The Story of Acetophenone: More Than a Chemical by Another Name
Historical Development
Acetophenone traces its roots back to the laboratory days of the 19th century. Early chemists, through a lot of hands-on trial and error, discovered this colorless liquid tucked away in the byproducts of organic reactions. It found an early place in the annals of synthetic chemistry, showing up in the stories of coal tar distillation and the busy days of expanding organic synthesis. If you flip through an old chemistry handbook, you’ll often stumble over references to it as methyl phenyl ketone — and for good reason. As those old-school labs searched for ways to map out the world of aromatic compounds, acetophenone set itself apart by proving to be surprisingly versatile for both academic and industrial applications.
Product Overview
What makes acetophenone stand out after all these years? This substance serves as both a building block and a flavor note in the world of molecules. It wears many hats, showing up in the labs for chemical synthesis and also finding a place in perfumes. Walking past a bakery or a candy shop, there's a fair chance you’ll catch a whiff of this compound’s almond-like scent, thanks to its use as a flavoring agent. In the chemical supply catalogs, acetophenone doesn’t put on airs — it’s a practical, straightforward workhorse that doesn’t shy away from getting put to use in new pharmaceuticals, resins, and even solvents.
Physical & Chemical Properties
This liquid doesn’t draw much attention to itself at first glance. With a melting point just below room temperature and a boiling point sitting a bit above water’s, acetophenone fits well into most lab environments. It dissolves well in alcohols and ethers, less so in water, making it easy to coax through a variety of reactions. Its refractive index and density land in the typical range for simple aromatics, but the real standout feature is the combination of a pleasant aroma with the right mix of chemical stability. It’s a familiar sight in organic chemistry classes, where students talk about carbonyls and the way they pull electrons across a molecule’s structure.
Technical Specifications & Labeling
For packaging and labeling, clarity matters a lot. Suppliers mark bottles clearly with the name, chemical formula (C8H8O), CAS number, purity percentage, hazard statements, and date of manufacture. Those details might sound routine, but they mean a great deal when safety and traceability step up as real-world concerns. Labels call out flammability, suggest PPE, and point to the kind of ventilation that keeps labs running smoothly. Batch numbers are more than just extra digits—they track origins and quality, covering both regulatory demands and the expectations set by good laboratory practices.
Preparation Method
The process for making acetophenone usually starts from benzene and acetyl chloride or acetic anhydride using a Friedel-Crafts acylation, that classic reaction that so many chemistry students wrestle with in the lab. Aluminum chloride acts as the catalyst, pulling the reaction forward. This straightforward path means the molecule remains accessible for all kinds of industrial-scale work. Some industrial producers shift to oxidation of ethylbenzene for larger batches, taking advantage of cheaper feedstocks. The flexible approach to synthesis gives producers the freedom to chase efficiency without compromising on purity.
Chemical Reactions & Modifications
The real value in acetophenone comes from its willingness to take part in further reactions. From there, chemists produce hydrazones, oximes, and even stretch the molecule into secondary alcohols after reduction. One trick involves halogenation, which allows for more functionalized derivatives. These modifications play a big role in medicine, where altered versions of acetophenone contribute to treatments or act as intermediates in larger pharmaceutical syntheses. The structure’s simplicity gives it a kind of flexibility, acting as a base to build more complex organic molecules.
Synonyms & Product Names
Chemists, regulators, and marketers refer to acetophenone by a range of names. Methyl phenyl ketone brings out its true structural identity, while phenyl methyl ketone and 1-phenylethanone pop up in older or region-specific documentation. Its EINECS and CAS registry numbers keep it sorted in chemical inventories. On perfume and food additive lists, it may even show up under specialized trade or aroma names. These many aliases mean keeping an eye on labeling and safety data sheets, as confusion can sneak in when switching between industries or regulatory systems.
