3-Hydroxypropionitrile: Looking Beyond the Lab Bench
Historical Development
The path to understanding 3-Hydroxypropionitrile, a chemical once buried in the back pages of organic chemistry texts, has reflected the changing priorities of science and industry. Early research, mostly tucked away in European chemistry journals, traced syntheses and cataloged reactions but rarely pushed toward practical uses. As industrial chemistry picked up speed in the twentieth century, interest in intermediates like 3-Hydroxypropionitrile slowly grew. I remember digging into older literature for a university project—much of the foundational knowledge arrived in fits and starts, sparked by advances in catalysis, interest in platform chemicals, and the broader quest for sustainable pathways. Chemical companies and university groups have built on this body of research, especially as green chemistry set new goals for efficiency and safety. Even today, much of what we know feels hard-won, the result of experimental persistence and the impulse to find cleaner, smarter chemical routes.
Product Overview
Talking about 3-Hydroxypropionitrile isn’t about listing its catalog number. It’s a grainy, colorless liquid, often tucked away as an intermediate, but there’s nothing minor about its place in organic synthesis. Those with experience in synthesis recognize it as a bridge compound—neither the starting point nor the final destination, but absolutely critical to getting from one to the next. The presence of both a hydroxyl group and a nitrile group in a single, small molecule gives it flexibility. Chemists value chemicals like this because they open up new avenues: a reaction with 3-Hydroxypropionitrile can move a synthesis forward smoothly or unlock more sustainable processes by reducing the number of steps or lowering energy demands. For manufacturers focused on specialty chemicals, these kinds of compounds fill a unique niche.
Physical & Chemical Properties
3-Hydroxypropionitrile melts just below room temperature but gets runny fast when the temperature rises. Its smell hints at the underlying chemical energy. The molecule itself holds significance—compact, with a reactive nitrile at one end and a hydroxyl on the other. Chemists appreciate its solubility in water and common solvents, the modest boiling point, and the potential for hydrogen bonding. These features don’t just show up in the handbook—they matter when you’re putting the molecule to work in a reaction vessel or scaling up to industrial volumes. This combination can mean less investment in exotic equipment, lower reaction temperatures, and fewer headaches if you’re working to minimize unwanted byproducts.
Technical Specifications & Labeling
Working with 3-Hydroxypropionitrile calls for at least a decent level of certainty in composition. Analytical standards require confirmation of purity, though real-world samples almost always carry some trace contamination from unreacted precursors. Suppliers sometimes vary the levels of trace hydrolysis products or remaining starting materials, which can influence yields or require extra purification. As a result, researchers and manufacturers tend to build quality checks right into their protocols. Proper labeling matters—there’s no room for error with a compound that can release toxic fumes or threaten worker safety in enclosed spaces. In many labs, barcoded vials or strict inventory audits pair up with clear hazard symbols and relevant hazard and risk statements.
Preparation Method
The most common approach I’ve seen to preparing 3-Hydroxypropionitrile relies on the reaction of acrylonitrile with water, typically under catalytic conditions. This hydration step, sometimes run under acidic or basic environments, splits the carbon-carbon bond in a predictable way. Modern refinements focus on maximizing yield while limiting the formation of side products. Newer catalytic systems streamline the process, but in my experience, it still takes careful monitoring to prevent over-reaction or product loss through evaporation or hydrolysis. If you’re synthesizing this at scale, managing reaction heat and waste streams becomes as important as the chemistry itself. Projects aimed at using biocatalysts or engineered microorganisms look promising, offering gentler conditions and fewer emissions.
Chemical Reactions & Modifications
With both hydroxyl and nitrile groups, 3-Hydroxypropionitrile supports a wide palette of transformations. Nitriles can be hydrolyzed further to carboxylic acids or amides, while the hydroxyl group can be protected or functionalized to guide more complex syntheses. I remember projects where the molecule acted as a launching pad for building blocks used in pharmaceuticals, thanks to its flexibility. Not all reactions proceed cleanly, though—side reactions such as reversion or oligomerization require a steady hand and strict reaction condition control. In R&D labs, teams often look to adapt this molecule to make it compatible with new synthetic strategies, such as green solvents or continuous flow processing, which can open up new efficiency gains.
Synonyms & Product Names
3-Hydroxypropionitrile goes by several names. Chemists often call it beta-hydroxypropionitrile or 3-hydroxypropanenitrile. CAS number references come up in technical discussions, but most working chemists and engineers recognize it by its standard name. What stands out in real-world usage is the potential for confusion — similar-sounding names and overlapping structural analogs mean label checks matter every time.
