5,6,7,8-Tetrahydroquinoline: A Close Look at an Influential Chemical Compound
Historical Development
Chemists began exploring quinoline derivatives in the 19th century, chasing natural alkaloids and practical synthesis routes. The partial hydrogenation of quinoline led to an array of hydrogenated analogs. 5,6,7,8-Tetrahydroquinoline emerged from this landscape as a versatile member of the family, with reports appearing in scientific literature at the start of the last century. Large-scale industries and universities kept pushing the frontiers, seeking routes that could deliver the compound in higher yield and cleaner isolation. These efforts helped build up not just the supply but also the understanding of its chemistry, cementing its place in organic synthesis, pharmaceuticals, and materials science. The demand for new molecules to meet changing scientific and medical challenges kept tetrahydroquinoline in focus, prompting researchers to refine old processes and invent new ones.
Product Overview
Interest in 5,6,7,8-tetrahydroquinoline springs from its unique structure. The partially saturated ring system lets the molecule exhibit both aromatic and aliphatic character, a trait valued in many research labs. It stands out for researchers engineering building blocks that join together into bigger, more complex frameworks. I’ve seen it star in projects where medicinal chemists tested new scaffolds for drug candidates, especially those targeting the central nervous system or infectious diseases. Synthetic chemists often turn to 5,6,7,8-tetrahydroquinoline to help bridge classic heterocycles with modern applications.
Physical & Chemical Properties
The molecule carries the backbone of quinoline but swaps out four double-bonded positions for saturated carbons along the B-ring, softening its reactivity compared to its parent. Under typical conditions, it appears as a colorless to pale yellow liquid, picking up slight scents reminiscent of its aromatic relatives. It dissolves well in common organic solvents like ether and dichloromethane and stands stable at room temperature, showing resilience against light and gentle heat. The unique mix of saturated and unsaturated character sets it apart, offering more flexibility in synthetic routes than either pure aromatic or pure saturated analogs.
Technical Specifications & Labeling
Suppliers usually offer 5,6,7,8-tetrahydroquinoline with a purity in the upper ninety percent range, disclosed by gas or liquid chromatography. Labels often list precise molecular formula C9H11N and molecular weight around 133.19 g/mol. Material safety data call out hazards typical of low molecular weight amines, such as irritation to skin and eyes, and the need for careful handling under clean bench conditions. In the lab, a tightly capped container and storage away from oxidizers help ensure its stability over several months.
Preparation Method
Many syntheses of this compound begin with a catalytic hydrogenation of quinoline itself. Using palladium, nickel, or platinum as catalysts under a steady hydrogen atmosphere, chemists can guide the hydrogen atoms to the right ring without over-reducing the structure. Temperature, pressure, and catalyst loading all play critical roles in shifting the balance between partial and total reduction, and batches need monitoring by thin layer chromatography or NMR. Common practice points to palladium on carbon as a favorite, both for its relative selectivity and for ease of recovery after the reaction. In my own lab experience, controlling the uptake of hydrogen and sampling small amounts across the run helps avoid overshooting the mark and ending up with octahydroquinoline or other deep-reduced derivatives, which often lack the utility of the tetrahydro version.
Chemical Reactions & Modifications
5,6,7,8-tetrahydroquinoline enjoys a range of classical transformations, each one opening doors to new analogs. The nitrogen atom at position 1 can accept acyl or alkyl groups, making N-substituted versions that tweak solubility or biological activity. The aromatic ring, while less reactive than pure benzene, still accepts electrophilic substitution under vigorous conditions. Oxidation or halogenation run smoothly with the right reagents, letting chemists tailor the final molecule. In medicinal chemistry, this flexibility feels like gold: one starts with the simple tetrahydroquinoline backbone, then builds out the molecular arms tailored for a specific target—anything from neuroreceptors to bacterial enzymes. In practical terms, this means that a single starting block can become host to dozens of related structures, each worth testing in its own right.
Synonyms & Product Names
Through scientific papers and catalogs, 5,6,7,8-tetrahydroquinoline often turns up under related names. You’ll see it called by its hydrogenated status—tetrahydroquinoline—or referenced specifically as decahydro-1-azananaphthalene in systematic chemical databases. Some older texts pin it as B-tetrahydroquinoline, highlighting the ring locations involved. In everyday research circles, people often trim the name to “tetrahydroquinoline” or even “THQ” if the context is clear, though strict publications will demand the full numerical prefix to avoid confusion with other isomeric forms.
