The Evolving Role of 3-Methoxybutyl Acetate in Industry and Science
A Look Back: Historical Development
3-Methoxybutyl acetate didn’t jump straight into the modern chemical market out of nowhere. Chemists set out decades ago to find better, safer solvents as manufacturing kept growing. Before anyone ever searched for this molecule, paint thinner and cleaning choices ran thin on green options. Early recipes for solvents often came laced with hazards that sent people hunting for cleaner, less toxic answers. With the push for safer chemistry and fewer environmental headaches, research labs dug into alternative esters. That’s where 3-methoxybutyl acetate found its stage. Its introduction in industrial catalogs followed detailed work in both Europe and Asia, driven by growing concern over old solvents polluting air inside factories. Markets needed something with similar solvency, slower evaporation, but better handling and safer storage. Demand for improved coatings, inks, and cleaning agents nudged companies to explore this compound's profile closely and make it more widely available.
Product Overview
You’ll see 3-methoxybutyl acetate listed as a clear, colorless liquid with a faint sweet scent. The stuff blends into daily life more often than folks realize, showing up in coatings for cars, electronics, and wood. Unlike older solvents that quickly gas off into the air, this one takes its time—useful for paints needing longer open time. I’ve noticed, especially in the hands-on tech world, that chemists gravitate to it because it performs well without the harshness or high odor of some competitors. The search for safer ingredients never ends, and this compound stands out as progress in the right direction.
Physical & Chemical Properties
3-Methoxybutyl acetate hangs out on the lower side of the volatility chart. Its boiling point settles above that of classic acetates—right around 170 to 180°C range—making it friendly to work with and not quick to disappear from open containers. Its flash point, which matters for storage and use in hot shops, measures much safer than many common ether or acetate solvents. Solubility sticks out: it blends easily with most organic solvents and can handle small amounts of water. Viscosity gives it enough heft to avoid splashing during mixing, but it pours quite nicely. Density sits just below 1 kg/L, so it neither sinks nor floats on water. Chemical stability helps explain its rising use; it resists breakdown during storage and under regular heat or sunlight exposure—nobody likes a container that goes sour after a week in the warehouse.
Technical Specifications & Labeling
Technical details don’t just live on a label for decoration; they steer safe handling. Labels flag key metrics—purity by percentage, boiling range, water content, and sometimes acidity. My own experience dealing with large drums means double-checking these specs before ever moving a container around a shop floor. Quality matters. Purity for industrial use needs to clock in at 99 percent or better, or residue gums up coatings and slows down drying. Regulatory labeling rules call out hazards, recommended storage temps, and emergency precautions, linking back to workplace safety routines. Proper labeling can be the line between a smooth day and an emergency call.
Preparation Method
Production typically involves esterification. Chemists start with 3-methoxybutanol and acetic acid, driving the reaction using a strong acid catalyst. The reaction needs tight temperature control to avoid unwanted side reactions—years spent in a research lab make you appreciate how careful attention to temperature, mixing, and post-processing can save huge headaches down the line. Once the reaction wraps up, a round of purification lifts out the end product, making it suitable for the demanding applications it fills. Waste stream management flows right along with this step, with modern setups recycling byproducts and minimizing environmental impact better than in decades past.
Chemical Reactions & Modifications
The chemistry world thrives on modifying molecules for specialized tasks. 3-Methoxybutyl acetate takes part in basic ester hydrolysis, freeing up methoxybutanol and acetic acid again—something manufacturers sometimes exploit for recycling. Reacting it with strong bases or acids unravels it, but under most conditions, it stays pretty sturdy. In synthetic labs, chemists often use it as a starting block for more complex molecules; its ester group opens doors to further chemistry. Nobody in the paint or cleaning business sticks to just the basic formula—additives or blending change things for each specific need, and this acetate accepts a range of adjustments without losing its core strengths.
Synonyms & Product Names
Names do shift from one industry to another. You’ll run across “3-methoxybutyl ethanoate” or “butanoic acid, 3-methoxy-, acetate” in regulatory documents, while European reports have listed it as “MBAc” and “1-butyl acetate, 3-methoxy-”. Anyone ordering this for a plant or lab needs to double-check codes and CAS numbers to dodge mix-ups—mislabeling has tripped up shipments in warehouse jobs more than once. Regional variations in name and documentation increase the odds of mistakes if teams don’t stay alert.
