3-Pentanone: Chemistry’s Versatile Middleman
Tracing the Footsteps of 3-Pentanone
Turning back the pages of chemical history, 3-pentanone shows up as an underappreciated yet resourceful ketone. Chemists first noticed it in the 19th century as they pieced together relationships among simple organic molecules. Before modern digital instruments, researchers relied on smell, boiling points, and hands-on reactions to map out the world of compounds. It became clear that this molecule, known as diethyl ketone, came with practical advantages—easy to purify and reactive in just the right ways. As modern labs evolved with NMR and chromatography, tracing and isolating 3-pentanone moved from hours at the bench to rapid results, but the compound still carried a whiff of those early, painstaking experiments.
Getting Acquainted with the Molecule
3-Pentanone, or diethyl ketone to many, slots neatly into the growing line-up of simple ketones. Carrying a five-carbon backbone with its carbonyl located at the third carbon, it fills a gap between more famous cousins like acetone and methyl ethyl ketone. In the bottle, it looks like a clear, watery liquid. Its odor—stronger than acetone but less biting—is a calling card for anyone who spends time around solvents. The molecular structure makes it less volatile than acetone, yet it dissolves many organic substances with ease.
Physical and Chemical Realities
This molecule boils at about 102 degrees Celsius and freezes somewhere around minus 40, making it usable across a fair range of temperatures. It doesn’t like water much; you’ll see it floating on top if the two end up together, which matters for separating mixtures in the lab. Flammability is part of its story too; a spill near a flame can spark excitement of the wrong kind. On the chemical side, that central carbonyl group is eager to react—offering a launchpad for condensation reactions, reductions, and nucleophilic attacks. Chemists appreciate how predictably it behaves compared to other similar ketones, giving some control in synthesis.
Specifications and Labeling Challenges
Lab bottles of 3-pentanone should carry clear, honest labels. Concentration, purity, and known contaminants matter to both technicians and downstream users. Relying on outdated or ambiguous tags can reroute an experiment or cause costly batch recalls. The presence of ethanol, water, or byproducts from synthesis gives clues about quality and source. Regulatory warnings flag flammability and exposure risks, keeping end-users aware of operational hazards. Any labeling strategy that skips details opens the door to confusion or unsafe handling—a lesson hammered home in lab safety seminars everywhere.
How Chemists Prepare 3-Pentanone
Traditionally, 3-pentanone has roots in distilling calcium salts of valeric acid mixed with calcium acetate, a process that echoes back to classic organic chemistry protocols. This heating splits the mix apart and emits the ketone, later separated by distillation. Industrial routes have grown more efficient over time, shifting toward more reliable, scalable approaches using Grignard reagents with propionyl chloride or other tailored methods. These new processes cut down on byproducts and waste, not just meeting demand but also heeding modern calls for green chemistry. Refinements in purification, often using fractional distillation or column chromatography, carve out the pure compound from the raw mix.
Reacting and Tweaking 3-Pentanone in the Lab
With its central carbonyl, this molecule acts as a good starting material for a slew of organic syntheses. Under the influence of strong bases, it undergoes aldol condensation, stringing new carbon chains together—a favorite trick for building complex molecules. Reducing agents like lithium aluminum hydride can turn it into secondary alcohols. Halogenation, if conditions suit, pops a chlorine or bromine on the alpha carbon, opening more doors for synthesis. I’ve watched students use 3-pentanone as a model molecule for teaching these reactions; its simplicity makes the lessons stick and leaves fewer unpleasant surprises compared to trickier or more sensitive substrates.
Names in the Marketplace
This single molecule goes by many handles. Diethyl ketone pops up in older literature, and international chemists sometimes call it propanone. Other synonyms, such as pentan-3-one, hover around the same structure. The differences aren’t just academic—ordering the wrong synonym from a supply house can mean days lost to reshipping or redoing procedures. Careful cross-checking gets more important as supply chains stretch around the globe.