Safety & Operational Standards
Anyone who’s handled nitriles in the lab knows the hazards go beyond a printed label. 3-Hydroxypropionitrile shares the typical risks of small, volatile organonitrile compounds, including the potential to off-gas hydrogen cyanide if overheated or mixed with strong acids. Adequate ventilation, local exhaust, and appropriate gloves are non-negotiable. This is where organizations like OSHA step in, setting exposure limits and mandatory training for all handlers. I’ve seen projects stall because management underestimated how quickly small spills could lead to a building evacuation. Industry trends lean toward automated handling and closed systems, reducing contact and keeping emissions in check. The need for rapid response plans, cyanide antidotes, and regular safety audits runs through every level of operation with this compound.
Application Area
Most people never realize how often they cross paths with chemicals like 3-Hydroxypropionitrile in finished products. It has carved out a role in fine chemical manufacturing, acting as an intermediate in the synthesis of pharmaceuticals, acrylics, and specialty polymers. In practice, its reactivity profile lets formulators create new compounds with tailored physical or pharmacological properties. These reactions don’t sit in a vacuum — the chemical finds its way into adhesives, coatings, and advanced materials. Its role as a building block for vitamin B derivatives and potential role in bio-based plastics gives it added appeal in a world shifting away from oil-based inputs. Each application brings a new slate of challenges around purity, regulatory compliance, and minimization of residual monomers, especially if the final product enters the medical or food supply chain.
Research & Development
R&D teams working with 3-Hydroxypropionitrile aren’t just interested in pushing yields—they’re out to transform how platform chemicals contribute to a more resilient supply chain. Efforts focus on cleaner preparation methods, especially routes that cut hazardous byproducts or tap into renewable raw materials. My conversations with colleagues often circle back to process optimization: automated controls that reduce human error, analytical tools that provide instant readouts, and scalable processes that behave just as expected in pilot plants as in the lab. Projects pushing biocatalysis or enzyme-driven synthesis hold promise, particularly when companies seek non-petrochemical feedstocks. Collaboration between universities, startups, and the chemical industry helps keep the pace steady, ensuring new research doesn’t get locked behind academic paywalls.
Toxicity Research
Toxicologists treat 3-Hydroxypropionitrile with respect. The compound carries acute toxicity risks, owing to the reactivity of the nitrile group and the metabolic potential to release cyanide in living organisms. Published studies show the importance of strict exposure limits—short-term contact can irritate skin and mucous membranes, and inhalation or ingestion carries real danger. Chronic exposure effects appear less studied, something that could draw more attention as production scales up for broader industrial use. Any claims of “safer” derivatives or green processes need real-world data and peer-reviewed toxicology, not just marketing boosts.
Future Prospects
Demand for 3-Hydroxypropionitrile could rise sharply, especially if biobased plastics, new adhesives, or pharmaceutical candidates enter mass production. Regulatory pressure on hazardous waste and carbon footprints forces companies and researchers to rethink every step, from raw material sourcing through distribution. Advances in process engineering—continuous flow reactors, recyclable catalysts, robust waste treatment—could cut costs and risks. Researchers also see potential in collaborating across industries, such as using agricultural byproducts or CO2-derived materials as inputs. A more complete toxicological profile would help guide new safety regulations and training programs. As the role of specialty intermediates expands in modern manufacturing, close ties between research institutions, producers, and regulatory agencies will shape the story of 3-Hydroxypropionitrile for years to come.
What is 3-Hydroxypropionitrile used for?
Where 3-Hydroxypropionitrile Shows Up in Real Life
Most people never hear about 3-hydroxypropionitrile outside a lab or a chemical catalog. This small molecule pops up in several places that matter a lot. Its structure allows it to serve as a building block for products most of us rely on every day, although we never see it mentioned on a label at the store.
I once toured a factory producing acrylamide, which shows up in plastics, papers, and even sewage treatment. The folks there talked about the chemistry like it was an art form, but beneath it all were these small, functional molecules doing the heavy lifting in the manufacturing chain. That’s where 3-hydroxypropionitrile comes in — not glamorous, but vital.
Paints, Plastics, and Medicine
This compound starts by supporting the synthesis of acrylic acid. Acrylic acid shapes up as a big deal in producing resins, adhesives, and paints. Think of how many surfaces at home or in the office count on coatings and finishes that last. The durability comes from molecules like this one working behind the scenes.
Moving further, researchers use 3-hydroxypropionitrile to build intermediates for pharmaceuticals. Some of these medicines anchor treatments for infections or metabolic diseases. There’s no mystery as to why science leans on simple molecules; they let drug designers piece together more complex structures with consistency.
