Acrylic Acid: Examining Its Role, Impact, and Future in Industry and Society
Looking Back: How Acrylic Acid Came to Shape Modern Materials
Over a century ago, the German chemist Georg W. A. Kahlbaum first introduced acrylic acid into scientific literature. His modest synthesis methods look quaint now, but they opened the door to a compound that would make its presence felt from the 1930s onward, as industry demanded better plastics, coatings, and adhesives. The journey from a laboratory curiosity to a chemical staple mirrors the industrial world’s relentless push for smarter, tougher, and more versatile materials. Acrylic acid's development tracked with the rise of polymer science, serving as a backbone for products like superabsorbent polymers in baby diapers, dispersants, thickeners, and more. As makers demanded greater performance and lower cost, production methods evolved, shifting from risky acetylene-based processes to safer, more scalable oxidation of propylene, a byproduct of petroleum refining that remains the core feedstock today.
What Acrylic Acid Brings to the Table
Acrylic acid stands out for its simple structure—a carboxylic acid group hooked to a carbon-carbon double bond—yet it packs surprising power, reactivity, and adaptability. Anyone who has spent time in a lab knows its sharp, irritating odor, and the way its clear, slightly runny appearance gives few hints about the complexity below the surface. The double bond makes acrylic acid almost eager to react, latching onto itself and other monomers to form long chains. These chains form the heart of acrylic resins, which can be tuned to create everything from pressure-sensitive adhesives for labels to flexible yet tough paints favored by artists and engineers alike. What surprises some folks is just how much we rely on these products, whether tracking mud into our homes across stain-resistant carpets or using hygiene products that quietly soak up moisture for hours on end.
Physical & Chemical Profile: Not Flashy but Uncommonly Useful
On the surface, acrylic acid doesn’t draw much attention: colorless, with a biting smell that hints at its acidity and reactivity. Safety data reveals its volatility and corrosiveness, with a boiling point just above water’s and a tendency to polymerize on its own if left unchecked. Many in manufacturing know that even small exposures irritate skin and mucous membranes, a problem made worse by its tendency to vaporize. Its chemical structure opens the door for endless modification—esters, salts, and even grafting onto other polymers—giving rise to a legion of related products used in everyday life. Its simple formula belies how it serves as the foundation for industries that stretch from automotive to agriculture.
Not Just Chemistry—Technical Standards and Practical Labeling
The minute acrylic acid enters a supply chain, the rules get strict. Technical standards cover everything from water content and inhibitor levels to the purity required for medical or food packaging. The industry labels acrylic acid with numerous codes and trade names, reflecting its use in countless end products. Companies ship it in drums outfitted to prevent runaway polymerization; a little heat or a dash of contaminant can trigger an exothermic reaction, turning a routine delivery into a serious hazard. A workplace veteran quickly learns to double-check every label and lean on tried-and-true protocols, rather than chance a shortcut. Compliance isn’t about paperwork—it’s about protecting workers and entire operations from the unexpected.
How Science Tames Production: Getting Acrylic Acid at Scale
The leap from lab-scale chemistry to industrial synthesis pushes the limits of chemical engineering. Most acrylic acid is made by oxidizing propylene in catalyzed reactors, pumping out hundreds of thousands of tons each year. These processes favor high yield while minimizing byproducts and emissions. R&D teams constantly improve reactor designs, catalysts, and waste handling, trying to squeeze every percent of efficiency while dealing with the environmental and economic pressure mounted by regulators and the public alike. Moving toward more sustainable propylene sources, including bio-based routes, has gained some ground but faces challenges with feedstock cost, consistency, and logistics.
From Raw Material to Engine for Transformation: Chemistry in Action
What makes acrylic acid truly valuable is its place at the crossroads of chemical reactions. Its double bond prefers addition reactions, snapping together with other small molecules to form diverse families of acrylic polymers. Chemists can stitch on all kinds of functional groups to make products as soft as a child’s finger paint or as tough as a marine-grade sealant. Downstream modifications give rise to polyacrylates, acrylamide, and even cross-linked networks found in hydrogels and advanced coatings. This power for transformation explains the ever-growing roster of synonyms and trade names, as manufacturers leverage similar base reactions to crop up in markets as diverse as medical devices and advanced batteries.
