Acetic Acid: An Editorial Reflection on a Common Yet Powerful Chemical
Historical Development
The story of acetic acid goes back thousands of years, woven into humanity’s earliest experiments with fermentation and preservation. Ancient Egyptians and Babylonians relied on vinegar—a dilute solution of acetic acid—not just to sharpen their foods but to safeguard them from spoilage. It wasn’t until the Renaissance that chemists like Johann Rudolf Glauber isolated impure forms of acetic acid from vinegar, starting the march toward industrial processes. By the 19th century, German chemist Hermann Kolbe revolutionized its production through chemical synthesis instead of biological methods, paving the way for the painfully efficient scale of today’s chemical plants. Modern acetic acid, more often produced from methanol, owes its existence to lessons learned over centuries, marrying the patience of ancient brewers with the relentless innovation of industrial chemists.
Product Overview
Acetic acid, known variously as ethanoic acid, vinegar acid, or E260, commonly appears in diluted form as the staple “vinegar” in homes and restaurants. Despite its common presence in the kitchen, industrial acetic acid clocks in at far higher concentrations, serving as a building block for everything from adhesives to pharmaceuticals. Chemists assign it the formula CH3COOH, a molecule simple enough to draw on a napkin yet powerful enough to anchor global industries. Its role straddles the worlds of food, textiles, plastics, and cleaning products, which makes it both versatile and essential for supply chains that touch daily life.
Physical & Chemical Properties
Acetic acid catches attention with its sharp, pungent aroma—a whiff instantly conjures up memories of vinegar or pickled vegetables. With a melting point just over 16.6°C, it freezes on a chilly day, forming ice-like crystals that fascinate anyone seeing them for the first time in a laboratory. In liquid form, it is colorless, and its high polarity lets it mix readily with water, making it easy to dilute or use as a solvent. Its acidity arises from the carboxyl group, which can release a proton, lowering the pH of solutions and lending acetic acid its cleaning prowess as much as its sour taste. The pure compound, known as glacial acetic acid for its tendency to solidify at room temperature, is hygroscopic and readily absorbs water from the air. This property shapes not just storage requirements but informs safe handling practices as well.
Technical Specifications & Labeling
Acetic acid demands respect in handling and storage. Concentration levels run from the dilute vinegar in salad dressing (around 5% by volume) up to glacial acetic acid, which exceeds 99% purity. Labeling laws vary region by region, but regulated substances like acetic acid must carry proper hazard warnings reflecting their corrosive nature and inhalation risks. Regulatory bodies, including the FDA and EPA in the US, set strict limits for residual acetic acid in foods, pharmaceuticals, and household products. Laboratories and process plants must label storage containers with concentration, hazard class, and unique identifiers not only for compliance but for the safety of every person who may interact with this compound.
Preparation Method
Industrially, most acetic acid originates from the carbonylation of methanol, a process that marries carbon monoxide with methanol under high temperature and pressure using a rhodium or iridium catalyst. This method replaced the slower fermentation process, which historically involved slow oxidation of ethanol by Acetobacter bacteria. Methanol carbonylation can crank out millions of metric tons annually, supporting global demand for acetate plastics, solvents, and feedstocks. The production method affects both cost and purity, shaping how different industries source their raw materials and even how sustainable their supply chains become, since methanol itself derives from either fossil fuels or, increasingly, biomass.
Chemical Reactions & Modifications
Acetic acid reacts with alcohols to form esters—compounds that show up in everything from fruit flavorings to solvents and perfumes. Combining with metals or bases yields acetates, salts with their own industrial uses. The carboxylic group makes acetic acid a prime candidate in synthesis, where its reactivity opens pathways for producing compounds like acetic anhydride, vinyl acetate, and various pharmaceuticals. In polymer chemistry, acetic acid modifies cellulose to produce cellulose acetate, which later transforms into fibers for clothing or film base in photography. Such versatility means that advances in acetic acid chemistry ripple outward, influencing sectors as disparate as medicine and plastics.
Synonyms & Product Names
Across markets and disciplines, acetic acid answers to many names. In supermarkets and kitchen cupboards, “vinegar” dominates. In chemical catalogs and lab benches, it’s “glacial acetic acid” at high purity. Regulatory documents and global trade channels might refer to it as E260, a food additive code, or ethanoic acid, following IUPAC conventions. Understanding these interchangeable terms avoids confusion—especially critical for importers, lab technicians, or anyone keeping compliance with product labeling and hazard communication standards.