Safety & Operational Standards
Some folks might remember a time when gloves and goggles got brushed aside during bench work, but things have shifted fast. Today, quality safety standards draw a clear line: handle acetophenone in well-ventilated spaces, store it away from ignition sources, and pay close attention to local environmental compliance. The material can irritate skin and eyes, and inhalation isn’t recommended. Spill response and waste disposal both demand real diligence, as regulations for aromatic solvents get stricter year by year. Training matters just as much as data sheets—a culture of safety rests on making real choices, not just checking off requirements on paper.
Application Area
Few chemicals wear as many hats as acetophenone. The perfume industry counts on its soft, floral odor to anchor sweet and powdery notes in fragrances. Food chemists drop it into flavorings, though only at trace amounts, after rigorous review for safety and quality. In plastics and resin manufacture, it acts as an intermediate, swinging from one process to another with little fuss. Medical researchers transform it into a range of drugs and agents, showing its core place in both basic and applied science. It stands as one of those compounds that keeps chemistry moving forward without ever taking the spotlight.
Research & Development
New uses for acetophenone keep showing up through the lens of academic and industrial R&D. Biocatalysis opens new routes for making its derivatives, focusing on greener chemistry with less hazardous byproducts. The fine-tuning of catalysts and conditions has given rise to routes that are more sustainable, fitting with both regulatory and ethical shifts. Teams working on advanced materials explore its potential in polymer modifications and specialty coatings, where subtle changes to its structure lead to real performance gains. The collaborations between universities and industry have a long history of unlocking fresh ways to coax more value—and fewer hazards—from old chemicals.
Toxicity Research
Safety research digs deep into how acetophenone acts in the body and the environment. Acute exposure tends to create irritation rather than severe systemic toxicity, though studies follow potential links to metabolic disruption or chronic effects with sustained contact. Regulatory agencies classify it with care, balancing workplace risk with downstream environmental safety. Teams keep a watchful eye on workplace exposure limits, train staff to respond fast if spills occur, and track waste streams to prevent lingering environmental impacts. Scrutiny continues on every step, from manufacturing floor to market shelf.
Future Prospects
Acetophenone holds steady relevance for both traditional synthesis and new fields looking for chemical versatility. Green chemistry continues to rework how companies make and modify this molecule, pushing for renewable resources and safer reaction conditions. Advances in biotechnology hint at biosynthetic pathways for producing acetophenone from plant or microbial sources, bypassing fossil fuel feedstocks entirely. As more applications line up in advanced materials, pharmaceuticals, and specialty aromas, the story of acetophenone grows richer. Ongoing research in toxicity and biodegradability aims to place responsibility side-by-side with innovation, setting a standard for how the next wave of chemistry interacts with the world.
What is acetophenone used for?
Real-World Purposes of Acetophenone
People outside of chemistry class rarely give much thought to a compound like acetophenone. It doesn’t get the headlines like plastics or miracle drugs, but walk down any perfume aisle or open a bottle of cola and its influence is right under your nose (sometimes literally).
Acetophenone’s scent lands somewhere between oranges and almonds, sweet with a spicy note. Perfumers have relied on it for decades to form the base notes in traditional fragrances. The aroma sticks around long after the lighter scents fade. It isn’t a luxury—just a way to help the perfume last through a busy day.
The food industry also gets mileage from acetophenone. It shows up in some artificial flavors, especially for items meant to copy the zest or tang of fruit. The US Food and Drug Administration (FDA) put acetophenone on its list of food additives considered generally safe, but industry sets strict limits on how much can go into products to avoid health concerns.
Acetophenone’s Footprint in Industry
The story doesn’t end at taste and smell. Manufacturers use acetophenone to help make resins and plastics harden in a certain way. Chemists call it a “precursor,” which just means it’s part of a chain reaction to produce other chemicals. One key product in that chain is styrene, the building block of polystyrene—think foam coffee cups and packing peanuts.
