Acetic Anhydride: A Cornerstone of Modern Chemistry
Understanding the Road So Far
Acetic anhydride didn’t spring up overnight; it comes with a long trail of discovery and use that mirrors the rise of industrial chemistry. In the 19th century, chemists in Europe isolated this compound and quickly realized that its potential stretched much further than early expectations. In factories of pre-World War eras, production lined up with surging demand for cellulose acetate—used in everything from photographic film to textiles. Government agencies paid attention since synthesis methods opened doors not just for legal manufacture but for less desirable activities too. In my own experience working in chemical research, it’s clear that few substances have sparked as much practical innovation and regulatory scrutiny as acetic anhydride. This history winds together advances in technology and emerging public health awareness, shaping the standards we see applied worldwide today.
What Acetic Anhydride Brings to the Table
Acetic anhydride looks like a clear, slightly volatile liquid with a biting, pungent vinegar odor. It boils just above water’s boiling point and doesn’t blend with water readily—mixing these prompts a feisty chemical reaction. Its molecular weight, density, and the way it reacts to heat and air all tie into handling issues. Chemists value its strong acetylating power, meaning it donates acetyl groups to other molecules in numerous synthetic pathways. In the lab, the sight of a sealed acetic anhydride bottle flags both its reactivity and the need for solid ventilation. I remember standing back, safety goggles on, watching as a batch of salicylic acid fizzed and released heat once acetic anhydride entered the flask—a vivid reminder that this is not a benign ingredient.
Technical Details and Labeling Practices
Regulators approach acetic anhydride with seriousness. Labels shout safety warnings: “Corrosive,” “Causes burns,” “Handle with care.” Storage calls for cool, dry areas, with tightly sealed containers, since moisture triggers decomposition into acetic acid, ramping up the fumes and potential for equipment corrosion. Industry guidelines outline purity benchmarks; pharmaceutical and photographic film producers keep an eagle eye on contaminants. Proper labeling matters not only for legal compliance but for the front-line workers around the stuff day in and day out. Any slip in process control, or a simple oversight on a container label, brings real risk—a lesson hammered home for anyone who’s spent hours tracing back the source of a failed batch or a safety scare inside the plant.
From Raw Materials to Bottle—The Preparation Side
Making acetic anhydride on a commercial scale usually means reacting acetic acid with either ketene or acetic acid anhydride—depending on the local regulations, plant design, and available feedstocks. The ketene process, for instance, involves heating acetic acid over a dehydrating catalyst, squeezing out water and shifting equilibrium toward the anhydride. These are high-energy, highly technical operations, run with close monitoring. In smaller-scale labs, synthesis often follows analogous routes but in glassware rather than reactor vessels. Each synthesis brings questions about yield, purity, and disposal of waste. From an operational view, efficiency sits right next to worker safety and environmental responsibility.
Chemical Reactivity and Ways to Tweak It
Acetic anhydride only needs a nudge to go to work. In academic labs and factories, it reacts with alcohols, amines, and phenols—transferring an acetyl group and forming esters or amides. Aspirin synthesis stands out: acetic anhydride meets salicylic acid, and in go the acetyl groups to make acetylsalicylic acid. The same story repeats in dyes, perfumery, plastic production, and explosives. Once in a while, process engineers look for ways to avoid using acetic anhydride altogether, especially if supply hiccups or regulatory barriers rear up. Alternative reagents or process tweaks might step in, but few do the job quite so thoroughly.
Synonyms and Naming Roundabouts
The chemical industry spawns a heap of synonyms for acetic anhydride, with “ethanoic anhydride” showing up in technical literature and “acetyl oxide” cropping up less often. Regardless of name, professionals recognize it by its chemical shorthand—(CH3CO)2O. Being able to decode synonyms keeps things clear, especially during international collaborations or when poring over research papers and safety data sheets. I’ve watched experienced colleagues double-check chemical identifiers to catch translation errors that could cause costly mistakes in sourcing or regulatory filings.