Concerns and Risks Around 3-Hydroxypropionitrile
Every time I visit a chemical plant, safety stands front and center. A compound that can spark new plastics can also bring hazards. For 3-hydroxypropionitrile, toxicity hits animal nervous systems hard, leading to questions about worker protection and environmental exposure. The folks I’ve interviewed in occupational health remind me how quickly things can head south without careful controls, from ventilated labs to good storage practices.
Accidental release into waterways presents another issue. Fish and small aquatic life prove sensitive to nitrile-based chemicals. Researchers track water quality down to trace amounts for a reason. Safeguarding against leaks matters as much as handling in the first place, and strict disposal protocols exist for a good reason.
What Solutions Actually Matter?
Here’s where the rubber meets the road: Proper training, strong oversight, and up-to-date safety systems make a real difference. Investing in closed systems pays off. One engineer told me their plant cut exposure rates in half by redesigning extraction processes with tight containment. These solutions come from those who work close to the danger, and they stay motivated by real-life safety wins rather than memos from faraway offices.
Policymakers usually aim safety standards at the highest-risk chemicals, but plenty of everyday molecules bring hidden threats. Adding stricter thresholds for occupational exposure and tighter storage regulations can help. Transparent reporting and access to public records help keep companies on their toes. At the same time, investing in greener alternatives may shift the industry away from the most hazardous compounds over time.
The Takeaway
3-Hydroxypropionitrile isn’t famous, but its reach stretches from plastic wrappers to pills on the pharmacy shelf. The work to keep things safe falls on both the companies that use it and the regulators who keep score. Behind every everyday product lies a world of hidden chemistry, and it’s always worth paying attention to what goes on inside that world.
What is the molecular formula and structure of 3-Hydroxypropionitrile?
Peeling Back the Layers: What’s in the Name?
The name 3-hydroxypropionitrile might sound intimidating, but its core idea is simple. This compound comes together with a three-carbon skeleton and mixes the world of alcohols and nitriles. Its molecular formula sits as C3H5NO. That means three carbon atoms, five hydrogens, one nitrogen, and one oxygen. The “hydroxy” part means there’s an -OH group, and the “nitrile” sings about a -C≡N group. The arrangement of these groups across three carbons shapes how it behaves and where it can be useful.
A Closer Look at Structure
If you draw it out, you start with a three-carbon chain – not too different from how simple kitchen ingredients begin. At one end (carbon one), you’ve got the nitrile group—an unyielding triple bond between a carbon and a nitrogen atom. The other end (carbon three) has the hydroxy group (-OH), which is the same motif found in familiar alcohols. Carbon two sits quietly in the middle, keeping both groups apart. Stack the atoms in order: HO–CH2–CH2–C≡N. This split personality—alcohol on one side, nitrile on the other—creates opportunities for reactors and chemists.
Importance Away from the Lab Bench
Many folks might never hear about 3-hydroxypropionitrile outside of chemistry circles. Still, it plays a subtle background role in industry and research. Its structure opens doors for making pharmaceuticals, fine chemicals, or even certain specialty plastics. That hydroxy-nitrile split means it reacts in ways other molecules can’t. I remember in a college lab, we searched for starter molecules with both reactivity and flexibility; 3-hydroxypropionitrile stood out as a tool for building larger, more complex structures. Its compact design and clear functional groups let chemists steer reactions toward useful products.
Real-World Impact and Challenges
One tough thing about molecules like this: Nitriles can be tricky to handle, sometimes toxic, and need careful attention in both storage and use. Lab safety routines always covered proper handling with gloves and good ventilation. Mistakes happen quickly with potent nitriles. On the other hand, the -OH group makes it easier to dissolve in water and manipulate in common lab environments, unlike many organic compounds that run away from water. This balance speeds up experimentation and streamlines purification steps.
Environmental responsibility asks another question: How do you manage wastes? Chemists have pushed greener approaches, shifting toward biosynthetic routes. Some recent studies use enzymes to make 3-hydroxypropionitrile, lowering the risk of hazardous byproducts. These greener processes not only reduce risk to people but also leave a smaller footprint on waterways and soil—something every responsible chemical business has to take seriously today.
Finding Progress One Molecule at a Time
It’s easy to overlook how basic molecules like 3-hydroxypropionitrile shape innovation outside of textbooks. Better routes for synthesis, safer handling procedures, and greener chemistry all start from understanding its formula and structure. Investing time in thoughtful research pays off by reducing hazards, improving product quality, and delivering benefits downstream, whether for a new pill, a cleaner material, or safer industrial chemistry.
Is 3-Hydroxypropionitrile hazardous or toxic?
