4-Methyl-2-Pentanol: A Closer Look at Role, Value, and Risks
Historical Development
For more than a century, 4-Methyl-2-pentanol, often recognized in industry circles as methyl isobutyl carbinol or MIBC, has carved out a place in chemical history. The molecule emerged during the surge in organic chemical synthesis that followed discoveries in petroleum refining and coal tar processing. Chemical engineers and process chemists experimented with various alcohols in the early 20th century, hoping to find compounds that could do more than the likes of ethanol or butanol. The story of MIBC highlights a time when chemical innovation raced to match the needs of growing industries, such as mining and synthetic rubber manufacturing. With a backbone built from basic carbon chains, the compound entered commercial networks through manufacturing in the United States and Europe, responding to demands from metallurgy and solvents.
Product Overview
Across industrial sectors, 4-Methyl-2-pentanol often pops up in applications that ordinary folks rarely see. It acts as a frother for mineral flotation processes in mining, which means its addition helps separate valuable elements like copper, zinc, and lead from rock, making the actual extraction work more efficient. Its moderate volatility and distinctive odor also place it in lubricants and specialized solvents, boosting the performance of paints or cleaning fluids in tight manufacturing processes. On the shelf, containers mark MIBC as a colorless liquid, but beneath the label lies a chemistry that cuts across factories, research labs, and sometimes even fragrance mixes.
Physical & Chemical Properties
MIBC comes off as surprisingly unassuming in a laboratory flask. At room temperature, it looks clear, with a faintly sweet odor that betrays its alcohol roots. Specific gravity lands it just under that of water, helping workers distinguish spills and measure quantities by eye. With a boiling point that tops 130°C, engineers rely on its stability under heat during mineral extraction, while its moderate miscibility with water allows it to form emulsions or separate layers as needed. What surprises people most about this compound is how a small shift in structure from related alcohols lets it take on uses they cannot handle.
Technical Specifications & Labeling
Labels for 4-Methyl-2-pentanol in storage or shipping seldom get flashy. Chemical purity, usually above 99 percent for industrial markets, signals reliability in results. Regulatory tags appear on containers, driven more by safety than by branding. Hazard diamonds warn against careless handling, and descriptions urge caution due to flammability and moderate toxicity. Certification for use often rests on compliance with REACH or similar chemical management guidelines in Europe, and the Globally Harmonized System supplies most of the icons that workers spot in storage silos and shipping yards.
Preparation Method
At the manufacturing end, MIBC arises through the hydrogenation of 4-methyl-2-pentanone (MIBK). In practice, hydrogen and a catalyst meet the ketone precursor in high-pressure reactors, turning one functional group into another and generating the target alcohol in high yield. Industrial facilities monitor temperature, pressure, feedstock purity, and catalyst life constantly, since stray contaminants can spoil an entire batch. Most chemical plants streaming out MIBC refine the product by distillation, isolating it from side products and recycling unused feedstock back into the process. This technology reflects a mature industry, shaped by decades of tweaks and tight safety oversight.
Chemical Reactions & Modifications
MIBC can undergo esterification with organic acids to generate esters, which see use in fragrance and flavor industries. In practice, chemists may oxidize it to form ketones, branching out its utility even further into synthesis work. The alcohol’s secondary structure enables creative transformations that lead to higher-value intermediates. Photon-driven reactions and specialty catalysts continue to expand the toolkit, allowing custom building blocks for pharmaceutical research or novel plasticizers. Despite its age, new ways of harnessing MIBC’s reactivity keep emerging, driven by the need to tailor chemicals for smaller niches and greener outcomes.
Synonyms & Product Names
People in research or procurement may stumble on aliases like methyl isobutyl carbinol, 4-methylpentan-2-ol, or even MIBC. International databases and regulatory documents list a half-dozen more synonyms, each echoing a different lingo from chemical nomenclature or trade. No matter the name, the underlying molecule remains, reminding buyers to double-check with CAS numbers to avoid mix-ups between different substances on invoices or import forms. This tangle of product names stems from decades of shifting chemical standards and world trade.