Balancing Safety and Daily Operations
No corners can be cut dealing with acetic anhydride. The compound’s corrosive power can damage skin, eyes, and the respiratory system. Labs and factories insist on gloves, eye protection, good ventilation, and immediate cleanup of leaks or spills. I’ve sat through enough chemical safety drills to know that assumptions spell trouble—every workplace incident analysis points right back to lapses in caution or training. Strong operational standards, frequent drills, and clear signage all play their part. Beyond immediate risk, regulators and environmental watchdogs keep tabs on emissions since release into water or air poses risks for wider communities. It takes more than handing out goggles; it’s about building a culture where people take these standards to heart.
Acetic Anhydride in Action
Look around modern industry, and acetic anhydride shows up in a surprising number of places. Drug makers rely on it for life-saving pharmaceuticals such as aspirin and some antibiotics. Textile plants use it for making acetate fibers, which weave into soft, lustrous fabrics. The compound’s acetylating punch makes it key for producing pesticides, dyes, and photographic films. Even in food packaging, modified starches trace back to acetic anhydride chemistry. Drawn by that versatility, my former research group once dug into greener pathways for acetylation, hoping to trim both costs and environmental impact. The challenge always comes down to balancing throughput with worker safety and long-term sustainability.
On the Cutting Edge: Research and Development
Research into acetic anhydride wades into deep questions about process efficiency, fewer unwanted byproducts, and ways to recycle waste streams. Green chemistry weighs heavily on development pipelines. Academics and industrial chemists tackle methods reducing energy input or exploring renewable feedstocks. Thinking back, conversations at conference poster sessions often come alive around clever catalyst choices or the bump in yield that tipped a process toward commercial viability. Process safety and continuous monitoring, using advanced sensors and analytics, make it possible to spot problems long before they become serious. Research teams focus not just on what works in the flask, but what can be scaled while slashing emissions or hazardous byproducts.
Getting to the Heart of Toxicity
Risk assessment of acetic anhydride starts with understanding its impact on people and ecosystems. Acute exposure can burn airways, eyes, and skin. If spilled or released recklessly, it doesn’t take long before that vinegar stench turns into a real threat, affecting both first responders and nearby communities. Toxicity research looks at short-and long-term effects, including possible links to respiratory illness or sensitization. Organizations such as the Occupational Safety and Health Administration (OSHA) and the European Chemicals Agency (ECHA) set strict exposure limits, reflecting both recent studies and lessons from accident case reports. Drawing from my own time handling process incidents, most scare stories come from badly ventilated rooms or neglected maintenance—it’s the human side of the equation that proves decisive.
On the Horizon: What Comes Next?
Acetic anhydride faces a crowded future. Producers grapple with tighter environmental laws, supply chain scrutiny, and demand swings from pharmaceuticals or textiles. There’s a real push for alternative synthesis methods that use fewer fossil-derived raw materials, spit out less waste, or pack less firepower in the hands of illicit actors. Digital systems—real-time sensors, predictive maintenance—promise fewer accidents and cleaner operations. As researchers chase lower-energy, lower-waste pathways, the line between practical chemical engineering and global sustainability grows thin. In my own work, every experiment and process review echoes this trend. The entire story of acetic anhydride—its history, dangers, and possibilities—underscores why smarter chemistry matters more than ever.
What is acetic anhydride used for?
Industrial Workhorse in Plain Sight
Acetic anhydride doesn’t jump out at most people when they walk through a grocery store or open their medicine cabinet, but this colorless liquid has more influence on daily life than you might guess. Over the years, I’ve seen how essential raw materials quietly steer whole industries, and acetic anhydride fits that mold. Factories rely on this chemical for making something as common as aspirin. The world turns to those little white pills for backaches, fevers, or heart health. The production process depends on acetylation: acetic anhydride attaches to an active ingredient called salicylic acid, transforming it into aspirin. The same goes for paracetamol, better known as acetaminophen.
Companies use acetic anhydride well beyond their labs. Take cellulose acetate, for example — this plastic shows up in eyeglass frames, photographic films, and some cigarette filters. I once replaced the arm of my reading glasses, and the smooth, flexible plastic comes from cellulose fibers treated with acetic anhydride. Manufacturing teams mix acetic anhydride with cellulose pulp, and after a few steps, turn it into a lightweight material that outlasts metal hinges or wood.
