4-Methylpyridine: Navigating Its Substance, Uses, and Impacts
Historical Development
A glance back in chemical history lands 4-methylpyridine among the persistent building blocks in the toolkit of modern synthesis. Researchers identified its structure in the late 19th century, during an age flush with experimentation on coal tar and nitrogen heterocycles. Back then, separating pyridines from a sticky, complex mixture demanded patience and a tough nose. German chemists working in industrial labs and university benches mapped out methylated pyridines, chasing new pharmaceuticals and coal tar dyes. Its rise intertwines with the expansion of chemical manufacturing, fuel additives, and the transition from dyes to pharmaceuticals during the 20th century. Over decades, labs kept turning to 4-methylpyridine, not because it was trendy, but because it was reliable, adaptable, and sometimes the only thing that would fit a reaction’s unique demands.
Product Overview
4-Methylpyridine delivers what its simple structure promises: a ready source of a nitrogen-rich ring system with a subtle tweak from a methyl group at the four position. Chemists value its reactivity—the ability to play both a guest and a host in molecular frameworks. This feature makes it a preferred starting point in pharmaceuticals and as an intermediate for other fine chemicals. Production often scales up in centralized chemical parks, where the compound leaves the reactors as a clear, colorless liquid, carrying the sharp, fishy aroma typical of many pyridine derivatives. Those who’ve worked with it in the lab quickly recognize the scent and rarely forget it.
Physical & Chemical Properties
This liquid boils at about 145°C and weighs in at a little above one gram per cubic centimeter. It dissolves easily in water and many common organic solvents, and it’s highly flammable. The nitrogen lone pair at the ring's core gives it an edge, letting 4-methylpyridine act as a base, grab protons, or match up with metals. The methyl group at the fourth slot doesn’t just sit there—it bends electron distribution in just enough to tweak both chemical reactivity and physical character. From direct experience, handling this substance calls for care. Spills evaporate fast and send their unmistakable signal through a lab. Staff get used to wiping surfaces fast, keeping airflow healthy, and keeping careless touches to a minimum.
Technical Specifications & Labeling
Any bottle or drum in industry or university should bear a hazard label for toxicity and flammability. Packing integrity and clear batch identification matter, as the material travels from synthesis plants to research labs and downstream production. Details such as the CAS number, purity percentage, and batch number aren't just paperwork—those links help trace out issues when something in the process heads sideways. From years of lab experience, mislabelled bottles create chaos—confusion wastes time and sometimes money, or worse, health. Workers know that clear labeling isn’t just a rule, but a lifeline.
Preparation Method
Companies haven’t stuck to just one recipe. Two broad routes define most production: isolation from coal tar fractions and, for industrial-scale needs, catalytic methylation of pyridine itself. Using catalysts like alumina, chemists can coax methyl groups into place. Scaling up demands reactor designs that keep heat even and handle the mix of gases without clogging or runaway reactions. Getting high yields often depends on subtle pressure and temperature controls, not brute force. Chemists and process engineers bring their own tricks here, often rooted in hard experience or closely-guarded patents rather than what textbooks offer.
Chemical Reactions & Modifications
4-Methylpyridine and its ring system rarely find peace in a flask. The molecule sticks out for its extra reactivity in both substitution and oxidation reactions. A straightforward path leads to 4-cyanopyridine or 4-pyridinecarboxylic acid, essential skeletons in drug synthesis. Scientists use it as a ligand in metal complexation, or as a precursor for stepping into bigger heterocycles. In pharmaceutical research, dozens of groups scan modifications at the methyl or ring positions, searching for new biological hits. Sometimes, the nitrogen lends itself to forming intricate ionic salts that behave very differently in solution than the parent molecule. The breadth of directions open for modification keeps chemists coming back.
Synonyms & Product Names
Many older chemists remember calling it gamma-picoline, a nod to the position of the methyl group. Whether the label says "4-Methylpyridine", "gamma-picoline", or less-often "pyridine, 4-methyl-", anyone familiar with the field knows the compound by nose and reactivity. Each vendor or publication can pick its own name, but the substance inside the bottle doesn’t change. In practice, staff and researchers tend to settle on the quickest phrase—sometimes shortening it to "4-MePy" in notes, so long as the meaning remains clear.