Safety & Operational Standards
Safety matters, and standard operating procedures hold strong here. The amine structure poses a risk of irritation if splashed on skin or eyes, and like other low-weight organic amines, the vapors can tickle sensitive noses and throats. Good practice keeps the bottle capped, the pipetting under a well-ventilated hood, and full PPE on hand. Waste disposal lines up with typical amines: collect the spent material and contaminated wash in a dedicated solvent drum, label for organic bases, and send for incineration according to local rules. Spills clean up well with absorbent pads and a dilute bleach solution, breaking down trace organics before wipe-down. These routines help avoid repeated exposure, not just for lab staff but for the next person who uses the same work space.
Application Area
Pharmaceutical research draws heavily from this molecule, thanks to its adaptable skeleton. Medicinal chemists dig into it to create analogs or as a bridge in constructing more elaborate heterocyclic drugs. Among anti-infective projects, chemists have tested related scaffolds for their ability to block key enzymes, researching everything from tuberculosis to resistant staph infections. Material scientists look at tetrahydroquinoline derivatives when searching for new dye or pigment systems, or as ligands in catalytic chemistry for polymerization and synthetic transformations. The partial hydrogenation also keeps its structure sturdy enough for high-performance applications that need both flexibility and strength, such as certain engineering resins or specialty adhesives. For synthetic organic chemistry as a whole, the ability to functionalize this backbone opens research possibilities impossible with most simple cyclic amines or benzenoids.
Research & Development
Across years in the lab, I’ve watched researchers test a steady stream of modifications to 5,6,7,8-tetrahydroquinoline, always with an eye to solving real-world problems. Teams turn to the scaffold to avoid pitfalls of metabolism or toxicity encountered with similar molecules, probing each analog for safer and more effective profiles. Current academic work continues to report new synthetic methods—cheaper catalysts, greener reagents, or energy-saving conditions—all chasing sustainability along with efficiency. Automated machinery now lets some groups try dozens of substitution patterns in parallel, using computational models to guide which analogs look most promising. This approach saves time and helps avoid waste, based on smarter screening up front.
Toxicity Research
Nobody wants to move forward blindly, especially when it comes to safety. Published studies point out that 5,6,7,8-tetrahydroquinoline comes with moderate acute toxicity, with risk factors tied mostly to inhalation or skin absorption of concentrated solutions. Chronic exposure data ranks it as an irritant, and the parent quinoline group can lead to concerns about longer-term liver or kidney effects at high levels. Industrial hygiene recommendations stress that gloves, eye protection, and proper ventilation matter for anyone handling decagram or kilogram lots. Lab animal studies supplied data for drug metabolism specialists, who hunt for differences between species—a reminder that animal results rarely translate one-to-one to people, but form the backbone needed for careful escalation.
Future Prospects
The appetite for smarter chemicals and more precise drugs continues to climb, and 5,6,7,8-tetrahydroquinoline stands poised to anchor the next round of discovery. Newer synthetic methodologies—think electrochemical reductions or flow chemistry reactors—let chemists make the molecule with less waste and lower energy. Emerging biotech startups look for heterocycles that plug seamlessly into peptide mimics or advanced materials, and the unique blend of stability and modifiability makes this scaffold a natural contender. Regulatory agencies now watch for cleaner manufacturing since drug impurities rank high on the list of audit concerns, pushing industry to up their game with both chemistry and safety. If history tells us anything, it’s that once a molecule finds a home in both medicine and manufacturing, new uses will keep turning up—one experiment at a time.
What is 5,6,7,8-Tetrahydroquinoline used for?
The Chemical Behind Many Breakthroughs
5,6,7,8-Tetrahydroquinoline stands out in the world of organic synthesis. In chemistry labs and pharmaceutical companies, this compound shows up often as a crucial building block. Its structure, with a partially saturated quinoline ring, makes it easy to modify for creating other useful molecules. That versatility shapes how researchers tackle hard problems in medicine and material science.
Pharmaceutical Applications
Drug development always looks for reliable starting points, and this molecule fits the bill. Chemists use it to assemble antihypertensive agents, antimalarial drugs, and even medications aimed at treating neurological disorders. One example comes from studies trying to block enzymes that help harmful bacteria thrive. Modifications to the basic tetrahydroquinoline skeleton have led to compounds with impressive action against tuberculosis and malaria, helping scientists inch closer to fighting resistant strains. I remember working in a synthetic lab where colleagues built dozens of variations from this core structure, each with the hope of finding a combination that would outsmart the bugs or outlast cancer cells.