Safety & Operational Standards
Storing and handling 4-Methyl-2-pentanol brings real-world considerations to the factory floor. Workers receive training on splash risks and the importance of local exhaust ventilation, since the vapors cause drowsiness and mild irritation if inhaled in quantity. Bulk storage calls for closed systems and flame-resistant tanks, since, like other medium-boiling-point alcohols, MIBC can ignite on a hot day near a careless spark. Safety Data Sheets for the compound spell out measures in plain language: rubber gloves, safety goggles, and, in some settings, air monitors. Waste management plans stress safe incineration or controlled recovery, since water contamination presents environmental headaches down the line. Plant operators need to pair this knowledge with worker discipline, since modern compliance rules don't forgive shortcuts or lapses.
Application Area
MIBC’s presence in mining stands out above all else, where small amounts alter the surface properties of minerals. In copper, platinum, or gold ore processing, it helps float the valuable bits while leaving waste behind. In metalworking, its solubility in both water and oil means it helps shape specialty lubricants, cutting fluids, and corrosion inhibitors that extend equipment life. Even the world of paints and coatings taps into its solvent power, since its evaporation rate enables better film formation or pigment dispersion. In fragrances, synthetic chemists sometimes use its derivatives in laboratory-developed scents, betting on its grounded chemical structure for regulatory acceptance.
Research & Development
University labs and company R&D units treat MIBC as a springboard for innovation. Environmental chemists investigate greener ways to produce or recycle it, aiming to cut the carbon footprint of what’s now a fossil fuel-derived compound. In mining engineering classes, researchers simulate flotation tanks, testing MIBC’s behavior with digital models or robotic arms instead of trial-and-error with raw ore, looking to reduce waste and cut the energy used per ounce produced. Analytical chemists refine detection limits for MIBC in water samples, prompted by tougher rules against industrial discharge. Pharmaceutical R&D workers check if subtle tweaks of MIBC’s skeleton could lead to new drugs or drug enhancers, betting on its balance of polarity and reactivity.
Toxicity Research
Toxicologists track the compound through controlled studies, eyeing potential effects on central nervous systems, aquatic life, and long-term tissue health. Animal studies and cell culture tests look for damage at both high and chronic exposures. Warnings about short-term dizziness or sluggishness reflect tests with volunteers or case reports from decades past. Regulators increasingly call on toxicologists to study breakdown products, since the slow release of alcohols in landfills or surface water lingers in the ecosystem. Long-term cancer studies have yet to link MIBC directly to major threats, but chemists and safety officers rethink concentration limits and permissible exposure every time new evidence surfaces. Advances in gene editing and stem cell work may deepen understanding of how these alcohols enter and disrupt the body’s defense systems.
Future Prospects
MIBC’s next chapter looks tied to changes in industry and pressure from environmental groups. Stricter environmental laws may spur cleaner manufacturing routes, less reliance on petroleum, or even biosynthetic pathways from engineered microbes. In flotation, miners now want smarter, less wasteful additives that leave fewer chemical residues in tailings ponds. New solvent blends must meet evolving rules for air and water quality, pushing plant managers to test alternatives and stay nimble. Researchers sense opportunities in customizing MIBC derivatives for pharmaceuticals or specialty plastics. Market volatility, spurred by shifts in mining, metals recycling, and green energy demand, keeps the MIBC business unpredictable. Sustainability remains the big challenge: companies balancing tradition with responsibility to avoid repeating chemical mistakes of the last century and leave behind a better playbook for the future.
What is 4-Methyl-2-Pentanol used for?
Understanding Its Place in Industry
4-Methyl-2-pentanol, which some call methyl isobutyl carbinol (MIBC), doesn’t get much attention outside of chemical circles, yet it’s got a quiet influence in industries that touch everyday life. Most people never see this clear liquid, but miners, lab workers, and manufacturing folks depend on it to keep operations running smoothly. There’s good reason why this compound keeps showing up on factory floors and in mineral plants around the globe.
Mining Relies on MIBC
Open-pit mines churn out enormous amounts of ore, but valuable metals like copper and gold don’t just fall out of the rocks; they need coaxing. Flotation is a standard method for separating out useful minerals from everything else, and MIBC is a star in this process. It acts as a frother, making sure bubbles form just right in the flotation tanks. These bubbles cling to metal particles and float them to the surface, making it easier to scoop them away. Get the froth right, and recovery rates climb, meaning less waste and more efficient use of resources. Years of fieldwork and lab studies back this up, which is why mining operations buy MIBC by the tanker truck.
