4-Methyl-3-Penten-2-One: Chemistry’s Quiet Workhorse
Historical Development
Chemistry books rarely feature 4-Methyl-3-Penten-2-One on their covers. Even so, people in research labs have appreciated this molecule since the mid-1900s. Early on, scientists noticed its structure—a ketone with an extra methyl group and a double bond—meant it could enter reactions easily. Chemists in the post-war era used the compound’s straightforward synthesis as a training ground for students, showing how slight tweaks to basic ketones could yield something more reactive. Back then, chemical supply was often inconsistent and research sometimes stalled just waiting for the right precursors. Reliable routes to compounds like 4-Methyl-3-Penten-2-One became small victories, with chemists trading notes on yields, purity, and new applications.
Product Overview
4-Methyl-3-Penten-2-One may not flood consumer shelves, but it shapes what goes on in many production floors and labs. With a clear, colorless body and a sharp, sweet-smelling aroma, the molecule stands out from simple references in dusty catalogs. Its five-carbon backbone, punctuated by a ketone and a double bond, punches above its weight in synthesis and serves as a pivot in building larger, more complex chemicals. When you think about the everyday plastics, rubbers, and specialty chemicals that fill warehouse drums, odds are this compound or one like it sits somewhere in the ancestor chain.
Physical & Chemical Properties
This molecule clocks in with a molecular weight of 98.15 grams per mole, carrying both oil-like volatility and a notable flammability. Its boiling point hovers near 114°C, and it shows a reasonable solubility in organic solvents. In the open air, the double bond and ketone group invite oxidation—a property that can trip up those who store it loosely. I’ve watched careless storage leave residue and off-odors in the back of a stockroom, a sharp reminder not to underestimate simple hazards. The low flash point means anyone handling it outside of well-ventilated labs risks safety, and I’ve seen too many cut corners because it’s “just another liquid.”
Technical Specifications & Labeling
Bottles usually wear labels warning of its flammable nature and the potential for skin and eye irritation. Batch purity varies depending on source, but color often signals freshness—clear remains good, yellow hints at polymerization or oxidation. In Europe, a GHS exclamation mark labels the risks, with Safety Data Sheets reminding users to glove up and use eye protection. Scientists using reagent-grade samples demand at least 97% purity to avoid side reactions, while industrial plants balance between cost and cleanliness.
Preparation Method
This compound doesn’t force chemists to jump through too many hoops. A traditional method, Claisen condensation, links acetone and methyl vinyl ketone under basic conditions, followed by a bit of tinkering to steer the product toward the right double bond. Tweaks involve using sodium ethoxide or potassium hydroxide to generate the enolate and then careful temperature control to keep side-products in check. Other, fancier approaches pop up in patent filings, but the industrial sector sticks to what works on a ton scale. Recently, green chemistry advocates push for water as a solvent and milder bases, though reaction times can crawl without organic solvents.
Chemical Reactions & Modifications
4-Methyl-3-Penten-2-One loves to play the electrophile. That carbonyl grabs nucleophiles, opening doors to aldol additions and other carbon–carbon bonding tricks. The double bond adds another handle—think hydroboration, epoxidation, or additions that lead to spicy intermediates with pharmaceutical potential. Labs regularly tweak it to build specialized flavors, scents, or active intermediates. The lesson comes clear: small changes in such “building block” molecules shape the downstream possibilities in medicines, fragrances, or specialty materials.
Synonyms & Product Names
In a chemistry lab, precision matters. This molecule pops up under several aliases: 4-methylpent-3-en-2-one, methyl prenolone, and sometimes the slightly clumsy IUPAC name 4-methyl-3-penten-2-one. Catalogs may call it by its CAS number, 141-79-7, to avoid confusion, and I’ve worked on projects where a mislabeled reagent led researchers down a dead-end until a quick re-check of the bottle made everything clear.
