3-Nitrotoluene: A Closer Look at Its Role and Impact
Historical Development
My earliest brush with the story of 3-nitrotoluene involved sifting through crumbling chemical catalogs and textbooks that chronicled the era when chemists first introduced nitrotoluene into the market. Back then, nothing rivaled the thrill of new synthetic routes. 3-Nitrotoluene found its place at the end of the nineteenth century, propelled by the explosion of interest in organic chemistry and the quest for new dyes. Those early years were marked by trial-and-error purification and hand calculations. Large chemical companies, especially in Germany, established backbone processes that still echo in today’s methodologies. Synthetic dyes, explosives, and intermediates in agriculture soon rode on the back of this discovery.
Product Overview
3-Nitrotoluene emerges as a pale-yellow liquid with a faint aromatic odor familiar to anyone who’s worked a shift in an industrial lab. I’ve always thought its straightforward formula, C7H7NO2, reflected its dependable character. Among the various nitrotoluenes, the meta isomer stands out for its versatility between synthetic and applied chemistry, bridging explosives, dyes, and agrochemicals. Its significance rests in how it supports the building of bigger molecules, serving as a springboard for specialty chemicals. Walking the factory floor, I’ve seen barrels labeled for specific downstream syntheses, each destined for a unique corner of the chemical world.
Physical & Chemical Properties
At room temperature, 3-nitrotoluene remains a clear, oily liquid, rarely mistaken for anything else due to its telltale yellow hue. It packs a melting point just below zero, so it stays liquid even in a chilly storage warehouse. Boiling begins well north of 230 degrees Celsius, a trait that impacts choices for distillation or process heating. Water doesn’t welcome it; the two split, sending nitrotoluene floating on top. Benzene rings plus a nitro group mean it carries some flammability, but the more urgent concern comes during storage and handling, as both vapor and liquid form present inhalation and dermal hazards. Its reactivity extends to its ability to undergo reductions; hydrogenation transforms that nitro group to an amino, a reaction with far-reaching consequences in drug and dye synthesis.
Technical Specifications & Labeling
Every factory worker and lab technician dealing with 3-nitrotoluene must fix their eyes on its labeling. Hazards, concentration, and batch info appear in bold. International standards demand clarity: flammable liquid, risk of toxic upon inhalation, and strict incompatibility with oxidizers. I’ve stood in receiving rooms and watched managers check drum seals, matching numbers and stamps to databases and regulatory cards. Getting this wrong never ended well, especially when regulators made their annual rounds. Precise labeling not only supports safety, but cements traceability from synthesis to end use.
Preparation Method
Preparation typically happens through direct nitration of toluene using a sulfuric and nitric acid mixture, favoring the meta isomer under controlled temperature swings. I remember running this process in a scale-up setting, watching as the acids hissed and a yellow product layer gradually separated. Operators keep a keen eye on temperature, since exotherms can spiral quickly. Once the reaction settles, separation and a round of purification steps strip out unreacted acids, giving that telltale yellow stream of freshly minted 3-nitrotoluene. Safety training always emphasized how this method brings risk; acids eat through almost anything, and one slip meant burns or worse.
Chemical Reactions & Modifications
Organic chemists love the direction nitrotoluene can take. Hydrogenation stands out; pass the compound across a catalyst in a hydrogen stream, and the reduction transforms the nitro to an amino group. This opens doors to the world of m-toluidine, a precursor for dyes and pesticides. Friedel-Crafts alkylations or acylations find utility here, although conditions have to be tweaked because the nitro group drags the molecule’s reactivity lower. Oxidizing the methyl group, the ring’s resilience faces another chemical challenge, generating meta-nitrobenzoic acid along the way. Each bench or industrial reaction brings a different set of concerns for yield, purity, and environmental impact.
Synonyms & Product Names
Call it 3-nitrotoluene, meta-nitrotoluene, or m-nitrotoluene—employees and suppliers around the world use these names interchangeably. I’ve sat through more than one meeting where confusion between isomers caused delays, so clear product identification still matters. In academic lit and trade papers, aliases help, but they also underscore how this substance hasn’t escaped the family tree of aromatic nitro compounds. These names show up on container drums, regulatory reports, and customs doc sheets in international trade.
