Acetamide: An Editorial Reflection on a Humble but Essential Compound
Tracing the Journey: Historical Development
Chemistry has a way of turning simple ideas into irreplaceable tools, and the story of acetamide speaks to that. Folks tinkering with organic acids in the 18th century found that acetic acid, which comes from vinegar, likes to make friends with ammonia. From this, acetamide was born, first isolated in laboratories where many discoveries came from curiosity or happy accidents rather than grand, orchestrated experiments. Early chemists didn’t have modern equipment, so their method boiled down to mixing, heating, and noting what happened next. By the 19th century, with organic chemistry flourishing, acetamide grew in value. Scientists saw it as a building block, a connector that quietly helped shape everything from medicine to plastics. The humble start and steady growth of acetamide mirror the progress of chemistry: patient, practical, and often underappreciated by those outside the field.
Product Snapshot: What Acetamide Really Is
Ask a chemist what acetamide looks like, and the answer is straightforward: a white, odorless solid that melts into a clear liquid with moderate heat. It doesn’t sting the nose like some of its rowdier relatives, and it dissolves with ease in water. These unassuming traits actually make it quite valuable. Its chemical formula—CH3CONH2—places it squarely between simple acids and more complicated organic molecules. I remember in the lab, acetamide served as a stand-in when we wanted manageable, predictable reactions. It kept things straightforward when other chemicals threw in surprises or proved difficult to work with. There’s comfort in a compound that rarely misbehaves.
Physical and Chemical Nature: More Than Meets the Eye
Acetamide has a soft spot for water and methanol and doesn’t show much interest in dissolving in fats or oils. Its melting point hugs 82 degrees Celsius, a number etched in my mind after watching it liquefy in countless practical sessions. The molecule’s structure places an amide group next to a methyl group, a pairing that makes it less reactive than its acid or amine cousins but significantly more flexible. It won’t explode, rust metal, or give off clouds of choking fumes, which is rare comfort for anyone who’s worked through chemical safety protocols. These qualities help it play a background role in synthesis yet pop up front when researchers seek manageable toxicity and stability. Acetamide hangs quietly in the corner but steps forward when called.
Technical Specifications & Labeling: Practical Demands in Real Use
Strict handling guidelines matter, especially in research and manufacturing settings. Acetamide usually arrives in sealed, moisture-proof containers with simple labeling naming its CAS number and chemical structure—just enough to avoid confusion with its close relatives. In my view, these labeling requirements do more than just tick boxes; they foster awareness. Good labeling helps avoid mistakes, and old chemists will tell you, “Most accidents start with a bad label.” Regulations focus on purity, limiting trace toxins—nitrate and acetic acid usually get the most scrutiny. Only the most fastidious labs chase pharmaceutical-grade purity, but even general chemical work demands reliable, tested sources to prevent unwanted surprises in results. Metering out respect for these technical rules pays off with safer workspaces and cleaner experiments.
Preparation Method: Simplicity and Ingenuity from the Lab Bench
Turning vinegar into acetamide is as much a rite of passage in organic chemistry as titrating an acid. The method, familiar to generations, runs acetic acid together with ammonia. Heat drives off water, leaving behind acetamide with a little coaxing. The practical side is often messier than textbooks suggest—impurities sneak in, yields drop if care lapses. Large-scale production in industry uses pressure reactors and precise temperature controls, earning higher purity and less waste. Some scientists experiment with alternative synthesis routes, but most stick with what works. From a practical standpoint, it’s a reaction that rewards patience, not cleverness, and that has always seemed fair to me.
Reactions and Modifications: Acetamide as a Middleman
Acetamide doesn’t grab headlines for wild chemistry, but its position lets it swap partners quietly and without fuss. In labs, it accepts substitutions at the nitrogen and carbon sites, producing a range of derivatives useful in dyes, medicines, and polymers. Scientists often hydrolyze acetamide to acetic acid or convert it to methylamine using strong acids or alkalis. For students, watching these transformations is a staple of organic chemistry courses. The practical applications of these reactions extend to industry, where small tweaks turn it into something new and valuable. The reliability of acetamide in these roles explains its quiet reputation as a backbone molecule—never glamorous, always useful.
