4-Nitroaniline: Balancing Tradition, Innovation, and Safety
Historical Development
4-Nitroaniline has a story that takes us back to the foundational years of organic chemistry, where curiosity shaped even the simplest of molecules into powerful building blocks for industry and science. Chemists in the late 1800s first examined this compound while searching for new dyes, as the textile industry demanded vibrant colors that could withstand the test of water, sunlight, and washing. Over the decades, the focus shifted from just color to new functions—pharmaceuticals, photographic chemicals, rubber additives—all built from the same base structure. This shifts the narrative from 4-nitroaniline as just another compound to a tried-and-tested workhorse that punched above its weight, not because of flash, but because of utility. Its track record in production, modification, and application leaves a trail of scientific papers and patents, but it also brings a legacy of toxicological concern and regulatory scrutiny.
Product Overview
Scientists and engineers alike know 4-nitroaniline for its pale yellow appearance and bitter, faint aromatic odor. Manufacturers sell it in crystalline or powder form. Its widespread adoption springs from a couple of things: its ability to act as a precursor for more complex chemicals and its reliability in industrial processes. I’ve noticed how companies often take advantage of this consistency in their own downstream syntheses, using it as a starter for dyes like azo compounds or as a stabilizer in rubber processing. In the chemical marketplace, its name comes up in catalogues for research-grade reagents and large scale chemical production, and it shows up regularly in fields as different as pharmaceuticals and agriculture—which speaks volumes about the intersection of tradition and adaptability in a single molecule.
Physical and Chemical Properties
Diving into structure, 4-nitroaniline packs both a nitro group and an amino group onto one benzene ring. This combination gives it a unique reactivity profile. The physical aspects—melting in the range of 146 to 149°C, low water solubility, and moderate volatility—shape the way it’s handled in both benchtop and industrial settings. With a molecular formula of C6H6N2O2 and a molar mass around 138 grams per mole, chemists can predictably chart how it interacts in solution or under heat. Its nitro group sits on the fourth position relative to the amino, which influences reactivity with reducing or oxidizing agents. These characteristics earned it a spot as a go-to intermediate compound in processes where you want selective reactions rather than unexpected side products.
Technical Specifications & Labeling
A bottle of 4-nitroaniline gets labeled with more than just a name and hazard code. Regulatory guidelines—especially in the EU and United States—require clear statements on purity, which can range from 98% to even higher for research use. Batch producers conduct rigorous quality checks to verify the absence of moisture and contaminants. Standards exist for labeling based on hazard class, chemical identity, and storage recommendations. This level of documentation didn’t appear overnight; it reflects years of hard-won lessons from chemical mishaps, shifts in workplace norms, and a deepening respect for transparency in the supply chain. End-users, whether working in R&D or manufacturing, depend on these labels not just for compliance, but for keeping people and processes safe.
Preparation Method
The synthesis of 4-nitroaniline most often involves starting with acetanilide, which is nitrated to make p-nitroacetanilide, then hydrolyzed to remove the acetyl group. It sounds straightforward, but controlling reaction temperature, pH, and stirring rates turns out to be anything but routine. Some chemists use variations, like direct nitration of aniline, but the classic route gives better yields and fewer by-products. Even so, the process throws off nitrogen oxides, acidic waste, and needs real attention to exothermic risk. Over time, scaling up production forced producers to design better reactors, safer scrubbers, and tougher personal protective gear—all of which show how something simple on paper needs robust systems to do safely in the real world.
Chemical Reactions & Modifications
4-Nitroaniline doesn’t just sit quietly on a shelf; it’s often dragged into new territory by research teams eager to synthesize dyes, drugs, or imaging agents. Its amino group acts as a nucleophile, allowing for acylation or sulfonation, while the nitro group can be reduced to create para-phenylenediamine. Tinkerers in both university and private labs wield 4-nitroaniline to test catalytic activity and to design new conjugated molecules. Every new functional group slapped on the backbone changes its color, solubility, or reactivity, opening doors for new products in places as far-flung as solar cell design or redox-active polymers. For anyone tuning reactions in the lab, knowing how to direct these modifications means you’re one step ahead in innovation.