Safety & Operational Standards
Any professional who has faced a splash or inhaled fumes of concentrated acetic acid knows it burns and irritates on contact. Handling this substance means donning gloves, goggles, and working in well-ventilated spaces or fume hoods. In workplace environments, exposure limits set by bodies like OSHA cap air concentrations to prevent chronic respiratory, skin, or eye damage. Proper training saves both skin and careers, and clear hazard labeling lets even the newest intern steer clear of avoidable accidents. Transport regulations govern container sizes, packing materials, and emergency response needs, enforcing a culture of respect around such a potent chemical.
Application Area
Acetic acid’s reach extends far beyond salad dressing. Industrial teams harness it to manufacture vinyl acetate monomer, an ingredient in paints, adhesives, and sealants. The textile world transforms cellulose acetate into fibers for comfortable, breathable clothing, while film industries long relied on this same polymer for movie reels. Candy makers add measured amounts to tweak pH during processing, ensuring the right texture and shelf life. In medicine, dilute acetic acid cleans wounds and acts as an ingredient in numerous pharmaceuticals. Farmers use acetic acid-based herbicides to keep weeds in check, highlighting both the chemical’s accessibility and its biological power. This versatility also means that disruptions in acetic acid production ripple through multiple sectors, as seen in supply chain crunches affecting everything from food processing to plastics.
Research & Development
Research on acetic acid keeps evolving. Chemists and process engineers focus on improving catalyst stability, reaction efficiency, and sustainability. With mounting pressure to reduce fossil fuel dependence, methods that upgrade biomass into acetic acid or utilize renewable methanol sources gain traction. Environmental scientists track emissions and residues, looking for ways to keep production clean. Medical teams explore acetic acid’s uses as an antimicrobial, eyeing its potential in fighting antibiotic-resistant pathogens. Even in food science, researchers study how acetic acid interacts with flavors and textures, expanding what chefs and manufacturers can achieve. Advances in process intensification and membrane technology could cut energy and resource costs, helping even legacy plants pivot to greener operations.
Toxicity Research
Acetic acid, despite its familiar reputation, poses significant health risks at high concentrations. Short-term exposure to concentrated fumes can burn eyes, noses, and throats. Prolonged skin contact brings blistering and pain. Acute ingestion of concentrated solutions, tragic though rare, can damage the gastrointestinal tract. Animal studies reveal dose-dependent toxicity, prompting regulatory agencies to routinely reassess acceptable levels in occupational and food settings. Chronic exposure data remain limited, pressing the need for more robust epidemiological studies. On the environmental side, concentrated acetic acid runoff lowers pH in waterways, although its rapid biodegradability means it rarely sticks around for long. Even in common use as vinegar, governments cap allowable concentrations in foods to keep consumers safe.
Future Prospects
The world’s thirst for acetic acid shows no signs of slowing. With demand for plastics, coatings, and synthesized flavors rising in step with population and consumption, researchers face the ongoing challenge of producing it more sustainably and safely. Green chemistry offers promising avenues: bioengineered yeast strains that convert agricultural waste to acetic acid, or efficient electrochemical routes using renewable electricity, give hope that future supply won’t depend on fossil fuels. Regulatory pressure on emissions in industry-heavy regions motivates plant managers to adopt cleaner catalysts and recovery systems. As more sectors—especially pharmaceuticals and advanced materials—seek specialty acetates and tailored solutions, flexibility and innovation in acetic acid manufacturing will drive industry upgrades. The humble sour taste of vinegar may one day represent the triumph of sustainable chemistry as much as centuries-old tradition.
What is acetic acid and what are its main uses?
The Simple Side of Chemistry
Acetic acid lands on my kitchen counter every day. It’s the sharp smell from vinegar, and it cuts straight through a salad dressing. This stuff isn’t rare or mysterious. It shows up in places that matter—inside pickles, next to cleaning supplies, sometimes even as a topic in my old high school science class. Chemically, it’s a pretty simple acid with two carbons, four hydrogens, and two oxygens. For most people, that counts as enough background. But this simplicity hides a bit of power.