Safety & Operational Standards
Labs have learned to respect this liquid for its flammability and reactivity. Fume hoods remain the tool of choice not just because of policy but experience—spilled drops evaporate fast, the vapors sting the nose, and with the wrong spark, flash fires follow. Protective gloves, face shields, and flame-proof lab coats stop being overkill when you consider how many accidents result from “quick” open-air transfers or forgotten hot plates. Industry has moved toward automated dispensing and closed systems to cut down exposure. To this day, legislation continues to rank ketones like this among high-priority chemicals for workplace injury prevention.
Application Area
Beyond academic exercise, this molecule carves a niche in synthetic organic chemistry and the industrial supply chain. It serves as a starting material in flavor and fragrance manufacture, especially where fruity or sweet notes matter. Because it can extend carbon chains and assemble into more complex structures, the pharmaceutical sector relies on it for intermediates, particularly in steroid and vitamin synthesis. Polymeric materials and specialty solvents owe part of their diversity to such alpha,beta-unsaturated ketones. Agricultural labs experiment with derivatives as potential crop protection agents, though regulatory scrutiny stays high for anything near food production.
Research & Development
While not every breakthrough gets a press conference, researchers keep pressing this compound into new service. Academic papers showcase new synthetic routes aiming for greener conditions and fewer byproducts. Techniques like microwave-assisted catalysis and flow chemistry have been tested to boost yield and cut hazards. Intellectual property databases fill steadily with patents for derivatives—each modification offering new surface-active properties, flavors, or biologically active molecules. In recent years, the bio-based industry experiments with fermentation-derived acetone to feed into its synthesis, aiming to displace some of the petroleum pedigree in the chain.
Toxicity Research
Toxicologists have put this molecule through the wringer. Animal studies suggest moderate acute toxicity; eyes and lungs burn with strong exposure, and repeat skin contact leads to dermatitis in lab staff. Inhalation remains the most significant route, with central nervous system effects documented at high concentrations. Regulators set workplace limits to reduce chronic exposure risks, and every well-run lab audits air measurements in storage and preparation spaces. Disposal gets tricky—burning its waste material calls for specialized incinerators, since breakdown products can include irritant gases. Researchers keep an eye on long-term environmental fate, especially as the molecule’s volatility encourages escape into the atmosphere.
Future Prospects
Looking ahead, 4-Methyl-3-Penten-2-One seems poised to deepen its role in both established and emerging technologies. Its simplicity and adaptability match the drive toward sustainable synthesis, offering a bridge between legacy fossil feedstocks and tomorrow’s biomass. Clever catalyst design and green solvents promise higher selectivity and lower hazards, while automation opens doors to safer, large-scale applications. Demands from the personal care, food additive, and pharmaceutical sectors drive innovation in both production and downstream chemistry. The challenge sits in ensuring rigorous health and safety controls track alongside increased use, preventing the old pattern of underestimating risk with “routine” building blocks. Researchers, regulators, and producers all have a stake in engineering safer, greener, and more efficient chemistry—4-Methyl-3-Penten-2-One may be a simple molecule, but its influence stretches further than most realize.
What is 4-Methyl-3-Penten-2-One used for?
Everyday Chemicals, Hidden Roles
4-Methyl-3-penten-2-one rarely shows up in big headlines, but its quiet influence touches a surprising range of products. I first heard about it while working part-time in a local lab. Someone spilled a little on the bench, and the sharp, fruity smell caught everyone’s attention. From that moment, I realized this isn’t just some obscure solvent; it’s a real workhorse in chemical manufacturing.
The Backbone of Chemical Synthesis
Many companies use 4-Methyl-3-penten-2-one as a starting point. It acts like a puzzle piece, linking together small chemical chains to make bigger, more valuable compounds. It fits into a variety of reactions that create flavors, perfumes, and even some medications. Pharmaceutical chemists value it for how well it reacts to create certain drug ingredients. Creating a medicine isn’t just about the final pill—each building block needs to get made reliably and affordably, and this chemical makes that job easier.
In the world of flavors and fragrances, companies look for molecules that stick around and smell pleasant but not overwhelming. The punchy aroma of 4-Methyl-3-penten-2-one brings out fruity notes in artificial flavorings for gum and candy. Essential oil manufacturers sometimes use derivatives of this molecule to bring out fruity notes in perfumes and oils.
