3-Methyl-1-Pentyn-3-Ol: A Down-to-Earth Look at Its Role in Modern Chemistry
Historical Development
Chemistry has a long memory, and 3-methyl-1-pentyn-3-ol stands as a reminder of how organic synthesis transformed in the twentieth century. In those early decades, chemists spent years refining methods to build complex structures around carbon-carbon bonds. Alkynes, with their triple bonds, captured interest for their reactivity and versatility, and lab notebooks from university research groups began to show entries about tertiary alcohols tethered to triple bonds. 3-methyl-1-pentyn-3-ol entered the picture as both an industrial curiosity and as a reference point in academic studies. Tasks once handled by crude distillates now saw cleaner, more controlled syntheses, and this compound lined up as a useful model for new reactions or as a precursor for bigger molecules. It’s easy to overlook such a compound in the shadow of more famous reagents, but as university libraries filled with research on oxidative couplings and alkynol derivatives, it played a supporting role, earning notice among specialists in fine chemicals and pharmaceutical intermediates.
Product Overview
The compound 3-methyl-1-pentyn-3-ol often appears alongside its descriptive synonyms, like 3-methylpent-1-yn-3-ol or 3-methyl-3-hydroxy-1-pentyne, showing up in catalogs for labs looking to trial unusual reactions. The molecule holds a unique spot due to the alcohol group attached to a triple-bonded carbon chain. This structure brings together the best and worst traits from both functional groups—reactivity from the alkyne, potential for catalysis or transformation from the tertiary alcohol. You find it available as a pure colorless liquid, something that points to careful manufacturing and thoughtful purification. Most researchers know it by its CAS number or its shorthand, but in the end, it’s the peculiar combination of properties that gives it staying power in synthetic routes.
Physical & Chemical Properties
Experience with 3-methyl-1-pentyn-3-ol in the lab teaches one thing above all: it’s not just another boring solvent. Its faint, somewhat sharp scent hints at the alkyne backbone lurking below the surface. At room temperature, the liquid form pours easily, with a moderate boiling range that puts it past the volatility of most common laboratory solvents, but you don’t need to set up elaborate apparatus to handle it either. The triple bond and tertiary alcohol define its chemistry, offering both nucleophilic and electrophilic interaction points. Its miscibility with many organic solvents matters during reactions and workups—a detail that saves headaches during purification. Seasoned chemists keep track of where the molecule absorbs on a chromatogram and remember the risk of dehydration or rearrangement if reaction conditions turn too harsh. Its flammability and tendency to volatilize at higher temperatures mean that keeping it under control in the lab won’t differ from protocols for similarly structured alcohols.
Technical Specifications & Labeling
The technical side of 3-methyl-1-pentyn-3-ol revolves around purity and accurate labeling. Suppliers must comply with industry standards that ensure both professional and academic chemists can trust the bottle’s contents. Clear labeling points out the molecular weight, structure, and any detectable impurities, usually trace amounts of related alkyne or alcohol contaminants. Any reputable source subjects the product to rigorous quality control—using gas chromatography, NMR verification, and moisture analysis. Sometimes manufacturers will specify a minimum purity threshold, such as 98 percent or higher, depending on the needs of the end user. As knowledge of this compound’s hazards grew, labels reflected updates from regulatory agencies, stressing proper handling and disposal to keep both staff and the environment safe.
Preparation Method
In practice, most chemists generate 3-methyl-1-pentyn-3-ol via nucleophilic addition reactions, often starting with propyne or similar materials. The go-to route involves reacting an appropriate alkyne anion with a methyl ketone such as acetone under controlled conditions, usually with an inert atmosphere and dry solvents to avoid side reactions. The Grignard or organolithium reagents play a major part here, taking advantage of the strong nucleophilicity needed to form the critical bond next to the alkyne. The resulting product gets isolated via extraction, distillation, and occasionally by crystallization, depending on temperature and scale. Lab hands familiar with the process learn how small changes in reactant ratios or temperatures shift both yield and purity. Plants producing it at scale invest heavily in process control and environmental safeguards to ensure efficiency and regulatory compliance, with waste streams neutralized and emissions kept within legal limits.