Risks and Responsibility
Some of the downside catches less attention but carries real consequences. Acetic anhydride can help in production of illegal drugs like heroin. Drug enforcement teams have seized it at borders for good reason; cartels use it to change morphine into heroin. Authorities keep a watchful eye on shipments and inventories. This kind of oversight complicates life for legitimate producers, who must follow rules set by the Controlled Substances Act in the US and international conventions. Factories face paperwork, audits, and background checks, but these hurdles stem from an honest intent to reduce the spread of drugs harming families and communities.
I talked to a friend in pharmaceutical manufacturing who described the extra work added by regulations, but he stressed that safety comes first. Oversight helps keep hazardous chemicals in the right hands, not those looking to profit off addiction.
Safety and Environment: Not Afterthoughts
Small spills of acetic anhydride can sting eyes and noses, triggering coughing or burning skin. On a shop floor, workers wear goggles and gloves because no one wants a sudden rush of fumes. Storage takes place in cool, ventilated areas. Accidents can bring fire hazards, so careful attention matters. Businesses can't cut corners — not just to stay within the law, but so their crew heads home healthy at the end of a shift.
Environmental impacts ask for strict controls too. If factories release acetic anhydride, it can break down in water and soil to acetic acid, which is less toxic but still can hurt fish and plants in high amounts. Strong safety programs and environmental management limit leaks. Upgrading equipment and training workers bring results. The European Union and US EPA require plants to report how much they use and dispose of, helping track and fix problems before they get out of hand. Lean operations lower waste, keep air cleaner, and avoid the fines that come up from shortcuts.
Weighing Needs Against Misuse
Balancing the good that acetic anhydride brings to health care and manufacturing with its potential for abuse isn’t easy. Vigilance through law enforcement, along with good corporate citizenship, makes a difference. Finding alternatives for some uses could ease the pressure. Until then, a focus on transparency, monitoring, and safety gives society the chance to enjoy helpful products and keep risks at bay. From aspirin to eyeglass frames, the reach of acetic anhydride stretches farther than most folks realize, carrying both benefits and responsibilities with every batch.
Is acetic anhydride hazardous or dangerous?
Understanding What Acetic Anhydride Is
Acetic anhydride isn’t something most people keep around the house. In a lab or industrial setting, it shows up as a colorless liquid with a sharp, vinegar-like smell. Across the world, chemists use it to make things like aspirin, certain plastics, and even perfumes. Most students running through their first chemistry labs never run into it. For folks outside of science, its name doesn’t ring many bells unless you’ve heard of its misuse in making illegal drugs.
Real Hazards in Everyday Use
My time working in a college lab left me wary of this chemical. Short exposure to even moderate levels irritated my eyes and sent coworkers into fits of coughing. If splashed on skin, you feel a painful burn — the kind that lingers throughout the day. Inhaling its fumes can make your throat feel raw and cause headaches, even long after you leave the room. Transporting or using acetic anhydride outside a controlled space opens up a risk for accidents, and the potential aftermath sticks with you. Think of what happens if it catches fire: thick, choking fumes fill the air, making rescue or cleanup much harder.
Environmental and Social Impact
Disposing of even small spills takes careful work. Acetic anhydride reacts strongly with water and many other chemicals. Just a drizzle down a drain, and someone could face a burst of hot, corrosive gas. I’ve seen what happens when waste handling isn’t strict—cleanup crews need special gear, and nearby wildlife can suffer. It’s not a chemical you want loose in a waterway.
Regulators track its sale and movement closely. That tight control stems from more than just workplace safety. Making illegal substances like heroin can call for large amounts of this chemical, which means criminals hunt for unguarded supplies. Law enforcement agencies have caught on, so industries and labs fill out paperwork and log every drop.
What Really Matters for Safety
Rules around acetic anhydride aren’t just red tape. Wearing gloves, eye protection, and working with strong vents or hoods forms the baseline for handling it. Companies invest in training and keep detailed records because a single mistake puts workers and neighborhoods at risk. Even disposal turns into a big task, handled only by trained professionals with secure procedures. Accidents involving this liquid rarely stay small.