Keeping Hands and Labs Safe
Handling 3-pentanone means paying attention to flammability and inhalation risks. I’ve seen senior chemists grow frustrated when undergraduates treat volatile solvents casually or skip eye protection. The compound evaporates fast, filling the air with fumes that can irritate eyes and lungs. Good ventilation isn’t just suggested—it’s essential. Written safety standards make the rounds in most workplaces, but in practice, repeated reminders and clear signage do the real work. Developing habits like capping bottles and keeping trays underneath stock containers lowers risk. Modern regulatory bodies require clear exposure limits, and experienced chemists keep Material Safety Data Sheets handy, not just as legal sleight-of-hand but as a road map for safe operations.
Working Solutions and Real-World Uses
Chemists rely on 3-pentanone for tasks ranging from solvent duties in syntheses to intermediate steps in the production of pharmaceuticals. Reactions with active methylene compounds produce tartronic acids and other building blocks. In fragrance industries, it helps dissolve and blend flavors and scents—its lower toxicity than several alternatives comes as a surprise to newcomers. Coatings and resins, too, find a place for this ketone as a handler of polymers at the blending stage. Its presence is less obvious than high-volume solvents like acetone or MEK, but in specialty work or fine-tuned resin systems, it fills an important gap.
Research Streams and Modifications
Research groups have dug into 3-pentanone’s reactive nature, using it as a standard in spectroscopic studies as well as a jumping-off point for exploring new synthetic techniques. Its reactivity with nucleophilic reagents does more than fill journals; the insights matter for real production-scale chemistry. Green chemistry advocates take a hard look at emerging synthesis methods, measuring atom economy and solvent alternatives. In academic labs, undergraduate courses use its reactions to anchor foundational concepts about ketones and organic reactivity. Patents involving derivatives of 3-pentanone show up in fine chemical synthesis and agrochemical intermediates, where every new functional group added to its core structure gets measured, documented, and analyzed.
Understanding the Risks: Toxicity Research
Work on 3-pentanone toxicity draws a mixed picture. At low concentrations, exposure mainly leads to mild irritation—eyes water, throat itches—but at higher levels, central nervous system effects kick in. Animal studies point to reversible symptoms, yet avoidance of direct contact and inhalation comes from years of cumulative lab experience, not just datasheet warnings. The odor, sharp and unmistakable, acts as an early warning system. Chemists in older generations remember scant attention paid to ventilation, but modern safety standards and ongoing toxicology updates keep risks within reason. Some studies have looked at long-term effects without turning up the carcinogenicity concerns linked to other industrial solvents, yet prudent handling remains universally recommended.
Looking Ahead: The Road for 3-Pentanone
The future of 3-pentanone depends on innovation both in lab technique and industrial scale-up. Researchers keep chasing greener, cleaner synthesis pathways, targeting reduced waste and lower environmental impact. Its role as a model ketone in teaching is unlikely to fade; students need simple, well-behaved molecules to cut their teeth on. Demand in niche areas like pharmaceutical intermediates or specialty coatings will persist, especially where tighter regulation edges out more toxic or environmentally unfriendly solvents. As sustainable chemistry pushes forward, the appeal of this molecule could expand—less because it grabs headlines, more because of its reliability, interactive chemistry, and the lessons it teaches for both newcomers and seasoned practitioners.
What is 3-Pentanone used for?
What Makes 3-Pentanone Stand Out
Most people don’t think about where the sharp aroma in some perfumes or the flavor notes in food products come from. I’ve walked through a fragrance factory and picked up more than a whiff of chemicals like 3-pentanone. This liquid brings a crisp, fruity smell, popping up everywhere from pear drops in sweets to musty notes in wine. Chemists call it an organic solvent, but anyone working in industrial labs knows it has a character you won’t find in every bottle on the shelf.
Everyday Factories and Labs
Down on the production floor, 3-pentanone works its magic as a solvent, especially for hard-to-dissolve stuff like resins and waxes. Paint manufacturers lean on it for its ability to blend smoothly and evaporate at just the right speed. It turns up in adhesives you’d find at a home improvement store. Even seasoned workers appreciate its knack for cleaning metal and glass, scraping away grease that stubbornly clings after other solvents give up.