Chemical Reactions & Modifications
3-methyl-1-pentyn-3-ol gives synthetic chemists all kinds of opportunities for creativity. The triple bond acts as an entry point for hydration, halogenation, or coupling reactions, shifting the molecule’s function for use in everything from pharmaceuticals to agrochemicals. Catalysts coax the molecule into oxidative couplings, sometimes building novel frameworks for natural product synthesis. I’ve seen seminar presentations where this compound serves as the launching pad for complex lactones and cyclic ethers—a testament to its flexibility. The accompanying alcohol group enables esterifications, oxidations, or even dehydration under acidic conditions. Chasing the right selectivity becomes a challenge if unwanted rearrangements threaten the outcome, so reaction conditions make the difference. Literature abounds with examples of subtle modifications, proof that this compound occupies a favored place for developing new organic methodologies.
Synonyms & Product Names
A tour through the chemical literature brings up names like 3-methylpent-1-yn-3-ol, 3-methyl-3-hydroxy-1-pentyne, or 3-methyl-1-pentyne-3-ol. The CAS number usually stays constant across catalogs, but international regulatory bodies may list the compound under local language equivalents. Some suppliers streamline sales by assigning proprietary codes or abbreviations, yet within the research community, the chemotype description stays consistent. This consistency matters, since mislabeling impedes safety and experimental reproducibility. The clarity of naming conventions prevents mistake-laden orders and laboratory mishaps, which is especially crucial for small companies or academic departments running on tight budgets.
Safety & Operational Standards
Working with 3-methyl-1-pentyn-3-ol brings familiar risks seen in tertiary alcohols and low-molecular-weight alkynes. Its flammability remains the leading concern; open flames and hot equipment ask for extra vigilance. Lab routines involve grounding vessels, storing the chemical away from oxidants and strong acids, and using splash-proof goggles during transfer. I’ve found that its vapor can cause mild irritation, prompting the use of fume hoods or well-ventilated work areas even during small-scale prep. Documentation from regulatory agencies breaks down limits for personnel exposure and details on fire-fighting approaches. Hazard statements summarize long experience with accidental spills or inhalation, requiring spill kits and training. Disposal routes must align with environmental rules in the region, often through certified chemical waste handlers.
Application Area
Chemists turn to 3-methyl-1-pentyn-3-ol for divergent reasons. In pharmaceuticals, its unique backbone stands in as a precursor for molecules under patent search or structure-activity relationship analysis, giving a route to test new biological scaffolds. Agrochemical labs favor it for developing novel growth regulators or pesticide intermediates. Universities use the compound in undergraduate and graduate training, especially in courses covering reaction mechanisms. In the specialty chemicals sector, it can slip into the background as a reactant for custom flavor, fragrance, or polymer applications. People who synthesize advanced materials sometimes look twice at the alkyne’s reactivity for inclusion in smart materials or coatings. The versatility, rooted in the dual alkyne and alcohol character, means it rarely sits unused on a shelf.
Research & Development
Research on 3-methyl-1-pentyn-3-ol continues to produce fresh chemistry as new catalysts and greener reagents take center stage. Graduate students publish studies showing improved yields under less hazardous conditions, testing the limits of transition-metal catalysis or photochemical activation. Analytical groups scrutinize derivatives for biomedical imaging or as standards in mass spectrometry workflows. Over the past decade, increased emphasis on sustainable chemistry drew interest in scaling production methods while cutting costs and lowering emissions. Academic and industrial chemists collaborate, sharing reaction data and process improvements at conferences and in open-access journals. Funding for research on related alkynol compounds typically flows to groups probing new medicinal applications or exploring structure-property relationships in advanced technology sectors. This pattern shows a healthy mix of curiosity-driven work and industry-guided need for better intermediates.