Overlooked Roles in Materials Science
The compound pops up in the creation of dyes and pigments, too. Its backbone helps form colorants with stable and vibrant hues, perfect for use in high-end paints or specialty inks. Researchers looking for new materials that react to light, heat, or other triggers often start with familiar structures. Adding functional groups to 5,6,7,8-tetrahydroquinoline lets them tune the properties of finished products, from better solar panels to advanced thermal sensors. I recall seeing simple tweaks producing dyes that outperformed commercial ones after a bit of molecular tinkering.
Fueling Advances in Synthesis
Organic chemistry often faces the challenge of making molecules efficiently. Here, this compound earns its keep by serving as both a starting material and a reaction helper. It lends itself to catalytic cycles, especially in the creation of chiral molecules. That matters because drug safety can depend on the exact shape of a molecule. With 5,6,7,8-tetrahydroquinoline, labs gain access to molecular frameworks that simplify how they reach those precious final products. Some startups building custom synthesis services advertise the efficiency gains they get from using this single core ingredient.
Challenges and Responsible Use
Every time I discuss synthetic building blocks, safety and environmental issues come to mind. Large-scale chemical production can spill solvents and waste, risking health and polluting water. Regulatory agencies in Europe and the US both monitor processes using 5,6,7,8-tetrahydroquinoline and related compounds. Labs now lean on greener methods—solvent recycling and catalyst recovery—so work moves forward without sacrificing environmental safety. Training and oversight minimize risks to workers handling any risky intermediates or final products.
Looking Ahead
Interest in this compound won’t fade. Drug resistance and new material demands push chemists to look again at molecules that can be tweaked or repurposed. As genetic medicine and smart materials keep evolving, compounds like 5,6,7,8-tetrahydroquinoline offer flexibility and reliability. The cycle of discovery—experiment, learn, refine—relies on a few well-chosen starting blocks. In my experience, success often comes from these small, underestimated pieces that help bring cures and advances closer to reality.
What is the chemical structure of 5,6,7,8-Tetrahydroquinoline?
A Look at Its Shape
5,6,7,8-Tetrahydroquinoline pops up in organic chemistry discussions, especially among people who dive into aromatic heterocycles. Its chemical structure starts from quinoline, which has a fused double-ring system — one six-membered benzene ring attached to a six-membered nitrogen-containing ring. The “5,6,7,8-tetrahydro” part hints at certain positions on the quinoline's second ring being fully saturated with hydrogen. So, the aromaticity stretches across the benzene ring, but the other ring loses its planarity because hydrogen takes over at positions 5, 6, 7, and 8. This changes how the molecule fits into some chemistry problems, especially those touching pharmaceuticals and materials science.
What’s Special About This Structure
The first time someone handed me a ball-and-stick model of 5,6,7,8-tetrahydroquinoline, it looked like an old friend wearing a new hat. Most aromatic compounds keep things flat. This molecule has part of its structure bumped out because of the extra hydrogens. That breaks up the delocalized electrons in the typical quinoline ring and adds flexibility. Chemists know this makes the molecule more likely to take part in specific transformations, such as hydrogenation or alkylation, without smashing apart the aromatic core.
Relevance in Research and Industry
Structure shapes function — that’s a lesson that never goes out of style. For 5,6,7,8-tetrahydroquinoline, the saturated portion changes both its reactivity and how it interacts with biological targets. Quite a few pharmaceutical researchers use this scaffold to build drug candidates. Antimalarial drugs, antihistamines, and even some antipsychotics share this backbone. In labs, its partially saturated ring often provides a starting spot for making even more complex molecules. Reports from the Journal of Medicinal Chemistry make it clear: adding hydrogens at positions 5 to 8 can tune how the molecule interacts with enzymes and other proteins, improving activity or reducing side effects.
Challenges and Possibilities
Synthesis leaves chemists rolling up their sleeves. Even though making tetrahydroquinoline derivatives looks straightforward on paper, selectivity can challenge experienced hands. Keeping control over hydrogenation so it targets only the right positions can be fussy without the right catalyst. Industry’s been searching for greener synthetic paths, as traditional hydrogenation needs metal catalysts that sometimes raise sustainability flags. Skipping over harsh reagents and finding more gentle ways to nudge the molecule along — that’s a push with real-world impact.
