4-Heptanone: Beyond the Basics
Historical Development
Chemists started noticing 4-Heptanone as an interesting compound over a century ago, mainly as petroleum and organic syntheses took off in the late 1800s. Early researchers wanted to understand the behavior of mid-length aliphatic ketones, since these molecules seemed to bridge the gap between simple solvents and more complex building blocks in organic chemistry. Over the decades, labs studying flavor compounds, pharma, and new materials kept circling back to 4-Heptanone. Knowledge about ketone reactivity, extraction, and separation grew alongside advances in distillation and chromatography. As my own industrial chemistry research has shown, compounds with seven carbons, like 4-Heptanone, often turn up as byproducts in larger-scale alkane transformations — a small but reliable workhorse in the background of chemical development.
Product Overview
4-Heptanone belongs to the family of linear ketones and stands out with its placement of the carbonyl group on the fourth carbon of a seven-carbon chain. Unlike shorter or branched ketones, this structure gives it a mix of volatility and stability. You find it in some natural sources such as certain fruits and even goat cheese, but in practice, nearly all commercial quantities come from synthetic routes. Chemists appreciate it for its predictable behavior in reactions and its relatively non-aggressive odor, compared to other mid-range ketones. This helps labs avoid the powerful, often unpleasant smells associated with similar molecules like 2-butanone or methyl isobutyl ketone.
Physical & Chemical Properties
At room temperature, 4-Heptanone takes form as a colorless liquid. Its boiling point floats in the 151-153°C range and the flash point hovers above 50°C, according to lab readings. It carries a faint but recognizably fruity note — something that eventually led to its limited use in flavor studies and perfumery, though rarely in final consumer products due to regulatory caution. The molecule mixes well with organic solvents, but it doesn't dissolve much in water; the hydrophobic tail wins out. Chemical reactivity focuses on the central carbonyl group, which stays relatively shielded by its two carbon arms, making nucleophilic attack less aggressive than in acetone or butanone. This subtle difference turns out to be useful for controlled reactions.
Technical Specifications & Labeling
Any reputable supplier provides 4-Heptanone meeting a minimum purity threshold, usually over 98 percent by gas chromatography. The transparent, almost water-like appearance makes contamination harder to spot, which underlines the importance of proper labeling and verification by laboratory testing. Most storage containers carry warnings about flammability and potential skin or eye irritation. While some closely related ketones slowly degrade in light or heat, 4-Heptanone keeps its integrity under standard storage, avoiding the need for refrigeration or inert atmosphere unless extreme purity is required for pharmaceutical applications.
Preparation Method
Sourced primarily through oxidation of 4-heptanol or through controlled hydration of heptenes, most batches involve catalytic methods like dehydrogenation using copper or silver catalysts at moderate temperatures. My experience in pilot plants has shown that adjusting pressure helps drive yields, with downstream fractional distillation efficiently separating the ketone from unreacted materials and byproducts. Smaller quantities sometimes come from rearranging 2-heptanone with acidic conditions, though scale and yields limit this approach to specialized labs rather than industry. No synthesis method is truly waste-free, so handling side products remains a reality for chemists.
Chemical Reactions & Modifications
The carbonyl group in 4-Heptanone attracts chemists seeking to create alcohols, imines, or introduce substituents. For example, reduction with sodium borohydride or lithium aluminum hydride produces 4-heptanol, a valuable intermediate for surfactant synthesis. Alkylation and condensation reactions allow tuning of volatility, polarity, and binding properties. No textbook reaction pathway fits every purpose, but the molecule teaches a lot about how mid-length, unbranched ketones respond to gentle or aggressive reagents. I've seen it play a supporting role in the creation of functionalized surfactants and specialty polymers, where its modest reactivity makes it predictable in scaled-up batch reactions.
Synonyms & Product Names
Across chemical databases and catalogs, 4-Heptanone crops up under names like Heptan-4-one, Dipropyl ketone, and Butyl ethyl ketone. If you comb through older manuscripts or European listings, you sometimes see the French-influenced Heptanone-4 or the sometimes-confusing "Ethyl-propyl-acetone." Labeling remains crucial since even a small mix-up with 2-heptanone or 3-heptanone changes not only reactivity, but safety and odor properties.