Safety & Operational Standards
The substance makes no secret about the risks it poses. Flammable vapors demand ventilation. Exposure can cause headaches, nausea, and even nervous system symptoms if inhaled over a workday. Direct contact irritates the skin and eyes. Regulatory agencies such as OSHA or EU REACH mandate careful handling and proper personal protective equipment—goggles, gloves, and well-maintained fume hoods. From years in research settings, institutional safety drills and regular hazard reviews keep the worst mishaps from happening, although even the most committed staff end up with the occasional sniffle or spill. The learning curve teaches respect, and safe labs foster a culture where getting careless is never an option.
Application Area
The reach of 4-methylpyridine extends across pharmaceuticals, agrochemicals, and as a flavoring ingredient precursor. Drug companies depend on it for routes into anti-tuberculosis medicines and antihistamines. Agrochemical producers use the compound as an intermediate for pesticides and soil treatment agents. Recent years have seen interest in its potential as a ligand for catalyst development, where the tailored reactivity opens new doors in green or more selective chemistry. Working downstream from these applications, users care most about reliability of supply and purity, as a minor impurity can play havoc in sensitive formulations.
Research & Development
Chemists rarely rest easy with “good enough.” Ongoing work keeps pushing 4-methylpyridine chemistry into new directions—finding pathways with higher atom economy, safer byproducts, or lower energy input. Efforts to swap out rare or expensive catalysts with abundant metals, or to run reactions under milder conditions, dominate current literature. Teams investigate greener solvents and “flow” approaches over traditional batch processing to minimize waste and spills. University groups hunt for new pharmacologically active derivatives, trying every possible modification a methyl allows—sometimes returning to basics, sometimes leaping off in wild new directions that only years later find their way into practical applications.
Toxicity Research
Toxicity sits squarely on the caution list for 4-methylpyridine. Repeated animal studies link it to nervous system effects, and at high doses, liver and kidney stress. Regulatory agencies keep limits strict for workplace exposure, rarely tolerating more than trace levels in air or finished food products. The compound does not build up in fatty tissue, but acute exposure brings rough symptoms: dizziness, vomiting, and, at worst, collapse. Several decades ago, the field learned these lessons hard, with tales in the literature of lab mishaps and factory exposures. Knowledge feeds safety policy—modern labs employ continuous air monitoring, real-time exposure alerts, and keep rigorous medical surveillance for staff who work with higher inventories.
Future Prospects
Thinking ahead, 4-methylpyridine won’t fade from chemical supply lists and research priorities. As green chemistry matures, more attention goes to methods that produce it with fewer byproducts and lower energy demand. There’s talk in research circles about biocatalysis or engineered enzymes perhaps replacing harsh catalysts downstream. Creative routes using renewable biomass over fossil sources may offer future breakthroughs if energy prices or supply shocks tighten traditional access. New uses can arise as scientists dig deeper into heterocyclic chemistry—one promising area is more efficient ligands that streamline catalysis for large-scale pharmaceutical and material synthesis. Current regulations and market forces favor suppliers who lead on safety, sustainability, and transparency—a trend that carries right into the future, where good stewardship meets the age-old demands of molecule-making.
What is 4-Methylpyridine used for?
Behind the Chemistry of 4-Methylpyridine
Four-methylpyridine rarely grabs headlines, but folks in different industries depend on it more than most realize. This small molecule, a derivative of pyridine, has a sharp, chemical bite to its scent and looks like a clear to yellowish liquid. Think of it as the quiet helper behind the curtain in many manufacturing processes, especially where chemistry gets a little complicated.
Key Role in Agrochemicals
Working a season on a Midwest farm taught me about the chemicals that help feed the world. Out in the fields, 4-methylpyridine starts its journey as an ingredient that helps protect crops. It gets used mostly to make chemicals like vitamin B3 (niacin) and certain herbicides. Niacin, in particular, plays a key part in animal feed and human supplements. Without folks making niacin this way, prices would jump and supply could tight up. Sometimes, small molecules like this quietly make nutrition accessible.
