The Real Story on Acrylamide: What Experience and Facts Show Us
How Acrylamide Entered the Picture
Acrylamide did not just appear overnight in chemistry labs or processed food headlines. In the 1950s, researchers discovered it during efforts to boost polymer science for industries like water purification and paper-making. Early work focused on its ability to form strong gels. As the years went on, manufacturers started using it more to create polyacrylamide, a material that helps clean up drinking water, improves oil recovery, and even serves needs in cosmetics. Over time, though, people came to see another side of the story: acrylamide can form naturally in foods cooked at high temperatures, like French fries, potato chips, and toast. Food scientists and public health folks have spent decades tracking what this means for health.
Decoding Acrylamide's Make-Up
If you look at acrylamide through the lens of chemistry, it's a small molecule, with the formula C3H5NO. It shows up as white, odorless crystals, and dissolves easily in water and ethanol. It's reactive, thanks to a double bond between two carbon atoms and an amide group that can latch onto other molecules during polymerization. That quality gives it the ability to build long chains, or polymers, with ease. Producers stick to specifications for things like melting point, residual solvents, and purity, because off-spec acrylamide plays havoc in manufacturing. Labeling now reflects health concerns, spelling out warnings about toxicity, especially after certain regions tightened up requirements based on animal studies raising cancer concerns.
How Chemists Prepare Acrylamide in Practice
Scientists have a standard approach: acrylamide springs from the hydration of acrylonitrile, usually with copper-based catalysts under basic conditions. The process happens at temperatures and pressures that balance safety with yield. Over the years, companies have tweaked this route, not just to scale up output but to minimize byproducts that could cause trouble downstream. These improvements did not come cheap or easy. Operators mix skill and know-how, because controlling all the variables — pH, temperature, catalyst regeneration — proves tricky in big reactors. Other side routes include enzyme-aided production, but those rarely compete cost-wise.
What Acrylamide Turns Into and Why It Matters
In lab settings, acrylamide becomes a workhorse for chemical reactions. Its double bond makes it eager for polymerization, letting it snap together into chains with water as a base, giving polyacrylamide its unique gelling power. Chemists can add co-monomers or modifiers to tweak things like strength or water absorbency. Additives alter flocculation rates for water treatment, or change viscosity for oilfields. While most focus lands on polymerizing acrylamide, reactions like Michael addition — where a nucleophile attacks the double bond — open doors for modifying its core structure. These avenues show up in specialty gels, biomedical carriers, and inventive materials only possible over the past few decades.
Other Names and Labels for Acrylamide
Chemists and industries give acrylamide a few aliases: 2-propenamide, acrylic amide, or even prop-2-enamide show up in journals and supply catalogs. These names all point to the same molecular backbone. Some food and public health authorities simply call it by its main name to keep messaging clear, since too many nicknames breed confusion outside the lab.
Staying Safe with Acrylamide: Hard Lessons Learned
Worker safety lessons came hard in the early years. Handling acrylamide without proper gloves, goggles, or ventilation led to reports of nerve damage and skin irritation. Regulations in regions like the EU and US pushed for closed handling, well-marked containers, and training beyond basic lab safety. Acrylamide easily slips through gloves and skin barriers. People running reactors or packing polyacrylamide drums get regular blood checks in some plants. Companies install scrubbers to capture dust or vapor. Wastewater clean-up prevents accidental releases since fish and aquatic life take a hit even at low exposure. These standards don’t come from bureaucracy alone — they reflect real, sometimes tragic mistakes made in workshops or factories decades ago.
Acrylamide in Action Across Industries
Everyday water you drink likely passed through polyacrylamide in a treatment plant. Municipal and industrial water processors rely on these polymers to settle out suspended solids. Oil fields use thickened polyacrylamide to wrestle more crude from hard-to-reach reservoirs, propping up global production in ways few realize. Paper mills squeeze out better product with fewer tree fibers, cutting waste. In cosmetics, acrylamide polymers thicken creams and lotions, making them feel rich and smooth. Food science carved out its own focus after acrylamide turned up in cooked potatoes and bread, sparking global efforts to find cooking methods or ingredients that cut levels down. Labs use acrylamide for electrophoresis gels, separating proteins or DNA fragments in the kind of tests at the heart of medical diagnostics and biological research. These wide uses keep demand high, pushing researchers to keep improving both the product and its safety profile.