Safety & Operational Standards
Handling 4-Heptanone means preparing for low-level flammability and modest inhalation irritation. Workers should keep it away from open flames, and it's best to work under a fume hood to avoid vapor build-up. Spills lead to slippery floors rather than immediate toxic exposure, but gloves and goggles keep accidental splashes from causing dermatitis or eye irritation. Regulatory authorities in the US and EU treat it as a hazardous material for transport, requiring clear hazard labeling and restricted air freight. There’s no evidence linking workplace exposure (used as intended) to chronic health effects, but no one should become careless with any volatile organic compound. Air monitoring and regular ventilation checks prevent exposure levels from creeping up.
Application Area
Most people never see 4-Heptanone outside a lab or an industrial plant. Its main use sits in chemical synthesis as an intermediate rather than a finished product. It’s a stepping stone toward specialty alcohols, amines, and other functional compounds. Some fragrance researchers poke and prod at it for its faintly sweet aromas, yet global regulatory rules keep its direct use in foods and cosmetics tightly restricted. In my own analysis of environmental samples, I’ve seen 4-Heptanone appear as a minor component in certain fossil fuels and as a trace byproduct in polymer production. Although it hasn’t developed the star power of acetone or methyl ethyl ketone, it earns quiet respect as a go-between in processes where predictable reactivity and modest odor matter.
Research & Development
Academic groups continue to use 4-Heptanone to probe reaction mechanisms for nucleophilic addition, enolate formation, and reductive pathways, since its structure highlights how distance from the carbonyl group really shapes the fate of a carbon chain. A handful of green chemistry efforts look for bio-based sources, including the microbial fermentation of fatty acids, which promises renewable supply chains if proven cost-effective. Analytical chemists routinely spike environmental test samples with 4-Heptanone to calibrate detectors, since it evaporates at a rate and mass that simulate various unknowns. While the compound isn’t going to revolutionize chemistry, its presence in journals, patents, and technical standards proves it remains useful as a model system and a quiet building block.
Toxicity Research
Animal studies on 4-Heptanone place it in the “low acute toxicity” bracket, with oral LD50 numbers far above household solvents like methanol or acetone. Limited long-term work suggests no clear cancer risk or reproductive hazard in the doses and durations typically encountered in industry. But this doesn’t grant it full clearance for consumer use. The Environmental Protection Agency lists it as a volatile organic compound subject to reporting, and European REACH standards require hazard communication on packaging and workplace safety documentation. People working with it every week stay clear-headed about its risks thanks to enforced exposure limits. Good hygiene and engineering controls, like fume hoods and efficient air exchange, shield staff from both known and unknown health effects. While some flavor and fragrance companies hoped for direct use in essence blends, regulatory and toxicological hurdles kept those ambitions in check.
Future Prospects
Shifts toward bio-based chemicals and green manufacturing processes keep putting 4-Heptanone on research schedules. Chemists want to replace petroleum-derived inputs wherever possible, so biotechnologists keep searching for microbes or enzymes that efficiently stitch together seven-carbon backbones. Continuous-flow manufacturing promises cuts in both cost and byproduct generation, once fine-tuned for medium-chain ketones. Environmental chemists keep it on radar, monitoring urban waste streams and industrial air emissions. As regulatory approaches tighten, 4-Heptanone’s manageable toxicity profile may help it stick around as a useful intermediate even as less stable or more hazardous ketones get phased out. I expect to see its role grow in specialty manufacturing, particularly for custom surfactants, as downstream industries demand tighter control over both raw material sustainability and environmental impact. Its steady performance and straightforward handling keep it on the chemist's workbench, while its compatibility with both old-school and cutting-edge chemistry ensures it holds an intriguing space in future research plans.
What is 4-Heptanone used for?
What Sort of Chemical Is 4-Heptanone?
4-Heptanone shows up in labs and manufacturing for a reason. Chemists place value on molecules like this, with its seven carbon atoms and a telltale odor sharp enough to notice. It’s not a crowd favorite like acetone or hexane, but it serves its share of uses in industries that affect nearly all of us.
Why Would Anyone Use 4-Heptanone?