Other Spots Where MIBC Matters
MIBC walks beyond mine gates. Solvents owe some of their effectiveness to this alcohol, especially for dissolving waxes, oils, and resins. Whether in paints, coatings, or synthetic rubbers, MIBC helps tweak viscosity and improve spreadability, all of which improve the final product. Having worked on a few industrial cleanup jobs myself, I’ve seen how critical solvent choice can be; one job went from a sticky mess to a clean finish just because the switch got made from a basic alcohol to MIBC-based blends.
It shows up in labs too, taking part in organic synthesis and chemical reactions that need a touch of selectivity. Some processes want just a bit of extra solubility or a certain reaction rate, and swapping in MIBC saves both time and raw materials. That means less waste—something every chemist can appreciate.
Health and Environmental Points
Every substance that’s useful comes with safety considerations. MIBC has a low odor and doesn’t evaporate as fast as some alcohols, so breathing it in big doses isn’t a daily hazard in most work settings, but it can still irritate the eyes or lungs if handled carelessly. The real concern comes from spills or disposal—local waterways don’t need more chemicals, and workers don’t want chronic exposure. Proper ventilation, gloves, and closed systems go a long way. Regulations already exist in most countries, but enforcement and training mean more than paperwork. I remember working alongside a seasoned safety officer who’d remind everyone to double-check seals and drums, arguing that a good process always starts with paying attention.
What the Future Might Hold
Mining stays essential, but public scrutiny keeps growing. People want less impact on land and water, so alternative frothers and greener processes could take some of MIBC’s role. Substitute chemistries get tested every year. Right now, nothing beats MIBC for reliability and price in big flotation setups. Over time, as companies invest in research, safer or more sustainable options might take center stage, but that change asks for real commitment from both suppliers and buyers. Until that day, most industries treat MIBC as a workhorse worth respecting and handling carefully.
Is 4-Methyl-2-Pentanol hazardous or toxic?
What’s in a Name: Understanding 4-Methyl-2-Pentanol
The world of solvents and chemicals throws plenty of big names at us, but 4-Methyl-2-Pentanol slips under the radar for many people. Some folks working with paints, cleaning agents, or even certain cosmetics might cross paths with this compound. It’s a clear liquid, gives off a mild odor, and helps as a solvent in many formulas. The question always comes up — is it dangerous, or just another chemical with a long label?
Check the Facts: Toxicity and Health Impacts
High exposure to many alcohol-based chemicals can put your health at risk. The main worry with 4-Methyl-2-Pentanol comes from inhalation and direct contact. Safety data sheets warn that breathing the vapors over a long period can cause headaches, dizziness, or respiratory tract irritation. Getting it on your skin repeatedly can dry things out, cause redness, and sometimes lead to dermatitis. Swallowing this chemical? That’s a trip nobody wants, with risks like nausea or trouble with coordination kicking in.
A real concern comes from its similarity to other industrial alcohols. The liver works hard trying to break it down, so drinking or inhaling large amounts piles stress on that organ. Animal studies hint at depression of the nervous system at high doses, but so far, day-to-day exposure in most workplaces doesn’t come close to these levels. Safety comes from good habits and knowing how to treat a spill or exposure before panic sets in.
Environmental Effect and Safe Practices
This solvent doesn’t tend to build up in the environment in the way that persistent pollutants like some heavy metals or pesticides do. It breaks down when exposed to air and sunlight, but with all solvents, careful storage and disposal matter a lot. Pouring leftovers down the drain or ignoring fumes can add up in closed spaces, especially for people cleaning with it day after day. I’ve seen small businesses skip proper ventilation, thinking they save money, but it just piles up risk over time. Responsible handling benefits everyone in the building.
The Occupational Safety and Health Administration (OSHA) doesn’t set an explicit permissible exposure limit for 4-Methyl-2-Pentanol, but it falls under rules for similar organic solvents. Companies put their own controls in place, like local exhaust systems and personal protective gear. Relying on a mask, gloves, and common sense beats rolling the dice with your health. People should always check labels and Material Safety Data Sheets before using anything new at work or home.
Reducing Risk: What Actually Works?