From Vinegar Jars to Factory Floors
The most familiar acetic acid use pops up in the kitchen. Folk wisdom treats vinegar as essential: for flavor, for food safety, and for chemistry experiments at the dining table. White vinegar typically clocks in around 5% acetic acid. Grandma’s pickling recipes wouldn’t work without it—trapping those crunchy cucumbers, beating back unwanted bacteria. Restaurants, home cooks, artisanal producers: all trust this acid to do the heavy lifting in everything from condiments to sauces.
Big factories see acetic acid differently. Industrial setups pump out huge volumes, feeding a hunger for plastics, paints, inks, and adhesives. One main job: turning raw acetic acid into vinyl acetate monomer, a core ingredient in the plastic films and glues that stick labels, seal windows, and protect food. Chemists count it as a “platform chemical,” meaning it sits early in the chain and shapes what follows. The food world gives acetic acid its sharp taste; the industrial world shapes everything from synthetic fibers to solvents.
Cleaning and Disinfecting
Most people trust vinegar for more than salads. Acetic acid stands among the favorite eco-friendly cleaners. It cuts through limescale on kettles, leaves bathroom tiles shining, and dissuades mold from taking over windowsills. Its low pH and ability to dissolve minerals make it a go-to for folks turning away from harsh chemicals under the kitchen sink. Hospitals sometimes use stronger versions to handle medical equipment, though precise concentrations for safety and disinfection need expert handling. It’s one of the few chemicals with equal value in a chef’s kitchen and a janitor’s cart.
The Risky Edge
No chemical with so many uses goes without risk. Undiluted acetic acid can burn skin and eyes. Mistaking bottle strength for kitchen strength causes plenty of accidents every year. Breathing in high concentrations can create coughing fits or lung troubles in workplaces. I remember seeing labels warning about mixing acids with bleach—words everyone should take seriously since the result can mean toxic gases in the air. Clear labeling and education go a long way. Regular folks need to pay attention when switching between household strengths and lab-grade acid.
Better Handling, Smarter Regulation
Safe use starts with education and access. Clear instructions on bottles remain crucial. In industrial settings, good training and protective equipment cut down on accidents. On the environmental side, some newer plants recover or recycle acetic acid to ease pollution and chemical waste, but adoption moves slowly. Thoughtful rules and better practices lower risks for everyone: workers, families, and even the planet. Whether stirring a jar of pickles or working near large reactors, respect for what acetic acid does keeps its benefits on our side.
Is acetic acid safe to handle and what precautions should be taken?
Kitchens and Labs: A Surprising Connection
Pour some vinegar into a salad and you’re dealing with acetic acid. Yet, anyone who’s ever opened a jug of concentrated acetic acid in a lab knows it’s a whole other level. The stuff in your pantry tops out around five to seven percent, but some bottles in the lab go well over 99 percent. That difference matters.
Why Concentration Changes Everything
I remember the first time my supervisor showed me glacial acetic acid. It looked harmless, but the smell burned my nose. A few drops and I instantly understood why keeping a fume hood running wasn’t just about following a rule. Pure acetic acid eats through cotton and gives off fumes that sting nose and eyes. Medical journals record cases of burns and lung damage when people used it with too little respect.
Direct Contact Isn’t Just Annoying—It’s Dangerous
Health authorities, including the CDC, point out that splash incidents cause serious burns. Glacial acetic acid really isn’t a household chemical. The skin burns are often slow to heal and leave scars. Eyes are even more vulnerable. The World Health Organization lists it as a high-risk chemical for accidental exposures. My chemistry professor kept a bottle wrapped in an extra layer for a good reason.
Vapors: The Invisible Threat
Fumes drift up even from open bottles, and nobody wants to breathe them in. In some makeshift setups, people have landed in the ER with coughing fits and chest pain. Anyone who has tried moving a bottle outside a lab’s ventilation system knows the whole room starts to sting your throat. People get nosebleeds and headaches if a small spill isn’t cleaned up fast.
Practical Protection Every Time
Protection isn’t complicated. Lab coats and gloves go on before a bottle gets opened. Nitrile gloves won’t dissolve, but some thin ones let the chemical seep through. I relied on thick gloves after seeing a classmate’s red hands. No one forgets the lesson of goggles either. Once I saw someone splash a diluted solution onto their face. Eyewash stations save eyes, but it’s best not to need one.