From Lab Curiosity to Health Headline: Crunching The Toxicity Data
Years back, nobody thought twice about acrylamide in food. Medical journals and government agencies changed that after animal studies showed links between high doses and cancer or nerve effects. Rats fed huge quantities developed tumors, spurring agencies like the International Agency for Research on Cancer to flag acrylamide as a probable human carcinogen. Still, actual risk at levels in cooked food sparked rows between toxicologists, nutritionists, and industry. Epidemiological studies in people jump between unclear links or minimal risk, while animal evidence looks much stronger. To anyone cooking at home, the advice comes down to straightforward steps: fry at lower temperatures, toast bread to gold rather than brown, soak potatoes before frying. Some food companies switched to potato varieties lower in the precursor asparagine, or tweaked recipes to control browning and acrylamide formation. Measuring levels in real-world food keeps improving as new analytical methods roll out. The gap between lab data and real grocery store risk still stirs debate, but efforts to cut down exposure reflect a clear respect for long-term health.
The Race for Better Ways: Research, Development, and What's Next
Every scientific advance on acrylamide, from safer manufacturing to detection in food, comes out of real worry for worker and public health. Polymer chemistry keeps inching forward, finding ways to reduce residual monomer inside finished polyacrylamide so that users face less risk long after the product ships. Food scientists chase new enzymes or processing tricks to slash acrylamide levels without gutting flavor or crunch, because nobody wants limp fries in the name of safety. Regulatory agencies fund long-term studies to pin down real-life harm in populations, combing through cancer statistics. Chemists experiment with greener catalysts and energy-saving synthesis routes, looking for scalable options that keep costs level. Labs building more sensitive tests expose hidden acrylamide in places like coffee and cereals, guiding manufacturers and food safety experts. Ethical sourcing, sustainable production, and open results let regular people — not just scientists — see progress up close.
Looking Forward: What Acrylamide's Future Could Hold
Acrylamide will not vanish from industry or food any time soon. Water needs cleaning, old oil wells do not pump themselves, and demand for affordable food keeps factories humming. The most realistic path lies in incremental change: sharper analytic tools, more transparent reporting, and steady pressure on producers to keep pushing levels downward where possible. Innovation comes from melding green chemistry with industrial scale, rolling out new catalysts or process tweaks that cut risk at the source. Consumer awareness calls the shots in food — as people get sharper on reading labels and asking questions, companies answer with safer, clearer products. In medicine and research, polyacrylamide gels undergird modern diagnostics, making ongoing safety checks essential. Instead of silver bullets, the story of acrylamide reminds us that progress stacks up in steps. Trust builds through plain facts, honest mistakes, and the determination to keep improving what we know and how we act.
What is acrylamide used for?
Where You’ll Find Acrylamide
Acrylamide gets a lot of attention these days, often for its link to food and potential health risks. Digging deeper, its usefulness shows up all over modern industries. Acrylamide plays a supporting role in everything from water treatment plants to the production of diapers. Its flexibility comes from the way it transforms into polyacrylamide, a material that can absorb water and help bind things together.
Behind the Scenes in Water Treatment
Anyone who cares about clean drinking water should look at how acrylamide-based polymers make filtering possible. Municipal plants use these compounds to help dirt and particles clump together. Once bound up, it gets easier to pull out unwanted bits from the water supply. That means faster and more efficient purification, which has a real effect on public health across the world. I’ve talked with operators who appreciate how these polymers keep up with tough regulations and rising demand.
The Power of Absorption in Everyday Products
Parents wouldn't go far without diapers or disposable hygiene products, and acrylamide polymers work quietly inside those products. The gel inside a diaper needs to lock in moisture fast. Polyacrylamide can absorb several times its own weight in liquid. Beyond hygiene, gardeners sometimes use polyacrylamide in soil conditioners to help hold water and improve drought resistance. In regions with low rainfall, this means better crop survival and smarter use of resources.