Safety & Operational Standards
Safety isn’t a buzzword here; it saves lives. Exposure risks start the moment a container opens. Liquid and vapor seep into skin and lungs—engineers and managers know to gear up: gloves, goggles, full-face shields, and air handling systems. Every warehouse or process junction needs firefighting media at hand, and spill response kits stay stocked. Industry standards across continents keep raising the bar for permissible exposure and worker training. I’ve seen standards evolve from fire extinguishers in dark corners to automatic vented storage and continuous air monitoring, reducing accidents and protecting health.
Application Area
Few compounds stretch across so many industries. Dyestuffs built from m-nitrotoluene color synthetic fibers used in clothing and upholstery. Agrochemical factories transform shipments into herbicides and pesticides, supporting farmers’ crop yields and global food chains. Explosives manufacturers count on its structure for controlled synthesis of m-toluidine derivatives, turned into stable detonators for mining operations. Each use creates its own demand for purity, safety, and regulatory compliance. In each application, workers and managers focus intensely on handling and disposal to avoid community and environmental impacts.
Research & Development
Labs all over the world continue to untangle new pathways using 3-nitrotoluene as a starting block. Every year brings reports of catalytic upgrades, greener nitration routes, or specialty intermediates for pharmaceuticals or polymers. In my experience, researchers hunt for lower-energy processes, maximizing yields with less waste. A surge of computational studies recently mapped the electronic quirks of nitroaromatics, offering blueprints for more selective coupling reactions. Funding trends push scientists to dig into biodegradable byproducts or innovative downstream uses, feeding the pipeline for next-gen dyes, medicines, or energy storage materials.
Toxicity Research
Concerns about toxicity hover over every conversation in facilities working with nitrotoluenes. Workers exposed to vapor or liquid have reported skin irritation, headaches, or worse, depending on contact time and care level. Regulatory agencies set clear exposure limits, but I’ve seen how lapses or tight schedules sometimes chip away at compliance. Modern toxicology digs deeper, mapping not just immediate symptoms, but tracking breakdown products in soil and water for months. Debate continues over long-term exposure effects—cancer risk, organ damage, and potential neurological impacts sit under the scientists’ lens. Every new finding fuels tighter rules and fresh PPE strategies.
Future Prospects
Looking forward, the fate of 3-nitrotoluene will likely depend on two forces: green chemistry and regulation. Chemists now race to cut hazardous waste before governments even finish rewriting the rules. Alternative solvents, catalytic breakthroughs, and recyclable process streams signal a future where each molecule’s environmental cost shrinks. Academic and industrial labs will push for multi-functional intermediates—transforming m-nitrotoluene into new drug scaffolds or polymer additives. Synthetic biology hints at fermentation approaches, potentially shaving away the harsh acids of traditional nitration. Regulations, especially in Europe and North America, probably will tighten, pushing everyone from factory managers to supply chain partners to engineer safer, more transparent processes. Learning from history and leveraging new technologies will shape where this small but impactful molecule fits into the coming chemical landscape.
What is 3-Nitrotoluene used for?
Stepping Into the Chemistry Workshop
A stroll through any working chemical plant reveals a range of specialty chemicals quietly powering bigger industries. Among these, 3-nitrotoluene stands out. Beneath its technical name, the molecule marks a key stop on several industrial roads, most notably in color, plastics, and an old staple—explosives.
The Real-World Path of 3-Nitrotoluene
People rarely talk about how a yellowish liquid can unlock bright colors in our lives. 3-nitrotoluene fits into the process that leads to dyes—bright, lasting ones found on clothes, leather goods, and sometimes even in the inks that put our ideas in print. Factories rely on it to produce intermediates, which then help to build azo dyes. Some of the reds, oranges, and browns popular in textiles owe their start to this single compound.
Beyond color, the molecule shows up in the world of plastics. It helps chemists build glossy coatings, resilient resins, and makes its presence felt in chemical research. Plastic manufacturers often turn to 3-nitrotoluene to craft specialty products that handle wear and tear or add chemical resistance. Consumer goods such as countertop laminates, or even small auto parts, often trace their performance back to an early step involving this substance.