Toxicity Research
Knowing what 3-methyl-1-pentyn-3-ol does to living systems ranks high on the list for regulatory approval in the EU, US, and Asia. Toxicity studies focus mainly on acute effects—exposing test organisms to vapors or solutions and monitoring for irritation, systemic absorption, or liver and kidney impacts. Results suggest general low toxicity at trace exposure levels, but higher concentrations can cause central nervous system depression, with drowsiness or headaches reported in poorly ventilated spaces. Long-term data remain sparse, so most regulatory recommendations err on the side of caution. Animal testing in the past showed minimal bioaccumulation and a modest breakdown rate in surface water, lessening concerns about environmental persistence. Synthetic chemists wear gloves and eye protection, avoid inhaling vapors, and treat waste responsibly to minimize risks for both workers and downstream consumers. The absence of severe toxicity makes the molecule a preferred starting point, but nobody relies on assumptions where health is concerned.
Future Prospects
As research targets more complex molecules with higher specificity, 3-methyl-1-pentyn-3-ol sits at a crossroads between old-school utility and new discovery. The rising cost of specialty chemicals drives innovation in process chemistry, with continuous-flow techniques or biocatalytic methods promising cleaner, more affordable syntheses. Medicinal chemists eye the compound for alkynylation or carbocycle synthesis, predictions bolstered by machine learning models that scan millions of chemical pathways. Environmental regulations push producers toward greener solvents, waste minimization, and lower carbon footprints, spurring incremental improvement every year. Younger scientists find untapped uses for such versatile building blocks, sometimes repurposing known reactions for material science or analytical technology. The future looks driven by collaboration—integrating process scale-up, computational modeling, and sustainable practice to keep 3-methyl-1-pentyn-3-ol in the toolbox long after more famous molecules fade into obscurity.
What are the main uses of 3-Methyl-1-Pentyn-3-Ol?
Finding Value in a Niche Chemical
In labs and factories, some compounds don’t get headlines, but they help push plenty of science forward. 3-Methyl-1-pentyn-3-ol fits right into that world. It’s a colorless liquid, catches attention from chemists because of the triple bond up against its alcohol group. Anyone who has tinkered with synthetic chemistry knows a thing or two about working with small molecules like this. You need compounds that offer a bit of versatility, and this one does just that.
Building Blocks in Organic Synthesis
For most, the main draw of 3-Methyl-1-pentyn-3-ol shows up in organic synthesis. It works as a useful intermediate during the creation of all sorts of complex molecules—think pharmaceuticals, agrochemicals, even advanced materials. The triple bond and alcohol group both make it valuable during chemical transformations. Laboratories reach for it during the production of propargylic derivatives because it can serve as a starting point for more complicated reactions. Experience in scale-up labs makes one appreciate how an intermediate like this saves trouble, cutting unnecessary steps during reaction sequences. Every stage you shave off brings real savings to manufacturers and research labs alike.
Research and Fine Chemical Development
Universities and R&D outfits order 3-Methyl-1-pentyn-3-ol for method development and to create model compounds. The compound’s structure lets researchers study how triple bonds play with other functional groups. This kind of hands-on work often leads to new reaction pathways or more efficient catalytic processes. Having spent time around bench chemists, it’s clear that practical, straightforward chemicals like this offer dependable results—the bread and butter of running reliable experiments. Data on the reactivity and selectivity in reactions involving compounds like this supports broader progress, especially in asymmetric synthesis.
Flavor and Fragrance Routes
Another use, though less flashy, pops up in the world of flavors and fragrances. Many flavor houses and perfumeries turn to small propargylic alcohols as key ingredients or as intermediates. While 3-Methyl-1-pentyn-3-ol isn’t a star on its own, it helps to create aroma compounds with a touch of uniqueness. The subtlety it brings depends on controlled reactions, where the structure can be altered just enough to produce nuanced notes. Small companies searching for bespoke molecules for luxury fragrances benefit from chemicals like this, since they open up creative routes for new products.