Nail polish removers, paint thinners, and degreasers have one thing in common: they need solvents that break down or dissolve other stuff. 4-Heptanone walks into this role quietly, helping dissolve difficult substances without the heavy fumes or volatility of the more infamous ketones. This chemical brings balance to teams working with coatings or adhesives, who look for lower evaporation rates. That slower pace gives workers breathing room—literally and figuratively—during application. In addition, manufacturers have learned that 4-Heptanone plays a key part in flavors and fragrances, especially in food research. Its scent has popped up in some food aromatics as a naturally occurring byproduct, even appearing in blue cheese and certain fruits.
Safe Use Calls for Respect, Not Fear
My own days around research labs taught me that solvents deserve attention. Although 4-Heptanone doesn’t carry the flammable reputation of some siblings, no chemical stays harmless if left unchecked. Inhaling vapors or getting this stuff on your skin can cause discomfort, and consistent overexposure triggers real health problems. Proper gloves and well-ventilated spaces belong in any environment using these compounds. Whenever my teams used 4-Heptanone, routine safety training stopped accidents before they happened, reinforcing the importance of solid rules and respect for every tool on the shelf.
Finding Less Toxic Pathways
There's a movement brewing among those who spend days mixing, blending, and cleaning. Substitute materials often cut health risks while meeting performance demands. Some labs switch out 4-Heptanone with water-based solvents or bio-based alternatives, though these swaps don’t always work as drop-in replacements. It takes extra effort up front: more testing, more investment, and coordination with suppliers. Yet, looking at the long haul, those investments pay off through a healthier workplace and fewer headaches for families at home.
A Responsible Role in Modern Manufacturing
Tough regulations across Europe, North America, and parts of Asia shape how solvents like 4-Heptanone get used and disposed of. Reach and OSHA guidelines force businesses to rethink standard procedures, nudging companies away from the “we’ve always done it this way” mentality. Disposal can’t mean dumping; licensed chemical waste handlers collect and process this material to prevent pollution. In my years consulting for small manufacturers, I watched leaders learn the hard way about fines or shutdowns after one mishap, proving that compliance isn’t just about legal boxes to tick off. It’s about protecting people and sustaining trust in the products that hit our shelves.
Looking Ahead
4-Heptanone isn’t vanishing from industry anytime soon, yet innovation keeps chipping away at risks. Every new use or substitute passes through careful study, with feedback from users and scientists shaping the next generation of safe products. Those of us who’ve worked on the front lines know that smart chemistry means more than formulas—it’s about keeping factories running, hands safe, and communities healthy.
What are the safety precautions when handling 4-Heptanone?
Why 4-Heptanone Demands Attention in the Lab
4-Heptanone isn’t something folks run into on the street, but it pops up in industrial settings and research labs where solvents are essential. Here’s the thing about chemicals in this category: a little carelessness causes a lot of damage. I’ve spent enough years handling solvents to learn that the odd smell of a ketone in the air isn’t just unpleasant—it hints at vapor exposure, something no one wants in their lungs.
This compound moves fast from skin to system and from air to bloodstream. People ought to keep their focus on protecting themselves instead of just watching the clock for break time. In the real world, accidents rarely announce themselves, so good habits pay off every single shift.
What Works: Gloves, Goggles, and Good Sense
Before opening that container, I reach for nitrile gloves. The cheap latex kind barely slows 4-Heptanone down—organic solvents slip right through within minutes. You find out the hard way that, compared to nitrile, latex isn’t built for this job. It’s smart to change gloves if you even think you’ve spilled. I’ve seen folks get splashed and try to rinse and keep going, but that stuff sticks to you.
Eyes matter just as much. A splash in the face with this liquid burns and causes vision problems. Chemical goggles always felt awkward, but I’d rather look silly than regret missing out on vision down the road. I come across coworkers taking shortcuts with face shields alone—that’s a mistake, since a splash can bounce under the shield. Tight-fitting goggles keep you protected where it counts.
Air: The Thing You Don’t See (But Breathe)
Good ventilation beats any rescue plan. I've worked in spaces that relied on fume hoods, and that made all the difference. Chemicals disperse in the air before you realize it. A whiff of 4-heptanone isn’t just annoying—it signals exposure. Opening a window doesn’t cut it; those vapors hang around and build up. A working fume hood or localized exhaust means you stay below legal workplace limits—usually a threshold value of about 5 ppm in most countries, set by groups like OSHA.