Building Roads and Wells
Acrylamide polymers don’t just stay in laboratories or homes. They make their way to roads and oilfields where they help with drilling and construction. In tunneling or building foundations, the material stabilizes loose soil. This can prevent settling and make heavy projects safer. In oil recovery, adding these polymers to water pumped underground can improve the amount of oil pulled out of wells. That sort of efficiency draws the interest of engineers looking to squeeze more from old wells and extend our current energy supplies.
Risks That Deserve Attention
For all its uses, acrylamide comes with tough questions about health and safety. The World Health Organization notes that exposure in large amounts can affect the nervous system and even raise cancer risk in lab studies. In food, acrylamide starts forming in some starchy foods cooked at high temperatures—think crispy fries, toast, or potato chips. Consumers should know why some governments and food producers keep looking for ways to cut those levels, such as lowering frying temperatures or changing recipes.
Responsible Approaches
Industries using acrylamide need close controls in place. Strong training for workers, upgraded protective gear, and regular monitoring can all help reduce workplace risks. In the public sphere, food science offers alternatives like switching out ingredients or adjusting cooking times to limit acrylamide in the foods people love. Public health leaders and manufacturers have started investing in education and research to help people understand and manage this risk.
Moving Forward
Acrylamide isn’t going away, and its practical uses touch lives every day. All the same, everyone involved—from engineers to parents to scientists—benefits from clear, fact-based information about how exposure happens and how to reduce it. Solutions grow from working together on safer processes, honest labeling, and more transparent communication. Trust comes from action and open talk about these chemicals that shape our world.
Is acrylamide toxic or hazardous to health?
What We Know About Acrylamide
Acrylamide comes up a lot when folks talk about cooking methods at home or the risks involved with certain foods and industrial environments. Basic chemistry tells us it forms in starchy foods—think fries or toast—when cooked at high temperatures, like frying or baking. Most people don't realize it also crops up in many industrial applications, from water treatment plants to paper manufacturing. Anyone curious about food safety or workplace hazards needs to stay up to date on acrylamide facts.
How Acrylamide Impacts Health
Plenty of studies show acrylamide can cause cancer in laboratory animals when they’re exposed to high doses. The World Health Organization and the American Cancer Society both point to this risk, but they also stress that animal studies don’t always translate directly to people. No large-scale studies in humans have yet shown a clear cancer link caused by day-to-day exposure from cooked foods.
Still, research on workers in industries that deal with acrylamide dust or solutions shows the need for caution. These workers often report skin irritation, neurological symptoms, and, in rare cases, problems with fertility. The U.S. Environmental Protection Agency has listed acrylamide as a probable human carcinogen, which means people shouldn’t dismiss prolonged, high-level contact with it.
Why the Way We Cook and Work Matters
Each time I browned a slice of bread or waited for fries to crisp up in the oven, acrylamide formation wasn’t top of mind until I started reading up on food science years ago. Little habits, like preferring darker roasted coffee or letting toast get a bit too crispy, can raise exposure. But for most people who eat a balanced diet, this exposure stays well below the levels scientists have found risky in animals.
In the workplace, the story changes. Factories and plants handle large amounts of this compound. Workers who breathe in dust or absorb it through their skin need proper protection. It’s not about panic, but about the right equipment and safety checks. Hearing stories from friends in those industries opened my eyes to just how vigilant people have to be.
Reducing Risk: Practical Steps
On the home front, simple choices help. Go for golden-yellow potatoes instead of letting them get too brown. Try boiling or steaming more often, mix up your grains, and eat plenty of fresh fruits and vegetables. Keeping an eye on cooking temperature makes a big difference. Bread and potatoes taste great when they’re not charring.
For those in industries, employers must provide safety training and personal protective equipment, and regular monitoring of acrylamide levels in the air and on surfaces. Open communication between management and staff, along with medical check-ups, keeps everyone safer.
What Science Recommends
Researchers still look for stronger answers about how much acrylamide people can tolerate without harm. In the meantime, most health agencies recommend a precautionary approach—reduce exposure where possible, keep up to date with guidance from health authorities, and pay attention to new research as it emerges. Health, after all, isn’t just about what we know now, but how willing we are to adapt as new facts come in.
How should acrylamide be stored and handled safely?