Flavor House Essentials
The food industry pays attention, too, though quantities stay low. 3-pentanone helps re-create authentic fruit flavors in candies, chewing gum, and drinks. Natural pears, apples, and cheeses all have small amounts. Regulations set strict limits—flavor chemists keep paperwork close, since overdoing it could turn a treat into a health hazard. The science on safety continues to get clearer, but right now, trace amounts for flavoring remain generally safe according to the FDA and European Food Safety Authority.
Perfume and Fragrance Design
Perfume makers call it "valerone" and treat it as a valuable base note. Its unique, sweet smell rounds out more intense scents, helping create a perfume that lingers but doesn’t overpower. Every time you catch a perfume that reminds you of fresh fruit or dry grass, there’s a good chance 3-pentanone played a part in the formula.
Tool for Scientists
In the chemistry lab, researchers turn to this compound as a building block for all sorts of experiments. Making new pharmaceuticals often starts with a simple molecule before adding more complex groups. 3-pentanone acts as an intermediate in the production of vitamins, agrochemicals, and even specialty plastics. Academic researchers push the limits, testing how this one solvent supports innovative reactions that shorten manufacturing steps and cut down on waste.
Navigating Concerns and New Directions
Handling solvents brings real risk, and 3-pentanone is no exception. It can irritate the eyes, skin, or lungs if you don’t use it with proper safeguards. Long-term exposure needs more study, but strict workplace rules—ventilation, gloves, goggles—protect workers in factories and labs. Industry leaders keep looking for greener options too. Bio-based solvents, safer handling guidelines, and better waste recovery can make a difference, but fast, effective solvents like 3-pentanone aren’t easy to replace.
The Bottom Line
Whether in perfumes or paints, food labs or research benches, 3-pentanone carries real value. Respecting its benefits without losing sight of safety and environmental impact takes teamwork from scientists, manufacturers, and regulators alike. As technology improves, safer and more sustainable options may start to take 3-pentanone’s place, but for now, it remains one of those behind-the-scenes heroes in products people use every day.
What is the chemical formula of 3-Pentanone?
Getting Straight to 3-Pentanone
3-Pentanone, known to many chemists as diethyl ketone, brings a formula that’s easy on the eyes: C5H10O. This single line tells us a lot about the substance. Break it down, and you spot a five-carbon chain, ten hydrogens, and one oxygen in the mix. It’s a simple compound, but its story is bigger than just numbers and letters.
How 3-Pentanone Shapes Industries
Back in college labs, I remember the sharp smell of ketones during organic chemistry practicals. 3-Pentanone’s strong odor stands out, and it’s no surprise that folks in the fragrance and flavor world have taken notice. This compound finds its way into perfumes—offering a unique, slightly fruity note. That carbonyl group (the oxygen double-bonded to a carbon) is the secret ingredient that lets it dissolve all sorts of stuff. Companies leverage this property to whip up coatings, inks, and adhesives.
The Science Behind the Formula
Understanding what C5H10O means isn't just for chemists. The structure of 3-pentanone, with its carbon backbone and oxygen, determines how it behaves. It doesn’t react with water the way some other ketones do, but it mixes well with organic solvents. This lets manufacturers use it as a medium for chemicals that don’t like water much.
Safety and Environmental Footprint
Chemical formulas point out not only the makeup of a compound but also the risks. 3-Pentanone, like many other ketones, can irritate eyes and the respiratory system. At one old job working with industrial glues, I watched coworkers handle all sorts of solvents every day. Gloves and goggles mattered just as much as a lunch break. Companies follow rules on proper ventilation for a reason—the vapor from ketones like this one makes headaches a reality if rules take a back seat.
On the environmental side, 3-pentanone is volatile. It can evaporate quickly, adding to air pollution if too much escapes. The Environmental Protection Agency in the United States sets exposure limits, keeping health and air quality top of mind. Waste from chemical plants needs proper treatment so the air and water outside the fence line stay safe for everyone nearby.
Bigger Picture: Responsible Chemistry
So why spend time talking about a formula like C5H10O? It connects to something broader: how society uses science responsibly. As someone who’s spent time on both the lab bench and the factory floor, I’ve seen how a clear understanding of molecular formulas leads to better choices—both in what gets made and how it’s handled. Making sure students really grasp the basics goes beyond passing a test. It builds a base for safer workplaces and cleaner communities.