Preventing Harm in the Future
Addressing the danger of acetic anhydride comes down to vigilance. Factories and labs benefit from regular audits and surprise inspections. Modern storage systems with automatic leaks alerts, plus secure cabinets, make theft and accidents less likely. Governments keep tabs through international agreements, like the Chemical Weapons Convention, to prevent both industrial disasters and criminal misuse.
For anyone near an industrial site, community education helps spot leaks or spills early on. Emergency response teams train for exactly this kind of event, so nearby residents know evacuation zones and warning signals. People forget just how much hidden risk rides with certain chemicals. It’s often those with deceptively simple names — not just the exotic ones — that need serious respect. Acetic anhydride falls right into that group.
What are the storage requirements for acetic anhydride?
Why Acetic Anhydride Demands Careful Handling
Few chemicals draw more nervous glances in a stockroom than acetic anhydride. Known for its pungent smell, this colorless liquid burns on the nose and lingers in the air. Those who’ve handled even a liter have learned how fast it finds water, jumps at the chance to break down, and will corrode hands or eyes in seconds. Acetic anhydride’s dangers aren’t just talk — the U.S. Drug Enforcement Administration keeps it in close watch, and incidents of improper storage have made headlines for causing dangerous workplace spills and fires. Losing focus around this chemical isn’t an option.
Stable Temperature, No Sun, No Surprises
Acetic anhydride doesn’t like surprises in temperature; sudden changes can ruin a barrel. Cooler conditions, somewhere under 30°C, put the brakes on its tendency to decompose and fume. Hot storerooms drive up vapor pressure, setting the stage for leaks and flammable atmospheres. I once saw a warehouse where the air shimmered with the smell of vinegar because someone left a drum near a boiler room vent. The acid burns in that air stood as a rough lesson: “Store it far from heat sources.”
Darkness works as insurance as well. Sunlight can nudge acetic anhydride toward breaking down into acetic acid, which not only ruins batches but also loads the air with more fumes. Keeping containers in a shaded or windowless space, away from direct beams, gives each barrel the best shot at a long shelf life.
Containers: Glass, Steel, and Trusted Seals
Choosing the right material for storage containers makes all the difference. Glass bottles or drums lined with fluorinated resin offer protection, since acetic anhydride tears straight through simple plastics, most rubbers, and even some metals. Stainless steel earns trust, though not every alloy holds up, especially if water might sneak in over time. Teflon-lined fittings stand up to the challenge, as do high-quality gaskets. I’ve watched less experienced techs try to pour off leftovers in a polyethylene bucket, only to watch the slow seep begin as the plastic distorted beneath the liquid.
Seal It Tight—Moisture’s the Enemy
Even small leaks invite disaster. Acetic anhydride grabs oxygen from the air, reacts with water vapor, and generates acetic acid — strong enough to eat through many shelf linings and harm the lungs. I’ve always spent extra time checking drum seals and vent valves. If a lid felt loose after one round of storage, that was a red flag to swap it or reseal the entire batch. Good practice keeps spill kits nearby, since a mop and bucket won’t fix a chemical puddle that can burn through shoe soles.
Spacing and Signage Save Lives
Colleagues in warehouse safety will back me up here: spacing out containers prevents cross-contamination if a leak appears. Chemicals that react with acetic anhydride — like bases, alcohols, or strong oxidizers — stay in different bays or separate storerooms if resources allow. Every drum, bottle, or container gets bold hazard labels, including corrosion and flammability icons. It’s not about bureaucracy; it’s about the rookie on a late shift knowing exactly what they’re up against at a glance.
Regular Checks and Better Training
Checking for corrosion, weakened seals, and strange smells gives everyone peace of mind. Training sessions on safety gear — goggles, gloves, aprons — cut down on accidents. Waste disposal plans need to be clear, since dumping acetic anhydride down the drain threatens both the building’s pipes and the local water table.
For any storage plan, reducing risks with simple steps makes the real difference. Smart storage for acetic anhydride comes down to respecting its power, investing in the right containers, managing the environment, and making training a priority every day.