Safe Handling at Work and Home
At home, vinegar belongs in food, not on skin and not in eyes. Heavy-duty acetic acid should stick to proper labs. Anyone who works with the concentrated stuff needs training—really. Safety Data Sheets spell out handling, storage, and spill control in plain language. Locked storage keeps curious kids out. Spills deserve attention before someone else gets exposed. I’ve seen people skip labels, only to grab the wrong bottle. Label everything. Never trust faded ink.
Fixing Common Problems
Accidents drop when people keep clear instructions and the right gear nearby. I’ve set up workspaces with extra gloves and warning signs. Quick cleanups with baking soda keep surfaces from corroding. Open bottles under fume hoods, not desks. Make a rule about double-checking labels before pouring. These steps cut down on emergency calls and ugly accidents.
Respecting the Chemical Means Respecting Yourself
Safety routines make daily life easier, not harder. Whether you’re running biology experiments or cleaning counters, using the right version of acetic acid matters. Take the precautions and the job always gets done with everyone walking away healthy.
What is the concentration or purity of the acetic acid product?
Packing a Punch in the Lab and Beyond
Acetic acid turns up everywhere you look—vinegar on salads, cleaner in kitchens, key ingredient in chemical plants. That clear, sharp-smelling liquid looks so unassuming, but any mix or measure carries real-world impact. What concentration sits in the bottle makes all the difference, whether you’re a chemist checking your process, or someone trying to make pickles at home.
Different Jobs, Different Strengths
Walk through a grocery store and you’ll find vinegar, that household staple, with around 5% acetic acid. Taste, preservation, and safety come together at this level—enough bite to flavor food and slow down bacteria, not so much that someone finds their tongue burning. But step into a chemistry classroom or industrial lab, and bottles carry labels showing 99% purity or higher. No one cooks with that stuff. High-purity acetic acid, often called “glacial,” freezes at temperatures below 16°C, and a drop can eat away at your hands.
The reason for such a wide gap often comes back to use case. Textile factories rely on high concentrations when dyeing fabric. Pharmaceutical plants use it as a key building block, requiring strict quality standards. Even in plastics production, like in making polyvinyl acetate for adhesives, tiny impurities change the outcome. Each of these industries knows exact concentrations matter—precision reduces waste, prevents side reactions, and can even protect workers.
Quality Control Builds Trust
From my time working in a food lab, I remember the strict controls. Even a small error in acidity could ruin a whole batch of pickled vegetables or create product safety questions. Most labs check batches by titration, measuring the acid’s true strength, not just trusting what the label says. Anything out of range gets rejected. In global markets, companies source acetic acid under strict certifications—ISO, USP, or FCC—to assure everyone from food inspectors to doctors that what’s inside the drum matches expectations.
Poor-quality acid opens doors to all sorts of trouble. In cleaning products, too little acid and germs stick around. Too much acid, pipes and hands can get burned. Even wineries, which use vinegar as a cleaning agent, pay close attention to purity, because residues can ruin the taste of a whole season’s work.
Improving Concentration Means Safer Products
Getting the right purity goes beyond just mixing and bottling. Reliable companies invest in strong analytical labs, staffed with trained technicians and modern gear. Gas chromatography or high-performance liquid chromatography provide quick and traceable results. This kind of oversight means fewer recalls and less risk to people handling the chemical.
If a mistake slips through, businesses own up to it promptly—pull products, offer refunds, update customers. Regulators also play a big role, conducting inspections and requiring clear labelling for anything reaching store shelves or entering factories. Anyone buying a drum of acetic acid, whether a chef or a pharmacist, looks to see these details before using it for something important.
Simple Solutions to a Complex Problem
Education stands out. People who understand the risks of different concentrations can handle and store acetic acid more safely. Safety data sheets, training sessions, and clear warning labels all act as defense lines. On the supply side, transparent testing, third-party certification, and digital traceability keep everyone in the loop if standards change or batches get compromised.
The future looks promising, with sensor-based checks and real-time quality notifications improving trust in the supply chain. Every step—production, testing, labelling, and use—builds confidence that acetic acid in the bottle matches what’s needed for every task.
How should acetic acid be stored and what is its shelf life?
Understanding Acetic Acid’s Quirks
Acetic acid forms the backbone of industrial chemicals, food production, and even home remedies. Anyone who’s worked in a lab or even walked into a janitor's closet knows the sharp bite of vinegar in their nose—that's dilute acetic acid. But the concentrated form demands real respect. Breathing in too much or letting it spill burns more than nostrils. So how acetic acid gets stored in any workplace or school lab becomes a detail that really counts.