Keeping People Safe: What Experience Teaches About Handling Acrylic Acid
Those who handle acrylic acid soon learn respect for its hazards. Inhalation and skin contact can lead to acute irritation, and there’s no shortcut around proper protective equipment and ventilation. Facilities rely on process automation to keep as few hands near open containers as possible. Emergency drills aren’t a sign of paranoia—they’re a lesson learned from incidents where simple mistakes led to fires, toxic exposure, or environmental releases. Ongoing training beats out complacency, and firms that cut corners often pay in lawsuits and community backlash. The history of chemical safety standards around acrylic acid reads like a casebook study in how tragedy drives regulation—from strict storage temperatures to redundant monitoring systems.
Where Acrylic Acid Makes a Difference: Life Beyond the Lab
Pick a modern convenience, and there’s a decent chance acrylic acid plays a behind-the-scenes role. Its biggest market lies in superabsorbent polymers, the backbone of disposable diapers and incontinence products that allow millions to live with dignity and ease. Water treatment plants rely on acrylate-based flocculants for clearer rivers and safer drinking water. Paints, adhesives, and specialty coatings—each depends on acrylic acid’s versatility, providing long-lasting finishes in everything from aircraft hangars to living room furniture. The agricultural sector values acrylic polymers as soil conditioners and slow-release agents, helping stretch precious water in drought-prone fields. Even the textile industry leans on it for soft, colorfast, and easy-care fabrics that stand up to daily life. These aren’t distant, abstract benefits—they land in homes, hospitals, and workplaces every day.
Looking Ahead: Pushing the Boundaries Through Research & Development
R&D in acrylic acid continues to push boundaries, often in response to the industrial world’s hunger for sustainability and efficiency. Teams invest heavily in greener pathways—whether through bio-based feedstocks, lower-emission catalysts, or recycling strategies for acrylate polymers. Startups and established firms alike race to design polymers that break down faster or use less energy across their life cycles. Academic labs study new catalysts aimed at pushing selectivity and yield higher, squeezing value out of every barrel of propylene. Open collaboration between research institutions and manufacturers accelerates the process, but pressure to balance cost, safety, and environmental friendliness keeps innovation on edge.
Understanding Risk: Toxicology and the Push for Safer Use
Toxicity studies on acrylic acid began decades ago, prompted by early reports of skin and eye irritation among exposed workers. Chronic inhalation brings respiratory distress; accidental ingestion, thankfully rare, harms stomach and kidneys. Regulators around the world list it as a hazardous substance, though most studies suggest it breaks down rapidly in environmental conditions—reducing the risk of long-term bioaccumulation. Still, advocacy groups and researchers push for better risk assessments, particularly as acrylic acid derivatives cycle through the environment or show up in recycled goods. Transparency around exposures, coupled with stronger workforce protections, can close the gaps exposed by each safety lapse.
Future Prospects: Navigating Change and Responsibility
Acrylic acid’s future looks both promising and fraught with challenge. Industry sees opportunities in renewable resources, advanced recycling, and next-generation formulations that deliver performance with a lighter environmental footprint. Market demand faces the test of tighter health and environmental rules, particularly in Europe and parts of Asia where consumer skepticism and political oversight run high. Companies lead with stories of cleaner processes and safer products, betting that public trust won’t wear thin under the weight of past chemical scares. As society demands accountability, the acrylic acid sector faces a reckoning; real progress will depend on persistent improvement, transparent practices, and a willingness to rethink chemical stewardship from the ground up. Scientists, workers, and end users all stand to gain if lessons from history guide choices for the decades ahead.
What is acrylic acid used for?
Everyday Products That Depend on Acrylic Acid
Acrylic acid might not show up on grocery lists, but it’s one of those chemical ingredients you bump into every day. At home, people use super-absorbent disposable diapers, soft tissues, and slick paints. Acrylic acid makes these possible. Large manufacturers transform it into polymers that soak up liquid and keep spills from spreading. Think of it next time a diaper holds wetness all night or a paper towel dries a stubborn mess.
Clean Homes and Tough Surfaces
Scrubbing the bathroom or kitchen, the cleaning liquid foams and lifts grime. Many household cleaners owe their thick, clingy feel to acrylic acid. It builds viscosity, gives gels and detergents that satisfying texture, and helps dirt break apart. Repairing scratched paint or roughened cement? Acrylic acid-based coatings step in with lasting strength. Industrial roofing and waterproofing materials rely on these coatings to resist harsh weather and sun. Painters and contractors value acrylic acid resins for fast-drying layers and tough finishes.