Improving safety means paying attention not only to what goes into a product, but also to how it gets used and disposed of. I’ve seen progress in workplaces using closed systems and better training, leading to fewer accidents and less long-term harm. Switching to safer alternatives works for some jobs, but sometimes nothing else matches 4-Methyl-2-Pentanol’s solvent power. In those cases, careful storage, fast clean-up of spills, and making sure no one works in unventilated rooms help keep dangers to a minimum. Reporting any exposure early can also prevent long-term health trouble.
Knowing the risks, acting with some caution, and reaching for the right protective gear go a long way. The tool itself isn’t the problem — it’s all about knowledge and respect for the label it carries.
What are the storage and handling requirements for 4-Methyl-2-Pentanol?
Basics Behind Safe Storage
Anyone who spends time in a lab or factory knows that chemicals demand respect. 4-Methyl-2-Pentanol has a strong smell and can irritate eyes, skin, or lungs. You won’t see it causing chaos on the evening news, but it deserves just as much attention as headline-makers like acids or strong solvents.
A flammable liquid, this compound should always be kept in tightly sealed containers made from materials that won’t react with alcohols. Metal cabinets or specially coated drums tend to do the job better than low-grade plastics. Shelving should sit in cool, well-ventilated spaces, away from heat, open flames, sparks, or direct sunlight. It’s no secret fire loves to find a shortcut; don’t risk leaving this liquid near electrical panels or hot pipes.
Humidity does more than fog up your goggles. High moisture can break down container materials or promote chemical changes. Even lab veterans run into trouble when a leaky cap or a sweaty drum seems minor, then later turns into a big mess. By limiting storage rooms to low humidity and steady, moderate temperature—think 15 to 25 degrees Celsius—you cut down on surprises.
Labeling and Leak Management
A fading marker line on a bottle can cost someone an afternoon of detective work. Use tough, chemical-resistant labels and replace them before smudging or peeling makes a label hard to read. Signs should warn about flammability up front.
Drips and small leaks might appear harmless, but this alcohol can spread quickly across smooth surfaces. Regular inspections mean fewer slipups. After each transfer or pour, check for rings, damp cartons, or corrosion on containers.
Cleanup kits help most if everyone using the chemical knows where to find them. Absorbent pads that handle alcohols make cleaning up puddles easier, while sand buckets or spill socks add another layer of defense. Sites that skimp on cleanup gear end up paying double when minor leaks go unchecked.
Ventilation and Personal Safety
People don’t always appreciate good ventilation until condensation or fumes show up. Open windows might seem enough on a cold day, but mechanical fans, fume hoods, or exhaust ducts truly keep vapor clouds from hovering. Try opening a poorly vented bottle in a small closet—within minutes, the sharp smell becomes overwhelming.
Safety goggles and chemical-resistant gloves belong on hands anytime this liquid leaves its storage shelf. Stories float around about someone “just opening the cap” without protection, only to end up with bloodshot eyes or rough skin for a week. Quality labs also keep lab coats and sometimes face shields nearby; these habits save money and health in the long run.
Disposal and Community Impact
Dumping leftover 4-Methyl-2-Pentanol down a sink or storm drain seems easy. This doesn't just break the rules—it risks local water. Waste must always travel to permitted waste handlers, and it gets tracked with paperwork to prove nothing slipped through the cracks.
Community relationships depend on the local industry following good practices. A neighbor’s complaint about strange smells travels fast and hurts trust. Regular training, inspections, and conversations with local fire marshals or environmental inspectors help keep everyone safer.
Looking Forward
Not everyone grows up learning chemical safety, but anyone handling 4-Methyl-2-Pentanol can pick up strong habits after a few real-world lessons. This isn’t just about following rules for the sake of it; storing and handling chemicals right protects people, buildings, and reputations.
What is the chemical formula and structure of 4-Methyl-2-Pentanol?