From Laboratories to the Field: More Than Color
Chemists have tapped into its structure to go further—3-nitrotoluene becomes a stepping stone for manufacturing certain pesticides and fungicides. By supporting the synthesis of agricultural chemicals, the compound indirectly protects crops and helps boost food production. Pharmaceuticals also show the fingerprints of 3-nitrotoluene, although stricter controls and vetting surround its usage here.
Working in a chemical lab, you gain a respect for these precursors. 3-nitrotoluene rarely makes headlines, but in the background, it makes the day shorter for manufacturers. In the 1940s and ‘50s, the arms industry knew it as one of the keys to making TNT. Today, demilitarized uses outweigh the legacy, but the explosive connection still lingers in regulations.
Health and Environmental Footprint
The health and safety angle never strays far from concern. 3-nitrotoluene comes with risks—safety goggles and gloves become basic gear anywhere it's handled. Long-term exposure brings dangers, including skin and respiratory issues. Environmentalists keep a close watch as toxic byproducts can leak into water or soil. Regulations encourage strict containment and promote best practices for disposal and spill response.
As tighter rules arrived, industries started chasing cleaner processes. Many chemical plants upgraded venting systems, swapped in safer solvents, or moved to closed systems. The trend to “green chemistry” makes slow inroads, but habits change with better alternatives and pressure from local communities.
Looking Forward
People in the wider world might never see a drum of 3-nitrotoluene, but their daily life still connects with its footprint. Cleaner colorants, tougher plastics, crop protection—each carries a legacy that started with tough chemistry decisions. The work never finishes, since safety, waste management, and environmental burden still matter. Supporting safer production, smart alternatives, and stronger oversight keeps bringing long-term benefits—on the factory floor and out in the world our families share.
What are the safety precautions when handling 3-Nitrotoluene?
Why 3-Nitrotoluene Demands Respect
Handling chemicals like 3-Nitrotoluene isn't a task for careless hands. I’ve worked in labs with materials that can mess up your skin, your lungs, or leave you dizzy from fumes. 3-Nitrotoluene ranks on the list of those you treat with real caution. Its main risks stem from its toxicity, flammability, and long-term health effects. The National Institute for Occupational Safety and Health lists 3-Nitrotoluene as a possible carcinogen. This isn’t something to brush off: inhaling or having it soak through your skin can do damage, and the body often holds onto nitrotoluenes longer than you’d expect.
Personal Experience Counts
Many folks in the lab world learn early that gloves aren’t a suggestion—they’re your first line of defense. While using 3-Nitrotoluene, I always use chemical-resistant gloves, a high-quality lab coat, and protective goggles. This combo keeps splashes from burning or irritating skin and eyes. One chemist I know ignored the rules, removed his gloves to make extra precise adjustments, and paid the price with red, itchy skin for days. After that, nobody questioned the rules.
The Right Setting Matters
Don’t even think about opening a bottle of this stuff without a good ventilation system. Fume hoods aren’t just fancy cabinets—they stop vapors from seeping into your lungs. I’ve smelled fumes leak from poorly maintained hoods, and the headache comes fast. Always check the airflow before working and swap out old filters often.
Fire Hazards and Explosion Risks
3-Nitrotoluene doesn’t need much of a spark to catch fire. I always keep it away from open flames or hot surfaces, and I never use metal tools that might cause static discharge. More than once, I’ve seen careless storage habits—bottles left unsealed near heat sources. One slip could lead to an explosion. Everyone in the lab should know where extinguishers and sand buckets are kept, and these must always stay within reach.
Spills Happen—Preparation Saves
Spill kits aren’t optional equipment. I’ve had to use one during a moment of bad luck, and I learned the hard way that a dry lab towel won’t cut it. Think ahead and keep absorbents and neutralizers handy. Once the mess gets cleaned, contaminated gloves, towels, and tools go straight into hazardous waste bins—never into regular trash.
Training Trumps Overconfidence
Nothing breeds mistakes like overconfidence. Proper training on chemical handling saves lives. Labs that skip regular safety drills often pay for it later. In my early days, I spent a dedicated week learning only about chemical risks and safety measures. This hands-on approach built habits that saved me more than once.
Keeping Safe Means Looking Out For Each Other
Peer checks can catch missed steps. Someone calling out a missed glove or forgotten goggles prevents accidents. Supervisors who make time for refreshers show their teams that safety isn’t a box to check; it’s about going home healthy.