Solutions That Move Us Forward
Switching to biocatalysts and mild reaction conditions has started to pay off. Researchers tapping into enzyme-catalyzed reductions steer past toxic byproducts and waste stream headaches. More accessible, efficient synthesis unlocks the potential of these types of molecules as building blocks for new medicines and advanced materials.
Why All This Detail Matters
The story behind 5,6,7,8-tetrahydroquinoline’s structure runs deeper than just textbook chemistry. It shows how a simple tweak — adding hydrogens to a familiar backbone — shifts everything from synthesis to possible uses. Studying and understanding this molecule brings insight into the world of drug discovery. Its flexibility and altered reactivity stand as a reminder that chemical structure isn’t just about atoms on a page. For anyone working at the intersection of organic chemistry and real-world applications, details like these mark the difference between theory and something that can really change lives.
Is 5,6,7,8-Tetrahydroquinoline hazardous or toxic?
Understanding What We're Dealing With
Chemistry often brings up complicated names like 5,6,7,8-tetrahydroquinoline, and most folks outside the lab probably haven’t crossed paths with it. It’s a heterocyclic compound built around a quinoline core with extra hydrogen atoms packed in. In industry and research, scientists use it as a building block for more complicated chemicals, including pharmaceuticals and agrochemicals. It's this practical use that puts the spotlight on possible hazards of the substance.
Looking at the Science
Direct information about 5,6,7,8-tetrahydroquinoline’s hazard or toxicity remains limited. Most chemical safety data sheets mention skin and eye irritation as common risks. Lab workers handling it are told to use gloves and goggles—standard practice in the lab, but always worth reinforcing for a reason. Our skin absorbs many chemicals faster than we tend to believe. If something causes irritation, there’s a chance stronger or repeated exposure could lead to more lasting harm—especially since some quinoline family members have shown carcinogenic properties in animal studies.
A big part of gauging risk involves understanding long-term exposure. Data for this specific compound isn’t widely published, but research related to similar chemicals in the quinoline family has linked repeated contact or inhalation to effects like liver damage or increased cancer risk. This doesn’t guarantee the same outcomes for every related chemical. Each slight tweak in a molecule shifts its behavior in the body. Still, with not enough research done on 5,6,7,8-tetrahydroquinoline, folks must take the precautious approach.
Workplace and Environmental Concerns
Factories and labs stand on the front lines for exposure. Inexperienced handling or inadequate protective equipment can lead to spills, splashes, or fumes escaping. Without strong ventilation and personal safety habits, people suffer the consequences. I’ve worked in research facilities where even minor negligence escalates quickly. Small mistakes—like skipping gloves—can add up.
Waste disposal poses challenges, too. Pouring leftover chemicals down the drain isn’t just bad form; it risks groundwater contamination and can harm aquatic life. The compound’s relative stability means it's not breaking down fast once outside the lab. That sort of persistence raises environmental concerns that should not go overlooked.
Solutions Start With Responsibility
Safe handling makes the biggest difference. Personal protective equipment, fume hoods, and proper disposal sites are more than checkboxes—they’re investments. Training sessions at workplaces and universities work best when treated as practical, hands-on instruction instead of tedious slideshows. Managers ought to back up these lessons by providing clear, up-to-date safety data sheets for every chemical in use, including this one.
For regulators and policymakers, the answer is investing in more toxicology research, particularly on compounds like 5,6,7,8-tetrahydroquinoline where information gaps remain. Comprehensive data means clear labeling and informed restrictions. Without that, businesses and workers operate in the dark—and the risks rise.
Chemicals with a "handle with care" tag ultimately call for a mix of know-how and respect. Training, strict clean-up, and an attitude of caution go a long way—not just for individual safety but also the greater good.
What are the storage conditions for 5,6,7,8-Tetrahydroquinoline?
Looking After Chemicals Starts with Simple Rules
Anyone who's spent time in a lab knows a bottle’s label can’t tell the full story. 5,6,7,8-Tetrahydroquinoline looks friendly enough as a transparent liquid or off-white crystal, but nature often hides fire where folks least expect it. Once I worked in a university lab that didn’t follow proper storage guidelines for aromatic amines—let’s just say, one afternoon, we found out why rules matter. Distinct fishy smells, sudden color changes, odd heat around containers—early warnings, but sometimes ignored.