Synonyms & Product Names
Walk through any chemical supplier’s listing, and you’ll spot this compound under a heap of other names: p-nitroaniline, 1-amino-4-nitrobenzene, para-nitroaniline, and more. Each name points to its structure—the nitro on the fourth position relative to the amino. In commercial catalogs, you might see it referenced as a dye intermediate, or by specific product numbers known mostly to procurement staff and compliance officers. These varying names have tripped up even skilled chemists, especially when the regulatory environments shift or suppliers change documentation practices. Still, it pays to learn these aliases because mistakes in ordering or storage can lead to costly, sometimes dangerous, mix-ups.
Safety & Operational Standards
No one spends long in a lab with 4-nitroaniline without learning respect for its toxic profile. It can be absorbed through skin, inhaled, or ingested, leading to serious health effects from methemoglobinemia to acute toxicity. In the plant, workers follow strict ventilation protocols and use gloves, goggles, and sometimes respirators. Regulators require specific labeling, emergency procedures, and waste disposal routines. Over the years, industrial hygiene has tightened up: fume hoods, spill kits, and regular safety drills stand as evidence of lessons learned from lax practices in decades past. Exposure limits now reflect better toxicology data, and compliance means looking beyond the shop floor—environmental releases must be monitored, and emergency response systems tested regularly. Emphasis falls on worker education; everyone handling 4-nitroaniline should know both the risks and the signs of exposure. Familiarity with the compound leads either to a safe day’s work, or a reminder about the price of shortcuts.
Application Area
Applications for 4-nitroaniline extend from color chemistry to the life sciences. The dye sector still represents a major market because it delivers that irreplaceable azo function needed for bold reds and yellows. In pharmaceuticals, it serves as an intermediate in specific APIs where the para relationship between amino and nitro injects the right chemical property at the right stage. Agricultural chemicals tap into 4-nitroaniline’s reactivity for select fungicides or herbicides. Researchers in analytical chemistry use it as a standard in spectrophotometric measurements, especially in kinetic studies. I’ve seen it show up in academic labs as a teaching tool for demonstrating chemical transformations or as a model substrate for catalysis research, where it helps illuminate mechanism pathways and reaction rates. The compound carves a path between workhorse practicality and creative experimentation.
Research & Development
R&D involving 4-nitroaniline never stops evolving. As new regulations drive the search for safer, “greener” syntheses, laboratories look for ways to minimize toxic by-products and avoid hazardous reagents in production. Research teams work at the intersection of classic organic chemistry and modern materials science—new derivatizations lead to advanced sensors, optical devices, or functional polymers. A drive toward digital chemical control—using software and real-time analytics—gives production teams sharper oversight on all processes involving 4-nitroaniline, from batch quality to emissions data. I’ve watched academic groups use this molecule to benchmark novel catalysts, track enzyme activity, or model electron transfer systems for energy research. This blend of old and new techniques keeps 4-nitroaniline in the research spotlight, always on the lookout for safer routes and smarter uses.
Toxicity Research
Toxicologists study 4-nitroaniline with a mix of wariness and rigor. Acute exposure risks like cyanosis and methemoglobinemia make medical staff and safety officers demand regular training and emergency supplies such as oxygen and antidotes. Animal studies have shown effects at both high and moderate exposure, with researchers reporting changes in blood chemistry and organ function. Chronic exposure remains a major concern in workplaces, prompting routine health surveillance and biological monitoring. Regulatory agencies draw on this research to craft stricter workplace exposure limits and more precise risk communication. Environmental scientists add another layer, digging into its fate in wastewater streams—degradation can be slow, and the potential for bioaccumulation leads to increased scrutiny. Teams continue searching for less toxic substitutes, creative encapsulation techniques to limit exposure, and process changes to reduce waste. Toxicology research rarely finds tidy solutions, but it drives progress inch by inch.