Safety & Operational Standards
Responsible workplaces don’t skip safety basics. 3-Methoxybutyl acetate still counts as flammable, so handling near sparks or open flames risks disaster. Its relative low toxicity has helped it gain approval in tougher work environments, but that doesn’t mean skipping gloves, goggles, or good ventilation. Years spent around mixing tanks taught me that “less toxic” never means “non-toxic.” Inhalation of concentrated vapors can irritate, and spills left on hands create dry, cracked skin. Key standards on storage—fiat drums, sealed caps, no direct sunlight, regular checkups on container condition—and clear spill kits make the difference between a safe operation and chaos. Training people to respect what’s in their bottles matters more than checking boxes on paperwork.
Uses Beyond the Obvious: Application Areas
This acetate solved real-world problems in coatings, plastics, and cleaning chemicals. Automotive coatings teams like it for compatibility with pigments, while people in the printing business count on it for even drying. I’ve watched it help wood finish operations hit that smooth wipe-on quality without choking up workers with harsh fumes. Many electronics manufacturers picked it to reduce worker exposure to heavier, more toxic solvents. Plus, labs use it as an extraction chemical when tricky separations need less water solubility. The demand for gentler industrial chemicals keeps broadening its reach. As global regulations keep squeezing out old-school solvents, promising esters such as 3-methoxybutyl acetate only grow in appeal.
Research & Development
Universities and private companies keep diving deeper with this compound. Research focuses on fine-tuning purity, mixing it with other green solvents, and examining byproducts for recyclability. A lot of lab time goes into evaluating how it behaves under heat, light, or in the presence of metal catalysts. Environmental impact studies have picked up pace since more countries flagged volatile organic compounds (VOCs) as an air-quality priority. Researchers now weigh solvent effectiveness against its carbon footprint, toxicological outcomes, and end-of-life options. Journals keep filling up with papers on improved synthesis, safer storage chemistry, and advanced use cases that stretch beyond just paint and ink. The momentum to find lower-risk, high-performance molecules won’t slow anytime soon.
Toxicity Research
Long-term exposure studies take center stage for any solvent aiming for broader acceptance. Initial tests showed relatively low acute toxicity, especially when compared with toluene or other high-profile industrial solvents. I’ve seen companies lean on these findings to help gear up safer workspaces. Even so, limited skin and respiratory irritation means personal protective equipment stays mandatory. Animal studies flag minor effects at high doses, but regulatory agencies keep pressing for more research into chronic, low-dose exposure. Carcinogenicity hasn’t shown up as a confirmed risk yet, and environmental persistence comes off as manageable given the compound’s breakdown profile. Still, letting down your guard around chemicals always comes with a price, so ongoing monitoring and updated health standards remain front and center.
Future Prospects
Looking ahead, industry shifts keep spotlighting 3-methoxybutyl acetate as a go-to alternative. Tighter environmental regulations close the door on harsher solvents, and manufacturing keeps chasing options that don’t just work but also carry fewer health risks. Companies investing in green chemistry find this molecule lines up with efforts to drop overall workplace hazards. Markets will keep testing its versatility—from automotive to electronics and pharmaceuticals—looking for broader formulations that bring better performance and safer disposal. I see researchers sharpening efforts to boost efficiency in synthesis, trim waste, and lower lifecycle environmental costs. As pressure mounts to rethink what goes into coatings, cleaners, and adhesives, this acetate gives the next generation of chemists, engineers, and workers plenty of reasons to keep experimenting and learning.
What are the main uses of 3-Methoxybutyl Acetate?
An Overlooked Workhorse in Modern Manufacturing
Years back, I took a summer job in an auto-body paint shop. My boss cared a lot about clean finishes—no streaks, no bubbles. It surprised me how much time we spent mixing solvents instead of paint. That’s where I saw 3-methoxybutyl acetate in action. This solvent isn’t a flashy name, but it plays an important role behind the scenes in coatings, inks, and cleaning fluids.
Coatings and Paints: More than Just Coverage
Most people buy a can of paint and think about color or finish. Manufacturers have to worry about how that paint flows, how it dries, and how it sticks over time. 3-Methoxybutyl acetate gives coatings the right viscosity. It helps paints spread without leaving brush marks and slows the drying just enough to prevent surface defects, which anyone who has spent an afternoon with a roller can appreciate. Without it, high-gloss automotive and industrial paints would streak and ripple.
This solvent handles both water-based and solvent-based systems. Brands often use it in automotive finishes, factory machinery paints, and wood coatings. It lets them design durable products that can be applied in real-world temperatures.