Synonyms and Other Names: Language Reflects Necessity
Acetamide gathers a handful of names over its career. The IUPAC name, ethanamide, reflects modern systematic nomenclature, yet most chemists, myself included, stick with acetamide as tradition. It gets called acetimidic acid amide or vinegar amide in older texts. These variants crop up in patents, regulatory documents, and safety data, showing how a single molecule can inhabit many worlds at once. Precision in naming helps everybody stay on the same page, especially in cross-border shipments or international research.
Safety and Operational Rules: Practical Hazards and Careful Handling
Working with acetamide doesn’t set off the highest alarm bells in labs, but its connection to toxicity research keeps everyone mindful. Long-term exposure, especially by inhalation or ingestion, raises red flags—rat studies link chronic exposure to liver and kidney issues, along with potential carcinogenic effects. Good lab practice says don’t eat compounds, avoid unnecessary inhalation, and always wash up after use. Proper gloves and splash-proof eyewear fall into the unbreakable routine. Storage demands dry, cool, sealed conditions—acetate forms if moisture sneaks in. Older colleagues warned me early on: “Treat every chemical like it’s trouble until proved otherwise." That mindset anchors operational safety.
Application Area: Beyond the Obvious Horizons
Acetamide creeps into more fields than most would guess. Recently, its solvent abilities found roles in plastic production and microelectronics design. Pharmacy benefits from its role as a starting material in developing drugs and fine chemicals. In agriculture, it offers routes to pesticide development without excessive risk to handlers, so long as it’s managed by experienced people. Some research teams borrow acetamide’s stable structure to model metabolic studies, tracking how simple organics metabolize in animal studies. These cross-disciplinary uses highlight its versatility. Having seen acetamide pop up unexpectedly in different projects taught me to keep an open mind about old compounds finding new work. Progress often comes from reimagining what’s already familiar.
R&D and Toxicity Work: Navigating Risks with Open Eyes
Toxicological research never really ends for compounds like acetamide. Animal testing continues to probe for subtle, long-term harm, especially around cancer risk, prenatal effects, and respiratory exposure. Most recent findings lean toward recommending continued caution, especially in workplaces with chronic low-level exposure. Modern labs invest in better air filtration and split up hazardous operations to limit risk, drawing lessons from years of occupational health studies. These safeguards cost money, but long-term benefits to worker health outweigh the initial expense. As rules evolve, scientists work directly with regulators to keep data transparent and recommendations sensible, avoiding the pitfalls of overreaction or complacency.
Outlook: Where Acetamide Might Go Next
Basic chemicals like acetamide don’t often make headlines, yet demand rarely fades. New application areas, from next-generation polymers to niche pharmaceuticals, depend on reliable intermediates. On the regulatory front, tightening rules push suppliers to ramp up purity, minimize contaminants, and adopt greener synthesis. Some researchers work to turn acetamide into a building block for biodegradable plastics or renewable chemicals, responding to environmental pressures that touch every industry. Every time I walk through a lab or manufacturing floor, evidence grows: old chemicals get new stories, shaped by new needs. Acetamide may not capture public imagination, but inside science, nobody underestimates what basic molecules can do.
What is Acetamide used for?
Pocketed in Industrial Shelves and Classroom Cabinets
Walking through a science lab in high school, you probably passed a bottle labeled “acetamide.” Few notice it. Most only hear about acetamide during experiments that introduce students to basic organic chemistry. This little compound, simple as it might look—just a carbonyl group and an amine hitched together—shows up in more places than most folks suspect.
Setting the Stage in Organics
I first encountered acetamide in a college chemistry lab. The instructor handed out safety sheets—acetamide listed. Its unpleasant, mousey smell stuck around long after the lesson ended. Some find it sharper than household vinegar. Scientists reach for acetamide mainly because it’s a perfect building block. You heat it, add a strong base, and it morphs into methylamine, a molecule with big industrial ambitions. Methylamine sits at the root of many crucial products like pharmaceuticals, pesticides, and tanning agents. Acetamide’s role here: precursor that people trust due to its reliability and simplicity.