Making Pharmaceuticals Happen
Walk through any pharmaceutical plant, and the chemical smell isn’t always pleasant. 4-Methylpyridine helps produce drugs that control everything from blood pressure to bacterial infections. It acts mostly as a building block—chemists use it to start, change, or finish off other molecules. For example, it’s part of the process for making pyridine-based drugs and some over-the-counter medicines. The Food and Drug Administration keeps a close eye here, but the roots start in industrial vats mixing up this chemical.
Solvent and Research Ally
Universities and private labs stock 4-methylpyridine bottles for a reason. Growing up near a retired chemistry professor, I heard stories about tricky reactions that only worked with the right solvent. This compound often fits that bill. It helps dissolve both organic and inorganic substances, useful in tricky syntheses or in finding the best conditions for new reactions. The clarity and speed that 4-methylpyridine brings are small wins in labs struggling to answer tough scientific questions.
Challenges and Safer Steps Forward
Many industrial chemicals demand respect, and 4-methylpyridine is no different. Exposure can irritate eyes, skin, and lungs, and high exposure causes much more serious health issues. Regulatory agencies like OSHA set limits for workplace air concentrations. Workers need strong training, solid personal protective equipment, and well-maintained ventilators. Companies using this substance should embrace safety training instead of cutting corners to boost profits. I remember a neighbor who switched jobs after a chemical exposure incident—safety isn't just a rule, it shapes real lives.
Safer alternatives to some chemical uses may emerge as green chemistry shapes the future, but industries aren’t quick to replace reliable materials unless new ones match their performance and price. Stronger regulations, development of safer production methods, and routine community health monitoring around chemical plants encourage a middle ground between progress and safety. Advocacy groups can keep pushing for more public disclosure and independent oversight.
The Takeaway from Everyday Experience
4-Methylpyridine doesn’t make the evening news or roll off the tongue, but it links fields from food to research labs. The story behind its use highlights how chemistry often runs behind the scenes, quietly shaping the world. Taking responsibility for handling it and exploring safer paths for its application keep people safer—and industries sustainable.
What are the safety precautions when handling 4-Methylpyridine?
Understanding the Risks Beyond the Label
4-Methylpyridine isn’t a household name, but in chemical plants and research labs, it finds regular use. This colorless or pale yellow liquid might not seem imposing, but I’ve worked in spaces where substance misjudgment led to nausea, dizziness, and much more serious long-term troubles. Its vapors hit fast, and a moment’s carelessness leaves a sharp, irritating taste in your airways. The risks reach further than skin or eye irritation—4-Methylpyridine may affect the nervous system, and long-term exposure brings up bigger health worries, including liver and kidney problems.
Personal Protective Equipment Makes a Difference
In my experience, trusting only fume hoods or basic gloves leads to regret. Chemical splash goggles prevent painful redness and vision blur that can last for days. Lab coats serve as more than a uniform—they keep those fast-evaporating droplets from your skin. Nitrile gloves hold up better than latex here, since pyridines slip through the wrong barrier easily. I’ve watched coworkers think a quick transfer wouldn’t matter. A rushed job led to burns that lingered longer than anyone expected.
Ventilation Isn’t Just a “Nice-to-Have”
Chemical vapors fill a room quickly, and 4-Methylpyridine brings its own sharp, distinctive smell that stings even in low doses. I’ve learned the hard way that a good fume hood lets you focus on the work, not the taste in your mouth or the headache creeping in. Relying on open windows or weak bench vents skips critical control. Poor ventilation pushes up the risk of inhalation exposure, and in my own work, poorly ventilated workspaces always led to sluggish team members, forgotten procedures, and preventable errors.
Storing Responsibly: More Than Locking Up
It’s tempting to stash chemicals out of the way and move on, but 4-Methylpyridine needs segregated, properly labeled storage. I saw a night-cleaner mistake an unlabeled bottle for a cleaning agent—luckily, a strong smell gave it away before real trouble started. Always store this chemical away from strong acids and oxidizers, and keep bottles tightly closed. Use secondary containers with clear hazard warnings. Spills leak through cardboard and thin plastic fast—contain leaks on metal or chemical-resistant trays.