Printing Inks: Keeping the Presses Rolling
Visit any print shop, and there’s a distinct aroma in the air—solvents at work. Ink makers value 3-methoxybutyl acetate because it dissolves pigment and resin evenly. The solvent keeps inks from drying too fast inside high-speed printers, which prevents clogged nozzles and rejects. That means sharper graphics, better color registration, and faster print runs.
Environmental rules keep getting stricter for ink makers. This solvent passes the latest guidelines for low odor and lower toxicity, making it a safer option for workers. There has been a shift in the last decade as companies seek options that balance performance and regulatory pressure. Data from the European Chemicals Agency notes 3-methoxybutyl acetate’s “comparatively low acute toxicity,” which helps employers breathe a little easier—literally and legally.
Industrial Cleaners and Degreasers
Welders, machinists, and mechanics work with greasy tools. Strong cleaning agents cut through the grime, and 3-methoxybutyl acetate shows up in these products. It breaks down oil and dirt quickly and rinses away without leaving an oily residue. That trait matters on electronic components or precision equipment, where leftover cleaner can cause failure down the line.
You won't see ads for this solvent, but any place relying on clean equipment has probably used it. Mixing facilities like its compatibility with surfactants and its ability to act at lower concentrations, which keeps costs down.
Looking Ahead: Better Choices for Industry and Environment
Companies face tough choices these days. They must deliver quality goods and limit impact on workers and the planet. Many older solvents vanished due to health or safety risks, but 3-methoxybutyl acetate has carved out a spot for safer, effective ingredients. Careful use, proper ventilation, and closed-loop manufacturing can further reduce risks from solvent exposure. As chemical engineers look for lower-impact formulas, this solvent stands out for letting industries keep vital processes running without hiking liability or harming air quality.
From paint shop memories to today’s push for cleaner chemistry, it’s clear that small chemicals like 3-methoxybutyl acetate deliver value quietly but consistently. By supporting production with fewer tradeoffs, it helps entire industries make products that last—and workplaces that stay safer.
What is the chemical formula and structure of 3-Methoxybutyl Acetate?
Getting to Know the Formula
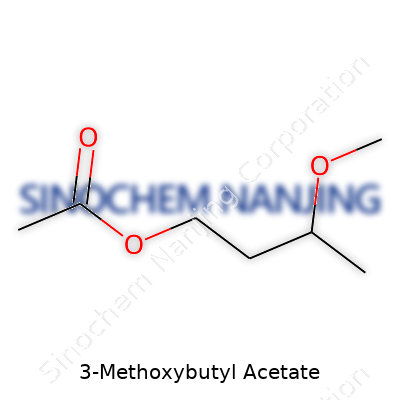
3-Methoxybutyl acetate catches attention because of its chemical backbone and how it blends properties useful for both scientists and regular users. The chemical formula for 3-Methoxybutyl acetate is C7H14O3. At first glance, this formula might just look like a string of letters and numbers, but each part holds meaning about the atoms packed into each molecule. There are seven carbons, fourteen hydrogens, and three oxygens working together to create a solvent valued in both industrial and specialty settings.
The Molecular Structure Explained
Drawing this out, you would see an acetate group (CH3COO–) attached to a 3-methoxybutyl chain. This chain has a methoxy group (-OCH3) stuck on the third carbon of a straight butyl group—so you get a main carbon chain: CH3–CH(OCH3)–CH2–CH2–O–C(O)CH3. This gives the molecule a tail that resists breaking down in standard reactions, granting durability and flexibility to the final blend it joins.
If you are picturing it, think part of the molecule supplies the "grease" so it dissolves other chemicals, while the other end makes it less harsh and more stable than many other esters or acetates used in paints or coatings.
Experience on Why It Matters
In practice, I’ve watched this solvent offer stronger performance in paint applications. Its balanced evaporation rate reduces streaks or visible brush marks, making it a favorite among those who care about the finish quality. Unlike harsh acetates that fill a room with sharp fumes, 3-Methoxybutyl acetate tends to be milder. That seems small, but if you have spent any afternoon in a workshop, it’s clear how much air quality matters for health and comfort.
Beyond paint, this chemical appears in cleaning products or ink formulas. The methoxy group not only affects how it mixes, but also lessens the aggressive edge so it won’t ruin sensitive components. Synthetic chemists appreciate that it plays nicely with a range of resins and binders, which gives more freedom in product design. Manufacturers don’t always broadcast the choice of solvent, but 3-Methoxybutyl acetate appears frequently on safety data sheets for wood varnishes, adhesives, and protective coatings.