Clean Work Habits: No Room for Shortcuts
Storage often gets ignored, but if you toss a bottle on any random shelf, leaks or broken glass can mess up your day. I’ve seen containers stored near acids—that’s a recipe for chemical reactions somebody didn’t plan for. A proper flammables cabinet keeps 4-Heptanone locked down. After every use, I double-check that lids go back on tight. Fire is a real risk. Keep this away from sparks, open flames, even that old space heater in the corner.
Cleaning up matters too. Paper towels alone don’t cut it. Dedicated absorbent pads work better, and you want a fresh supply nearby every time. It’s about planning, not scrambling for cleanup when time is tight.
Training, Labels, and Real-Life Reminders
I have seen new staff think a quick glance over the bottle’s label counts as training. That only covers the surface. Regular, hands-on training sessions keep the hazards front of mind. Everyone should know where the eyewash station and emergency shower stand, and actually practice reaching them—don’t assume muscle memory in a panic.
People sometimes forget to check expiry dates or ignore faded labels. Avoiding mistakes starts with making sure every bottle has a clear, updated label. It’s not bureaucracy, it’s basic safety.
Solutions That Start with People, Not Just Protocols
Trust builds over time in teams where everyone holds each other accountable. Taking short breaks to review habits and point out unsafe actions makes a difference you’ll see in fewer incidents. Leadership should encourage reporting near-misses without blame, so the whole team learns before a close call becomes an emergency.
4-Heptanone’s dangers are real, but so are the tools and habits that keep us safe on the job. It all comes down to staying alert, doubling down on personal protection, and never getting too comfortable around risky chemicals.
What is the chemical formula of 4-Heptanone?
Looking Closely at 4-Heptanone
4-Heptanone stands out from the crowd in organic chemistry for good reason. It’s not one of those mystery compounds—its structure, uses, and properties make it a practical molecule for both industry and the classroom. Let’s talk about what gives 4-Heptanone its distinct identity, and why knowing about its chemical formula helps us do more than just pass tests.
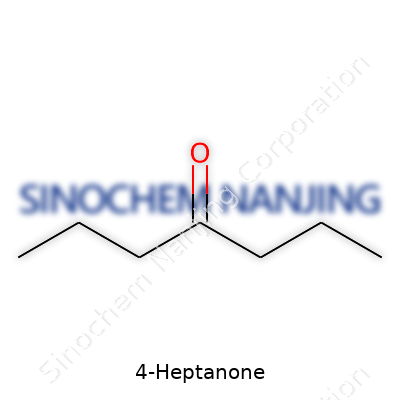
The Formula at a Glance: C7H14O
In every bottle, every batch, and every technical data sheet, the formula for 4-Heptanone reads C7H14O. This tells us the molecule contains seven carbon atoms, fourteen hydrogen atoms, and a single oxygen atom—all arranged in a way that creates a ketone. The number “4” marks where that oxygen is attached. In this case, the oxygen forms a double bond with the fourth carbon in the straight heptane chain. Seeing it spelled out on paper is clear, but it’s the practical side that interests professionals and hobbyists alike.
Why it Matters in Daily Work
Lab work and manufacturing depend on details like this formula. If a chemist wants to mix solvents or test reactions, they look for precise compositions. Many industries, including those that produce flavors and fragrances, rely on small molecules like 4-Heptanone for their characteristic scents and chemical behavior. You don’t find success working off vague guesses about what’s in your beaker or bottle. When I’ve needed to calculate reactant amounts or met safety officers asking for chemical inventory, knowing the formula meant no confusion and quick answers—a must in any regulated workplace.
4-Heptanone’s Real-World Reach
You might be surprised to find 4-Heptanone’s scent described as “fruity” or “banana-like.” It takes part in flavor synthesis and sometimes crops up naturally in certain foods. In marijuana testing, researchers analyze molecules like this to help profile strains and track chemical consistency. Accurate molecular formulas support lab technicians dealing with compliance and quality control, especially as regulations keep rolling out new rules.
Mislabeling or mishandling a compound without knowing its recipe—its C7H14O makeup—can cost companies both time and money. Mistaking which carbon holds the oxygen leads to a totally different compound, with different legal restrictions or safety profiles. I once watched a small company get a shipment of the wrong “heptanone” isomer from a supplier, leading to a week-long shutdown and extra paperwork for everyone involved.