Pushing Paints and Plastics Forward
I talked to a friend in coatings manufacturing who says this chemical pops up in plenty of paint shops. Paints need chemicals that help them spread smoothly, dry quickly, and last long. 4-Methyl-3-penten-2-one gives paint labs a thinner that evaporates just fast enough for a decent finish, but not so fast that brush lines set in. Companies producing coatings and adhesives often reach for it for its balanced evaporation rate. The plastics industry sometimes uses it to help form specialty polymers as well.
Handling and Health Concerns
Like many strong-smelling solvents, this one isn’t the sort you want to breathe in all afternoon. I remember wearing a mask and gloves anytime I worked with chemicals like this, even if the bottle label looked harmless. Prolonged exposure to 4-Methyl-3-penten-2-one can cause irritation or headaches. Agencies like the National Institute for Occupational Safety and Health (NIOSH) recommend using good ventilation and proper gear when handling it. Companies monitor exposure closely. Over the years, more businesses have turned to greener methods and better containment, following the lead set by California and Europe in restricting hazardous solvent use.
Making Safer, Smarter Choices
The growing push for safer manufacturing standards means companies keep looking for less risky alternatives. Green chemistry also calls for chemicals that break down harmlessly and avoid polluting water or air. This isn’t a wishful trend—regulators and consumers demand it. Researchers keep working on substitutes that deliver the same properties without the same risks, which helps move industries forward. In a world full of invisible ingredients, it’s the people who notice and handle these compounds responsibly who really shape the future.
What are the physical and chemical properties of 4-Methyl-3-Penten-2-One?
The Traits That Set It Apart
Living in a world built on chemistry, it’s easy to walk past something like 4-Methyl-3-Penten-2-One and not think twice. Once you peel back the curtain, this compound shows some memorable features. It offers up a bright, sharp odor—some say it lands between fruity and somewhat acrid. Not your average household scent. If you work with organic solvents or synthetic intermediates, catching that strong aroma gives a quick clue you’re dealing with a ketone.
As a colorless liquid, it slips easily into the hands during handling. The boiling point clocks in around 106 to 107°C, which places it in a comfortable range for distillation and purification without making the job dangerous. Its flash point drops to about 7°C. This means it catches fire on a chilly day, so safety demands attention. Toss it in with most organic liquids, and it offers decent miscibility. On the flip side, bring water into the mix and it refuses to blend, just like oil at a salad bar. If left in an unventilated space, it evaporates quickly thanks to a low molecular weight and high volatility.
Chemical Character and Reactivity
This compound carries two big features on its back: a methyl group and a double bond, both alongside a strongly reactive ketone group. That gives folks working in labs a lot of options. Nucleophilic additions go off smoothly—think of simple Grignard reactions cooking up new alcohols. The double bond, being conjugated, can shift reactivity. This makes hydrogenation fairly straightforward; the right catalyst will pull up and help saturate the molecule.
Under the hood, conjugation between the double bond and the carbonyl oxygen helps stabilize the molecule somewhat, but you can’t ignore how easily it can polymerize under acidic or basic conditions. It won't just sit quietly if heat or sunlight hit it for too long. The industrial value comes from this lively chemistry: making flavors, fragrances, or more complex chemical building blocks. The carbonyl sits front and center, making this a solid candidate for anyone looking to extend carbon chains or introduce new groups onto a backbone.
What’s at Stake, and Better Handling
Using this compound in the workplace calls for solid safety habits. Its strong odor isn’t just an irritant; inhaling the vapors at higher concentrations will irritate the nose and eyes and, if ignored, could hit the respiratory system. Over the years, more plant managers and lab techs started insisting on better ventilation and personal protection, especially gloves and goggles. Accidents never wait, so storing this ketone in tightly sealed cans out of sunlight and heat keeps things smooth. Spill kits on hand aren’t just good practice—stories from small labs show that a single lapse led to evacuations because the liquid flashed before anyone could react.
With raw materials like this, waste handling deserves as much respect as use. Sending 4-Methyl-3-Penten-2-One to landfill isn’t an option. Chemical waste centers handle these loads by incineration, breaking down organic molecules so they don’t add to broader pollution. Cleaner disposal keeps the water table safe—stories from manufacturing sites that cut corners led to costly groundwater remediation later. Science and responsible management walk side by side.