From Factories to Farmland
Industrial production leans on acetamide for more than making methylamine. It works as a solvent, helping dissolve other chemicals smoothly without leaving much residue. Paint manufacturers, textile factories, and even some plastics companies mix acetamide to help blend threads of product together or improve processing. Its ability to blend and smooth things out makes life easier for workers trying to avoid sticky messes or uneven finishes.
Small Doses, Serious Talk
Anyone looking deeper will find acetamide listed in some herbicide formulations. Farmers and manufacturers have found it helps buffer or adjust chemical properties for better performance on crops or soil. At the same time, toxicologists warn about overexposure. The International Agency for Research on Cancer labeled acetamide as “possibly carcinogenic.” The fact that agencies keep tabs on its use underlines how safety governs modern industrial chemistry. Companies must follow rules about how much acetamide can be used or released. I remember a safety officer drilling into our team about gloves, goggles, and good ventilation.
Home and Health
Acetamide rarely shows up directly in homes outside student chemistry kits. It never becomes a household cleaner, and regulators keep it away from food. Even so, its industrial cousins—products relying on acetamide derivatives—reach into everyday life: pharmaceuticals shaped from those basic building blocks, dyes used in clothing, and some components in plastics. The science behind this relies on precise control, careful storage, and well-trained hands on the production line. Handling steps matter, from warehouse to workbench.
What’s Next?
Allowing continued use of a versatile chemical like acetamide relies on balance. Responsible handling, strong ventilation in workspaces, regular monitoring, and clear health guidelines all matter. Schools and labs train students to respect substances, not fear them. Workers get updated training year-round in factories. Innovators chase safer alternatives or better containment methods—real answers come from commitment to improvement, not shortcuts. The best solutions grow from daily experience and attention to detail, not panic.
Is Acetamide toxic or hazardous?
A Closer Look at Acetamide’s Safety Concerns
Acetamide doesn’t draw much attention in everyday conversation, but anyone dealing with chemicals at home or work ought to know what makes a compound safe or risky. Acetamide is a white, odorless solid at room temperature, frequently found in lab settings and sometimes in industrial processes for manufacturing plastics and solvents. If you’ve ever had to handle chemicals, you probably already understand the pressures that come from keeping people safe while getting work done.
The Science Behind the Worry
Let’s break it down. Scientists have classified acetamide as a possible human carcinogen. The International Agency for Research on Cancer (IARC) found sufficient evidence of its ability to cause cancer in animals, though data in humans is less clear. The real challenge comes from long-term exposure to large amounts, especially if inhaled or ingested. There’s reason to take that seriously, because cancer risk in workplace environments has been linked time and again to repeated contact with certain chemicals.
Short-term risks shouldn’t get overlooked either. Breathing in acetamide dust can irritate your respiratory system, and getting the powder on your hands might lead to skin or eye discomfort. A friend of mine, who worked in a chemistry lab in college, remembers developing itchy eyes and a cough after a careless afternoon spent transferring powders between bottles. He bounced back after a wash and some fresh air, but that small incident hammered in the habit of keeping gloves and goggles on whenever possible.
What the Numbers Say
Safety data sheets, like those required by OSHA, report toxicity figures measured in rats and rabbits. Ingesting several grams per kilogram of body weight causes health issues. That’s a lot more than a person would meet in one careless touch, but people in workplaces can end up facing small doses repeatedly—and that’s where trouble starts. Cancer agencies and toxicologists urge caution, even at low doses over time, because predicting rare outcomes isn’t easy.
Best Practices Make All the Difference
The rules around chemicals have gotten tighter over the years for good reason. Hoods that whisk odors and dust away, gloves that block skin contact, and eye protection stop most potential accidents. If you’re storing or using acetamide, keep containers tightly sealed, away from heat, open flames, or moisture. Washing hands after use and cleaning up spills with a damp cloth (never dry sweeping) all go a long way. These steps echo the habits we picked up in industrial training—cutting corners leads to trouble.