Personal Care Matters
Many people have smeared on a lotion that promises silky skin. The smoothness and hydration often come from acrylate polymers, born from acrylic acid. Thick creams, styling gels, and even sunscreens use it to deliver a certain slip and softness. Without it, the feeling would be runny or gritty. Dentists and doctors tap into acrylic acid in dental fillings, tissue adhesives, and even contact lenses — it helps craft materials stable enough for the human body, yet gentle enough for sensitive use.
Agriculture and Water Treatment
Crop fields and gardens all need water. Farmers use water-retaining polymers made from acrylic acid in powders and crystals sprinkled in soil. They trap moisture, keeping roots hydrated longer between rains. Acrylic acid also finds its way into municipal water treatment. It helps remove pollutants and keeps sludge from clumping, so water stays cleaner. Industrial waste streams benefit when acrylic acid-based solutions bind with heavy metals and unwanted particles.
The Environmental Question
There’s a catch — making acrylic acid demands fossil fuels and churns out emissions. Communities near factories worry about safety and air quality. Many scientists are exploring ways to make acrylic acid greener. One approach involves using corn, sugar, or plant waste instead of oil and gas. Early efforts in Europe and North America show that fermentation and biological processes can create acrylic acid with a lower carbon footprint. In my experience growing up in a town with both chemical plants and farmland, I learned that new tech often starts small, but every factory that shifts to bio-based acrylic acid can calm neighbors and protect air and soil.
Choosing Better, Living Smarter
Acrylic acid links modern life to science, industry, and environmental responsibility. It’s woven into everything from home repairs to medical care and farm fields. Companies and communities have a shot at cleaner production by pushing for renewable technology, updating old equipment, and enforcing clear safety rules. Shoppers can stay aware by reading ingredient lists and asking brands about sourcing. Honest conversations between chemical makers, local families, and policymakers help steer change in the right direction. Acrylic acid tells a story of progress—one that everyone has a hand in shaping.
What are the main properties of acrylic acid?
A Look at What Makes Acrylic Acid Stand Out
Acrylic acid shows up in a surprising number of everyday products, but most people probably never stop to think about what it really is. It’s a colorless liquid, carrying a sharp, acrid smell. For folks who’ve worked in labs or chemical factories, that smell lingers in memory: a warning that this isn’t the safest stuff to handle barehanded. Touching acrylic acid can lead to skin burns; breathing in its vapors may irritate lungs. The hazardous side draws plenty of attention from regulators and workers alike.
Most of its danger comes from the double whammy of being both acidic and able to react with lots of chemicals. It belongs to the group called carboxylic acids: you find these in vinegar and citrus juice, but acrylic acid adds a double bond—making it more likely to participate in quick chemical reactions. This is why manufacturers like it so much. The double bond enables reactions to create plastics, paints, adhesives, and absorbent materials. Acrylic acid sits at the crossroads of utility and risk: necessary for industry, but nobody should take its handling lightly.
What Makes Acrylic Acid Useful?
The thing about acrylic acid is its ability to turn into polymers. Anyone who has pulled on a disposable diaper or a hydrogel medical pad has met its handiwork. Water swells these materials thanks to cross-linked polyacrylates, built from acrylic acid molecules. This swelling power means more absorbency for hygiene products and better moisture-handling for gardening gels. Manufacturers count on its reactivity: that double bond comes alive during polymerization, making it a favorite for designers of new plastics.
Even though it carries risks, acrylic acid remains easy to customize. Chemists can tweak how it forms chains, whether they want something tough, flexible, sticky, or absorbent. Its structure helps producers dial in the right texture or properties for thousands of different products. That sort of flexibility helped the material sneak into paints, textiles, and coatings without most of the public ever noticing.
Handling Risks and Safety
Acrylic acid isn’t friendly to amateur chemists. Spill some on the floor, and you’ll notice the fumes almost right away. Skin contact leads to irritation and can cause burns if ignored. Once someone realizes how much of this chemical flows through supply chains, safety becomes a top concern. Workers and communities living near factories ask for better ventilation, sealed containers, and spill control plans. Accidents and leaks highlight the importance of hazard training and protective gear.