Making Safety Stick
Storing chemicals properly, wearing protection every time, and staying alert mark the difference between smooth experiments and disasters. Safety needs to become second nature. Accidents remind us how quickly things can spiral. With 3-Nitrotoluene and other hazardous chemicals, the small details matter most. Stay mindful, and look out for your team.
What is the chemical formula of 3-Nitrotoluene?
Getting to the Roots of 3-Nitrotoluene
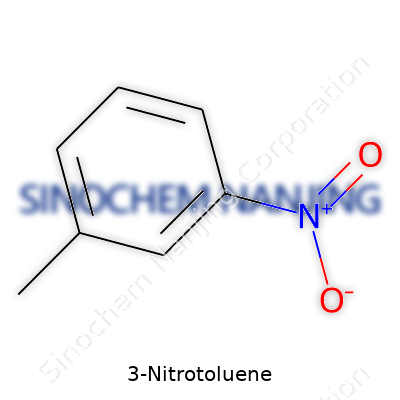
Ask anyone who's spent time in a chemistry lab—clarity helps, especially with organics. 3-Nitrotoluene carries the chemical formula C7H7NO2. That strips the compound down to the basics: seven carbons, seven hydrogens, one nitrogen, and two oxygens. This arrangement is not just a collection of atoms. Each atom’s position and neighbor shape what this molecule does in the world.
What’s in the Structure?
Toluene forms the base, which happens to be a benzene ring holding a methyl group. Sticking a nitro group (-NO2) onto the third spot—that’s what makes this 3-nitrotoluene instead of its relatives, like the 2- or 4- nitro versions. It may sound like splitting hairs, moving one group over, but subtle changes flip the script. Organic chemists—myself included—quickly learn this is not just book trivia. Properties like melting point, boiling point, and even how a molecule reacts with others hinge on that nitro group’s location.
Why Chemists and Industry Care
Studies and safety data catch my eye for a reason. Adding a nitro group reacts differently with the environment and with people. As a precursor for dyes, 3-nitrotoluene gets directed down industrial pipelines, finding its way into colorants, pesticides, and specialty chemicals. The chemical formula isn’t just a string of numbers and letters for a test—it acts as a sort of address. Regulatory bodies cross-check this formula to enforce safety and manage risks. In fast-paced production settings, precise identification keeps plant workers safe, especially since nitro compounds are known for volatility or toxicity.
Real-World Impacts and Concerns
One story I still remember involves a shipment mislabeled with the wrong isomer of nitrotoluene. The product didn’t match specs, and the batch, worth thousands, turned useless for the intended pigment application. This underscores how one position change, reflected in the formula, triggers cascading effects in manufacturing and waste.
Research on 3-nitrotoluene’s health profile offers fact-based warnings: repeated exposure links with organ toxicity. Proper labeling through the formula C7H7NO2 helps safety managers enforce training and controls. Wastewater treatment, emissions, workplace handling—all build from this. On my visits to plants, I’ve seen how environmental audits comb through records to check chemical usage against inventory lists by formula. Accuracy on this line saves time during inspections and, more importantly, prevents accidental releases into water or soil.
Finding Better Solutions
Improving education about chemical formulas benefits everyone—from high school labs to multinational corporations. Basic refresher courses help employees recognize and handle 3-nitrotoluene appropriately. Labeling systems using formulas, not just chemical names, reduce confusion. Technology can help as well: databases and digital logs track chemical stock and movement, slashing human error.
Anyone responsible for chemicals in a warehouse or a research group should drill down on formulas like C7H7NO2. Chemistry doesn’t leave a lot of room for guessing, and as experience has reminded me, a good understanding of these formulas builds a sturdier foundation for safety and progress in the lab and beyond.
How should 3-Nitrotoluene be stored?
Safety Always Comes First
Not every chemical on the shelf brings the same level of risk. Some, like 3-nitrotoluene, show up in big manufacturing plants and research labs for good reason: their use helps create dyes, explosives, and other chemicals. But with strong power comes real responsibility. 3-nitrotoluene won’t explode just by looking at it, but one wrong step with temperature, light, or a careless container can spell trouble. Drawing from personal experience, walking through cramped supply rooms full of mismarked drums, I can’t forget the relief of seeing clear, consistent labeling and cool, ventilated storage—simple steps, but a world of difference.