A personal example: If you’ve ever walked through a construction site, that glue-like smell near some foam insulation usually traces back to the same line of chemicals—including ones derived from acetophenone. That part of the process isn’t glamorous, but it keeps homes insulated and shipping crates lightweight but durable.
Pharmaceutical companies depend on acetophenone too. Drug developers have turned to it to help anchor certain medications. In rare cases, acetophenone itself was used as a sedative in medicine, but today it mostly paves the way for synthesizing more complex pharmaceuticals. That shift happened because newer medicines proved more effective or had fewer side effects, but acetophenone’s chemical ease still helps launch fresh solutions in drug labs.
Safety and Solutions
No one should ignore safety with this chemical. In large amounts or over long periods, exposure can be rough on the nerves or cause skin irritation. That means factories need good ventilation and careful storage to protect workers. Scientists and safety crews monitor airborne levels tightly, drawing on research from the National Institute for Occupational Safety and Health (NIOSH) and similar agencies. In my experience working with lab teams, spills meant immediate action—gloves, fume hoods, and a healthy respect for what you’re handling.
Across all these uses, acetophenone supports industries people count on daily—perfumes, flavors, plastics, medicine. Each application benefits from careful regulation and responsible use. Technology will keep changing what we expect from basic chemicals, but learning where they show up in the world makes it easier to spot the risks and value in what we use every day.
Is acetophenone safe to handle?
The Realities of Acetophenone in the Real World
Anyone who has worked in a lab or a factory knows some chemicals demand more respect than others. Acetophenone, with its sweet, floral odor, can trick the senses. On a quick sniff, it might not scream danger. But trust me—handling chemicals safely is not about what the nose tells you. Experience has taught me to read the data and not the scent.
What the Science Says: Hazard and Harm
Acetophenone turns up in everything from perfumes to industrial processes. Its low boiling point means it can slip into the air with ease. The CDC and many material safety data sheets point out the risks: skin and eye irritation, drowsiness, headache, and even respiratory tract issues. No one wants to cut corners and end up with a health scare that could have been avoided by following simple safety measures.
Working around volatile organic compounds brings stories—sometimes hard-learned ones. I remember one shift after forgetting a glove change led to tingling and redness that lasted hours. Mild, maybe. Still, enough to remind me skin absorption is real and gloves are not for show.
Why Proper Handling Matters—For You and Everyone Around You
It’s tempting to view products used in commercial flavoring or scents as “safe” by default, just because they eventually end up in consumer products. The workplace is a different beast than a bottle of soda. The concentration, the exposure time, the risk of accidental spills—everything scales up. That’s especially true for acetophenone, which can affect the central nervous system with repeated exposure.
Direct contact isn’t the only way for health problems to show up. Vapor is the silent partner here, drifting around faster than we notice. This stuff doesn’t need to leave burns or scars to cause damage, either. Some evidence ties repeated exposure to adverse effects over time, including effects that might take weeks or years to notice.
Building Safety Into Routine
Out in the field, the basics prove their worth daily: gloves, goggles, good ventilation, and following standard operating procedures. Ventilation makes a difference. A good extractor fan or fume hood is not a luxury—it’s a shield. Respirators have saved my lungs more times than I care to count. Anyone working with acetophenone should have easy access to a safety shower and eye wash. Accidents happen, and a quick response can make all the difference.
Knowledge makes the backbone of safe workspaces. Training is more than a box to check—it’s a way to make sure everyone knows the warning signs and the escape plan. If someone feels lightheaded, has irritated skin, or can’t shake a nagging cough, that’s a sign the system might need an upgrade. Regular refreshers help catch mistakes before they become emergencies.
Looking Ahead: Solutions and Smarter Standards
Substituting safer chemicals stands out as a strong solution. When that’s not possible, smart engineering controls and personal protective equipment need top billing. Every year brings new safety gear and better workflows. Supporting a transparent, open culture around safety helps everyone speak up if something feels off or a process looks risky.