Better ventilation, strict storage protocols, and regular training all make a difference. Even small efforts like double-checking labels or following up-to-date safety data sheets can keep accidents from happening. It’s less about memorizing chemical formulas and more about respecting what they represent: real substances that touch real lives, for better or worse.
Is 3-Pentanone hazardous or toxic?
What is 3-Pentanone?
3-Pentanone, sometimes called diethyl ketone, pops up in labs and industrial workspaces. It acts as a solvent in the making of pharmaceuticals, inks, and paints. With a sweet, fruity smell, it often sneaks its way into processes that need something to dissolve or react without changing the substance too much. Folks working in science or manufacturing tend to run across this chemical more than most.
Exposure Risks and Health Concerns
No one walks around worrying about mystery chemicals until research points out what can go wrong. The story with 3-pentanone doesn’t read like a horror novel—yet anyone who deals with it should show some respect. Vapor from 3-pentanone can irritate your eyes and nose. Some people get headaches and dizziness after breathing it in for long stretches or at high concentrations. It’s easy to end up with a skin rash if you spill the stuff and let it soak in.
Scientists running tests on this chemical have found that its toxic effects rank lower than those of more famous troublemakers, like benzene or toluene. But “less toxic” doesn’t mean safe in any situation. NIOSH, a respected health agency, set recommended exposure limits for workers. Chronic overexposure to solvents like 3-pentanone raises concern for the nervous system and may affect memory and mood as time adds up.
Accidental Release and Environmental Impact
Spilling 3-pentanone inside a poorly ventilated building makes air tough to breathe. Its vapors like to hug the floor, moving along surfaces, which raises the odds of exposure—especially in closed environments. Outside, a spill could seep into soil or waterways. It breaks down in air over a few days, but fish and bug life in creeks don’t appreciate exposure to chemicals. Contamination may lead to local ecosystem strain, though it doesn’t seem to stick around or build up like some legacy pollutants.
Protecting Health and Safety
People who handle 3-pentanone at work need real solutions, not just warnings. Wearing gloves, using splash goggles, and keeping chemical-proof aprons handy reduces the risk of skin and eye problems. Chemical fume hoods or local exhaust fans pull away vapors, so workers aren’t forced to breathe them in. A solid training program goes a long way towards building habits, so nobody gets sloppy around containers or open vessels. Relying on clear labels and locked storage rooms helps separate dangerous ingredients from visitors or unsuspecting staff.
On job sites, I’ve seen how regular air monitoring builds confidence for everyone on the floor. Managers who trust the numbers can spot problems with ventilation or leaks early. Workers get peace of mind knowing someone checks to keep the air safe—not just for legal reasons, but to protect their own well-being.
Informed Choices and Responsible Use
Most of us want to know that safety comes before profit in any workplace. Solid science and real-world reports show that 3-pentanone isn’t among the deadliest chemicals, but it shouldn’t be brushed off as harmless. Using engineering controls, solid protective equipment, and taking the time for training helps prevent trouble before it starts. Government guidelines back up these efforts with evidence from years of chemical research. Respecting those facts keeps people and the planet in better shape, even with compounds that seem modest at first glance.
What is the boiling point of 3-Pentanone?
Understanding 3-Pentanone’s Behavior in the Lab
3-Pentanone, known as diethyl ketone, turns up a lot in chemical labs and industry projects. Its boiling point comes in at about 102 degrees Celsius. If you've done any distillation, you know how crucial it gets to track exact numbers like this one. Overheating or guessing the point means losing yield, burning up the compound, or even cranking out faulty results. Those who have sat over a flask, watching for those first rapid bubbles, know the relief of landing on a number that matches expectations.
How Temperature Shapes Handling and Storage
Temperature influences how we manage chemicals from storage to disposal. For 3-pentanone, a boiling point over 100°C means you’re not handling something that turns to vapor in a warm room, but it won’t take intense heat to push it into gas either. On a summer day, a poorly ventilated storage room creates enough trouble without adding something that evaporates in a flash. Airborne ketones can lead to headaches and dizziness for workers, so recognizing at what point vapor really starts popping off matters a lot.