What Happens Around 3-Hydroxypropionitrile
3-Hydroxypropionitrile, known in some labs as 3-HPN, doesn’t show up in headlines often, but it definitely earns a spot on the list of chemicals that raise concern. Scientists come across it mostly in the context of organic synthesis, polymer science, or when tracing metabolic byproducts in certain fungi and bacteria. Despite its niche reputation, it deserves close attention.
Personal Experience Meets Real Risk
Anyone who’s spent time in a chemistry lab learns to respect nitriles. I remember the clear rule hammered into us: respect these bottles, wear gloves, work in a hood. With 3-Hydroxypropionitrile, the danger isn’t hidden. Just smelling it is risky — it gives off a sharp odor, a clear warning signal.
Data backs up those first impressions. Run through the toxicity information from the European Chemicals Agency and you’ll see danger signs flagged all over its dossier. Acute toxicity ranks high, meaning just one misstep can put your health in jeopardy. Lab tests say a few milligrams per kilogram can harm mammals; it irritates the skin, blows up your gut if swallowed, and brings on symptoms you really don’t want to experience. One of its nastier tricks is how it disrupts the body by releasing cyanide, which undercuts the ability for cells to use oxygen.
Balancing Industrial Use and Human Health
No doubt, chemistry gives us raw ingredients for new medicines, plastics, and even food science. But the rush to innovate can bulldoze safety if nobody’s watching. It helps to remember how chemicals like 3-Hydroxypropionitrile find their way out of test tubes: accidental spills, poorly ventilated spaces, or careless disposal. That last scenario keeps me up at night. Once dumped in water or soil, its breakdown products still pose risks, threatening fish and invertebrates long after we forget about it.
Laws, Labels, and Real-World Steps
Safety labels and official warnings matter only if people actually read and follow them. Too often, manufacturers skip detailed training, leaving workers to learn the hard way. Even in university settings, accidents happen because tired students rush through clean-up or ignore glove tears. This leaves a preventable trail of harm.
Government agencies already require hazard labels and spill procedures, but plenty of small suppliers or workshops fly under the radar. Tighter controls, regular training, and reliable monitoring really do work. Standard safety goggles and nitrile gloves shield workers, and a working fume hood drops exposure risks sharply. Spill kits near every workbench, combined with regular disposal checks, cut down on long-term exposure for both people and the wider environment.
Responsible Choices for the Future
Companies and research groups can find alternatives or refine their processes to use less hazardous chemicals where possible. Chemical users who insist on high safety standards raise the bar for everyone else, pushing the industry away from old habits. Environmental scientists urge investment in real-time sensors for air and water, alerting us to leaks early. Public sharing of incident records (without fear of backlash) fosters a culture of openness and constant improvement.
Chemistry does amazing things for society, but its benefits fall apart the moment health and environment take a back seat. 3-Hydroxypropionitrile isn’t just a harmless tool; it demands respect and smart procedures. Taking its risks seriously means less damage, fewer regrets, and a future where science and safety actually go hand in hand.
How should 3-Hydroxypropionitrile be stored and handled safely?
The Risk: More Than Just Science
3-Hydroxypropionitrile isn’t something you want roaming free in a storage closet or handled like ordinary solvents. Anyone who’s spent even a little time near a chemical lab knows that compounds like this bring two big worries: toxic vapors and the threat of accidental spills. Companies dealing with it aren’t just ticking off boxes for a safety audit; they’re protecting workers from lasting health problems.
Understanding What We're Dealing With
The chemical is volatile and often releases fumes that can irritate the eyes and respiratory tract. Plenty of us have read stories—sometimes shared in hushed tones in the break room—about the havoc inhaled nitriles can wreak. Direct contact could burn your skin or eyes, and inhaling a concentrated dose won’t just give you a cough for the afternoon.
Storing 3-Hydroxypropionitrile: No Room for Corners Cut
Anyone storing this chemical should use tightly sealed, clearly labeled containers made of materials that don’t react with it. I’ve seen more than one lab skip proper labeling, only to spend hours scrubbing down benches after a spill. Keeping containers in a cool, well-ventilated space far from direct sunlight and heat sources pays off. No budget shortcut makes up for the cost—financial and human—of an accidental release.
A separate, dedicated storage cabinet makes a big difference, especially in larger labs or warehouses. It shouldn’t sit next to acids, bases, or oxidizing agents. Mixing chemicals leads to surprise reactions, sometimes violent ones, and nobody wants to explain a preventable incident during an inspection or, worse, in the hospital.