Safety and Environmental Notes
Any discussion about industrial use would be incomplete without covering safe handling. Compounds in this family often catch the eye of regulators, because handling alkynes with alcohol groups can introduce risks like flammability or unusual toxicities. Taking inspiration from my own factory tours, the best-run shops put time into training workers on safety measures and proper storage. It keeps employees safe and chemicals contained. On the environmental front, 3-Methyl-1-pentyn-3-ol doesn’t introduce many persistent risks, but any spill or waste needs fast cleanup because runoff could damage local ecosystems. Factoring in compliance with regional environmental standards always matters to responsible operators.
Outlook and Sustainable Practices
One smart route to improvement involves finding renewable feedstocks or optimizing reaction efficiency. Catalytic research has cut down on chemical waste and energy demands. In my experience, the smaller specialty chemicals field keeps moving toward greener chemistry, not just because of regulation but because it saves costs in the long run. Sustainable practices—like process intensification or careful recovery of solvents—give companies more control and peace of mind.
Put simply, 3-Methyl-1-pentyn-3-ol gives modern chemistry a dependable, flexible tool. It plays a hidden but essential role in research, manufacturing, and the race for innovative consumer products—always pushing things forward, one reaction at a time.
Is 3-Methyl-1-Pentyn-3-Ol hazardous to human health?
The Real Impact Behind the Chemical Name
3-Methyl-1-pentyn-3-ol doesn’t show up in daily life for most people, but it plays a part in certain chemical processes and industrial settings. The conversation around its safety comes down to real-world exposure and risk management—not just technical jargon.
What Science Tells Us
Scientific literature, including safety data sheets from reputable chemical suppliers and regulatory agencies, lists 3-Methyl-1-pentyn-3-ol as an irritant. After reading several material safety data sheets and toxicology sources, it’s clear that contact with skin or eyes leads to irritation. Short-term inhalation of vapors doesn’t sound pleasant either and may make your nose or throat burn. Handling it without gloves or goggles—something I once saw in a poorly ventilated student lab years ago—lets you feel those effects in a matter of minutes.
As with plenty of alcohols and alkynes in its chemical family, the risk ramps up if the stuff gets swallowed, inhaled, or sits on the skin too long. The National Center for Biotechnology Information lists acute effects like headache, nausea, and sometimes confusion after significant exposure. No credible source shows evidence of cancer or chronic diseases linked to it, but the fact it acts as a central nervous system depressant in big doses can’t be ignored.
Real-World Hazards and Recognized Risks
Looking at case studies and workplace incident reports, issues pop up less because of catastrophic toxicity and more from accidental splashes or spills, especially in confined labs and factories. Even reputable labs sometimes cut corners with basics like fume hoods. I’ve watched even experienced chemists start coughing and reach for eye washes during unexpected incidents. This chemical's vapors prove heavier than air, which means they can hang out close to surfaces and make breathing a challenge if not ventilated.
The main route of harm shows up through skin exposure or inhalation. As with related solvents and chemical intermediates, sensitive individuals develop redness or rashes. Anyone asthmatic or with a history of allergies in my experience faces bigger problems than the average person. Pouring or mixing this compound indoors without proper air exchange adds up quickly—the smell alone starts a headache for many.
Response and Prevention in the Workplace
Safe handling boils down to respect for basic lab safety. Personal protective equipment—like gloves, goggles, and lab coats—stops most minor accidents from becoming ER visits. Ventilation systems protect everyone, especially pregnant workers or those with respiratory issues. Reading real incident reports on regulatory sites points to a pattern: Many minor injuries stem from carelessness or pressure to speed through routines.
If exposure happens, immediate washing of skin and flushing of eyes make all the difference. Supervisors hold a responsibility to keep material safety data sheets visible and make sure staff take annual safety training seriously. Emergency eyewash stations, showers, and clear labeling mean fewer surprises.
Moving Toward Safer Workplaces
There’s growing momentum for safer substitutes in science and manufacturing. Some labs move toward less volatile or less irritating compounds. Companies start pilot programs to track employee health and local air quality. These aren’t fluffy extras—they build trust in the workplace and prevent slips that can cost more than time or product loss.