Spills, First Aid, and Planning for Trouble
I’ve never seen a chemical spill turn out good without a fast, confident plan. Small spills on counters need immediate absorption with spill pads, not paper towels. Larger spills get contained with commercial absorbents, then collected in sealed, labeled waste containers. Only trained people using proper protective gear should handle cleanups—watch one person downplay a splash, and soon another ends up in the nearest eyewash station. For skin or eye contact, plenty of running water—15 minutes or more—makes all the difference. Inhaling vapor? Fresh air wins, but if someone struggles with breathing, emergency services take over. Every team should review the safety data sheet together before new work starts—skipping over the details puts everyone at risk.
Building Safer Habits Every Day
Expert handling comes from experience, ongoing training, and the wisdom to never cut corners. New hires bring a set of fresh eyes, but even the seasoned pro can make a mistake after a distraction. Regular safety drills, fresh signage near storage areas, and honest conversations after incidents do more to prevent harm than a dusty manual on the shelf ever could. Reducing accidents means building habits—the sort of habit where each glove and mask goes on without a second thought and every odd smell or unexplained splash gets dealt with right away.
What is the chemical structure of 4-Methylpyridine?
Understanding 4-Methylpyridine
4-Methylpyridine tends to show up in both industrial labs and academic lectures. You see this compound pop up in conversations about organic synthesis, pharmaceuticals, and even the flavor industry. Someone curious about its structure will notice it looks a lot like the basic pyridine ring, just with a twist—a methyl group attaches itself to the ring, changing properties and uses.
Breaking Down the Chemical Structure
At its core, 4-Methylpyridine holds a six-membered ring. It’s made of five carbon atoms and a lone nitrogen atom taking one spot on the ring. Instead of remaining a simple pyridine, a methyl group—CH3—hooks onto the fourth carbon atom, counting from the nitrogen. That “4” signals where the methyl group replaced a hydrogen. Chemists call this a substituted pyridine. Its formula reads C6H7N. Picture that ring: pyridine by itself looks like benzene, but swap one carbon for a nitrogen and tack the methyl group opposite the nitrogen—now you have 4-Methylpyridine.
The Structure’s Role in Real Life
One 4-Methylpyridine's practical uses pops up in the pharmaceutical world. My research and work around lab benches have taught me that this structure changes how a molecule behaves. While an extra methyl group might seem minor, it signals a new set of properties. It affects how the body processes a compound, solubility, and even toxicity. Pyridine rings are found in drugs that target everything from cancer to mental health. Shifting a methyl group changes molecular fit in enzymes and receptors, which pharmaceutical chemists use to their advantage.
Industrial and Environmental Impact
Smaller chemical makers craft 4-Methylpyridine to serve as a building block for vitamins, especially B6 (pyridoxine), herbicides, and corrosion inhibitors. The presence of nitrogen makes pyridines chemically flexible, but the methyl tweak changes its reactivity in various synthesis routes. Factories release it sometimes as a byproduct, which brings environmental concerns. Studies from the EPA reveal that contamination in soil and water can pose challenges, since these nitrogen-containing rings resist breakdown. In my university lab experience, I remember the strong, sharp odor from even dilute solutions, which matched the reputation pyridines have for being tenacious in the environment.
Solutions and Improvements
Cleaner production stands at the center of progress. Newer synthetic methods aim to cut down on waste and improve yields. Catalysis using less hazardous metals or even renewable feedstocks could reduce unwanted emissions. Companies and regulators have also swung into action on spill management and site cleanup. Soil microorganisms that specialize in nitrogen chemistry show promise for breaking down rings like 4-Methylpyridine, but the research sits at early stages. The push for green chemistry keeps driving safer substitutes with fewer environmental footprints.
Why Structure Matters
4-Methylpyridine’s impact stretches from atomic arrangements to real-world consequences. Its chemical structure, though simple on paper, offers a window into how a small change can shape uses, impacts, and our approach to safety and sustainability. My work in chemistry has shown again and again that digging into structure opens doors to better solutions, smarter regulations, and safer products.
What is the CAS number of 4-Methylpyridine?
Digging Into 4-Methylpyridine
In chemical research circles and industry labs, knowing substances by name isn't always enough. Reliability depends on precision. 4-Methylpyridine’s Chemical Abstracts Service (CAS) number is 108-89-4, which works like a fingerprint.