Looking at Problems and Realistic Solutions
Not everything about 3-Methoxybutyl acetate shines. Like most organic solvents, it carries some health risks—especially if ventilation falters or skin exposure becomes regular. Regulatory groups in the US and EU have set occupational exposure limits for a reason. From my own time managing inventories, relying on the right protective gear and following local disposal rules stopped most headaches. Correct labeling and staff education made all the difference, preventing slips that could lead to unnecessary contact.
Some chemical engineers push for greener solvents, aiming to cut down on volatile organic compounds (VOCs). While 3-Methoxybutyl acetate isn't the worst offender in its class, it isn’t entirely benign. I’ve began seeing blends with bio-based alternatives or waterborne versions of the same coatings. Progress comes slowly, but sharing technical data between labs and end-users drives better options.
Why Transparency and Skill Matter
The key with specialty chemicals like 3-Methoxybutyl acetate isn’t just about having a new solution—it's about keeping products safe and reliable. Sharing science-based insights across companies, and making safety protocols clear for everyone from entry-level workers to experienced chemists, builds real trust. Labels on drums, supplier safety sheets, and clear training sessions turn complicated risks into manageable tasks. Responsible handling saves money, health, and the reputation of everyone involved.
Is 3-Methoxybutyl Acetate hazardous or toxic?
Everyday Encounters with Industrial Solvents
It’s not every day someone picks up a conversation about chemicals like 3-methoxybutyl acetate at the dinner table. Still, whether in paints, coatings, or cleaning agents, this solvent turns up in more places than most people realize. Coming from years of working near industrial finishing operations and staying curious about what goes into simple things like office paint, I've seen plenty of folks turn a blind eye to safety labels. Risks rarely get the spotlight until an accident or a health scare happens.
The Actual Hazards
3-methoxybutyl acetate shouldn't be shrugged off as just another long chemical word. Breathing in too much can irritate the nose and throat. Eyes might sting or water after a quick splash. Workers who ignore gloves and proper gear can develop headaches, dizziness, or even skin redness after regular handling. Take it from someone who once worked with solvents in a stuffy warehouse: nausea or lightheadedness after poor ventilation doesn’t feel trivial. Chronic exposure in confined spaces ramps up those risks, creating a real health headache for whoever ends up cleaning up the mess.
The EPA lists 3-methoxybutyl acetate as a volatile organic compound. In the world of workplace safety, that definition sets off a few sirens. State regulations and the Occupational Safety and Health Administration (OSHA) classify it as harmful if inhaled or absorbed through the skin. Material Safety Data Sheets (MSDS) warn against careless use. Universities and manufacturers reinforce protective steps, knowing that one misstep could spark an expensive clean-up or land someone in the hospital.
How Much Is Too Much?
Toxicity depends on dose, timing, and protection. According to the National Institute for Occupational Safety and Health (NIOSH), even low levels can trigger irritation or mild symptoms during heavy use. Higher concentrations lend themselves to more severe reactions. Long-term, repeated exposure without serious controls can inch toward more significant health effects. Besides, few workplaces monitor their air quality every hour or enforce gloves and masks during every task. This isn't alarmism—real accidents come from overlooked details.
Steps That Make a Real Difference
Solvent hazards remain real, but practical steps can lower the risk. In factories I’ve visited, a few changes made a night-and-day difference: better ventilation, stronger training for new hires, and supplies of gloves or masks at every station. Supervisors who skip routine checks pay the price with higher sick days or safety violations. Some employers swapped out older solvents for less hazardous alternatives. Schools and DIY spaces post bigger warning signs and enforce rules about handling and disposal. Habit builds safety culture just as much as any poster on the wall.
What to Watch Out For
Anyone handling 3-methoxybutyl acetate should expect more than just a quick fix. Spills can quickly turn an office into a hazardous mess. Getting into the habit of using personal protective equipment (PPE) provides more protection than relying on luck. Read labels, follow safety data sheets, and keep ventilation systems running. Even if regulations do not strictly demand eye-washing stations or spill kits in every room, experienced users will want those tools close by. In short, this solvent deserves respect—and a healthy sense of caution.