Moving Forward With Confidence
Understanding the properties of chemicals in use guides real-world decisions, from safety policies to new product design. 4-Methyl-3-Penten-2-One may seem a specialty item, but its reactivity shows up in daily processes, influencing both productivity and safety. Its volatility, flammability, and unique odor keep everyone on their toes, and using knowledge instead of luck is always better in the long run.
Is 4-Methyl-3-Penten-2-One hazardous or toxic?
Hard Facts About 4-Methyl-3-Penten-2-One
Some chemicals go by names that slip through daily conversations, never making headlines until an incident forces attention. 4-Methyl-3-penten-2-one, a colorless liquid with a sharp, pungent odor, slots into this category. Used in industries connected to flavoring agents, fragrance, and chemical synthesis, this compound has sneaked past most public scrutiny. But if you look closer, its properties and risks shouldn’t get brushed aside.
My Experience with Chemical Safety Rules
I spent years working near labs dealing with similar ketones and unsaturated compounds. No hazmat suit walks or gas masks unless you really need them, but a whiff of something sharp in the air tells you some chemicals don’t belong in lungs or eyes. Chemicals like 4-Methyl-3-penten-2-one sting much more in practice than any safety data sheet can warn. Most people don’t see what irritant effects mean until their skin tingles, eyes water, or headaches start after careless exposure.
The Science Behind the Hazards
Data collected from peer-reviewed research and safety data sheets shows that 4-Methyl-3-penten-2-one can irritate skin, eyes, and respiratory passages quickly at relatively low concentrations. The vapor isn’t something you’d want to breathe without good ventilation. Overexposure may cause headaches, dizziness, and possible nausea, especially without personal protective equipment. The compound also has a flashpoint low enough to rank it as a fire risk, with vapors heavier than air that travel along surfaces to find ignition sources.
Animal studies suggest repeated exposure may affect organs like the liver and kidneys. Actual effects on humans outside industrial contexts are less well-documented, but prudent risk management always means treating these possibilities with respect.
Why Should Anybody Care?
It’s easy to slip into the belief that chemicals handled in factories or labs don’t matter to most people. But poor storage, accidental spills, or improper disposal allow these substances to leak into places they shouldn’t be — water sources, the air around work sites, sometimes even products. Industrial accidents from mishandled containers or inadequate ventilation systems endanger both workers and the wider community. The overuse or careless handling of volatile compounds like this one contributes to pollution, health care costs, and long-term environmental damage.
Clear Steps That Make a Difference
Regulations only work if workers, managers, and local residents build good safety habits. Regular training, well-maintained equipment, and all the fresh-air fans you can get help stop accidents before they start. Workers need gloves, goggles, and safe storage for anything as irritating and flammable as 4-methyl-3-penten-2-one. Small improvements, like real-time air monitors and proper labeling, cut down confusion and panic if something does spill.
Health and safety officers should keep up with data and research, pushing for closed systems and substitution with safer chemicals whenever they can. Cleanup responses should be swift and follow best practices, not just for the company, but to protect surrounding neighborhoods. Reporting near-misses and actual incidents is better than hiding them. Practical vigilance does more for safety than corporate slogans or a poster on a wall.
For everyone from plant workers to policy makers, treating 4-Methyl-3-penten-2-one with caution isn’t just red tape — it’s real protection against avoidable harm.
What is the recommended storage condition for 4-Methyl-3-Penten-2-One?
Real Risks in Chemical Storage
Anyone working around industrial chemicals knows the dangers go well beyond what’s printed on the label. 4-Methyl-3-Penten-2-One doesn’t stand out in a warehouse, yet let it sit unsupervised in the wrong environment, and you’ll have more than spilled dollars on your hands—health and safety issues pile up pretty quickly. This particular solvent brings its own quirks: it’s volatile, has a sharp odor, and can irritate eyes, skin, and lungs if exposure isn’t strictly managed.