How should acetic anhydride be disposed of?
Understanding the Problem
Acetic anhydride isn’t exactly something you want lying around your shed. With its sharp smell, volatile nature, and ability to corrode skin and eyes, it takes real respect. Used for making pharmaceuticals, plastics, and even explosives, this chemical gets a bad rap in the wrong hands. Law enforcement keeps a close watch on it for good reason: it’s a major ingredient in illicit drug synthesis, and environmental regulators see the potential for real harm if it ends up in places it shouldn’t go.
Why Proper Disposal Makes a Difference
Dumping leftover chemicals like acetic anhydride down the drain or tossing them in the trash isn’t just lazy — it poses a real risk to people cleaning up after you, to water systems, and to wildlife downstream. There’s a reason disposal rules exist: acetic anhydride mixes with water to produce acetic acid, which can eat through pipes and harm aquatic life. Think of rivers, fish, and even farming land that relies on clean water—no one wants to be the reason for a local disaster just because they cut corners with chemical waste.
My own experience working in a college laboratory meant seeing, firsthand, just how easily hazardous substances could slip through the cracks if folks aren’t trained well. There were horror stories floating around of someone ignoring warning signs and ending up with chemical burns or an emergency evacuation. The real issue isn’t just about one person’s safety, but about protecting whole communities and our shared water and land.
What the Experts Recommend
The Environmental Protection Agency (EPA) and other agencies keep disposal guidelines pretty clear. They point out that all unwanted acetic anhydride should go to certified chemical waste handlers. These companies know how to neutralize and destroy hazardous chemicals without releasing dangerous fumes. In most places, authorities ask chemists and businesses to segregate acetic anhydride from other waste streams, label it, and log it for pickup by professionals. Even in smaller labs, working out a contract with a licensed disposal provider beats risking fines or injuries.
Room for Improvement
Small businesses and home experimenters sometimes get left out of these the loop, facing complicated paperwork or high fees from professional waste handlers. That gap encourages shortcuts — and there’s the real risk. Community-run collection events or subsidized hazardous waste drop-offs could ease this. Some cities have programs where residents can drop off left-over chemicals once or twice a year. More resources like this, paired with better education on why it all matters, keep disasters at bay and costs lower for everyone.
Smart Decisions Count
Modern life means dealing with chemicals we barely think about, but everyone has a stake in making sure waste doesn’t poison water or hurt people. My own rule: if I wouldn’t want something in my backyard, I don’t want it flushed away or buried somewhere to become a problem later. Acetic anhydride reminds us that responsibility isn’t just a label on a bottle—it's a daily choice with long-term impact.
What is the chemical formula of acetic anhydride?
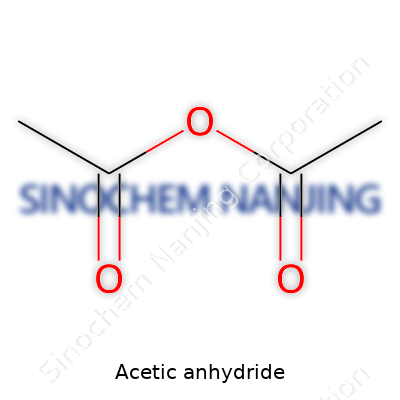
Getting to the Heart of Acetic Anhydride
Acetic anhydride, carrying the formula (CH₃CO)₂O, shows up all over in modern manufacturing, chemistry labs, and even in places people don’t always notice. If you’ve used aspirin, you’ve brushed up against this chemical—pharmaceutical makers depend on it to produce acetylsalicylic acid. The formula may look like a jumble at first, but it brings together two acetyl (CH₃CO-) groups attached by an oxygen atom. It catches chemists’ attention because it reacts with other molecules easily, making it a handy tool for making new compounds.
Why Formula Familiarity Matters
Getting the formula right in the lab isn’t only about penmanship—mistakes can lead to wasted chemicals or, in a worst-case scenario, dangerous reactions. At school, I once added what I thought was acetic acid to a mixture, only to realize by its smell (a much harsher, almost nasal-burning odor) I’d picked up acetic anhydride. The formula set me straight, and a patient chemistry instructor walked through why mixing it with water sends up plumes of acetic acid vapor. In labs and factories, correct formulas don’t just keep experiments on track—they keep people safe.