Safety and Workplace Realities
Ignoring boiling points means setting aside basic lab safety. Spills and uncontrolled boils put people and experiments at risk. As a former technician, I’ve seen what happens when focus shifts away from these benchmarks. Vented hoods, fire blankets, and spill kits are not just extra steps. They’re daily habits built around numbers like 102°C. Regular training helps staff remember that the boiling point isn’t a distant piece of trivia; it shapes the emergency plan printed on every wall.
Industrial Use and Environmental Impact
Industries use 3-pentanone for making pharmaceuticals, paints, and resins. The boiling point becomes a line engineers watch for setting up distillation columns or blending it with other solvents. Missing the mark by a handful of degrees sends fumes into scrubbers, which then need more frequent replacement. Environmental compliance means keeping emissions low, and that ties right back to understanding these physical constants. There’s a story in every air-quality report and every batch record about who paid attention to the boiling point and who didn’t.
Improving Knowledge: Training and Reference Materials
I’ve seen new hires struggle with basic data gaps. There’s no excuse for this once the fundamentals appear on wall charts and safety sheets. Companies that invest in easy-to-read reference materials set their teams up to avoid disaster. Good training makes a difference, especially during onboarding. Anyone working with solvents like 3-pentanone should have the boiling point locked into memory, right alongside common first aid for exposure or fire.
Solutions for Precision and Safety
Digital thermometers, boiling point tables posted at workstations, and regular safety drills turn numbers like 102°C from trivia into daily practice. Chemical management apps can send reminders or alerts as temperatures rise, so workers never get blindsided. Equipment with digital readouts leaves less to chance compared to old analog dials. This clarity boosts trust between management and staff while lowering near-misses and lost product.
Why Boiling Points Aren’t Just Numbers
For many outside the lab, a boiling point sounds boring. Yet those who’ve faced ruined batches or struggled through the aftermath of an evaporated solvent know that real-world attention to basics keeps people safe and businesses running. The more teams talk about numbers like these, the fewer emergencies roll across the factory floor or the lab bench. That’s where experience meets science, and everyone benefits.
How should 3-Pentanone be stored?
Why Safe Storage Matters
Anyone who’s spent time in a lab knows chemical storage isn’t something you leave to chance. I’ve seen the messes that come from forgetting basic rules, and nothing stings like realizing an accident could've been avoided. 3-Pentanone may not be as notorious as some solvents, but it has enough hazards to command respect. Liquid ketones like this catch fire easily and give off fumes strong enough to make you cough even through a cracked mask. Knowing how to handle it starts with knowing how to store it.
Conditions That Keep Everyone Safer
The first storage rule I learned about volatile chemicals: keep them cool and well-ventilated. 3-Pentanone gives off vapors, and in a warm, closed room, those vapors build up until a static shock spells real trouble. Don’t put the bottle on a hot shelf or anywhere near a sunny window. In my alma mater’s storeroom, every flammable liquid sat in a metal cabinet away from direct heat. That lesson stuck.
Simple steps cut down on accidents. Keep the container tightly closed when you’re not pulling from it. A screw cap works if you thread it properly—none of that half-closed nonsense. Someone last year left a 3-Pentanone bottle loosely capped, and we spent two days airing out the store room because of the stench and irritation complaints.
Chemical compatibility also plays a big part here. 3-Pentanone shouldn’t sit next to oxidizers. All it takes is one leaky bottle, and the risk of a fire jumps sky-high. Keep acids and bases somewhere else, preferably on different shelves or even in separate cabinets. This isn’t just lab paranoia—materials safety sheets back this up with hard data from incident reports over the decades.
Containers Matter More Than Most People Think
Don’t dump 3-Pentanone into plastic and hope for the best. Glass or steel containers hold up better since 3-Pentanone doesn’t chew through these the same way it weakens some plastics. I remember a budget-conscious boss who tried to reuse old plastic jugs for solvents. We caught a softening container before it leaked meaty-smelling liquid on the floor, but the point stuck: chemical compatibility beats thrift every time.