What Proper Storage Really Looks Like
Anyone thinking a plastic water bottle will do just hasn’t seen the damage acetic acid does to flimsy containers. This clear liquid eats through some plastics, not to mention paint and metal. Most facilities keep concentrated acetic acid in tightly sealed bottles made from glass or thick, high-density polyethylene. Sturdy, tightly closed containers keep the fumes from leaking and damaging nearby labels, equipment or worse, a person’s lungs.
My old university chemistry storeroom kept the stuff on lower shelves, so spills didn’t land on faces or upper bodies. Now I do the same at home, keeping household vinegar locked away from curious hands and mismatched lids. In workplaces, signs pop up reading ‘corrosive’ or ‘flammable’. Teams set up dedicated acid cabinets lined with plastic, far away from bases or oxidizers—mixing them creates serious hazards. Stainless steel and galvanized iron don’t belong anywhere near acetic acid, either. Corrosion wins every time.
A good habit: check those caps before storing bottles away. Even a half-turn loose, and the acid’s fumes drift out, slowly rusting anything metal nearby. Every seasoned lab tech has scrubbed away brown rust stains left behind after a tiny leak went unnoticed for a week. Keeping everything upright and in well-ventilated areas stops fumes from building up indoors. Flammable storage cabinets work well, since concentrated acetic acid catches fire at a lower temperature than some folks realize. NIOSH and OSHA both stress these points, based on decades of accident data.
Acetic Acid Doesn’t Last Forever
Vinegar bottles in your pantry won’t go bad any time soon, but strong acetic acid won’t hold its edge forever either. High purity batches in closed, undamaged containers run stable for about two years, sometimes three, if they live in cool, dry, dark rooms. Heat and sunlight nudge acetic acid toward decomposition, with strong batches giving off more fumes as time passes. Humidity speeds up water absorption, diluting the acid and changing its handling risks.
After years working next to chemical cabinets, I’ve seen what happens if forgotten bottles live too long. Caps corrode, the liquid inside gets cloudy, and labels fade. Every safety data sheet recommends marking down opening dates, tracking how long acids sit unused. Regular audits save headaches by catching these before they become safety risks or impact lab results.
For schools and businesses, training everyone who touches acid bottles pays off. Extra time spent reviewing storage steps and shelf life rules means fewer spills, injuries, or ruined experiments. Following responsible guidelines really roots itself in those small day-to-day decisions—checking a label, tightening a cap, moving a bottle down a shelf. Years of evidence and experience show these habits work, keeping workspaces safe and reliable for everyone who steps through the door.
Can acetic acid be mixed or diluted with other chemicals?
Everyday Life and Labs Collide
Mixing chemicals looks easy on TV, but behind the scenes, the risks run deep. Acetic acid, known to most as the bite in vinegar and a reliable lab staple, pops up in cleaning agents, food processing, and even textiles. Almost everybody mixes it with something at some point. It sounds simple—just add water or another solvent—but simple mistakes invite surprises no one wants. I once splashed some on my hand while juggling tasks early in my career, thinking “It’s just vinegar's big cousin, it’ll be fine.” Angry red irritation taught me to give acids the respect they demand, regardless of concentration.
Understanding the Chemistry, Avoiding the Burns
Splashing acetic acid into water doesn’t just dilute the acid; it releases heat. Toss it into the wrong chemical—say, a strong oxidizer— and you might spark a reaction far faster and hotter than you’re ready to handle. It’s never enough to check the label once and shrug—it pays to know each substance in the mix. Glacial acetic acid becomes quite dangerous thanks to its high concentration and love of giving off flammable vapors. More than one news story has started with someone ignoring that fact and leaving an “unlabeled container” in the corner.
Stats from poison control centers in the US point to hundreds of acetic acid exposures each year. Most are accidental, often from household cleaning blends or DIY projects that spin out of control. I recall a neighbor who mixed vinegar with bleach to tackle mold, expecting double the cleaning power. Instead, she got a blast of nasty fumes and a tough phone call to the emergency line. Even simple-sounding reactions release compounds like chlorine gas, strong enough to send healthy folks running outside, wheezing.