What is the chemical structure of 3-Methyl-1-Pentyn-3-Ol?
A Look Beneath the Surface
Plenty of us hear a chemical name and the eyes start to glaze over. Digging into what makes up these compounds, though, uncovers some fascinating details—especially when you see how small tweaks to a molecule’s shape can swing its usefulness and risks in the lab or in industry. 3-Methyl-1-pentyn-3-ol stands out as a compact molecule with interesting properties, driven by its unique mix of functional groups.
Building Blocks: What’s There?
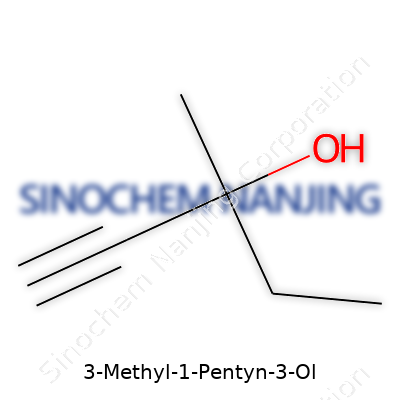
3-Methyl-1-pentyn-3-ol goes by the formula C6H10O. That backbone hosts six carbons, and this specific line-up matters. Start with a five-carbon chain, then attach a methyl (CH3) group and a triple bond (an alkyne) – all while tacking on an alcohol (-OH) group at just the right spot.
If you lay it out, numbering begins at one end where the triple bond lands, so position 1 holds a carbon-carbon triple bond. Down the line, at position 3, both the methyl group and the alcohol group branch from the same carbon. The molecule draws structure from both its triple bond and its alcohol: the alkyne adds reactivity, making the molecule quick to jump into further chemistry, and the alcohol end gives it the ability to form hydrogen bonds and dissolve in water.
Why Chemistry Folks Care
It helps to see beyond the symbols and letters. That triple bond gives chemists a foot in the door for building longer, more complex molecules—lots of widely-used drugs and advanced materials start from small alkynes like this one. The presence of the alcohol function brings solubility, which opens more doors for mixing and reactions in water.
You’ll rarely find 3-methyl-1-pentyn-3-ol in home cabinets, but it finds a home in certain specialty syntheses and research labs. Researchers use it as a stepping stone in developing new reactions. Its structure means it can serve both as a source of reactivity (from that triple bond) and as a way to anchor a molecule in water-based solutions (from its -OH).
Safety and Environmental Questions
Chemical structure calls for a discussion on safety. Small alkynes like this can sometimes cause headaches if handled carelessly—triple bonds add both reactivity and, often, flammability. Alcohol groups also mean a molecule can slip into water systems, raising questions about its handling and disposal. Labs working with such chemicals follow clear rules—good ventilation, correct storage, proper waste collection—because that’s just good lab sense.
Looking outside the lab, the structure ties into questions about lifecycle and waste. Molecules with triple bonds don’t always break down easily in nature, so researchers keep tabs on how these chemicals travel in water or air.
Where This Connects to Everyday Life
Few people outside research circles know the ins-and-outs of an alkyne-alcohol hybrid like this, but every modern material and medicine starts with someone puzzling over a molecular structure. Building up a deep understanding, block by block, lets chemists make safer products, cleaner processes, and more targeted medicines. The choices about how to arrange those carbons and hydrogens decide how a substance interacts with our world—sometimes for the better, sometimes not—and always with a story behind the structure.
What precautions should be taken when handling 3-Methyl-1-Pentyn-3-Ol?
Understanding the Risks
3-Methyl-1-Pentyn-3-Ol belongs to a group of chemicals that bring both usefulness and risk straight to the lab bench or factory floor. It's often used in organic synthesis, and it doesn’t play nicely if handled carelessly. From my own days working in a chemical plant, safety comes up in daily conversation because taking shortcuts leads to trips to the nurse—or worse. If you’ve got this compound in your workspace, treat it seriously.