The Bigger Picture: Regulatory Action and Safer Choices
Regulators in the US keep acetamide on hazardous chemical lists. Some industries have worked to phase out or replace it with safer alternatives, often driven by worker complaints and costs tied to workplace illness. As chemical safety moves further into public view, both employers and regulators look for chemicals that offer the same performance with fewer risks. Laboratories have switched to less toxic amides or water-based solvents. This isn’t just compliance in action—it’s a response to real health impacts tracked over decades.
New research continues to watch acetamide closely. Improved detection methods and long-term health studies will keep shaping safety standards. From my time around labs and factories, I can say that following basic safety habits and choosing less hazardous materials always pays off, both at the bench and at the bottom line.
What are the storage conditions for Acetamide?
What We're Dealing With
Acetamide, a simple amide of acetic acid, finds its way onto lab shelves, chemical storerooms, and even some industrial sites. The stuff looks harmless—a white, odorless solid with the tendency to absorb moisture from air. You’d think it’s just another jar with a complicated name. But if you skip the right storage steps, small lapses scale up into messy, even hazardous situations both for workers and the environment.
Why Humidity Stirs Up Trouble
Acetamide doesn’t just sit quietly. This chemical draws in moisture like a sponge, especially in humid lab environments. Left out, even with a loosely sealed cap, it clumps, dissolves, or changes consistency over time. Anyone who’s worked in a hot, muggy storeroom knows how quickly powders can cake together—what starts as a textbook sample can end up as a solid block, tough to handle and difficult to weigh accurately. Worse, those changes hit the reliability of experiments, throwing off key results.
Safe Temperatures and Fresh Air
Acetamide doesn’t burn easily, yet heating it above 220°C kicks off decomposition, sending out ammonia and acetic acid vapors. At room temperature, it won’t explode or burst into flame, but storing it near heat sources or in direct sunlight can stir up trouble by slowly breaking down the compound, letting off irritant gases. A cool storeroom with good ventilation shields staff from any accidental fume releases and helps maintain its purity over time. I’ve seen chemicals stored next to radiators or in sunlit windows degrade before their expiry date, costing both time and money for replacements.
Why Containers Make the Difference
Acetamide may look stable, but air and moisture creep in wherever they find a chance. Sealed, airtight glass bottles with screw caps block that out best. Plastic containers don’t always hold up; some plastics let air or vapor pass through, especially over long periods. Jars should get labeled with the date received and opened, keeping track of shelf life and identifying anything that’s spent too long outside controlled conditions. In my own lab days, unlabeled jars often led to mistakes—mixing up chemicals or using outdated stocks that threw off months of work.
Fire Safety Isn’t Optional
Even though it won’t spark flames at room temperature, Acetamide remains a combustible solid. Dust in the air or spilled powder near ignition sources can go up, especially in large quantities. Keeping it away from oxidizing agents, open flames, or electrical outlets helps reduce those risks. Cleanliness counts too—powder tracked around the storeroom creates fire hazards and contaminates other bench work.
Responsible Disposal and Environmental Concerns
Unchecked, acetamide can seep into water or soil. Don’t toss leftovers into the trash or down the sink. Follow hazardous waste procedures: use sealed waste containers, label them clearly, and arrange pickup by trained disposal services. This cuts down environmental exposure, keeps local wastewater safe, and complies with chemical regulations. Labs ignoring these steps endanger not only their staff but also the community around them.
Keeping Track for Better Results
Effective storage doesn’t just prevent spills or health risks; it boosts accuracy, protects investment in supplies, and keeps labs running smoothly. Staff need training with real stories and simple rules—not just policy binders. Getting conditions right means acetamide stays ready for research, and everyone goes home safely at the end of the day. That pays off beyond a single container.
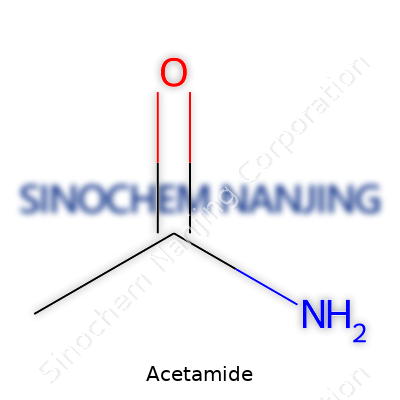
What is the chemical formula of Acetamide?