Taking Safety Seriously
Goggles and gloves aren’t just for chemistry teachers trying to scare high school kids straight. Anyone handling acids, including acetic acid, needs real protection, no matter how quick the job appears. Fume hoods and ventilation become non-negotiable in labs. At home, an open window and rubber gloves go a long way toward keeping skin and lungs safe. Major chemical supply companies echo the same warnings: always add acid to water—never water into acid. Anyone who’s watched that sizzling reaction up close knows this tip saves skin and glassware.
Solutions Built on Knowledge
Mixing acetic acid with other chemicals starts and ends with research and planning. Modern safety data sheets online hold all ingredients, dangers, and recommended handling methods. Before grabbing bottles, reading through intended reactions could spare a doctor’s visit. At work, most companies push regular safety refreshers for good reason. Home users could benefit from simple guides or safety apps—something to consult, even on a cleaning day. Even a quick internet search to double-check combinations is better than guessing.
Why it Matters for Everyone
People in factories, teachers in science classrooms, and even parents using vinegar for chores all face the same truth—acetic acid reacts with its environment, and not always in gentle ways. Getting hands-on with chemicals demands curiosity, sure, but also respect for the risks and tireless attention to safe habits. Each story of mishap I’ve seen could have played out differently with a minute's thought and basic protection. Safety isn’t about following rules just for compliance; it’s about understanding what’s really in that bottle and what happens when you start mixing. That knowledge pays itself back with peace of mind, clean results, and health intact.
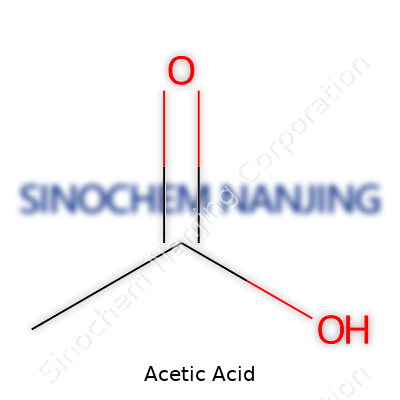
| Names | |
| Preferred IUPAC name | ethanoic acid |
| Other names |
Ethanoic acid Vinegar acid Methanecarboxylic acid Acetyl hydroxide |
| Pronunciation | /əˈsiː.tɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 64-19-7 |
| 3D model (JSmol) | `/wiki/Special:EntityData/Q47512.ttl` |
| Beilstein Reference | 1713884 |
| ChEBI | CHEBI:15366 |
| ChEMBL | CHEMBL: CHEMBL153 |
| ChemSpider | 175 |
| DrugBank | DB03166 |
| ECHA InfoCard | ECHA InfoCard: 024-002-00-9 |
| EC Number | EC 200-580-7 |
| Gmelin Reference | 635 |
| KEGG | C00033 |
| MeSH | D001097 |
| PubChem CID | 176 |
| RTECS number | AF1225000 |
| UNII | Q369O8VY0H |
| UN number | UN2789 |
| Properties | |
| Chemical formula | C2H4O2 |
| Molar mass | 60.052 g/mol |
| Appearance | Clear, colorless liquid with a pungent, vinegar-like odor. |
| Odor | Pungent, vinegar-like |
| Density | 1.049 g/cm³ |
| Solubility in water | Miscible |
| log P | -0.17 |
| Vapor pressure | 1.5 kPa (20 °C) |
| Acidity (pKa) | 4.76 |
| Basicity (pKb) | 14.5 |
| Magnetic susceptibility (χ) | -5.8×10⁻⁶ |
| Refractive index (nD) | 1.371 |
| Viscosity | 1.22 mPa·s (at 25 °C) |
| Dipole moment | 1.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 159.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −484.3 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -875.0 kJ/mol |
| Pharmacology | |
| ATC code | S02AA03 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H226, H314, H318 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P260, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P312, P321, P363, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 39 °C |
| Autoignition temperature | 463 °C (865 °F) |
| Explosive limits | 4% - 19.9% |
| Lethal dose or concentration | LD₅₀ Oral Rat: 3,310 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3,310 mg/kg (oral, rat) |
| NIOSH | NA213 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Acetic Acid: "10 ppm (25 mg/m³) TWA |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | IDHL: 50 ppm |
| Related compounds | |
| Related compounds |
Acetate Acetyl chloride Acetic anhydride Acetylsalicylic acid Citric acid Formic acid Propionic acid |