Room Temperature Doesn’t Always Mean Safe
Manufacturers say, “store at room temperature.” Scientists know “room temperature” is a sliding scale. June in southern India isn’t January in Finland. Temperatures over 30°C can speed up unwanted reactions for tetrahydroquinoline. I’ve watched this compound degrade over weeks in humid stockrooms, forming dark impurities, all while the thermometer sat quietly nearby. Sealed, airtight containers and dry cupboards give some peace of mind, especially through seasonal swings.
Why Dry Air Matters
Moisture creeps in if you leave a bottle loosely capped. For this compound, even mid-level humidity brings on slow hydrolysis or lets byproducts sneak in. Keeping it bone-dry preserves both quality and yield. Anyone with a history in chemical synthesis will recognize the value of adding desiccants to the storage area, whether it’s little silica gel sachets or specialized drying cabinets.
Stability Is Worth Investing In
Folks sometimes ask, “Do I have to use an inert atmosphere?” Not every lab needs a nitrogen glovebox, but minimizing air in the bottle helps, especially on a shelf where chemicals see little use. We capped ours tightly and purged with dry nitrogen every few uses, which proved practical and strengthened reliability downstream. There’s nothing worse than prepping for a big synthesis and discovering your only batch has oxidized.
Light Turns Progress Backwards
Some compounds shrug off a little sunlight. 5,6,7,8-Tetrahydroquinoline? Not so lucky. Visible light and UV rays break down bonds over time, creating nasty tars or dangerous byproducts—even inside plastic. Anyone who’s worked under bright lab lights has noticed darkened residues build up around bottle lips after weeks in the open. We solved this problem by moving stock to amber bottles and keeping them deep in cabinets.
Fire Hazards and Safety Precautions
This isn’t acetone, but 5,6,7,8-Tetrahydroquinoline will ignite if exposed to the right spark. I’ve seen near misses in poorly ventilated cupboards. Local fire codes require flammable storage units, which are worth the investment for any lab manager. Keeping smaller working quantities at the bench and the main supply locked up guards against disaster.
Solutions That Last Beyond One Lab
Word travels fastest through shared experience—mentors and colleagues who have seen firsthand what happens when corners get cut. Documenting storage routines in plain language—on the bottle, the shelf, and in digital inventory—reduces human error and waste. Weekly checks for container integrity and regular staff briefings kept our lab running smoothly for years. The real investment is in small habits—cheaper than replacing an entire contaminated batch or, worse, clearing out after an accident.
What are the main applications of 5,6,7,8-Tetrahydroquinoline in research or industry?
What Stands Out About 5,6,7,8-Tetrahydroquinoline?
In the world of organic chemistry, certain building blocks keep popping up where hard problems need smart answers. Among these, 5,6,7,8-tetrahydroquinoline grabs attention for its versatility. This compound shows up in the lab and the industrial plant, serving as a reliable backbone for discoveries and better processes. I’ve seen it not only stir curiosity but also solve practical challenges in drug design and other areas.
Paving Paths in Pharmaceutical Research
Pharmaceutical chemists chase compounds that promise activity against tough diseases. 5,6,7,8-tetrahydroquinoline plays a key role as a scaffold in the hunt for new drug candidates. Medicinal chemistry leans on it for the right mix of stability and reactivity, making it a solid choice for tweaking molecular structures. Many bioactive drugs owe their origins to quinoline analogs, and the tetrahydro version offers a balance between flexibility and shape. Researchers focus on its skeleton to create anti-cancer, anti-inflammatory, and antimicrobial projects. I’ve watched students and scientists alike jump into synthesis projects, trying to boost what this scaffold brings to the table.
On top of that, the process of modifying 5,6,7,8-tetrahydroquinoline can introduce features that make molecules latch onto biological targets with more precision. This isn’t just a theory—real-world clinical candidates trace their roots back to simple building blocks like this one. It’s possible to change outcomes just by inserting or swapping a group on this base structure, and that’s a rare quality.
Catalysts and Materials: Thinking Beyond Drugs
Chemists don’t stop at pharmaceuticals. 5,6,7,8-tetrahydroquinoline steps into catalysis as well. Researchers adopt derivatives as ligands in metal-catalyzed reactions, where controlling the environment around a metal ion can push a reaction to go faster, become more selective, or save steps. Efficiency like this means less waste, and that hits home in industry—less clean-up, lower cost.