Real-World Uses and Risks
The world leans on chemicals like acetic anhydride for more than creating medicine. Textile makers use it to turn cellulose into acetate fibers, which end up in clothing and even cigarette filters. Food businesses use it behind the scenes, not directly in what you eat, but to make things like flavors and fragrances. Yet, there’s a darker underside. Illicit drug labs covet acetic anhydride for turning morphine into heroin. This shadow market means governments clamp down on sales, and chemical suppliers need strict tracking protocols.
Responsible Handling, Smarter Policy
Open discussion on chemical safety and policy has to move beyond paperwork and into everyday life. Anyone who’s spent time in a university or commercial lab hears the emphasis on safety goggles and fume hoods. Yet, talking safety means connecting students and employees to the real dangers—not just rattling off hazard labels. Workshops with stories, rather than dry lists, get people’s attention. Strong regulations—such as licensing and real-time reporting for buyers, and surprise inspections—produce better compliance than just legal threats. Suppliers also benefit from tech-based inventory controls that flag suspicious purchases.
Looking Forward: Knowledge Builds Trust
Big issues with chemicals like acetic anhydride, from accidental exposure to illegal trafficking, don’t fade on their own. Clear education with real-world stories sticks. Walking through the implications of a napkin-sized formula links classrooms to headlines—and keeps people alert. For those in the trenches of chemical production or public health, understanding both the power and risk crammed into a simple formula, (CH₃CO)₂O, lays the groundwork for smarter decisions.
| Names | |
| Preferred IUPAC name | acetic anhydride |
| Other names |
Acetic oxide Ethanoic anhydride Acetyl oxide Acetanhydride |
| Pronunciation | /əˈsiːtɪk ænˈhaɪdraɪd/ |
| Identifiers | |
| CAS Number | 108-24-7 |
| 3D model (JSmol) | `3D model (JSmol)` string for **Acetic Anhydride**: ``` C1(=O)OC(C)=O ``` |
| Beilstein Reference | 12045 |
| ChEBI | CHEBI:17647 |
| ChEMBL | CHEMBL14078 |
| ChemSpider | 6088 |
| DrugBank | DB00741 |
| ECHA InfoCard | 3292445820120 |
| EC Number | 203-564-8 |
| Gmelin Reference | 821 |
| KEGG | C01407 |
| MeSH | D000004 |
| PubChem CID | 176 |
| RTECS number | AK1925000 |
| UNII | 7OV03QG267 |
| UN number | 1715 |
| Properties | |
| Chemical formula | C4H6O3 |
| Molar mass | 102.09 g/mol |
| Appearance | Colorless liquid |
| Odor | Pungent, vinegar-like |
| Density | 1.082 g/cm³ |
| Solubility in water | Reacts with water |
| log P | -0.02 |
| Vapor pressure | 0.4 mmHg (20°C) |
| Acidity (pKa) | 9.0 |
| Basicity (pKb) | 3.58 |
| Magnetic susceptibility (χ) | -43.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.370 |
| Viscosity | 1.08 mPa·s (at 25°C) |
| Dipole moment | 2.49 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 188.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -703.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | “-1410.0 kJ/mol” |
| Pharmacology | |
| ATC code | J01XX08 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H226, H302, H314 |
| Precautionary statements | P210, P233, P234, P240, P241, P242, P243, P261, P264, P271, P272, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P312, P314, P321, P330, P337+P313, P362+P364, P363, P370+P378, P403+P233, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 49 °C |
| Autoignition temperature | 316°C (601°F) |
| Explosive limits | 2.7% - 10.3% |
| Lethal dose or concentration | LD50 oral rat 1780 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1780 mg/kg |
| NIOSH | NIOSH: AK2625000 |
| PEL (Permissible) | 10 ppm |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Acetyl chloride Acetic acid Isobutyric anhydride Phthalic anhydride Propionic anhydride |