What Acetamide Means in a Lab and Daily Life
Acetamide pops up more than you might think. In labs, the formula C2H5NO tells you exactly how it stacks together: two carbon atoms, five hydrogens, one nitrogen, one oxygen. Growing up near a family member’s pharmacy, I saw plenty of strange bottles in the back room. Some had labels like “acetamide.” Most folks never wonder what’s inside those jars, but anyone mixing chemicals in a lab depends on formulas like C2H5NO to avoid mistakes.
Why the Formula Matters
A chemical formula isn’t just a string of letters and numbers. It’s a roadmap. In a chemistry class, a formula like C2H5NO turns into a model where every atom matters. Spill the wrong thing, mix the wrong compounds, and you can get problems fast. I learned this lesson working summers at a wastewater testing lab. If we forgot the difference between acetamide and a similar compound, analysis failed—wasting hours.
Beyond textbooks, the formula protects health and safety. Acetamide shows up in pharmaceuticals, plastics, and even in cigarette smoke. Knowing its formula, you can check safety data, understand exposure risks, and keep teams out of harm’s way. The Environmental Protection Agency keeps a close eye on chemicals like these for good reason. Too much exposure, and studies link it to health issues in some animals. People running industrial operations double-check the C2H5NO formula to track emissions and stay within safety rules.
Why Accuracy Counts
Think about baking a cake with salt instead of sugar—small mistake, ruined result. That’s what happens in a research project that swaps chemicals by mistake. A simple slip with a formula turns synthesis or industrial production expensive and dangerous. Years ago, a friend developed a nerve rash after a chemical spill in his college lab. Investigation pointed to mishandling acetamide because someone misread a label. The right formula linked the substance to the issue and pointed toward urgent solutions.
Precision also supports scientific growth. Researchers who focus on cancer drugs, plastics, and agriculture depend on C2H5NO. Without clear, accurate formulas, collaboration falls apart. If everyone follows the same system, advances happen quicker. My work with science writers taught me respect for peer-reviewed sources, since mixing up chemical names and formulas grows risk for readers and consumers.
Better Chemical Safety and Communication
People can do a few things to strengthen safety around chemicals like acetamide. Continuous training, plain-language labels, and double-checking references stop mix-ups before they start. Many schools and factories have begun to teach employees more about common compounds—not just for science majors, but for everyone in a building. Investing in real-world, scenario-based learning helped me remember critical formulas beyond exam dates.
Acetamide’s formula, C2H5NO, won’t win awards for complexity, but it keeps everyone on track in labs, industry, and medicine. Anyone who’s ever read the fine print on a cleaning product or worked in a school’s science room understands the hidden importance of these little combinations of letters and numbers.
How should Acetamide be handled safely?
A Closer Look at Acetamide
Acetamide often shows up in labs and industrial settings. Over the years, many of us working with chemicals pick up a certain respect for compounds like this. It’s not flashy, but it earns its space on the shelf—a building block in research and chemical syntheses. It looks harmless, but treating it with casualness causes real trouble. For those who haven’t worked with it before, acetamide is a white, water-soluble solid with a faint scent—not exactly alarming, but don’t let appearances fool you.
The Risks Behind the Calm Surface
I’ve seen what happens when safety protocols get ignored. Heads turn when someone opens a chemical container without gloves, or skips a face shield, and for good reason. Acetamide can irritate skin and eyes, even with brief contact. Its dust lingers, and inhaling it brings headaches, nausea, and dizziness. Extended exposure or ingestion causes far worse health issues, up to serious neurological harm. The International Agency for Research on Cancer lists it as a possible human carcinogen, so it’s clear: no one should take it lightly.
Every spill, every missed step, gets filed away as a lesson for next time. Early in my lab days, a friend of mine relaxed a bit during cleanup and paid the price with a week-long skin rash. Some only learn by suffering through consequences—better to listen, and act on what experience teaches.