Future Prospects
Challenges and opportunities both frame the future of 4-nitroaniline. Stringent environmental, health, and safety rules continue to shape production, pushing companies to lean heavily on real-time monitoring, automation, and advanced waste treatment. Researchers keep adapting its chemistry for new uses, especially in specialty dyes, photonic materials, and as diagnostic tools in medicine. Green chemistry demands cleaner production: the hunt for milder reagents, solvents with lower environmental footprints, and processes powered by renewable energy all play a part. The expectation for greater transparency in supply chains compels producers to adopt blockchain tracking or advanced analytics, tightening controls from raw material to finished product. Community demands for safer workplaces push education and regulatory enforcement, turning chemical safety from an afterthought into a daily habit. 4-Nitroaniline stands at a crossroads where tradition meets accountability. Its story will keep unfolding as long as innovation is matched by responsibility.
What is 4-Nitroaniline used for?
A closer look at 4-Nitroaniline
4-Nitroaniline, a yellow organic compound, often grabs attention in manufacturing and academic labs, but it rarely appears on the radar of most people. This compound shows up in places where chemistry acts as a bridge to real products—like dyes, pharmaceuticals, and other common chemicals. My past years working in a small chemical plant gave me a close-up view of why 4-Nitroaniline ends up in various industrial recipes, and what that means for those who use the finished goods.
Dyes and pigments: How color gets made
Walk through a textile factory and color swatches line the shelves. Much of the brightness in clothing today depends on azo dyes, and 4-Nitroaniline often forms a key building block for those. Chemists treat it as a stepping stone—mixing, reducing, and coupling to form stable, vivid colors. The compound helps bring bold reds and deep oranges to synthetic fabrics, making fashion more vibrant without relying on expensive or unsustainable natural dyes. Textile workers depend on predictable batches, and 4-Nitroaniline’s reliability helps with consistent results.
Pushing medicine forward
Pharmaceutical chemists value starting materials that offer reliability and easy modification. 4-Nitroaniline fits that profile, giving researchers an anchoring point for more complex molecules. It often turns into intermediates for producing drugs, including painkillers and antibiotics. Hospitals and clinics around the world count on these medications, many of which trace their synthetic roots back to simple molecules like 4-Nitroaniline. I remember seeing shipments marked for research universities and generic drug makers more than once, reminding me that this unassuming yellow powder influences real treatments and health outcomes.
Other uses: Chemicals, rubber, and agricultural products
Factories use 4-Nitroaniline for products most people never see directly, but feel the impact in daily life. Producers in the rubber industry count on this compound when making antioxidants to keep car tires from failing early. Seed coatings and other farm chemicals sometimes include substances built from 4-Nitroaniline to resist harsh conditions and pests. And in specialty chemicals, workers blend it into agents for photography, corrosion inhibition, and even lubricants. It sits quietly in formulas, but its presence keeps other systems running safely or efficiently.
Environmental and safety concerns
As a former chemical plant worker, I saw firsthand how handling 4-Nitroaniline requires care. Exposure risks include skin irritation and possible organ damage, so personal protective gear becomes essential. Regulators enforce strict rules for storage and disposal, aiming to avoid leaks into soil or water. Scientists have raised questions about its persistence in the environment. Companies that use it need to track waste and upgrade equipment to avoid accidental releases. Finding newer, safer alternatives isn’t simple, but investment in green chemistry promises hope.
Better choices for safer workplaces and products
Improving safety means more than just following rules. Plant managers need strong training programs. Engineers keep equipment in good repair. Research teams look to create new dyes, drugs, and additives less risky to workers and ecosystems. Some companies now invest in biobased starting materials. Working alongside regulators, chemical makers stay alert for new evidence so they can improve production standards and protect neighbors near their facilities. That’s a real step toward reducing chemical hazards in everyday life.