From personal experience, using acrylic acid in a research lab reminds me that no shortcut is worth the risk. Proper gloves, ventilation, and storage make the difference between a routine day and a dangerous emergency. Regulations set by OSHA and EPA give guidelines, but real protection comes from taking those standards seriously in daily practice.
Looking Forward
Worldwide demand keeps climbing, especially as new applications emerge for disposable goods and specialty polymers. Still, the industry faces pressure to cut emissions, reduce waste, and find safer workarounds. One way forward comes through greener chemistry. Some labs explore routes that use renewable feedstocks, aiming to reduce reliance on fossil fuels. Others push for tighter containment, recycling leftover chemicals, and shrinking the footprint of waste.
Acrylic acid matters because it sits at the intersection of chemical usefulness and human safety. Knowing its hazards, strengths, and how to manage both lets industry deliver the astounding variety of modern products while keeping workers and neighbors safer.
Is acrylic acid hazardous or toxic?
Looking Beneath the Surface
Acrylic acid shows up in places most people never consider. It’s found in paints, adhesives, diapers, and water treatment products. This stuff isn’t just another chemical in a big warehouse; it shapes everyday products and makes modern life easier. Here, convenience and risk bump into each other. Anyone working with or living near acrylic acid production probably wonders: is it dangerous?
Health Concerns That Deserve Attention
Coming from a background in industrial safety, I've seen how overlooked hazards end up collecting dust in safety briefings. Acrylic acid’s sharp smell hits the nose before it even touches the skin. It irritates eyes and airways right away. I remember a young coworker once dismissed a warning about vapors—he learned the hard way when his eyes started burning after a pipe leak. Medical research confirms this: even brief exposure can leave people coughing and struggling to catch their breath.
Direct contact leaves red, blistered skin, and breathing in too much may cause headaches, drowsiness, or even lung damage. Lab workers and factory operators carry the biggest burden. The US National Institute for Occupational Safety and Health (NIOSH) and OSHA both rate acrylic acid as hazardous at certain levels. Chronic exposure, even in small doses, can stress the liver and kidneys over time.
Folks at home probably don’t handle drums of acrylic acid. Still, accidents happen. Improper disposal or storage brings risks closer to everyone, especially kids, who may not recognize the dangers.
Environmental Impact Still Matters
After working near a chemical plant for years, stories circulate quickly if something spills into the local river. Acrylic acid, if dumped in large amounts, can pollute water and soil. Even small leaks matter, hurting fish and aquatic life. The Environmental Protection Agency (EPA) monitors acrylic acid closely for this very reason. Once it’s out in the open, it breaks down quickly under sunlight and air, but before then, it’s a potent irritant. Landfills are another trouble spot, as acrylic acid doesn’t mix well with other chemicals.
Weighing Practical Steps Forward
The cavalry isn’t coming. It falls on chemical companies, regulators, and local communities to be vigilant. Employers need to give proper masking equipment, good ventilation, and ongoing health checks. Regular drills make a real difference when something goes wrong. In one shop I visited, the difference between minor irritation and a hospital trip came down to one person knowing where the emergency shower was.
At the policy level, strong rules keep problems contained. Enforcement by OSHA and EPA keeps companies honest. Fines hurt, but the bigger incentive comes through a strong work culture that values safety beyond checklists.
For the public, clear labels and proper household disposal explain the story that a chemical plant’s fence cannot. Town halls, school programs, and responsible chemical sales work better than hoping for the best. It pays to ask questions and expect answers from local factories or suppliers.
Balance and Respect, Not Panic
Acrylic acid is no doomsday threat, but treating it with casual indifference creates problems. It earns both respect and care because health and the environment hang in the balance. Whether in a giant plant or a corner workshop, small steps—worn goggles, ventilation fans, informed choices—carry the weight of safety, not just individual comfort. Product innovation shouldn’t come at the cost of human health. Safety decisions close to the ground turn a risky tool into a manageable part of life.
How should acrylic acid be stored and handled?
Don’t Underestimate Acrylic Acid’s Fire Hazard
Acrylic acid brings a punch to industries that use paints, adhesives, and plastics, but the risks it carries are overlooked too often. While I’ve spent time walking through chemical warehouses, one of the clearest lessons is this: acrylic acid acts like a loaded gun if treated carelessly. Its boiling point sits lower than you’d expect, and it catches fire much more easily than people assume. Once, I watched a drum stored a few steps from a loading dock—sun pouring in all morning, warming the metal. Even with ventilation, you could smell the tension. Too much heat, a bit of static, and that story could’ve ended badly.