Respecting the Tools: Practices That Matter
Work in a fume hood whenever possible—ventilation pulls those invisible dust particles away before anyone breathes in trouble. Standard nitrile gloves work well, along with safety goggles or a face shield for splash protection. In the times I didn’t double-check my gear, I regretted it.
Don’t eat, drink, or touch your face during work. It sounds like common sense, but hunger or habit wrecks a safety streak quickly. I bring snacks and coffee, but they stay in a separate area until I’m washed and out of my lab coat.
Label storage containers clearly, and keep acetamide away from acids and oxidizers. I’ve seen labels fade in damp, busy labs, so I update them often. Spilled crystals or dust gets cleaned up with a damp cloth—no sweeping, since brooms kick up particles—and solid waste goes straight into a hazardous waste container, never down a sink or into regular trash.
Training and Awareness
No safety protocol works if people ignore it. Refresher training stays on my calendar, and I encourage colleagues to speak up about near misses and accidents. Story-sharing drives the point home better than lectures—real-world examples stick in the mind.
Having clear emergency procedures beats panic every time. I check where eyewash stations and showers are before each shift. When new team members join, I walk them through the steps. Knowing what to do transforms a potential catastrophe into a manageable incident.
Better Solutions On the Horizon
Researchers search for safer alternatives to hazardous chemicals like acetamide. Substitutions don't always fit every process, but whenever possible, I lobby for less toxic options. Until those come online, vigilance remains the best protection. Companies need to keep investing in better ventilation, improved personal protective equipment, and regular training. Everyone who works with chemicals should have a voice in shaping safety culture.
Years of hands-on experience prove one thing: respect wins out over convenience. Routine, preparation, and learning from each misstep help keep teams safe and let the work move forward.
| Names | |
| Preferred IUPAC name | ethanamide |
| Other names |
Acetic acid amide Ethanamide Acetimidic acid Acetamid Ethylamide |
| Pronunciation | /əˈsiːtəˌmaɪd/ |
| Identifiers | |
| CAS Number | 60-35-5 |
| 3D model (JSmol) | `/graphics/3D-structures/Acetamide-JSMol.png` |
| Beilstein Reference | Beilstein Reference: 0636805 |
| ChEBI | CHEBI:28347 |
| ChEMBL | CHEMBL951 |
| ChemSpider | 270 |
| DrugBank | DB03913 |
| ECHA InfoCard | ECHA InfoCard: 100.001.064 |
| EC Number | 200-473-5 |
| Gmelin Reference | 715 |
| KEGG | C06125 |
| MeSH | D000079 |
| PubChem CID | 175 |
| RTECS number | AF3870000 |
| UNII | F4XH9G8UWC |
| UN number | 2810 |
| Properties | |
| Chemical formula | C2H5NO |
| Molar mass | 59.07 g/mol |
| Appearance | white solid |
| Odor | Faint mouse-like |
| Density | 1.159 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | -1.26 |
| Vapor pressure | 0.08 mmHg (at 20 °C) |
| Acidity (pKa) | 15.1 |
| Basicity (pKb) | pKb = 13.96 |
| Magnetic susceptibility (χ) | -35.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.417 |
| Viscosity | 0.948 mPa·s (at 80 °C) |
| Dipole moment | 3.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 127.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -238.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –1267.7 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N01BX04 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H351 |
| Precautionary statements | P210, P280, P264, P301+P312, P302+P352, P305+P351+P338, P330, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 0, Special: - |
| Flash point | 79 °C (174 °F; 352 K) |
| Autoignition temperature | 221 °C |
| Explosive limits | Explosive limits: 7-17% |
| Lethal dose or concentration | LD50 Rat oral 1,130 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1,000 mg/kg (oral, rat) |
| NIOSH | ``` AS5250000 ``` |
| PEL (Permissible) | 250 ppm |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 300 mg/m3 |
| Related compounds | |
| Related compounds |
acetamidine acetamidoxime acetamidomalonate N-methylacetamide |