Why Proper Conditions Are Non-Negotiable
Chemists and warehouse staff who’ve handled 4-Methyl-3-Penten-2-One can tell you that stories about ruined stock and work injuries often come down to sloppy storage. A stable, cool, and well-ventilated place is crucial. Heat turns this substance into a genuine hazard, with pressure building up in sealed containers and vapors increasing both inhalation risks and flammability. Fire fighters respond to more incidents every year tied to improper handling of volatile organics. Not only does mishandling put people in danger, it leads to regulatory headaches, with agencies like OSHA and the EPA expecting tight practices—and holding facilities accountable.
Storage Strategies that Work
First, use containers made from chemically resistant materials—stainless steel, polyethylene, or glass. 4-Methyl-3-Penten-2-One eats through less robust plastics, leaking its way into surrounding products. Tight-fitting lids prevent vapor leaks and keep neighboring materials safe. Keep containers away from heat sources: direct sunlight, steam pipes, even warm radiators cause vapor pressure to spike, which leads to swelling lids or ruptures.
A chemical that picks up moisture isn’t just a loss for inventory; contaminated stock brings unpredictable reactions down the line. Always store in a dry spot, with a humidity monitor if climate control’s in question. Use proper ventilation, such as a flameproof exhaust hood or a ventilated chemical cabinet certified for volatile organics. Warehouses dedicated to hazardous goods keep these materials on clearly labeled shelves, never on the floor, and isolated from incompatible substances like oxidizers and acids—the kind of mixing disaster that gets coverage for all the wrong reasons.
Why Safety Practices Pay Off
Relying on clear labels and updated SDS sheets means everyone from shipping staff to emergency responders knows what’s in each container. Well-trained employees spot container bulging, leaks, or odd smells early on. I’ve seen the difference between warehouses where everyone reads the SDS and ones where training is just a box-ticking exercise. The first group reports fewer workplace accidents, and when an inspector shows up, nobody is scrambling for paperwork or hiding cracked bottles.
On a busy workday, it’s tempting to skip over protective gear or ventilation checks. Yet just one incident—whether a small fire or a chemical splash—can force days of downtime and pile on medical bills. Over the years, companies that stick to best practices rarely face the fines or lawsuits that come from ignoring storage basics. Good storage, in this sense, isn’t just about protecting a chemical: it’s about protecting people, reputations, and even the bottom line.
What is the CAS number and molecular formula of 4-Methyl-3-Penten-2-One?
Knowing the Numbers: CAS and Formula
4-Methyl-3-penten-2-one carries the CAS number 141-79-7 and its molecular formula reads as C6H10O. For chemists and manufacturers, these details matter. CAS numbers serve more than just tracking. They bring order to a crowded landscape. Nearly anybody who has rummaged through safety data sheets or tried to order a compound online sees the value of a single, universal identifier. Names get mixed up. One producer calls it methyl pentenone. Another drops in a hyphen or two. The CAS number sticks, keeping things honest and direct.
Why Even Simple Chemicals Deserve Respect
A lot of folks see formulas like C6H10O and see a jumble of letters. Those who've spent time in labs or worked on product development know there is more behind the symbols. Each molecule brings certain quirks. Take 4-Methyl-3-penten-2-one. As an alpha, beta-unsaturated ketone, it hints at both utility and risk. Its double bond and methyl group make it pop up in synthesis work—especially for flavors, fragrances, and some pharmaceuticals. The presence in daily-use products underscores why details matter, even for compounds that seem basic.
Working with chemicals like this brings both opportunity and responsibility. I remember getting a shipment in with a properly labeled drum, right down to the 141-79-7 number, and thinking about all the hands it passed through. Every link in the supply chain counts on those numbers. Without them, you end up with costly mistakes or, far worse, safety problems.
Small Compound, Big Picture
Regulation depends on accurate identifiers. Agencies track environmental and workplace exposure to compounds like 4-Methyl-3-penten-2-one through CAS numbers. Reporting hazards, shipping, and disposal all rely on that system. There have been cases—especially in smaller operations—where missing or incorrect CAS tags led to mix-ups that cost both money and time, never mind the legal headaches.