A Tough Chemical with Risks Scientists Can’t Ignore
Acrylamide is tough stuff. People use it for making polyacrylamide gels, treating wastewater, and plenty of industrial processes. But this powder or colorless liquid has a dangerous side. It carries health risks—skin contact, inhalation, even accidental eating—if people skip the right steps. For anyone who cares about safety, keeping acrylamide locked down and properly handled should never just be an afterthought.
Easy Steps Count for a Lot
Once, in college, I watched a lab technician shrug off a spill and wipe it up with a random cloth, skin barely covered. Lucky for him, nothing happened. But he rolled the dice. Acrylamide acts quickly through skin or lungs. Repeated exposure can tangle up nerves, cause tingling in fingers, and might even push someone closer to cancer, according to the National Institute for Occupational Safety and Health (NIOSH). Treating acrylamide with respect means suiting up—always use nitrile gloves, eye protection, a well-fitted lab coat, and avoid touching anything you don’t need to. Fume hoods should be a steady companion when weighing or mixing solutions.
Not All Containers Work—Pick Carefully
Forget about reusing coffee jars or storing hazards in mystery bottles. Acrylamide needs an air-tight, well-marked container—preferably one with a screw cap and chemical-resistant material like HDPE or glass. Keeping it away from heat, sunlight, and water stops it from breaking down or releasing toxic fumes. I’ve seen enough confusion in crowded chemical shelves to know how easy it is to grab the wrong jar. Good labeling—bright, bold, clear—saves a lot of loyalty to memory. A faded label risks expensive mistakes.
Temperature Tells the Story
Room temperature might work for a few hours, but acrylamide lasts longer in a cool, dry place. Heat speeds up its breakdown and can turn a safe powder into a messy hazard. Experienced lab techs will tuck acrylamide into dedicated chemical fridges, not next to solvents or acids, because cross-contamination has ruined more than one routine workday. Store it separately, keep moisture out, and check the expiration date. Never underestimate what an old or clumped-up bottle can do to an experiment.
Training Faces Real-World Problems
Training matters more than protocols written in a thick binder. People need to see real-life mistakes and solutions. Some of the best safety routines grow from discussions about what almost went wrong. Younger lab members watch senior staff wash their hands—before and after using the chemical—and start copying that habit. Spill kits, clear step-by-step emergency guides, and regular drills make a difference. A dry run with absorbent powder, shared cleaning gear, and non-panic teamwork makes the response to real spills cleaner and swifter.
Disposing of Waste Means Thinking Ahead
Pouring acrylamide down the drain is simply out of the question. Local hazardous waste rules almost always call for sealed, labeled waste bottles and clear logs of what goes in. In my lab, staff kept a calendar next to the chemical disposal area, so nobody forgot pick-up days or left problematic waste waiting too long. Sensible precaution beats shortcutting every time. Lab workers protect not just themselves but the ecosystem around them.
Small Habits Make the Big Difference
Safe handling of acrylamide is less about grand gestures and more about habits—checking gloves for holes, reviewing labels each use, scrubbing up, storing everything correctly. People who work with acrylamide share quiet responsibility with each other, never trusting luck or shortcuts. Setting up safety at the start saves headaches at the end. Protecting health, research outcomes, and the wider environment all starts with these daily, careful steps.
What are the main safety precautions when working with acrylamide?
Direct Risks Nobody Should Ignore
Acrylamide has become a staple for researchers building gels, but its toxic reputation is well-earned. Health agencies point to strong evidence showing cancer risks in animals and classify acrylamide as a probable human carcinogen. It’s also a neurotoxin, meaning too much exposure harms nerves. My early lab days drove this home: headaches and tingling should not follow a workday. There’s never a good reason to gamble with persistent chemicals like acrylamide—your body does not get better at handling this level of hazard over time.
Practical Ways to Stay Safe
Handling powdered acrylamide or its liquid monomers, skip the open bench. Use a certified chemical fume hood. Even carefully pouring acrylamide mixes can send invisible dust or vapors into the air, and fume hoods handle those well. Goggles, a snug-fitted lab coat, and nitrile gloves form your real armor. I still recall colleagues who believed “double gloving” solves everything—yet acrylamide can slip through certain glove materials. Nitrile or neoprene outperforms latex here, a small but crucial detail.
