runaway reactions. So it’s smart to regularly check for expiration dates and confirm stabilizer content in every batch; don't just trust the supplier. Some organizations keep logbooks to record receiving dates, inspection results, and opening times. This kind of recordkeeping feels tedious, but there’s no question it catches small issues before they get ugly.
Ventilation and Emergency Prep
Reliable storage isn’t just about containers and temperature. I’ve seen old-school fume hoods make the difference between a safe scrape and a hospital trip when cleaning up a spill. Good ventilation systems keep vapors from building up. Proper signage—bold, with clear hazard warnings—makes it hard for even the newest intern to miss the risk. Eye wash stations and spill kits never gather dust in a well-run storeroom. Every lab tech gets annual refreshers on what to do in an emergency. Those few minutes of drills pay off big if anything happens.
Taking Safety Seriously
Many of the tightest labs manage alphabet soup inventories day after day without shortcuts, because a lax attitude with allyl methacrylate ends badly. OSHA and EPA don’t just publish guidelines out of habit; they’re built on real accidents. For anyone handling this chemical, consulting authoritative sources like the material safety data sheet is as basic as locking the door at night. Following proven storage methods, tracking stock thoroughly, and treating every bottle with respect keeps both people and property intact.
What are the potential health hazards of Allyl Methacrylate exposure?
Everyday Exposure in the Workplace
Allyl methacrylate is no stranger in factories handling plastics, adhesives, paints, or coatings. I’ve seen friends wear goggles and gloves every day just to shape or pour this stuff. What sticks out is how the chemical’s sharp, almost eye-watering smell lingers in the air, making even quick jobs uncomfortable. Breathing in these fumes can sting the nose and throat. For many who work on the floor or manage barrels, red, irritated skin grows common, especially if gloves tear or sleeves ride up.
Long-Term Effects on Health
Shortness of breath and cough aren’t just annoying side effects. Over time, repeated exposure can dig deeper. The lungs might start to lose some strength, and persistent inflammation can set the stage for respiratory problems that last decades. Even a single spill can cause serious burns. As anyone who’s watched a coworker dart to the eye-wash station knows too well, eye contact burns and blurs vision for days.
Laboratory studies echo what workers quietly complain about. According to research published by the National Institute for Occupational Safety and Health, exposure can trigger asthma-like symptoms and can sensitize the skin. Allergic reactions get worse with each repeated contact. Some people wind up reacting so badly after a few months on the job that they have to leave their positions entirely.
Concerns Beyond the Factory Floor
These health risks don’t end in the plant. Poor ventilation or improper disposal can let this chemical drift into outdoor air or into water streams. Communities living near chemical plants face measurable upticks in eye irritation, coughs, and in rare cases, chemical burns from accidental releases.
Stats from the Agency for Toxic Substances and Disease Registry show that workplace exposure isn’t the only concern: accidental spills and leaking drums contribute to broader public risk. Environmental groups push for routine soil and water checks, which helps, but cleanup lags behind leaks. The compound may break down under sunlight and heat, but not fast enough to keep it from harming wildlife or people in the area.
Practical Steps Toward Protection
Relying on personal protective equipment sets the first line of defense. I remember my uncle, a plant supervisor, waking up coughing after a mask leaked during a late maintenance shift. Routine mask fits and replacing worn gloves help, yet management’s commitment to regular air monitoring and safety training matters even more. Signs and training videos fade unless bosses set a clear expectation for reporting small spills and symptoms.
Upgrading ventilation makes a clear difference, especially in crowded workspaces. Portable air purifiers and local exhaust setups offer practical relief where permanent upgrades stall due to budget. Medical check-ups at least twice a year catch early symptoms and support claims for workplace modifications. Workers have shared stories of clinics catching lung irritation early, giving them a chance to move jobs or switch tasks.
Safe storage and disposal cut back on wider environmental risks. Spill kits near every workstation let teams tackle leaks before they spread. Earning certification from occupational health agencies acts like a reality check—the workplace cannot ignore repeated mistakes if an outside body audits them. For communities, eyes on local water and air readings, not just company statements, keep companies honest about what gets released.
Toward a Safer Future
Every chemical carries risks, but the hazards of allyl methacrylate linger well beyond a shift’s end. Long-lasting health and environmental threats call for more than routine safety posters. Real improvements grow from listening to workers’ stories, timely medical follow-ups, and holding companies to clear, publicly reported standards.
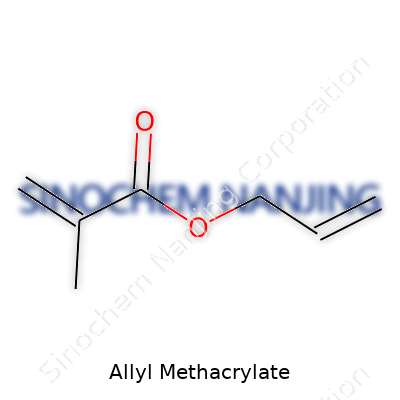
| Names | |
| Preferred IUPAC name | 3-methylbut-2-enoic acid allyl ester |
| Other names |
2-Propenoic acid, 2-methyl-, allyl ester Allyl 2-methyl-2-propenoate Allyl methacrylate Methacrylic acid, allyl ester AMA |
| Pronunciation | /ˈæl.ɪl mɛˈθæk.rə.leɪt/ |
| Identifiers | |
| CAS Number | 96-05-9 |
| Beilstein Reference | 87852 |
| ChEBI | CHEBI:88577 |
| ChEMBL | CHEMBL47478 |
| ChemSpider | 21513 |
| DrugBank | DB14036 |
| ECHA InfoCard | 03bfaea9-9a14-4e1a-9d5e-bb3c3c9f7d13 |
| EC Number | 203-379-2 |
| Gmelin Reference | 7947 |
| KEGG | C01770 |
| MeSH | D000581 |
| PubChem CID | 8776 |
| RTECS number | AS3150000 |
| UNII | V9F1UV845K |
| UN number | UN1098 |
| CompTox Dashboard (EPA) | DTXSID6043664 |
| Properties | |
| Chemical formula | C7H10O2 |
| Molar mass | 100.12 g/mol |
| Appearance | Colorless liquid |
| Odor | Strong, acrid. |
| Density | 0.936 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 1.95 |
| Vapor pressure | 3.2 mmHg (20 °C) |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | 6.69 |
| Magnetic susceptibility (χ) | -7.09·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.417 |
| Viscosity | 0.944 mPa·s (20 °C) |
| Dipole moment | 1.43 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 355.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -192.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2080 kJ/mol |
| Pharmacology | |
| ATC code | V09AX20 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H315, H317, H319, H331, H335, H400 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P273, P280, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378, P403+P235 |
| NFPA 704 (fire diamond) | 2-3-2-W |
| Flash point | 51 °F (11 °C) |
| Autoignition temperature | 346 °C (655 °F; 619 K) |
| Explosive limits | Explosive limits: 1.5–11% |
| Lethal dose or concentration | LD50 (oral, rat): 820 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1025 mg/kg (rat, oral) |
| NIOSH | SN4295000 |
| PEL (Permissible) | PEL: 1 ppm (5 mg/m³) |
| REL (Recommended) | 35 mg/m³ |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Allyl acetate Methacrylic acid Methacryloyl chloride Methyl methacrylate |