Accidents may not grab headlines, but chemical exposure leaves marks. I remember walking into a workspace with a lingering fruity odor and getting the sense that ventilation was being overlooked. Volatile ketones like this one tend to evaporate fast, which means unnoticed exposure builds up. Occupational health professionals count on accurate documentation to establish baselines and enforce proper limits. Protecting workers and nearby communities means knowing exactly what’s coming out of every bottle, bag, or drum.
Solutions to Reduce Risk and Confusion
Clear chemical records form the first line of defense. Using CAS numbers correctly builds trust. Checking the number on incoming materials, updating inventory systems, and training staff around those details helps avoid error. Digital inventory systems, combined with routine audits, close common loopholes. Even small manufacturers benefit from investing the effort upfront rather than chasing compliance or cleaning up mistakes later.
Suppliers have a responsibility to keep information transparent. Any updates to safety protocols or regulatory changes need relaying down the supply chain fast. Modern labeling technology—QR codes that link to digital data sheets, batch-level traceability—brings old-school inventory management into sharper focus.
Understanding What’s on the Label
Behind every clean label sits a network of regulations, expertise, and shared accountability. For 4-Methyl-3-penten-2-one, its CAS number and formula may look simple, but the story runs deeper. Getting those details right protects businesses, workers, and communities alike.
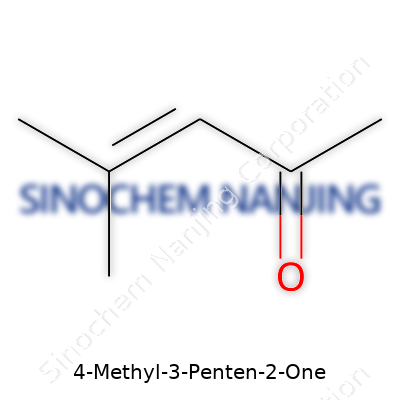
| Names | |
| Preferred IUPAC name | 4-methylpent-3-en-2-one |
| Other names |
Mesityl oxide Methyl isobutenyl ketone Isobutenyl methyl ketone 4-Methylpent-3-en-2-one Isobutylene methyl ketone |
| Pronunciation | /ˈfɔːr ˈmɛθɪl θriː ˈpɛntən tuː oʊn/ |
| Identifiers | |
| CAS Number | 141-79-7 |
| Beilstein Reference | 1209243 |
| ChEBI | CHEBI:51158 |
| ChEMBL | CHEMBL16261 |
| ChemSpider | 10903 |
| DrugBank | DB13867 |
| ECHA InfoCard | 03c5a395-b2a9-4026-8b69-7eb2fa0e0b89 |
| EC Number | 203-620-1 |
| Gmelin Reference | 109542 |
| KEGG | C00961 |
| MeSH | D017364 |
| PubChem CID | 7903 |
| RTECS number | RG3700000 |
| UNII | 6P92XON5TI |
| UN number | UN No. 1229 |
| Properties | |
| Chemical formula | C6H10O |
| Molar mass | 98.143 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Sweet, fruity |
| Density | 0.847 g/mL |
| Solubility in water | Slightly soluble |
| log P | 0.9 |
| Vapor pressure | 2.8 kPa (at 20 °C) |
| Acidity (pKa) | pKa ≈ 19.2 |
| Basicity (pKb) | Its pKb is 6.99. |
| Magnetic susceptibility (χ) | -8.62 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.422 |
| Viscosity | 1.267 mPa·s (20 °C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 316.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -124.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2934 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H319, H335 |
| Precautionary statements | P210, P243, P261, P271, P280, P304+P340, P312, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 33 °C (91 °F) |
| Autoignition temperature | 225 °C |
| Explosive limits | 1.2–9.7% |
| Lethal dose or concentration | LD50 oral rat 930 mg/kg |
| LD50 (median dose) | LD50 (median dose): 700 mg/kg (rat, oral) |
| NIOSH | SN2980000 |
| PEL (Permissible) | PEL: 100 ppm (350 mg/m³) |
| REL (Recommended) | REL (Recommended Exposure Limit): 1 ppm (4 mg/m³) |
| IDLH (Immediate danger) | 200 ppm |