No chemical handles itself safely. Acetophenone is no exception. The combination of respect, skill, and up-to-date information keeps jobs safe and reputations intact. For anyone in the lab, the factory, or even the hands-on classroom—taking care with this chemical benefits more than just the person holding the beaker.
What is the chemical formula of acetophenone?
What’s in a Formula?
Acetophenone, a name that rarely pops up in daily conversation, has made its mark in industries I never thought would link up—perfumes, pharmaceuticals, and even the world of plastics. Its formula looks simple: C8H8O. Eight carbons, eight hydrogens, and one oxygen. Short and sweet. But behind that string of letters and numbers sits a compound that’s shaped more than a handful of household products.
The Science Behind the Simplicity
Seeing C8H8O on a label doesn't give away much at first glance. That changes quickly when you understand the backbone—an aromatic ring (like the skeleton of benzene) with a chunk of acetyl group spliced on. I remember the first time I came across acetophenone working at a local pharmacy during college. No, I wasn’t formulating drugs in the back room, but I got curious about the stuff that went into allergy medications. The label didn’t exactly shout 'C8H8O,' but it played its part in the mix.
Everyday Encounters
Perfume makers love acetophenone for its faint, sweet, and almond-like smell. A bottle of cologne or aftershave might owe part of its character to it. Some artificial flavors and scents in processed foods can’t dodge its touch—C8H8O slips in under the radar but leaves a recognizable hint. I once found it listed in an ingredient deck for a vanilla-scented candle. My interest piqued, I dug around more and found out it didn’t just add scent; it brought stability, blending well without breaking down under heat.
Safety Comes First
Before getting too comfy with acetophenone in workplaces or homes, it’s important to remember the safety side. The chemical world never guarantees complete safety, even with familiar names. Research, like studies from the Environmental Protection Agency, shows that acetophenone carries moderate toxicity. Inhaling it for long periods or exposing skin to big doses can set off headaches, skin irritation, or worse. I always tell my friends and family to wear gloves and keep rooms ventilated when handling products with a strong chemical odor, no matter how ‘normal’ they seem.
How Should We Handle It?
Chemists and factory workers have detailed guidelines for handling C8H8O safely—safety goggles, gloves, and fume hoods don’t go unused in labs. It shocked me the first time I saw just how serious this gets at the industrial level compared to home use. Regulators play a part too. Agencies like OSHA and the EPA have helped keep exposure in check. Consumers can read the safety data sheets that most companies list online for chemicals that make their way into makeup, cleansers, and fragrance oils. These steps reduce guesswork, which matters when trust in science and transparency seem to be in short supply.
Trust and Transparency
Most people don’t memorize chemical formulas, but knowing C8H8O means more than just being able to answer a trivia question. Every ingredient holds a story—of innovation, risk, benefit, and responsibility. My own respect for everyday chemistry grows every time I notice just how much a simple molecule can mean to so many parts of life. So next time you catch the whiff of almond in a candle or cologne, remember that it might just be acetophenone, hard at work behind the scenes.
How should acetophenone be stored?
Understanding Why Storage Matters
Walking into a chemical storeroom, the labels and scents highlight just how varied and potent lab chemicals can be. Acetophenone shows up in perfumes, pharmaceuticals, and some chemical processes, but don't let its pleasant smell fool you into thinking it offers fewer risks. I’ve seen labs ruined by poor chemical storage, with costs that go far beyond money. Acetophenone won’t set the world on fire, but mishandling creates real problems for health and the environment.
Real Hazards to Watch For
A main concern is inhalation. Breathing in acetophenone vapor irritates the eyes and throat, so a good, tight container makes a big difference. Over time, poor seals or cracks can let vapors leak out, slowly raising exposure. In a hot room, its vapor pressure climbs, so I always keep it away from heat and direct sunlight. Years back, I saw a storage closet near a window cause a slow, sticky mess as chemicals warmed up. For acetophenone, losing control of temperature makes things riskier for skin contact and accidental breathing.