What are the safety precautions for handling 4-Nitroaniline?
Getting To Know 4-Nitroaniline
Handling 4-Nitroaniline reminds me of every moment in the lab where the line between a routine day and a dangerous one felt razor-thin. The yellow-orange powder seems harmless at first glance, yet its risks demand respect. Exposure through skin, inhalation, or even a careless spill can set off a mess of problems: eye and skin irritation, methemoglobinemia that brings on headaches and a blueish tint to skin, and even bigger complications for those who aren’t prepared.
The Gear That Makes All The Difference
All those fancy gloves piled up in storerooms aren’t there for show. Nitrile or neoprene gloves hold up much better than those loose latex ones—especially when chemicals seep through if you’re not paying attention. A snug pair of chemical splash goggles goes a long way. Just about every chemist I know has had the “one drop in the eye” scare, and face shields come in handy if there’s a risk of a bigger splash or splatter. Sturdy lab coats help, but make sure they’re buttoned and sleeves cover your wrists. Closed shoes, not sandals. You don’t grasp the value of good footwear until you fumble a beaker and worry about what you’re stepping in.
Good Habits Around The Lab
Safe lab work relies on habits, not just posters on the wall. Fume hoods stay open for a reason: 4-Nitroaniline can send up dust and fumes that seem invisible but pack a punch. I always remind newcomers—never sniff directly or try to waft this compound. Simple tasks like pouring or weighing out this substance demand a steady hand and focus, because accidental spills are tough to clean and can hang around as a danger for anyone who passes by.
One habit that saves skin and nerves: washing hands after every session, even after glove removal. Compounds can cling in places you don’t notice, and skipping the sink is one surefire path to unintended exposure. Waste goes in properly labeled containers, and containers must stay tightly closed. I’ve seen what happens when open bottles gather on crowded shelves—smells, confusion, and sometimes dangerous reactions.
Dealing With the Unexpected
Accidents show up in labs that seem the most prepared. Knowing your emergency showers and eye washes matters more than you think. Spills call for absorbent pads, not paper towels, and avoiding contaminated areas until cleaned helps everyone stay healthy. Every year, at least one colleague gets caught by surprise, and the lesson sticks: reading the Safety Data Sheet isn’t just a formality. It keeps panic in check when something goes sideways.
Looking After Everyone On The Team
Training sticks better when leaders walk the talk. I picked up good safety habits from mentors who let me ask questions, learn from small mistakes, and always kept up open communication. Stories of close calls often drive home proper handling better than long lectures. If you see someone skipping a step—like not capping a bottle or leaving glassware unwashed—call it out. Building a culture of accountability beats relying on rules and paperwork.
Labs stay safe when people share responsibility, look out for chemical storage rules, and treat substances like 4-Nitroaniline not like enemies, but like risks that deserve full attention. Good habits pay off, not just in fewer accidents, but in a workplace where everyone backs each other up. Ultimately, safety grows from every action—one careful pour, one glove swap, one reminder at a time.
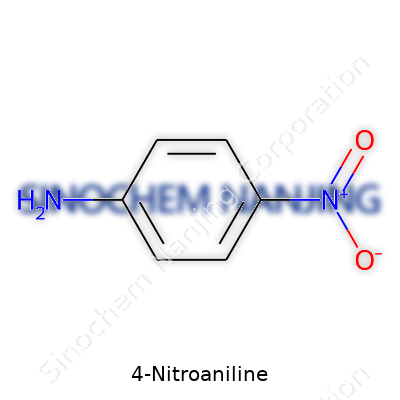
What is the chemical formula and structure of 4-Nitroaniline?