What Science Books Don’t Always Say: The Real Chemistry of 4-Methyl-2-Pentanol
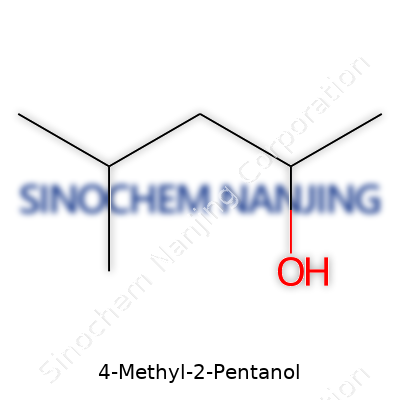
4-Methyl-2-pentanol is a clear example of chemistry sneaking into our daily life while staying out of the spotlight. You won’t see this compound grabbing headlines, but its practical significance stretches from laboratories through to the paint cans people keep in their garages. If you care about pure fact, the chemical formula is C6H14O. This tells the story of six carbons, fourteen hydrogens, and one oxygen, coming together in a way that looks simple on paper but turns heads in organic synthesis.
4-Methyl-2-pentanol—often called methyl-sec-butyl carbinol—builds itself around a six-carbon skeleton. I’ve worked in labs where recognizing this backbone quickly becomes second nature. One carbon, sitting at the second position, binds to a hydroxyl group (-OH), turning this molecule into an alcohol. At the fourth position, a methyl group (CH3) branches off the main chain. Show that to any organic chemist, and they’ll sketch it in their sleep: start at carbon 1, attach -OH to carbon 2, draw the chain through to carbon 5, then branch a methyl group off carbon 4. This branching changes more than the way it looks—it also shapes properties like solubility and boiling point, affecting how it behaves in the real world.
Walking Through the Structure
The structure looks like this: CH3-CH(OH)-CH2-CH(CH3)-CH3. Years ago, in college lab classes, I remember trying to see how this kind of branching changed the substance’s personality. Branching in molecules tends to drop the boiling point compared to straight-chain alcohols, which matters if you’re trying to separate it using distillation. That matters not just for students, but for professionals making specialized solvents or intermediates for manufacturing.
This alcohol doesn’t get as much attention as its close relatives, but its structure directly ties to how it’s used. It has a moderate level of toxicity—enough to keep chemists alert, not enough to scare people away from its industrial uses. You’ll find it as a solvent in certain coatings and plasticizers, where the slight tweak of the methyl branch can spell the difference between a smooth finish and a streaky mess.
Why This Specific Arrangement Matters
The way molecules bend and twist changes how they act, and 4-methyl-2-pentanol’s structure is no exception. That lone oxygen bonded to the hydrocarbon skeleton provides reactivity and a handle for biotransformation or chemical reactions. Down in the field—not just academic theory—producers and users know that changing branch points or shifting that hydroxyl group affects things like flammability, viscosity, and even how the substance smells. Chemical safety data sheets on this compound make it clear: always handle in well-ventilated spaces and respect what a small change at the molecular level can bring to physical health and safety.
Challenges and Thinking Ahead
Any discussion about chemicals like 4-methyl-2-pentanol eventually turns to managing safety, environmental impact, and waste. Labs and factories can improve the handling process by keeping up with the latest in air filtration and storage. Substituting with greener solvents gets discussed more often as regulations tighten worldwide. The facts point to proper training, clear labeling, and strict adherence to chemical handling best practices to keep risks low while making the most of its practical uses. That’s just responsible chemistry in action, whatever the size of the operation.
Where can I buy 4-Methyl-2-Pentanol?
Understanding the Substance
4-Methyl-2-Pentanol, often called methyl isobutyl carbinol (MIBC), shows up mainly in laboratories and industrial settings. It doesn’t belong on a home shopping list alongside groceries or hobby chemicals. You’re more likely to see it put to work as a frother in mineral flotation or as a solvent in specialized manufacturing than anywhere in the average household. This is not just a technicality—it changes the purchasing process completely.
Who Can Buy and Why It Matters
As someone who’s worked in research labs and manufacturing environments, getting substances like 4-Methyl-2-Pentanol has never involved a simple online checkout. Chemical suppliers, whether they’re giants like Sigma-Aldrich or smaller operations, ask a lot of questions. They want to know about professional credentials, shipping addresses (no P.O. boxes), and intended use. In practice, you need a business or institutional account. Even educational groups, local workshops, or independent researchers meet tight restrictions.