Some labs also dig into using this compound as a starting point for specialty materials. Its structure lets scientists link it into polymers and other advanced materials, sometimes chasing conductive or sensing properties. I’ve heard from researchers exploring these materials for electronics and sensors, hunting for that next breakthrough component.
Making Synthesis More Practical
Any chemist who’s worked at the bench knows that the right starting block can make life easier. 5,6,7,8-tetrahydroquinoline stands out for being manageable to handle and modify. Its chemical robustness lets researchers test new reactions, fine-tune conditions, and scale up without starting from scratch. That reliability pays off, and makes it suitable for big-picture projects as well as daily lab work.
Addressing the Challenges
No compound works alone. Moving from a test tube to something people use, scientists tackle safety, environmental, and economic issues. Using 5,6,7,8-tetrahydroquinoline, teams look for greener synthesis methods, aiming for lower waste and safer solvents. It’s clear that practical, scalable routes help keep both costs and risks in check. I’ve seen green chemistry push researchers to rethink old habits and test novel approaches with compounds like this one.
It’s not just about making more—it’s about making better. Collaboration between industry labs and universities keeps fueling progress, with a close eye on regulatory and health requirements.
Looking Forward
5,6,7,8-tetrahydroquinoline combines usefulness with adaptability. In my experience, its greatest impact comes from how well it fits into different research goals. Pharmaceuticals, catalysis, advanced materials—they all find a way to use this compound to build or enable something new. That’s why it keeps drawing attention from those who want not just to discover, but to solve real-world problems.
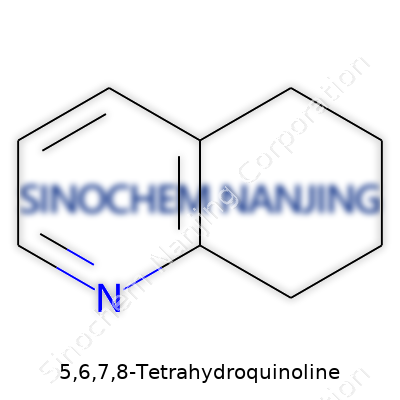
| Names | |
| Preferred IUPAC name | 2,3,4,4a-tetrahydro-1H-quinoline |
| Other names |
Hydroquinoline Tetrahydroquinoline Hexahydroquinoline 5,6,7,8-Tetrahydro-2-quinoline |
| Pronunciation | /ˈtɛtrəˌhaɪdrəʊ.kwɪˈnəʊliːn/ |
| Identifiers | |
| CAS Number | 635-46-1 |
| 3D model (JSmol) | `C1CCc2ccc(N1)cc2` |
| Beilstein Reference | 1720872 |
| ChEBI | CHEBI:35658 |
| ChEMBL | CHEMBL22139 |
| ChemSpider | 14209 |
| DrugBank | DB04182 |
| ECHA InfoCard | 100.011.198 |
| EC Number | 202-154-7 |
| Gmelin Reference | 84740 |
| KEGG | C06600 |
| MeSH | D015847 |
| PubChem CID | 10231 |
| RTECS number | VA8400000 |
| UNII | K7P4933AFH |
| UN number | UN2672 |
| CompTox Dashboard (EPA) | DTXSID0020383 |
| Properties | |
| Chemical formula | C9H11N |
| Molar mass | 173.26 g/mol |
| Appearance | Off-white to yellow solid |
| Odor | amine-like |
| Density | 1.093 g/mL at 25 °C(lit.) |
| Solubility in water | Slightly soluble |
| log P | 2.28 |
| Vapor pressure | 0.23 mmHg (25 °C) |
| Acidity (pKa) | pKa = 9.17 |
| Basicity (pKb) | 5.16 |
| Magnetic susceptibility (χ) | -57.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.604 |
| Viscosity | 1.215 cP (25°C) |
| Dipole moment | 2.33 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 165.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −23.3 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3710.9 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| Flash point | 104°C |
| Autoignition temperature | 480 °C |
| Explosive limits | 1.0-6.6% (V) |
| Lethal dose or concentration | LD50 oral rat 800 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 600 mg/kg |
| NIOSH | GW4200000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Quinoline 1,2,3,4-Tetrahydroquinoline Decahydroquinoline Isoquinoline Indoline |