Understanding the Risks
3-nitrotoluene doesn’t just give off a pungent odor; it carries toxic vapors with it. Long-term exposure can damage the liver, kidneys, or nervous system. Skin and eye contact easily cause irritation or worse. In open air and under sunlight, oxidation speeds up, creating even more dangerous byproducts. It reacts with strong oxidizers, strong acids, and heat sources, opening the door to possible fires or explosions. Companies have faced major fines, fires, and even hospitalizations from accidents tied to poor chemical storage. Just last year, the U.S. Chemical Safety Board cited improper chemical segregation as one of the causes of a multi-million dollar fire in a Midwest factory.
Best Practices That Actually Work
Proper storage depends on three key factors:-container, climate, and placement. Skip the fancy sales pitch for “universal chemical cabinets;” instead, focus on practical containment—airtight glass or HDPE bottles clearly labeled in block letters. I’ve never seen real value in skimping on lined shelving or sacrificing visibility for “efficiency.” Storing 3-nitrotoluene in a dedicated, flame-proof cabinet with spill containment helps cut the risk of leaks mixing with other chemicals or vapors building up. Good air flow makes a difference; a well-ventilated area drastically reduces fume buildup, which protects workers and cuts long-term maintenance costs.
Keeping the temperature steady belongs high on the priority list. In warmer months, fans and climate control can keep storage areas below 25°C (77°F). I recall a summer power outage turning an uninsulated lab closet into a chemical oven—damage control took days and cost thousands. Keep chemicals away from direct sunlight and away from sources of ignition, such as space heaters, electrical outlets, and matches.
Regulation and Common Sense
It’s tempting to cut corners, but standards exist for a reason. OSHA, NFPA, and local fire marshals lay out clear rules for storing hazardous materials. Following these guidelines in the workplace not only avoids fines but saves lives and property. Maintaining records about purchase, inventory, and use makes audits go faster and builds a safety culture. Strong policies—and the willingness to double-check them—go hand-in-hand with healthy, accident-free workplaces.
Staff training rounds out the picture. Posters and reminders help, but nothing replaces hands-on instruction. Cleaning up spills, using protective equipment, and recognizing warning signs demand real practice. Investing time in these habits means you don’t rely on luck or hope when something goes wrong.
Practical Solutions for Better Storage
Using secondary containment trays, locking cabinets, and clear signs helps prevent mix-ups, even during a hectic day. Rotating stock and keeping a close eye on expiration dates helps avoid surprises when opening bottles that have sat too long. Teamwork between safety officers, supervisors, and workers closes the loop, making sure chemicals like 3-nitrotoluene are respected, not feared.
Every workplace brings its own set of challenges, but hard-earned, practical experience counts just as much as official rules. Taking a few extra steps to store chemicals correctly pays off not just in insurance premiums and regulatory compliance, but in keeping everyone safe and healthy.
Is 3-Nitrotoluene hazardous to health or the environment?
The Real Risks Behind 3-Nitrotoluene
For anyone who's worked in a lab or spent time around chemical manufacturing, the name 3-nitrotoluene raises a few flags. Used mainly to make dyes, explosives, pharmaceuticals, and even some pesticides, this compound finds its way into more products than most folks realize. Yet, the conversation about its impact still stays under the surface, away from daily chatter outside of industrial spaces.
Digging into Human Health Concerns
Prolonged contact with 3-nitrotoluene puts workers at risk. Inhaling the vapors, getting the liquid on your skin, or handling it without the right gear delivers some real health consequences. Studies—including one from the National Institute for Occupational Safety and Health (NIOSH)—highlight how this chemical causes skin and eye irritation, headaches, dizziness, and in higher exposures, affects how blood carries oxygen. Methemoglobinemia becomes a concern, which can leave folks tired, short of breath, or worse, grappling with more serious complications.
Personal experiences at chemical plants echo this. Protective gloves, eye shields, and fume hoods don’t just tick boxes; they're daily necessities that keep workers safe. Accidents remind us that just a little carelessness around 3-nitrotoluene can sideline someone with symptoms or send them to urgent care. In places where regulations get ignored or oversight falls short, risks multiply.