There’s a good reason. 4-Methyl-2-Pentanol isn’t just another bottle of rubbing alcohol. Acute exposure causes headaches, irritation, drowsiness, and long-term misuse can have much nastier effects. Countries and regions make their own rules about buying and storing, often lining up with international chemical safety protocols. Sometimes local customs even hold shipments for inspection. That protects not just the buyer but anyone else around the material.
Online Listings and Red Flags
Searching for “where to buy 4-Methyl-2-Pentanol” from a general retail mindset often leads to a maze of resellers, some credible and others not so much. Well-known industrial distributors (like Thermo Fisher Scientific, VWR, Fisher Scientific) keep everything above board: safety data sheets are always available, product quality is well documented, and support lines are staffed by professionals. They deliver only to registered businesses or labs.
Any website offering this compound to the public, no questions asked, raises major red flags. I’ve seen fly-by-night operations vanish after collecting orders, leaving buyers empty-handed and out a lot of money. In serious cases, I’ve heard stories of unexpected law enforcement interest after someone tried sidestepping regulations, thinking it was all just red tape.
Best Practices for Responsible Sourcing
Before trying to source this chemical, step back and ask why it’s needed. If a process truly requires it, a professional setup is already in place. That means dedicated storage, proper labeling, staff wearing PPE, ventilation systems—everything spelled out in the safety protocols that get taught in certified environments. Ethical sourcing supports public trust in research and commercial chemicals, and avoids unnecessary risks.
Alternatives exist for many projects. If you’re a small workshop or educator, other solvents or teaching materials meet safety standards and often give the same results without the hoops or hazards. Joining a university or working with a local lab also opens access to chemicals under professional guidance, without shortcuts or sketchy sources.
Taking shortcuts in chemistry doesn’t just risk a failed experiment—it risks real harm. Choosing trusted suppliers, following regulations, and taking the training seriously keeps everyone safer and builds a culture of responsibility in science and industry.
| Names | |
| Preferred IUPAC name | 4-Methylpentan-2-ol |
| Other names |
Methyl isobutyl carbinol MIBC 4-Methylpentan-2-ol Isobutyl methyl carbinol |
| Pronunciation | /ˈfɔːr ˈmɛθɪl tuː ˈpɛntænɒl/ |
| Identifiers | |
| CAS Number | 108-11-2 |
| Beilstein Reference | 1751809 |
| ChEBI | CHEBI:77716 |
| ChEMBL | CHEMBL44330 |
| ChemSpider | 68210 |
| DrugBank | DB13925 |
| ECHA InfoCard | ECHA InfoCard: 100.003.954 |
| EC Number | 200-746-9 |
| Gmelin Reference | 82136 |
| KEGG | C02475 |
| MeSH | D013746 |
| PubChem CID | 11541 |
| RTECS number | SA4925000 |
| UNII | 4M6DP3I36E |
| UN number | UN2053 |
| Properties | |
| Chemical formula | C6H14O |
| Molar mass | 102.18 g/mol |
| Appearance | Colorless liquid |
| Odor | Characteristic odor |
| Density | 0.818 g/cm³ |
| Solubility in water | soluble |
| log P | 1.51 |
| Vapor pressure | 0.76 mmHg (at 25 °C) |
| Acidity (pKa) | 16.37 |
| Basicity (pKb) | 5.34 |
| Magnetic susceptibility (χ) | -7.74 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.419 |
| Viscosity | 3.9 cP (20°C) |
| Dipole moment | 2.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 224.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -396.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –3894.0 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P260, P264, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P233, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | Flash point: 52°C (125°F) |
| Autoignition temperature | The autoignition temperature of 4-Methyl-2-Pentanol is "375°C (707°F)". |
| Explosive limits | 4.1-8.0% |
| Lethal dose or concentration | LD50 oral rat 2480 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 2500 mg/kg |
| NIOSH | SA1750000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 4-Methyl-2-Pentanol: 100 ppm (360 mg/m³) |
| REL (Recommended) | REL (Recommended Exposure Limit) of 4-Methyl-2-Pentanol is "25 ppm (skin), 125 mg/m3 (skin) |
| IDLH (Immediate danger) | IDLH: 400 ppm |
| Related compounds | |
| Related compounds |
2-Pentanol 3-Pentanol 2-Methyl-2-butanol 4-Methyl-2-pentanol acetate 4-Methyl-1-pentanol |