The State of the Container Matters—A Lot
Not all containers give the same protection. The acid eats at some plastics over time, and cheap seals let fumes slip into the air. It’s straightforward: stainless steel or lined drums hold the chemical for the long run, so investing in these pays off. I remember one facility swapping to poly-lined drums because of cost, then pulling them out three months later when seals started leaking. A leak from one drum opens up real risk for both the crew and the neighborhood.
Temperature Control Isn’t Just For Show
Acrylic acid keeps stable if stored under 25°C. Above that, you start walking a thin line. I can’t count how many times I’ve heard, “It’s just a couple of degrees above target.” Those small slip-ups add up. If it gets too warm, polymerization kicks in, sometimes without warning, leading to heat and pressure that bulky containers can’t hold. Cooling rooms with temperature alarms and backup fans play a bigger role than managers often think. Cutting corners to save on electricity doesn’t work—one accident costs more than a year’s worth of energy.
Human Training Puts Out Fires Before They Start
Relying on a storage plan means little if folks don’t recognize what they’re handling. In every plant I’ve visited, the sites with regular safety briefings had fewer close calls. I’ve seen what happens when you bring in a temp to shift barrels, and they miss the red danger label. Wearing gloves, goggles, and a face shield gets old, but a splash to the skin will leave a mark for life. It helps to remind everyone on the crew—seasoned or not—how fast things turn wrong.
Separate Storage: Neighbor Chemicals Can Turn Deadly
Mixing acrylic acid with oxidizers or amines turns a regular shift into a chemical nightmare. Shelf space costs money, but separating acrylic acid from chemicals with which it reacts isn’t optional. During one audit, I saw someone stack bleach and acrylic acid five feet apart in a new warehouse. They fixed it fast, but complacency breeds disaster.
Venting and Spills: Nobody’s Perfect
Fume hoods and local exhaust fans draw away vapors before they drift to ignition sources. I once watched a team use just an open door for ventilation—a shortcut that led to complaints about headaches and a near-miss with a delivery truck’s spark. Spill kits need to remain close, and every worker needs to know how to use them, not just the full-time staff. Small leaks often go ignored until someone slips or starts coughing.
Invest in Better Practice, Not Just More Rules
Acrylic acid doesn’t give second chances. Reliable storage containers, real temperature checks, practical staff training, careful chemical separation, and solid venting stop disasters before they start. Cutting corners or relying only on paperwork puts both business and people in harm’s way.
What are the common grades or purity levels of acrylic acid available?
Why Purity Matters in Acrylic Acid
Everyday products like diapers, coatings, adhesives, and superabsorbent polymers all trace back to acrylic acid. If you peel back the layers of these industries, you’ll find decisions about quality start with the grade of acrylic acid in use. It’s not just a number on a label—the choice shapes everything from stability of final goods to the safety of the workplace. Pure acrylic acid can be clear and colorless, but if you let impurities creep in, the whole process can run off-track. Over time, I’ve seen small differences in purity create big messes on production lines and headaches for quality managers.
Main Grades of Acrylic Acid on the Market
Glacial Acrylic Acid counts as the most common form used in manufacturing. It usually carries purity above 99%, clear in appearance. Glacial doesn’t mean it’s cold—it’s just the industry way of saying this material is free from water. Even trace moisture can throw off chemical reactions, so this level of purity keeps things predictable in polymerization or esterification. Having worked with plant engineers, I’ve watched them double-check specifications for glacial quality, knowing any deviation can compromise entire batches of product.
Technical Grade Acrylic Acid comes with lower purity, often around 94–98%. This grade often finds its way into less demanding applications, like certain cleaners or agricultural chemicals. Technical grade carries more residual acids or colored impurities, things that would stand out in high-end coatings or hygiene products. In operations where cost matters more than pristine performance, technical can get the job done. But I’ve seen customers disappointed when they missed this distinction, winding up with sticky finishes or odd odors in finished goods.
High Purity or Ultra-Pure Acrylic Acid often exceeds 99.5% purity, targeting electronic or pharmaceutical industries. Here, trace metals, aldehydes, or other byproducts might only exist at parts-per-million levels. Even a microscopic contaminant could wreck sensitive applications. These grades demand rigorous packaging and transport, with constant checks along the way. In my experience, teams handling high purity grades almost treat them like medical supplies—everyone’s alert to cross-contamination.