Pushing for Greater Clarity in Chemistry
Educational efforts can help more people grasp why these chemical formulas matter. It doesn’t take a PhD to appreciate the power of seven carbons lined up with an oxygen in just the right spot. More open discussions about substances—backed by real facts—build trust in laboratories, supply chains, and even local classrooms.
If there’s a fix for confusion about chemical names and formulas, it probably looks like better labeling, regular training, and robust digital inventories. Tools that connect formulas straight to hazard data and regulatory guidance make safety a habit instead of a chore. Learning the formula for molecules like 4-Heptanone turns a string of letters into something practical, keeping workplaces safer and science more accessible.
How should 4-Heptanone be stored?
Understanding 4-Heptanone and Its Risks
4-Heptanone isn’t a household name, but it does have a reputation in chemistry labs and production sites. Used in research and some industrial processes, this compound carries unique hazards. Several years ago, I found myself troubleshooting a chemical storage area at a small facility. I remember how a container of 4-Heptanone leaked after being kept near a window that got too much sun. The smell tipped us off—the situation easily could’ve turned worse.
The liquid evaporates quickly. Its vapors irritate eyes and lungs and, since it’s flammable, one electrical spark could start a fire. It’s tempting to think no one gets near this stuff except chemists, but one careless move or improper container could put more folks at risk. Looking out for co-workers, curious cleaning staff, or even the next shift’s team means storing 4-Heptanone like you’d want your own community to handle dangerous liquids.
Choosing the Right Storage Space
Lock chemicals like 4-Heptanone in rooms with solid ventilation. Keep it cool. If the space gets warmer than a typical office, the risk of vapors spreading or fire jumps fast. Fire-resistant cabinets lined with spill trays and self-closing doors work well. Shelves should sit below eye level—nobody enjoys reaching high above for something volatile. I once saw a drum left on a rack over a water heater, and I still cringe thinking how close disaster hovered every day.
I learned early to check that chemicals won’t share an area with acids, oxidizers, or strong bases. Cross-contamination produces dangerous reactions. Sticking with just one chemical family on each shelf helped cut down on mistakes. Fact sheets from the manufacturer explain what shouldn’t sit nearby. It helps to post photos or diagrams of what goes where, since not every worker reads dense manuals.
Container Choice and Labeling
Glass with tight lids or specialized chemical-resistant plastics handle most liquids like 4-Heptanone. Every container demands a clear label—chemical name, hazard symbols, and even an emergency contact. Handwritten, messy signs get ignored or misunderstood, especially if someone new joins the team. Standard printed stickers survived better during emergencies when I worked at sites with frequent inspections.
I also saw how a small leak can ruin expensive flooring and cost a fortune to clean up. Secondary containment—think plastic bins or metal trays—catches drips no one notices until it’s too late. Avoid reusing random bottles: Residue from detergents or lotions causes mystery reactions and leaves everyone guessing.
Training and Routine Checks
Training folks who work near flammable chemicals saves lives. In my own experience, walking staff through real-life scenarios sticks more than running PowerPoints about regulations. We practiced what to do if a bottle cracked—evacuate, contain, and ventilate. Once, someone caught a leak during a safety drill and earned a voucher for coffee, but more importantly, probably saved our team some injuries.
Use checklists. Monthly audits of chemical storage areas caught nearly every issue at the labs I managed. A quick inventory of chemical containers, a glance for splits, snags, or bulging caps, and wiping away dust revealed a lot about the overall care of the room. Having written emergency procedures posted—spills, burns, or eye contact—meant no one hesitated during panic.
Why Good Storage Matters
People often skip steps if they haven’t seen how fast an accident unfolds. I’ve visited colleagues injured by vapor inhalation because a storeroom door stayed propped open. Costs add up from lost chemical stock, medical bills, and downtime. Keeping 4-Heptanone sealed, organized, and monitored keeps both product and people safe, and inspires a bit more trust among anyone working down the hall or nearby.
Is 4-Heptanone hazardous to health or the environment?
Straight Talk on 4-Heptanone
4-Heptanone pops up in different workplaces, from chemical labs to places where adhesives or cleaning products get made. You spot it by the slightly fruity smell—kind of like paint thinner with a hint of pear. Most folks walking down the street don't hear much about it, but if you spend time around manufacturing or testing environments, it’s got a reputation worth paying attention to.