Physical hazards stack up if bottles aren’t marked clearly and stored apart from incompatible chemicals. Mixing with oxidizers or strong acids creates dangers nobody wants to deal with—one slip, and you’re reaching for the spill kit or, worse, the emergency shower.
Best Practices from the Trenches
In my experience, keeping acetophenone in a cool, dry, well-ventilated spot brings peace of mind. A flammable chemicals cabinet works well, even though acetophenone isn’t the most flammable compound in the room. I choose cabinets that resist corrosion, with shelves that stop bottles from tipping easily. I never store glass bottles right at the edge. If possible, secondary containment—trays or bins—catches leaks before they reach other bottles or the floor.
Separation counts. Acetophenone should sit away from acids, oxidizers, and bases. In a busy lab, it’s easy for bottles to migrate between shelves, so regular checks become routine for good reason. Proper labeling—one that even a tired coworker can read—helps everyone avoid mistakes after a long day.
Health and Environmental Considerations
I’ve seen gloves dissolve after too much exposure, and skin irritation may sound mild, but it can easily escalate. The vapor isn’t something anyone should get used to. Acetophenone waste deserves careful handling. Never pour leftovers down the drain. Use clearly marked waste bottles and schedule pickups with professionals to avoid accidental releases into water systems.
Potential Improvements for Safer Storage
Better storage begins with training, not more rules. Too many accidents start because someone assumed clear liquid meant harmless liquid. Stocking spill kits close to storage areas and reviewing emergency plans regularly keep small mistakes from turning big. Labs and companies can use sensors for volatile organic compounds, spotting leaks before the nose catches them. Rotating stock—using older bottles before newer ones—prevents forgotten acetophenone from degrading in some unused corner.
From my perspective, safe chemical storage doesn’t stop with a locked cabinet. It’s about understanding real risks, sharing responsibility, and designing spaces that help people do the right thing—even on the busiest days.
What are the hazards associated with acetophenone?
Understanding Acetophenone’s Risks
Acetophenone pops up in many places, from laboratories to fragrances and even some pharmaceuticals. While it helps shape flavors and scents, this chemical hides some risks that deserve attention. Having worked around lab chemicals, it’s easy to overlook everyday hazards, but missing a safety step can flip a routine shift upside down.
Health Hazards: Eyes, Lungs, Skin
Acetophenone attacks the body right at entry points. If vapors waft too strong, they sting the nose, make breathing tough, and leave throats scratchy. Eye contact leads to nasty irritation and blurred vision, something nobody wants to deal with at work or at home. This isn’t rare–one slip with unfiltered ventilation and you’ll remember it for days. Handling liquid acetophenone without gloves gives skin red, itchy patches. Prolonged exposure can worsen rashes or even trigger chemical burns.
Acetophenone’s annoyance doesn’t stop on the surface. Over time, inhaling or absorbing too much allows the chemical to seep deeper. Headaches, sluggishness, and dizziness creep in after breathing it at moderate levels. High exposure can hit the central nervous system, leaving hands shaky and reflexes dulled. Chronic low-level exposure has reportedly affected memory and concentration in some workers.
Fire and Environmental Hazards
This liquid sparks easily and burns with an invisible flame. One coworker set off an alarm by storing acetophenone too close to a heat source. Flames grew out of a round-bottom flask while the fume hood struggled to keep up. Even a drizzle on a hot plate releases plenty of fumes—a reminder that fire risks demand respect.
Acetophenone leaks or spills run to sewers or soil and can upset waterways. Fish gulping contaminated water fail to thrive or die off in bigger spills. Once in the environment, acetophenone moves through the air and soil, making the cleanup job complicated.
Shortcomings in Current Safety Culture
People sometimes act as if gloves and goggles turn them invincible. Shortcuts add up and invite accidents. Employers skimping on ventilation or cutting back on safety education create an atmosphere where complacency thrives. Emergency plans often sit on paper but rarely get practiced. Even regular labeling slips through the cracks as containers get reused or improperly stored.