Personal Protective Equipment: Start With the Basics
Let’s keep it simple. Splash goggles should cover your eyes—chemical burns on the eyes land folks in the ER every year. A lab coat and chemical-resistant gloves like nitrile or butyl rubber shield your skin. I remember a colleague who once thumbed through safety sheets and dismissed thick gloves as overkill. He got a splash on his hands, spent hours in discomfort, and learned the lesson the hard way. You don’t forget that sort of thing.
Don’t handle it barehanded or wear thin latex if that’s all you have. Chemical fumes tend to irritate, so working beneath a fume hood isn’t optional—good airflow removes fumes before they hit your lungs. Respirators don’t just hang from the wall for decoration in well-run labs; grab one if you notice any trouble with fumes or if your work area ventilation falls short.
Storage and Handling—Think Ahead
One careless move can mean an accident. Store 3-Methyl-1-Pentyn-3-Ol in a tightly sealed bottle away from acids, oxidizers, and open flames. In my past lab setups, someone always tried stacking bottles to save space or pushed things toward the edge of a shelf, both bad ideas. Don’t store glass on high shelves, as that increases the risk of spills. Clear labeling keeps others in the loop about what’s sitting in a container—no guessing games with hazardous chemicals.
Dealing With Spills and Disposal
A spill is more than an inconvenience. Most chemical accidents start small but escalate fast if you’re not ready. Keep spill kits within arm’s reach. Use absorbent pads and not paper towels, which don’t hold up under solvents. Clean up only with gloves on and wrap up any waste, marking it for hazardous disposal. Dumping leftover chemicals down the sink is illegal in most places for a reason; groundwater contamination has real health consequences. Environmental agencies have strict guidelines here, so always call your Environmental Health office if you feel out of your depth.
Don’t Skip the Paperwork
Every chemist and technician should read the Safety Data Sheet from top to bottom. It’s not busywork. The SDS gives you what to do if someone’s exposed or if the bottle shatters. Keep a printed copy handy instead of trusting your memory or an online lookup that might lag at the wrong moment.
Building a Safer Routine
Precautions are more than hoops to jump through. A team that spends a few minutes before each new shift talking over tricky steps tends to run into fewer problems. Nobody remembers all the rules until practicing them together feels like muscle memory. In experience, the safest workspaces belong to folks who share what they’ve learned, ask questions, and admit when something looks off.
Handling chemicals such as 3-Methyl-1-Pentyn-3-Ol never gives “second chances.” Every day, double-check your gear, know your exits, review your labels, and trust your gut if something smells wrong. Respect for safety beats bravado every time.
How should 3-Methyl-1-Pentyn-3-Ol be stored and disposed of?
No Shortcuts with Storage
Working around chemicals like 3-Methyl-1-Pentyn-3-Ol always reminds me of my college lab days. Good storage isn’t a box-ticking exercise; it’s real safety practice. This isn’t a household cleaner. It brings fire risks and health hazards if handled like any other bottle on the shelf. Having seen spills and minor fires happen, I learned quick that a padlocked chemical storage cabinet can save an entire workplace from trouble.
This compound catches and spreads fire easily. Flammable liquids belong in marked, flame-resistant cabinets — far from heat, sunlight, or sparks. Never tuck a container under a workbench or near a power strip. It’s easy to think “it won’t happen here,” right up until the moment it does. Personally, I prefer using small, labeled containers to limit exposure during handling. Always check the seal after pouring or transferring; fumes and vapors should stay in, not leak into your workspace.
Air and moisture speed up chemical breakdown. Humid labs or warehouses can trigger reactions, especially for compounds like this. I’ve always trusted low-humidity storerooms, with dedicated ventilation fans to keep fumes away. Labeling matters: with shifting staff and busy days, clear hazard stickers and expiry dates kept more than one rookie from grabbing the wrong bottle. Never store acids, oxidizers, or food nearby. Surprises can turn dangerous.
Personal Protection Isn’t Optional
Back in my early lab jobs, I learned through a few skin rashes that gloves and goggles aren’t for show — they prevent long-term health issues. 3-Methyl-1-Pentyn-3-Ol can hurt your eyes and skin fast. Keep gloves on, use a lab coat, and swap out gear if you spot any rips or leaks. Don’t rely on a splash not hitting you; always shield up before touching or pouring.