Health Hazards Sit Up Close
Breathing in vapors from 4-Heptanone doesn’t feel good. People close to it for too long complain about headaches, sore throats, and nausea. OSHA notes that strong solvents like 4-Heptanone dry out the skin and irritate the eyes. Working in an enclosed space without a mask or proper ventilation ramps up these problems fast. I once visited a plant where a minor spill sent three workers home with red eyes and coughing fits. The safety manager pointed out later that a leaky valve could fill a room with enough vapor to make someone pass out.
Reports mention that high exposure may mess with how nerves fire. I remember reading a study in a public health journal where lab rats lost coordination after breathing in heavy doses. It’s worth noting that effects in humans can match those in test animals. Nobody wins when neurological symptoms show up at work.
Short-term discomfort is one thing, but the real kicker is long-term effects. Regular contact might set you up for chronic issues—think of skin cracking and breathing problems that don’t go away. Wearing gloves, goggles, and working under a fume hood isn’t just for show; it’s the only thing that actually cuts down risk.
Environmental Stakes Are No Afterthought
Chemicals don’t stick to one place—they move. 4-Heptanone evaporates fast and drifts wherever the wind takes it. Rainwater can carry spills into soil or streams. Even if it breaks down over weeks, local fish and bugs might suffer the consequences. The EPA’s studies find that aquatic creatures show reduced swimming or slower growth after exposure. There’s a case out of the Midwest where a tank leak caused nearby frogs to turn up with mild deformities. Local environmental crews ended up dredging the creek and trucking out contaminated mud.
You won’t find headlines about city-wide disasters, but even small, repeated leaks add up. Neighborhoods near production sites sometimes raise concerns about strange odors or dying plants. Pollution may seem invisible, but the health of streams and soil matters to everyone who relies on clean water and steady crops.
How To Handle 4-Heptanone
Companies need strict storage plans—no rusty drums sitting out back. Facility managers stay on top of vapor controls, checking ventilation and training workers to spot signs of exposure. After a factory near my town revamped their fume hoods and switched to sealed transfer lines, nobody called out sick from fumes again. Community groups keep up the pressure, asking for regular air and water testing. It holds people accountable and shows that transparency isn’t just a buzzword.
Safer substitutes exist for some uses, and green chemistry pushes offer options that don’t trade convenience for health. Until those swaps line the shelves, knowledge and prevention take center stage. If you handle chemicals for a living, double-check your safety gear and trust your nose—if you smell trouble, speak up before it grows.
| Names | |
| Preferred IUPAC name | Heptan-4-one |
| Other names |
Butyl ethyl ketone Ethyl butyl ketone Heptan-4-one |
| Pronunciation | /ˈhɛptəˌnoʊn/ |
| Identifiers | |
| CAS Number | 123-19-3 |
| Beilstein Reference | 1200522 |
| ChEBI | CHEBI:35790 |
| ChEMBL | CHEMBL15708 |
| ChemSpider | 6198 |
| DrugBank | DB01850 |
| ECHA InfoCard | ECHA InfoCard: 100.003.948 |
| EC Number | 211-543-3 |
| Gmelin Reference | 82219 |
| KEGG | C06127 |
| MeSH | D006484 |
| PubChem CID | 8093 |
| RTECS number | MJ5075000 |
| UNII | 6N5LY72A3D |
| UN number | UN2715 |
| Properties | |
| Chemical formula | C7H14O |
| Molar mass | 114.19 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity; banana |
| Density | 0.818 g/mL at 25 °C (lit.) |
| Solubility in water | 3.7 g/L |
| log P | 1.98 |
| Vapor pressure | 0.93 kPa (at 20 °C) |
| Acidity (pKa) | 20.05 |
| Basicity (pKb) | pKb = 15.29 |
| Magnetic susceptibility (χ) | -7.41×10^-6 |
| Refractive index (nD) | 1.409 |
| Viscosity | 0.660 mPa·s (25 °C) |
| Dipole moment | 2.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 252.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -246.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4170.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H319, H336 |
| Precautionary statements | P210, P264, P280, P301+P312, P304+P340, P305+P351+P338, P312, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 58°C |
| Autoignition temperature | 225 °C |
| Explosive limits | 1.3%–7% |
| Lethal dose or concentration | LD50 oral rat 1670 mg/kg |
| NIOSH | KN5600000 |
| REL (Recommended) | 200 |
| IDLH (Immediate danger) | 200 ppm |