Ventilation Shouldn’t Be an Afterthought
Lingering vapors get more people into trouble than splash accidents. Those who store chemicals at home or in a crowded workspace often forget about fume buildup. Regular air changes in the storage area keep vapor levels below irritation or explosive limits. I open up windows and use exhaust fans, especially before anyone else walks in. If you’re in a bigger facility, dabbling with a fan and a fume hood cuts the risk down to practically nothing.
Anyone managing a serious collection of chemicals ought to pick up a decent flammable storage cabinet and check that it meets local fire codes. Employers and schools face fines for skipping this step, and for good reason. Once 3-Pentanone escapes its bottle, it finds ignition sources nobody even considered, from light switches to static from synthetic lab coats.
Staying Ready for Spills
Spill kits aren’t optional. The right absorbents and neutralizers prevent a small mishap from becoming a costly cleanup job. I’ve watched someone panic with paper towels and make a bigger mess—never again. Write up a clear spill plan with absorbents, gloves, and clear instructions taped inside the cabinet.
Don’t Ignore the Labels
In my experience, the best storage habits start with a well-marked bottle. Label everything clearly with both name and hazard symbols. Faded or missing labels are a guessing game I’d rather not play. If you can't read the label, you shouldn’t open the bottle.
Moving Toward Safer Habits
Safe storage isn’t glamorous or exciting. It’s a series of small, common-sense decisions that add up. Treat 3-Pentanone and any other flammable solvent with the same respect you’d give gasoline—bottled potential that belongs locked up, marked clearly, and kept away from anything that sets it off.
| Names | |
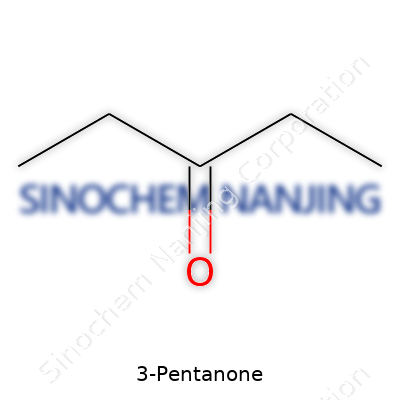
| Preferred IUPAC name | pentan-3-one |
| Other names |
Diethyl ketone Ethyl propyl ketone |
| Pronunciation | /ˈpriː.pen.təˌnoʊn/ |
| Identifiers | |
| CAS Number | 96-22-0 |
| 3D model (JSmol) | `3DStructure.CC(=O)CC` |
| Beilstein Reference | Beilstein Reference: 1209287 |
| ChEBI | CHEBI:80361 |
| ChEMBL | CHEMBL16339 |
| ChemSpider | 8186 |
| DrugBank | DB01941 |
| ECHA InfoCard | 03e24b8d-6a39-4d89-b8d9-cfd8f2d5774a |
| EC Number | 203-633-9 |
| Gmelin Reference | 106291 |
| KEGG | C01816 |
| MeSH | D010406 |
| PubChem CID | 7909 |
| RTECS number | SA4925000 |
| UNII | YC2Q1O94PT |
| UN number | UN No. 1090 |
| Properties | |
| Chemical formula | C5H10O |
| Molar mass | 86.13 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet, acetone-like |
| Density | 0.809 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | 0.97 |
| Vapor pressure | 3.4 kPa (at 20 °C) |
| Acidity (pKa) | 19.2 |
| Basicity (pKb) | 6.96 |
| Magnetic susceptibility (χ) | -7.58 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.397 |
| Viscosity | 0.455 mPa·s (20 °C) |
| Dipole moment | 2.76 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 232.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -249.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2296 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319, H336 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | -7°C |
| Autoignition temperature | 460 °C |
| Explosive limits | Explosive limits: 1.5–8.5% |
| Lethal dose or concentration | LD50 oral rat 1600 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1600 mg/kg |
| NIOSH | KTR |
| PEL (Permissible) | 200 ppm |
| REL (Recommended) | 700 mg/m³ |
| IDLH (Immediate danger) | 700 ppm |