The Chemistry Unpacked
Anyone who has spent time in a lab knows the importance of precision, both in measurements and understanding what’s on the bench. Take 4-nitroaniline as a classic example. This organic compound shows up not just in textbooks but in real-world applications all over chemical manufacturing and research. It carries the chemical formula C6H6N2O2. That formula alone tells a big story—six carbons, six hydrogens, two nitrogens, and two oxygens come together in a rather memorable way.
Chemists quickly recognize its structure: a benzene ring where the amino group (–NH2) sits directly opposite the nitro group (–NO2). By tradition, we call this the para position. That arrangement might look simple, yet it changes the way the entire molecule interacts—both in theory and out in industry.
Why the Structure Matters
Many people overlook how those functional groups affect the bigger picture. I’ve spent a fair share of time watching reactions hinge on those tiny details. The nitro group draws electrons away from the ring, making the molecule less reactive toward electrophilic aromatic substitution compared to plain aniline. Swing over to the amino side, and things look a bit different: that group can donate electrons, setting up a balancing act across the ring.
Textbooks point out the resonance and electron flow, but in a flask, these electronic effects decide what happens next. For dye makers, those groups make 4-nitroaniline an ideal intermediate for vibrant azo dyes. In the pharma world, it’s a stepping stone for making medications, where every notch of reactivity gets tuned to perfection. In every role, the structure’s symmetry makes purification by crystallization a breeze, contributing to its popularity in research and production labs.
Safety and Responsible Use
A little experience goes a long way with safety, and 4-nitroaniline demands attention. Its yellow appearance might look harmless, yet the compound carries toxic potential—especially if inhaled or ingested. Lab workers keep the material sealed, and those vent hoods aren’t just for show. Chronic exposure can impact blood oxygen transport, leading to methemoglobinemia. Every bottle comes with warning labels for good reason.
Many companies emphasize good ventilation, proper gloves, and eye protection, ensuring exposure remains minimal. In my years managing storerooms, strict inventory control and regular staff training have proven effective at keeping accidents at bay. Adding real-time monitoring systems gives another layer of protection, picking up any leaks early.
Addressing Environmental Concerns
Waste from nitroaniline production doesn’t vanish into thin air. Improper disposal threatens both soil and water health. Environmental regulations now insist that facilities treat their waste—oxidation processes and advanced filtration significantly reduce the risk of contamination. On-site remediation cells and community monitoring stand out as practical ways to track and fix issues before they spill out beyond plant boundaries.
Paths Forward
Bringing new eco-friendly technologies into chemical manufacturing has shown promise. Green chemistry teaches that even time-tested intermediates can become safer and cleaner with the right process tweaks. Ongoing research into alternative reaction pathways, safer solvent systems, and effective recycling practices will help protect both workers and the world outside the lab gates.
Facts That Stick
Looking at 4-nitroaniline’s formula and structure opens up more than theoretical insights—it provides a real-world case study in chemistry’s impact. Learning from day-to-day handling, safety protocols, and waste management creates not only better science but a more responsible future for the whole industry.
How should 4-Nitroaniline be stored?
Why Pay Attention to 4-Nitroaniline Storage?
4-Nitroaniline is pretty common in labs where chemists make dyes, drugs, or test chemicals. It brings with it some solid risks, being both toxic and a potential pollutant. Even if nobody talks much about 4-nitroaniline in everyday conversation, anyone who spends real time with chemicals knows how easy it is for small mistakes to grow into big headaches. I've seen spills and mix-ups—from broke students to seasoned techs. Almost always, the culprit is poor organization. So, good storage isn’t just some rule in a dusty binder; it’s what keeps people safe.
Straightforward Storage Steps
To keep things safe, start with a lab-grade container made from glass or high-quality plastic. Leaves no room for leaks, breaks, or unexpected reactions with the chemical. 4-Nitroaniline comes as a yellow powder, so you spot it easily if it spills. Snug-fitting lids stop moisture from creeping in—this chemical doesn’t love water, since it can degrade or clump up.