Don’t underestimate contaminated surfaces. Pouring, mixing, or even weighing acrylamide leaves spots you can’t see. I saw spills on benchtops that stayed sticky for weeks because no one treated them as urgent. I always recommend lining the workspace with absorbent pads and wiping surfaces with wet paper towels after every use. Toss those towels with gloves right into a specially-marked hazardous waste bin.
Know Your Chemical Storage and Labeling
Dry acrylamide easily absorbs moisture from air, so cap containers tightly and stash them in cool, well-ventilated cabinets away from heat or sunlight. Label every bottle with purchase and open dates, chemical name, and warning signs. I find this routine protects everyone in the lab. If you inherit someone else’s supplies, treating dusty or yellowing powder as dangerous waste saves a lot of headaches—literally and legally.
Spill Response Without Panic
Spills of dry acrylamide or the liquid form usually spark panic. The best response isn’t improvisation. Keep a spill kit right in the work area: gloves, absorbent pads, and a dustpan for powder. Scoop up powder (avoid brushing), damp-wipe the spot, seal everything in chemical waste bags, then wash up thoroughly. Report the spill even if you think you cleaned everything—this protects your coworkers, too. The worst mistake is not telling anyone or trying to ignore the problem.
Don’t Overlook Training and Documentation
Lab safety officers are your best line of defense. New researchers should never handle acrylamide without at least one run-through with someone experienced. Find the latest Safety Data Sheet before every new project—protocols change, and suppliers sometimes reformulate chemicals. Routine check-ins help everyone catch bad habits before someone gets sick. In my experience, the most dangerous mistakes come from shortcuts or simple forgetfulness, not ignorance.
Respect the Hazard, Protect Your Future
There’s nothing trivial about acrylamide. The health stakes are real and permanent. Wearing the right gear, working inside a fume hood, cleaning carefully, and following strict storage rules become habits that keep you—and everyone else down the hall—safer. This is one chemical that won’t forgive a casual approach. Give acrylamide the respect it demands, and your research won’t cost more than it should.
What are the regulatory limits for acrylamide in food and water?
Tough Questions About a Forgotten Chemical
Most folks never think about acrylamide on their grocery trips. It belongs to that list of lab words that rarely make it past the world of food technologists or environmental scientists. Still, this chemical turns up in many fried, baked, or roasted foods, often right on the dinner plate. You’ve tasted it in golden French fries, coffee, and toast more times than you can count.
The Science: Acrylamide Isn’t a Flavoring
So why worry? Acrylamide forms when certain foods cook at high heat—think above 120 degrees Celsius. The Maillard reaction, which gives those crispy brown edges we love, does more than just build flavor. This reaction in carbohydrate-rich foods leads to acrylamide formation. Drinking water can pick up acrylamide, too, though the main culprit involves how food is prepped.
Studies link long-term exposure to higher cancer risk in animals, and those findings have nudged governments into action. As a parent who checks ingredient lists and relies on the tap at home, it’s tough to brush aside that nagging question: what limits keep us safe?
How Regulators Draw the Line
In the US, the Environmental Protection Agency (EPA) doesn’t just hand out numbers. They limit acrylamide in drinking water to no more than 0.0005 milligrams per liter (mg/L), about half a part per billion. This level includes a safety margin, considering folks who drink and cook with water every day over decades. After reading EPA reports, this number stands out as a careful compromise—tight enough for protection, loose enough for modern treatment facilities to hit their targets.
Europe’s approach to food works differently. The European Food Safety Authority (EFSA) dug through the research and didn’t simply set a blanket limit for all foods. Instead, they push for “benchmark levels” tailored to product groups. For instance, bread, potato chips, baby food, and coffee each get their own maximum levels, often ranging from 40 to 850 micrograms per kilogram. Regulations nudge food makers to keep levels as low as they can, given the food type and recipe. I’ve seen more manufacturers in Europe brag about “acrylamide reduction” on packaging or websites, reflecting these benchmark shifts.
You won’t find one magic number that applies to every meal or snack. The US Food and Drug Administration hands out voluntary guidance rather than kicking down the door with mandatory limits. Food companies, especially those shipping worldwide, find themselves balancing local rules, customer preferences, and what’s possible in real-world kitchens.