Challenges in Achieving Purity
Bringing acrylic acid up to these standards isn’t a simple job. Distillation removes water, but separating out minor organic impurities like propionic acid or acetic acid often stumps even seasoned plant operators. Small oversights—the temperature a distillation column runs at, or how long it sits in storage—can tip the scale from glacial back to technical grade. I’ve heard plenty of stories where batches sat too long in a tank, picking up color or losing clarity, forcing a restart.
Finding the Right Grade for the Task
Most everyday uses fall on some spectrum between technical and glacial grades. Shifting to higher purity usually brings higher costs from extra distillation, tighter storage controls, and careful transport. Lower grades appeal when budget wins over performance, but cutting corners creates hidden risks. Manufacturers picking grades for new products do themselves a favor by running hands-on tests before locking in specifications. Building relationships with suppliers helps, making sure each shipment meets expectations and doesn’t throw off production lines.
Raising the Bar with Best Practices
Training plant staff, investing in real-time monitoring, and demanding full certificates of analysis all support getting the right purity. Anyone overseeing a facility where acrylic acid flows knows routine lab checks catch problems early. Robust storage—using inert gas blankets, controlling heat, minimizing air exposure—keeps product from degrading before use. Even warehouse staff can spot red flags, like yellowing containers or strong odors pointing to decomposition. By setting standards and sticking to them, teams across industries avoid the dangers of guesswork.
| Names | |
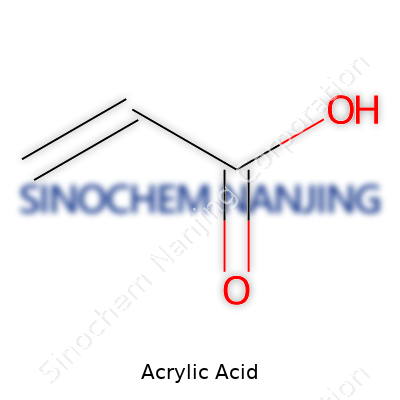
| Preferred IUPAC name | prop-2-enoic acid |
| Other names |
2-Propenoic acid Acroleic acid Vinylformic acid Propenoic acid Acrylic acid solution |
| Pronunciation | /əˈkrɪlɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 79-10-7 |
| Beilstein Reference | 635102 |
| ChEBI | CHEBI:30751 |
| ChEMBL | CHEMBL277500 |
| ChemSpider | 528 |
| DrugBank | DB03193 |
| ECHA InfoCard | 100.003.278 |
| EC Number | EC 201-177-9 |
| Gmelin Reference | 821 |
| KEGG | C00483 |
| MeSH | D001017 |
| PubChem CID | 657 |
| RTECS number | AT0700000 |
| UNII | 1DIY18LUO0 |
| UN number | UN2218 |
| Properties | |
| Chemical formula | C3H4O2 |
| Molar mass | 72.06 g/mol |
| Appearance | Clear, colorless liquid with a pungent odor |
| Odor | Pungent |
| Density | 1.051 g/cm³ |
| Solubility in water | miscible |
| log P | 0.35 |
| Vapor pressure | 4 mmHg (20°C) |
| Acidity (pKa) | 4.25 |
| Basicity (pKb) | pKb ≈ 9.25 |
| Magnetic susceptibility (χ) | -12.2e-6 |
| Refractive index (nD) | 1.422 |
| Viscosity | 1.3 mPa·s (at 25°C) |
| Dipole moment | 1.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 86.74 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -486.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -486.60 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D01AE22 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS02,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H314, H332, H335 |
| Precautionary statements | H314, H332, H312, H302, H335, H319, H317, H402, H412 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 124 °F (51.1 °C) |
| Autoignition temperature | 400 °C (752 °F) |
| Explosive limits | 2.3% - 12.5% |
| Lethal dose or concentration | LD50 oral rat 340 mg/kg |
| LD50 (median dose) | LD50 (median dose): 340 mg/kg (oral, rat) |
| NIOSH | ATY35 |
| PEL (Permissible) | PEL: 2 ppm |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 750 ppm |
| Related compounds | |
| Related compounds |
Acrylate Acrylonitrile Polyacrylic acid Methacrylic acid Propionic acid Acrylic anhydride |