Location matters. Don’t stash it near heat, open flames, or anything that sparks. 4-Nitroaniline can burn under the right conditions, and nobody wants to run a fire drill because something got too warm near a hotplate. Stick it in a cool, dry cabinet marked for toxic organics. Make sure the cabinet sits out of direct sunlight; light can help chemicals break down faster or act weird. Ventilation counts, too—a regular wooden cabinet won’t cut it for volatile or hazardous stuff.
Label Everything—No Exceptions
In my early days, a missing label could turn a standard day into a guessing game. Label clearly and honestly, using the chemical’s full name and hazard info. The extra few seconds could stop someone from grabbing the wrong stuff in a rush. Hazard pictograms help a lot—they catch the eye when words blur together after a long shift.
Keep It Separate
Some folks cram as many bottles as they can on one shelf to save space. That’s trouble. Store 4-nitroaniline away from oxidizers, acids, and anything that might set off a reaction. Incompatibility doesn’t sound exciting, but it sits behind most “unexplained” chemical shelf fires.
Emergency Prep Counts
Accidents happen—no matter how careful people act. Seeing firsthand the panic in a lab after a spill taught me the value of planning. Stock up on spill kits designed for organics. Train everyone on the location and use of eye wash stations, showers, and ventilation shutoffs. Review emergency procedures once a quarter so they don’t collect dust.
Waste Management
Throwing hazardous waste in a regular trash can is a fast track to fines and contamination. Collect solid or powder waste in sealed, labeled containers. Partner with a proper hazardous waste disposal vendor—many universities and research sites have these set up, so use them. Pouring anything down the drain or in the dumpster invites a call from the city or state.
Stay Up-to-Date
Safety rules shift as research and best practices get updated. Stay tuned into institutional or government guidelines, such as those from OSHA or your local environmental authority. A yearly check of storage practices can catch bad habits before they cause trouble.
Nothing Fancy—Just Careful Habits
Safe 4-nitroaniline storage isn’t about fancy gadgets or high-tech tricks—it’s about setting up good habits and sticking with them. People in the lab stay safer, equipment lasts longer, and research moves forward with fewer delays. Every time someone skips a step, risk climbs. Careful storage turns chemical handling from a risk into a routine.
Is 4-Nitroaniline hazardous to health or the environment?
What 4-Nitroaniline Brings to the Table
If you’ve worked in chemical labs or peeked into industrial processes, you might have heard about 4-nitroaniline. This yellowish, crystalline powder shows up in dye production, research, and even in some pesticide manufacturing. Being around chemicals like this means you learn early to respect their potential risks. 4-nitroaniline carries some real hazards — some that you won’t notice till it’s too late.
Human Health Risks: More Than Skin Deep
From my years in the lab, handling powdered anilines always brought home the need for proper gloves, masks, and ventilation. Inhaling or touching 4-nitroaniline can lead to headaches, nausea, or dizziness. Acute exposure sometimes causes something scarier — methemoglobinemia, a condition that reduces the blood’s ability to carry oxygen. This isn’t some rare theory; documented cases link symptoms like cyanosis (bluish skin) directly to nitroaniline exposure, especially if safety gear is ignored.
Many workers underestimate risks, thinking, “It’s a small amount, what harm?” Data from occupational safety agencies suggests otherwise. Chronic exposure — day after day, even at low levels — might damage the liver, kidney, or even cause changes in the blood. Wearing goggles and gloves doesn’t just keep the boss happy, it protects you from long-term harm you can’t sense or see.
What 4-Nitroaniline Does to the Environment
This chemical doesn’t just vanish after use. Once released into wastewater or soil, 4-nitroaniline tends to stick around. Soil bacteria have trouble breaking it down, so it lingers, moving through water sources and making its way into animals and plants. Researchers who monitor environmental contaminants point out that it can poison aquatic life. Even small amounts threaten fish, snails, and insects by upsetting the delicate balance of local ecosystems.