Living With Acrylamide: Practical Solutions
Reading about all this is one thing, but it’s natural to wonder if everyday choices can help. Major food companies, under pressure from governments and consumers, revamp recipes and cooking methods to keep acrylamide down. Restaurants tweak frying times or lower the toaster setting to keep both flavor and safety in mind.
Back home, things stay pretty simple—drop oven temps a bit and keep potatoes out of the fridge (which increases sugar level and leads to more acrylamide as they brown). Nobody really wants regulators to ban toast or coffee, but that doesn’t mean the risks deserve to be ignored. The back and forth between public health research, policy, and what we’re willing to change in the kitchen keeps this issue from getting stale.
Looking Ahead
Acrylamide sits at the intersection of science, industry, and what we enjoy eating. By paying attention to both water standards and food practices, health officials keep the worst risks at bay, and smart regulation keeps pushing for safer habits on all sides. Every batch of fries, every loaf of bread, tells part of an ongoing story, shaped by the limits we set and our daily decisions.
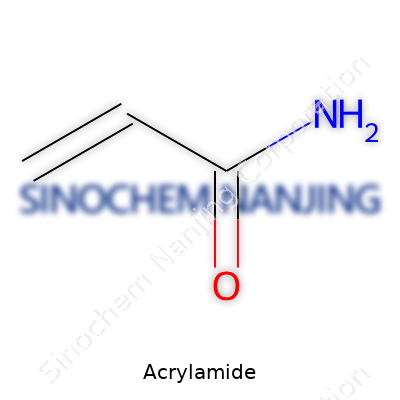
| Names | |
| Preferred IUPAC name | prop-2-enamide |
| Other names |
2-Propenamide Acrylic amide Ethylene carboxamide Propenamide |
| Pronunciation | /əˈkrɪl.ə.maɪd/ |
| Identifiers | |
| CAS Number | 79-06-1 |
| Beilstein Reference | 635 |
| ChEBI | CHEBI:28980 |
| ChEMBL | CHEMBL1566 |
| ChemSpider | 356 |
| DrugBank | DB00742 |
| ECHA InfoCard | 100.001.043 |
| EC Number | 200-852-2 |
| Gmelin Reference | **923** |
| KEGG | C14882 |
| MeSH | D000198 |
| PubChem CID | 6579 |
| RTECS number | AS3325000 |
| UNII | 40D5V8D4TY |
| UN number | 2074 |
| Properties | |
| Chemical formula | C3H5NO |
| Molar mass | 71.08 g/mol |
| Appearance | White, odorless, crystalline solid |
| Odor | Odorless |
| Density | 1.122 g/cm³ |
| Solubility in water | 2150 g/L (20 °C) |
| log P | -0.67 |
| Vapor pressure | 0.007 mmHg (25°C) |
| Acidity (pKa) | 15.1 |
| Basicity (pKb) | 15.1 |
| Magnetic susceptibility (χ) | -7.6e-6 cm³/mol |
| Refractive index (nD) | 1.450 |
| Viscosity | 0.8 mPa·s (25 °C, 50% aq. sol.) |
| Dipole moment | 3.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 137.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -182.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1769 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | J01XE01 |
| Hazards | |
| Main hazards | Toxic if swallowed, suspected of causing cancer, causes skin and eye irritation, may cause damage to nervous system |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H301, H311, H331, H340, H350, H360Df, H372, H400 |
| Precautionary statements | H260, H301, H311, H315, H319, H330, H340, H350, H360FD, H372, P210, P222, P260, P264, P270, P271, P273, P280, P281, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P311, P321, P330, P361, P363, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2-P |
| Flash point | > 138 °C |
| Autoignition temperature | 424°C |
| Explosive limits | 3–16% |
| Lethal dose or concentration | LD50 oral rat: 124 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Acrylamide: "107 mg/kg (oral, rat) |
| NIOSH | AS3325000 |
| PEL (Permissible) | 0.03 mg/m³ |
| REL (Recommended) | 0.0001 |
| IDLH (Immediate danger) | 60 mg/m3 |
| Related compounds | |
| Related compounds |
Acrylic acid Acrylonitrile N,N′-Methylenebisacrylamide Polyacrylamide |