The Environmental Angle
Factories pumping out large quantities of 3-nitrotoluene can spill it, sometimes without meaning to. The chemical doesn’t break down quickly in soil or water, so leaks stick around. Runoff from spills pushes it into rivers and groundwater, choking aquatic life and spreading through the food chain. The U.S. Environmental Protection Agency classifies it as a potential contaminant, and for good reason: even small levels harm fish, insects, and microorganisms that keep waterways healthy.
Growing up near an old dye factory, I saw the local creek lose frogs and dragonflies, then show a thick green film every summer. Only in hindsight—and by digging into the chemicals used out back—did I piece together how 3-nitrotoluene leaks lined up with those declines. Environmental testing and community science projects helped track the source, but cleanup took years because the chemical clings to sediment and resists normal treatment.
Safety Doesn’t Have Shortcuts
The need for tight controls on 3-nitrotoluene gets overlooked too often. Inspections, air tests, and spill response plans take investment, yet those steps keep plant workers and whole communities healthier. Emergency showers, real training, and strong personal protective equipment can make all the difference. Substituting with less toxic alternatives works for some processes, cutting long-term exposure and waste.
At the larger scale, companies that recycle or clean contaminated groundwater show what’s possible. Turning off the tap on new pollution, then investing in cleanup and habitat restoration, returns balance to local ecosystems. The public has a role here, too: pushing for right-to-know laws and better reporting forces transparency and makes sure industry stays accountable beyond the factory fence.
Looking Ahead with Responsibility
Protecting people and natural spaces from chemicals like 3-nitrotoluene comes down to responsibility at every level. Those making it, using it, or living nearby deserve information and safeguards—not just handshakes and promises. Experience and data both underline the need for respect, vigilance, and steady work toward safer alternatives and cleaner outcomes.
| Names | |
| Preferred IUPAC name | 3-Nitrotoluene |
| Other names |
m-Nitrotoluene meta-Nitrotoluene 1-Methyl-3-nitrobenzene |
| Pronunciation | /ˈnʌɪtrəʊtəˌluːiːn/ |
| Identifiers | |
| CAS Number | 99-08-1 |
| 3D model (JSmol) | 3D model (JSmol) string for **3-Nitrotoluene**: ``` CC1=CC(=CC=C1)[N+](=O)[O-] ``` |
| Beilstein Reference | 1209317 |
| ChEBI | CHEBI:34662 |
| ChEMBL | CHEMBL1122 |
| ChemSpider | 6367 |
| DrugBank | DB11341 |
| ECHA InfoCard | 100.008.360 |
| EC Number | 202-204-7 |
| Gmelin Reference | 81567 |
| KEGG | C01724 |
| MeSH | D017346 |
| PubChem CID | 10482 |
| RTECS number | XZ9625000 |
| UNII | B5M80L7AOG |
| UN number | 1664 |
| Properties | |
| Chemical formula | C7H7NO2 |
| Molar mass | 137.13 g/mol |
| Appearance | Yellow liquid |
| Odor | Aromatic odor |
| Density | 1.162 g/mL at 25 °C (lit.) |
| Solubility in water | Slightly soluble |
| log P | 1.93 |
| Vapor pressure | 0.27 mmHg (25 °C) |
| Acidity (pKa) | 10.8 |
| Basicity (pKb) | 4.65 |
| Magnetic susceptibility (χ) | -62.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.553 |
| Viscosity | 2.03 mPa·s (25 °C) |
| Dipole moment | 3.82 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −24.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3685.3 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H332 |
| Precautionary statements | Precautionary statements of 3-Nitrotoluene: "P210, P261, P273, P280, P301+P312, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 104 °C |
| Autoignition temperature | 644°C |
| Explosive limits | Explosive limits: 1.3–7% |
| Lethal dose or concentration | LD50 oral rat 1070 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 3-Nitrotoluene: 1010 mg/kg (oral, rat) |
| NIOSH | SN41750 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 3-Nitrotoluene: 2 ppm (10 mg/m³) |
| REL (Recommended) | 13 mg/m³ |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
2-Nitrotoluene 4-Nitrotoluene Toluene Nitrobenzene |