Factories sometimes release effluent containing 4-nitroaniline into rivers. Over time, those living downstream might notice fewer fish or bugs. Reducing such pollution asks for more than a simple filter; strong regulations, regular inspections, and smart technology all play roles in stopping 4-nitroaniline from reaching open water. From what I’ve seen touring chemical plants, investments in closed-loop recycling and better waste treatment actually pay off — workers get a safer workspace, and rivers don’t turn sickly brown.
Reducing Risk — Practical Steps and Smart Choices
Better awareness makes the biggest difference. Training helps workers spot early warning signs, like odd smells or colored dust, before accidents happen. Reading up on the latest safety data is worth the time; regulations often change as new research comes out. MSDS sheets and government guidelines spell out what’s needed for safe storage and disposal.
Community watchdogs play a big role, too. People living near chemical plants deserve to know what risks exist and what measures keep neighborhoods safe. Environmental monitoring, published openly, holds companies accountable. Choosing simpler, less toxic alternatives for industrial processes would sidestep many problems, but until then, vigilance and continued research remain necessary.
Final Thoughts
4-nitroaniline isn’t just “another chemical.” Its risks for both people and the planet demand respect, careful handling, and ongoing attention. My own experience taught me that teamwork, honest communication, and practical safety steps make all the difference — at work and in the wider world.
| Names | |
| Preferred IUPAC name | 4-nitrobenzenamine |
| Other names |
p-Nitroaniline para-Nitroaniline 1-Amino-4-nitrobenzene 4-Nitrobenzenamine |
| Pronunciation | /ˌfɔːrˌnaɪ.trəʊ.əˈnɪl.iːn/ |
| Identifiers | |
| CAS Number | 100-01-6 |
| Beilstein Reference | 87350 |
| ChEBI | CHEBI:7475 |
| ChEMBL | CHEMBL1409 |
| ChemSpider | 801 |
| DrugBank | DB14107 |
| ECHA InfoCard | ECHA InfoCard: 100.003.205 |
| EC Number | 202-806-2 |
| Gmelin Reference | 789 |
| KEGG | C06180 |
| MeSH | D009577 |
| PubChem CID | 7416 |
| RTECS number | BY6125000 |
| UNII | N08Z77WVVU |
| UN number | 1661 |
| CompTox Dashboard (EPA) | EPA-HCV0021042 |
| Properties | |
| Chemical formula | C6H6N2O2 |
| Molar mass | 138.13 g/mol |
| Appearance | Yellow to orange crystalline powder |
| Odor | Odorless |
| Density | 1.437 g/cm3 |
| Solubility in water | Moderately soluble |
| log P | 0.36 |
| Vapor pressure | 0.000071 hPa (25 °C) |
| Acidity (pKa) | 18.68 |
| Basicity (pKb) | 9.38 |
| Magnetic susceptibility (χ) | -45.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.675 |
| Viscosity | 1.15 cP (25°C) |
| Dipole moment | 3.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 63.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1496 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic if inhaled, causes skin and eye irritation, may cause methemoglobinemia. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H312, H332, H400 |
| Precautionary statements | P261, P280, P301+P312, P302+P352, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-OX |
| Flash point | 152°C (306°F) |
| Autoignition temperature | 525 °C |
| Lethal dose or concentration | LD50 (oral, rat): 750 mg/kg |
| LD50 (median dose) | LD50 (median dose): 750 mg/kg (oral, rat) |
| NIOSH | CN6125000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 4-Nitroaniline: "Methemoglobinemia: 1 mg/m3 (skin) |
| REL (Recommended) | 0.2 mg/m³ |
| IDLH (Immediate danger) | 200 mg/m3 |
| Related compounds | |
| Related compounds |
p-Aminophenol p-Phenylenediamine 4-Nitrophenol 2-Nitroaniline m-Nitroaniline |