Think about it: Common chemical names sometimes overlap. Trade names crop up, languages change spelling, and technical jargon chains itself into impossible knots. The CAS number cuts through confusion. Once you see 108-89-4 on an inventory sheet, the guesswork drops away. Anyone—whether sorting bulk shipments or digging through a digital library—knows what's in front of them.
CAS Numbers Build Trust and Safety
I’ve run into the wrong compound shipped under a similar-sounding name more than once. Even in professional spaces, this causes real trouble. A wrong chemical might trigger unexpected reactions or slip through safety checks unnoticed. Someone reading the wrong data sheet might underestimate toxicity or miss handling hazards. Recent safety studies from academic chemistry labs tie most small accidents to labeling errors or ambiguous records. A CAS number avoids that mess. With the right identifier, rules on labeling aren’t just “good practice”—they’re what keep people safe.
How Industry Leans on 108-89-4
Almost every reference you check—Material Safety Data Sheets, inventory control programs, procurement requests—assigns data to 4-Methylpyridine through 108-89-4. Dyes, rubber compounds, or pharmaceutical intermediates using 4-Methylpyridine depend on sourcing that exact molecule. Regulations from agencies like the EPA or OSHA often mention the CAS number straight away, because legislation needs crisp, clear language. I’ve also seen import bans and transport restrictions lifted, held up, or created simply because a paperwork line listed a name instead of a number.
Knowledge Fuels Safe Work
Learning about CAS numbers gave me more confidence when I first started working around chemicals. I memorized numbers for the compounds I handled most often. I encouraged the same habit among folks starting out. The reason is clear: if you reach for a chemical, search a database, or double-check a shipment, you want to know for sure what you’ve got. Miscommunication doesn’t always just slow things down—it sometimes sends people to the hospital.
Beyond the lab, consumer groups use CAS numbers to check what ends up in finished products. If you’ve ever typed a number into an ingredient-check app, the info you see is tied to regulatory databases that don’t trust names or abbreviations. Clear labeling builds trust, and trust builds repeat business. For anyone with allergies, safety concerns, or ethical questions, transparency starts with a number.
Better Systems for a Safer Tomorrow
Relying on the CAS number 108-89-4 for 4-Methylpyridine brings efficiency and safety. Digital inventory systems should require a CAS number every time someone logs a new chemical. Manufacturers could improve labels by printing the number larger and near the product name. Schools and workplaces need to train new team members to use these numbers, not just “name and formula,” and tie them to safety data and regulations. Policymakers writing rules about chemical use should rely on CAS numbers in public databases, not just region-specific trade names.
How should 4-Methylpyridine be stored?
The Real Risks on the Shelf
Anyone around chemicals for a living knows things can go south fast. 4-Methylpyridine, a colorless to yellowish liquid, falls into a group that keeps plenty of folks on their toes. Its strong, fishy smell hints at trouble: the stuff flares up easily, catches fire at lower temps than you might expect, and its vapors carry some real punch if you breathe them in.
What Safe Storage Actually Looks Like
My time working in industrial labs taught me it’s not enough just locking things away. 4-Methylpyridine likes its space cool and out of sunlight. This doesn’t mean stuffing it beside the air conditioner at home. Think climate-controlled rooms that keep temps steady—below 30°C really keeps risks down. High heat invites the liquid to turn into vapor, making an accident much more likely.
Plastic won’t cut it for the long haul. Over the years, I’ve seen more than a few containers start to degrade or leak, which invites a mess nobody wants to clean up. Metal drums with proper linings or tough glass bottles with sturdy, sealed lids stand up far better. Make sure every container is clearly labeled. The sharp odor might warn people something’s off, but relying on smell alone isn’t safe or smart.
Fire Hazards: Not Just on Paper
Everyone talks about flash points in training. 4-Methylpyridine lights up around 45°C (that’s not much warmer than a hot summer day outdoors). If a spark hits an open drum, forget it—the fire can spread before you have time to grab an extinguisher. Class B fire extinguishers (think foam or CO2) lined the shelves at every job site I worked on, and we kept a clear route to the door at all times.