Some businesses store multiple volatile chemicals alongside acetophenone. This mixing raises the risk of complex chemical fires or release of dangerous fumes, especially in cramped stockrooms.
Better Habits and Smarter Rules
Keeping acetophenone in check starts with a real look at habits. Pick up the safety goggles and seal gloves before touching the bottle. Install and run good exhaust fans and don’t let coworkers skip routine maintenance. Each regular practice matters more than a one-off safety poster.
Train teams to spot symptoms of exposure, not just follow a checklist. Invest in spill kits and practice drills. Separate chemicals that react poorly together. Employers can offer periodic checkups for those exposed daily, catching symptoms before they turn serious. The government can push for clearer labeling, stricter rules on air monitoring, and shorter allowable exposure limits. Community groups and environmental agencies should monitor water near factories for traces of these chemicals.
Small improvements in storage, training, and cleanup make a massive difference in health outcomes. Taking hazards like acetophenone seriously keeps workplaces—and communities—safer for everyone.
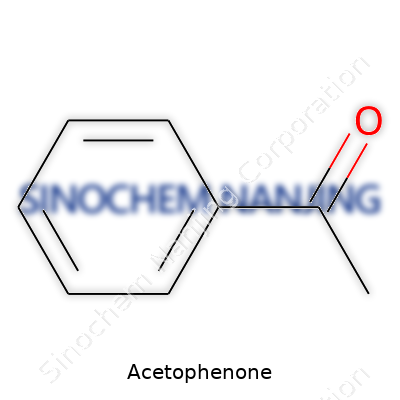
| Names | |
| Preferred IUPAC name | 1-Phenylethan-1-one |
| Other names |
Methyl phenyl ketone Phenyl methyl ketone Acetylbenzene Phenacetin impurity A |
| Pronunciation | /əˌsiːtəˈfəʊnəʊn/ |
| Identifiers | |
| CAS Number | 98-86-2 |
| Beilstein Reference | 635158 |
| ChEBI | CHEBI:27856 |
| ChEMBL | CHEMBL515 |
| ChemSpider | 550 |
| DrugBank | DB01961 |
| ECHA InfoCard | 100.09.067 |
| EC Number | 202-708-7 |
| Gmelin Reference | 1460 |
| KEGG | C02207 |
| MeSH | D000073 |
| PubChem CID | 7410 |
| RTECS number | AL3150000 |
| UNII | 1O8WYG0G7Q |
| UN number | UN1990 |
| Properties | |
| Chemical formula | C8H8O |
| Molar mass | 120.15 g/mol |
| Appearance | Colorless liquid with a sweet, pungent odor |
| Odor | fragrant; sweet; jasmine; almond; cherry; penetrating |
| Density | 1.03 g/cm³ |
| Solubility in water | Moderately soluble |
| log P | 1.7 |
| Vapor pressure | 0.4 mmHg (20°C) |
| Acidity (pKa) | 20.0 |
| Basicity (pKb) | pKb ≈ 14.0 |
| Magnetic susceptibility (χ) | -47.5e-6 cm³/mol |
| Refractive index (nD) | 1.533 |
| Viscosity | 0.969 cP (20°C) |
| Dipole moment | 3.07 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 186.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -48.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3227 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N05CM07 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P280, P305+P351+P338, P304+P340, P312 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 2, Instability: 0 |
| Flash point | 77 °C |
| Autoignition temperature | 615 °C (1139 °F; 888 K) |
| Explosive limits | Explosive limits: 2 - 10% |
| Lethal dose or concentration | LD50 oral rat 815 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, Rat: 815 mg/kg |
| NIOSH | KW3675000 |
| REL (Recommended) | 250 mg/L |
| IDLH (Immediate danger) | IDHL: 200 ppm |