Workplaces can create safer spaces by adopting smarter practices. Regulators can help by sharing clear limits and offering updated guidance. End-users who ask questions and follow instructions safeguard not just themselves but everyone sharing their air. In my own experience, small, regular changes shape a workplace you’d actually want to step into every day.
What are the storage and handling recommendations for 3-Methoxybutyl Acetate?
Why Everyone Working with Industrial Chemicals Should Care
Standing in a warehouse lined with chemical drums changes how you think about storage. Chemicals like 3-methoxybutyl acetate aren’t just bottles on a shelf—they call for careful choices every step of the way. One slipup can affect not only the quality of the product but also the safety of everyone around.
Key Storage Conditions
Storing 3-methoxybutyl acetate requires a cool, well-ventilated spot, somewhere away from direct sunlight or heat sources. Heat sneaks up on a storage area and can quickly change the stability of the chemical. Workers have shared stories about chemicals breaking down just because of an unexpected temperature spike—something easily avoided with good site planning.
Keep this solvent in tightly closed containers, using materials compatible with organic acetates. Metal drums tend to work well, but checking for any signs of rust or corrosion matters; leaks bring unwanted risks and headaches. High humidity also causes trouble. Everything stays safer when the area is dry, so moisture barriers or dehumidifiers pay off over time.
Labeling may seem simple, but it often gets overlooked. Mislabeling causes mix-ups that can result in chemical reactions or at least major cleanup jobs. Make each drum or container easy to identify, and use clear hazard signs—nothing slows down a job like guessing what’s in a barrel.
Good Handling Habits
Pouring or transferring 3-methoxybutyl acetate calls for common sense and good tools. A chemical splash in the eye or on the skin is more than just an inconvenience. Wear safety goggles and gloves rated for organic solvents. Cotton, nitrile, or neoprene gloves stand up better than vinyl, which doesn’t always cut it. Long sleeves and chemical-resistant aprons help cover exposed areas.
Spill kits should sit close to workstations. Time and again, I’ve seen quick action keep a bad day from getting worse. Workers should know where the nearest eyewash station and safety shower are—it’s one of those things you want to memorize before you need it. After any spill, ventilate the area well to clear fumes before restarting work.
Static sparks can light up vapors from this solvent. Always ground metal containers during transfer. Plastic containers build up static electricity, so try to limit their use and stick with recommended storage drums.
Minimizing Hazards for Long-Term Safety
Fire risk presents a real concern, even in places with strict precautions. No smoking, no open flames anywhere in the storage or handling areas—signs help, but daily reminders during shift meetings tend to stick better. Keep fire extinguishers accessible, and train workers to use them, not just point them out during an inspection.
Good ventilation keeps harmful vapors from building up. Chemical exposure creeps up on people, and headaches or dizziness often show up without warning. Regular checks of exhaust fans and air quality monitors help spot problems before they turn into emergencies.
Disposal needs attention too. Don’t pour 3-methoxybutyl acetate down the drain or toss it into general waste. Follow local regulations and use licensed disposal services. Back in my early days, the team had to redo a whole storage shed because of old, unmarked chemicals left behind. It cost time and money—proper disposal could have prevented it.
Supporting Safe Workplaces
Every storage decision affects both people and the environment. Reliable storage and well-planned handling practices set the standard for safety in any chemical-dependent industry. With a proactive approach—grounding containers, watching labels, controlling temperature—accidents and long-term damage drop dramatically. Regular reviews, clear policies, and shared experience go a long way in keeping workers and the workplace healthy.
What is the boiling point and solubility of 3-Methoxybutyl Acetate?
Why Boiling Point Matters in Everyday Labs and Factories
3-Methoxybutyl acetate has a boiling point around 154°C. This temperature speaks volumes about its practical uses. In paint shops or ink plants, solvents with lower boiling points may evaporate too quickly, risking inconsistent drying and application. A mid-range boiling point such as this offers a better shot at controlled evaporation—nobody wants paint drying too fast with brush marks everywhere. It also brings some peace of mind when working in spaces without fancy climate controls; the solvent won’t just flash off and vanish, but it won’t linger and slow things down either.
For me, handling chemicals in a small workshop setting, boiling points sit among the first numbers I check on a safety data sheet. Higher boiling solvents stick around to level out the finish, reducing ugly streaks. Still, too high of a boiling point leads to headaches—literally and figuratively—as the fumes can linger, and surfaces remain tacky for hours. 3-Methoxybutyl acetate hits a sweet spot for varnishes and coatings. Not too volatile in summer heat, not stubborn in winter chill.