Don’t stash this stuff with oxidizers, strong acids, or anything else that might start trouble in a chemical reaction. I once walked into a closet where someone stored ammonia bottles nearby. Mixing up chemicals in a tight space is asking for disaster.
Ventilation Makes a Big Difference
Some folks believe screwing the lid tighter will solve every problem. From what I’ve seen, good ventilation does even more. The vapors from 4-Methylpyridine can sneak out and irritate eyes, lungs, and even skin. A fume hood or special vented storage cabinet handles those fumes without dumping them into the building’s air. After someone tried storing similar liquids in a regular janitor’s closet, half the place smelled like chemicals for days, and people started reporting headaches. That’s not something you ever want repeated.
Practical Solutions and Smarter Choices
Training is non-negotiable. Folks who haven’t seen a chemical fire or aren’t used to handling hazardous liquids can make honest but dangerous mistakes. Routine checks on containers, labels, and temperature need to be part of daily habits—not something reserved for audits or inspections.
Spill kits—absorbent pads, neutralizers, protective gloves—should sit a few steps from where containers get stored and opened. Leak trays under bottles or drums keep small mistakes from turning into a cleanup nightmare. Local fire codes and OSHA rules rarely set the bar unnecessarily high; nearly every rule in the book has saved someone’s skin before.
It gets tempting to cut corners with storage, especially in cramped labs or busy shops. Take it from someone who’s been called in to mop up chemical accidents: a careful, by-the-book approach really is worth the hassle. Promoting a safety-first culture protects people, property, and keeps the air a whole lot cleaner for everyone nearby.
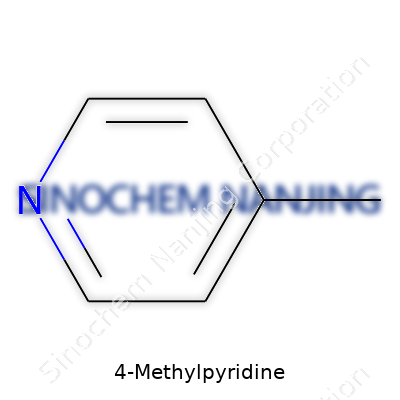
| Names | |
| Preferred IUPAC name | 4-Methylpyridine |
| Other names |
4-Picoline gamma-Picoline gamma-Methylpyridine 4-Mep |
| Pronunciation | /ˌfɔːrˌmɛθɪl.pɪˈrɪdiːn/ |
| Identifiers | |
| CAS Number | 108-89-4 |
| Beilstein Reference | 58348 |
| ChEBI | CHEBI:45879 |
| ChEMBL | CHEMBL1407 |
| ChemSpider | 6199 |
| DrugBank | DB03255 |
| ECHA InfoCard | 100.007.148 |
| EC Number | 207-838-8 |
| Gmelin Reference | 734 |
| KEGG | C01744 |
| MeSH | D008937 |
| PubChem CID | 1047 |
| RTECS number | UJ1050000 |
| UNII | A4B1D18S78 |
| UN number | UN2313 |
| Properties | |
| Chemical formula | C6H7N |
| Molar mass | 93.13 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | pungent |
| Density | 0.957 g/cm³ |
| Solubility in water | miscible |
| log P | 0.82 |
| Vapor pressure | 5.3 kPa (at 20 °C) |
| Acidity (pKa) | 5.98 |
| Basicity (pKb) | 8.77 |
| Magnetic susceptibility (χ) | -44.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.498 |
| Viscosity | 0.851 cP (20°C) |
| Dipole moment | 2.32 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 168.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 83.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3253 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H302, H312, H315, H319, H332 |
| Precautionary statements | P261, P280, P301+P312, P302+P352, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 68 °C (154 °F; 341 K) |
| Autoignition temperature | 560°C |
| Explosive limits | 1.3%–11% |
| Lethal dose or concentration | LD50 oral rat 1290 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1290 mg/kg (oral, rat) |
| NIOSH | JN7875000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 4-Methylpyridine: 2 ppm (7 mg/m³) |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Pyridine 2-Methylpyridine 3-Methylpyridine 2,4-Lutidine 2,6-Lutidine 3,4-Lutidine 3,5-Lutidine |