Disposal: Don’t Cut Corners
Disposing of this compound isn’t like emptying mop water. Hurrying disposal always ends badly. Pouring solvents down a drain clogs pipes, damages water sources, and puts waste facility workers at risk. I’ve seen drains back up and trigger more headaches than anyone expected.
Use a dedicated, clearly labeled hazardous waste container. Chemists I respect make their disposal choices based on local environmental laws. Every city and university has different guidelines, so ignoring them can lead to fines or, worse, environmental impact. Working with a university safety officer, I saw how even modest labs build separate systems for solvent waste — it really keeps toxic runoff out of landfills and rivers.
Never mix 3-Methyl-1-Pentyn-3-Ol with incompatible compounds during disposal. Some combinations set off reactions or create toxic gases. If the waste drum fills up, call a registered hazardous waste disposal service. Skipping the paperwork can risk both the team’s health and the environment. Record all waste amounts and hand off copies to your safety coordinator.
Solutions and Smarter Habits
Smart procedures often stop a disaster before it starts. Training staff regularly keeps everyone on the same page — no one should learn about chemical risks for the first time under pressure. Documenting every purchase, storage spot, and disposal event provides accountability, not just compliance. Keeping proper gear stocked and chemical storage updated feels tedious some days, but it supports safety every time.
From my experience, clear steps and honest habits make complex chemicals manageable. Even if you only handle 3-Methyl-1-Pentyn-3-Ol rarely, treat every interaction like it could matter — because it might. Today’s minor error can become tomorrow’s headline if you take shortcuts. It isn’t about paranoia, it’s practical respect — for your people, your facility, and the environment you share.
| Names | |
| Preferred IUPAC name | 3-methylpent-1-yn-3-ol |
| Other names |
3-Methylbutynol 3-Methyl-1-pentyn-3-ol Methylbutynol 3-Methylpent-1-yn-3-ol 3-Methyl-3-hydroxy-1-pentyne |
| Pronunciation | /ˈθriːˈmɛθɪlˈwʌnˈpɛntɪnˈθriːˌɒl/ |
| Identifiers | |
| CAS Number | 77-75-8 |
| Beilstein Reference | 1351487 |
| ChEBI | CHEBI:34274 |
| ChEMBL | CHEMBL1488930 |
| ChemSpider | 68298 |
| DrugBank | DB14078 |
| ECHA InfoCard | EC Number 201-233-5 |
| EC Number | 203-667-6 |
| Gmelin Reference | 82251 |
| KEGG | C03015 |
| MeSH | D010464 |
| PubChem CID | 11661 |
| RTECS number | SA7125000 |
| UNII | LDN39R04X5 |
| UN number | UN1987 |
| Properties | |
| Chemical formula | C6H10O |
| Molar mass | 100.16 g/mol |
| Appearance | Colorless to yellow transparent liquid |
| Odor | Strong acetone-like odor |
| Density | 0.832 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 0.5 |
| Vapor pressure | 0.17 mmHg (25°C) |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | pKb = 15.44 |
| Refractive index (nD) | 1.423 |
| Viscosity | 2.38 cP (20°C) |
| Dipole moment | 1.1775 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 353.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –71.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3887 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302, H312, H315, H319, H332 |
| Precautionary statements | P210, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-3-1 |
| Flash point | 31 °C (88 °F; 304 K) |
| Autoignition temperature | 405°C |
| Explosive limits | Explosive limits: 1.5–8.5% |
| Lethal dose or concentration | LD50 oral rat 1870 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 8210 mg/kg |
| NIOSH | SN8520000 |
| PEL (Permissible) | 50 ppm (240 mg/m3) |
| REL (Recommended) | REL: 5 mg/m3 |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
1-Pentyne 3-Pentyn-1-ol 2-Methyl-3-butyne-2-ol 3-Hexyn-2-ol 1-Hexyne |