Solubility: Not Just a Line on a Chart
3-Methoxybutyl acetate dissolves well in water, with a solubility near 26 grams per liter at twenty-five degrees Celsius. For anyone mixing waterborne paints, inks, or cleaning solutions, this number matters. A solvent that blends well with water helps suspend pigments and resins, leading to smoother colors and less separation. In eco-conscious workshops aiming to cut dangerous emissions and clean up with less fuss, water solubility supports safer disposal and faster wash-ups.
I’ve lost count of how many times friends working in small print shops have cursed at stubborn solvents that refuse to leave the rollers. A bit of water compatibility means less hassle, less time scrubbing, and fewer nasty residues on hands or brushes. Large factories save money, too, trimming hazardous waste costs and keeping things safer for workers.
Facts and Practical Solutions
If a product combines a manageable boiling point and moderate water solubility, it usually stands a chance to balance performance with safety. According to the European Chemicals Agency (ECHA), 3-methoxybutyl acetate proves less volatile than classic solvents like ethyl acetate, making its fumes more bearable. OSHA’s safety standards highlight how solvents near this boiling range keep chemical exposure lower in enclosed spaces. Less drifting solvent vapor means safer working air.
On the solubility front, higher water mixing capacity allows for more flexibility in cleaning methods and greener disposal. Environmental Protection Agency (EPA) reports mention that switching to water-miscible solvents can shrink harmful emissions and reduce fire risk. Not every paint or adhesive can get away from heavy-duty solvents, but leaning toward options like 3-methoxybutyl acetate often leads to safer workflows.
Working Smarter, Not Harder
Getting the most out of solvents takes some trial and error in the garage or the factory floor. I’ve found that sticking with chemicals that match practical boiling points and offer genuine water compatibility makes for easier product formulation and safer teamwork. Safety data—paired with hands-on trial—helps dial in a solvent’s fit. If the aim is balance, less-hazardous workspaces, and smooth performance, 3-methoxybutyl acetate earns consideration in paint, coatings, printing, and cleaning jobs. Small improvements in properties often make a big difference over hundreds of gallons or thousands of cans in real-world use.
| Names | |
| Preferred IUPAC name | 3-methoxybutyl ethanoate |
| Other names |
1-Acetoxy-3-methoxybutane 3-Methoxybutyl ethanoate Acetic acid 3-methoxybutyl ester Butanoic acid, 3-methoxy-, 1-acetate |
| Pronunciation | /ˈθriː mɛθ.ɒk.si ˈbjuː.tɪl əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | 4435-53-4 |
| 3D model (JSmol) | `3d:CCCCOC(=O)C` |
| Beilstein Reference | 613873 |
| ChEBI | CHEBI:89643 |
| ChEMBL | CHEMBL3184602 |
| ChemSpider | 173413 |
| DrugBank | DB14670 |
| ECHA InfoCard | 100.108.186 |
| EC Number | 203-576-3 |
| Gmelin Reference | 8119 |
| KEGG | C19644 |
| MeSH | D051642 |
| PubChem CID | 87790 |
| RTECS number | OJ9625000 |
| UNII | XU4LUV7096 |
| UN number | UN number: "UN 3272 |
| CompTox Dashboard (EPA) | DTXSID2022228 |
| Properties | |
| Chemical formula | C7H14O3 |
| Molar mass | 160.21 g/mol |
| Appearance | Colorless liquid |
| Odor | Mild, pleasant |
| Density | 0.940 g/cm³ |
| Solubility in water | 8.5 g/L |
| log P | 1.04 |
| Vapor pressure | 0.38 mmHg (20°C) |
| Acidity (pKa) | 13.05 |
| Basicity (pKb) | -3.5 |
| Magnetic susceptibility (χ) | -52.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.408 |
| Viscosity | 1.0 mPa·s (at 20°C) |
| Dipole moment | 2.92 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 417.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -576.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4787.3 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H336 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P312, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 63 °C |
| Autoignition temperature | 340 °C |
| Explosive limits | 1.2-7.0% (V) |
| Lethal dose or concentration | LD₅₀ (oral, rat): 2400 mg/kg |
| LD50 (median dose) | 1,740 mg/kg (rat, oral) |
| NIOSH | RN 4435 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Methoxybutyl Acetate: 50 ppm (240 mg/m³) |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
2-Methoxybutyl acetate 3-Methoxybutanol Butyl acetate Methoxyacetic acid Ethyl acetate |