Personal Safety: More Than Gloves and Goggles
Protective gloves and splash-proof goggles form the bare minimum line of defense. In my own lab days, I learned that nitrile gloves give a much better shield than latex here—chemicals seep through latex quicker. Lab coats prevent accidental splashes from soaking through to regular clothes, but it’s also smart to use fume hoods or local exhaust ventilation every time this chemical comes out. Chemical-proof aprons and face shields become necessary when pouring or transferring larger volumes.
Clean air matters. It’s not enough to open a window—good labs invest in a working fume hood because ordinary fans just push vapors around. Even a brief slip-up without a fume hood left my old supervisor coughing for hours, proving ventilation isn’t negotiable.
What To Do If Things Go Wrong
Anyone around this chemical should know exactly where spill kits, eyewash stations, and emergency showers stand. I’ve seen panic in a colleague’s eyes during a sudden spill, and clear training turned chaos into calm cleanup. Spill kits for 3-hydroxypropionitrile include absorbent materials and neutralizers that don’t react dangerously, and everyone nearby should know how to use them before the day ever goes sideways.
What Makes a Safe Workplace
A culture that prioritizes practical training, clear labels, regular safety checks, and open reporting reduces accidents. Relying on luck runs out fast in a world full of hazardous chemicals. Smart storage, protective gear, and true teamwork offer the best shot at coming home unharmed—that’s something every lab hand or facility manager can agree matters most.
Where can I purchase 3-Hydroxypropionitrile and what are its typical applications?
Where to Find 3-Hydroxypropionitrile
Anyone looking for 3-Hydroxypropionitrile will run into some hurdles beyond just finding a supplier. This isn’t something you buy like aspirin at a pharmacy. Instead, actual chemical vendors serve biotechnology firms, pharmaceutical labs, and materials science groups—think names like Sigma-Aldrich, TCI, or Alfa Aesar. These companies require a business account and clear documentation just to discuss a purchase. Access often gets limited to qualified researchers who agree to safety standards and regulatory checks.
Ordering goes beyond money and a shipping address. Suppliers demand proof of legitimate research need or industrial application, sometimes down to the project’s grant number or publication plans. This kind of gatekeeping helps keep hazardous chemicals out of the wrong hands. It isn’t just red tape. Chemicals like 3-Hydroxypropionitrile pose real risks if mishandled—call it the price of safeguarding health and security.
What Sets This Chemical Apart
3-Hydroxypropionitrile draws interest because of its unique makeup. This molecule, also called 3-HPN, contains both a nitrile group and a hydroxyl group, which opens up a range of chemical possibilities. I remember working on a project in graduate school where the starting molecules always dictated which products we could make. Once you have both a nitrile and a hydroxyl, the synthetic pathways multiply fast—one such possibility is in making acrylamide derivatives.
How Industries Actually Use It
Pharmaceutical researchers turn to this chemical for building more complicated molecules. They rely on its structure to create precursor compounds for active pharmaceutical ingredients. The presence of both reactive groups allows chemists to add on or swap out pieces of a molecule, enabling the design of new drugs from the ground up.
Monomer production ranks among the more industrial-scale uses. Some plastics and polymers get their properties from a carefully selected starting point like 3-Hydroxypropionitrile. These materials often end up in coatings, adhesives, or specialty packaging. The unique chemical structure allows for manipulation during polymerization, giving industry control over toughness, flexibility, or solubility. And while this might sound like niche chemistry, the results often touch everyday life—think flexible medical tubing or scratch-resistant films.
Risks and Responsible Use
Chemicals with reactive groups, including nitriles, need extra caution. Regulatory bodies, including OSHA and the EPA, set limits for employee exposure and waste disposal. In my own lab experience, these restrictions weren’t just theory—safety officers checked our logs weekly, and we kept cyanide antidote kits on hand, reserved for worst-case scenarios. Government agencies treat some nitriles as hazardous, and the rules for storing and transporting them reflect those risks.
Information gaps exist, especially for smaller operations jumping into specialty chemical sourcing. Not every lab has a dedicated safety officer, nor do all researchers keep up with regulations or disposal rules. I’ve seen plenty of confusion over these rules, ranging from import permit confusion to disposal mistakes. Better training and supplier vetting offer a way forward. Suppliers with technical support staff can walk new buyers through legal and safe handling, reducing the odds of accidents or regulatory trouble.
Paths Toward Safer, Smarter Chemistry
As long as strong regulations, supplier vetting, and staff training remain in place, chemicals like 3-Hydroxypropionitrile will stay available to those who need them for discovery and invention. Keeping everything aboveboard isn’t about gold-plating bureaucracy—it’s what stands between innovative science and the hazards sometimes hiding in a bottle marked with a simple formula.
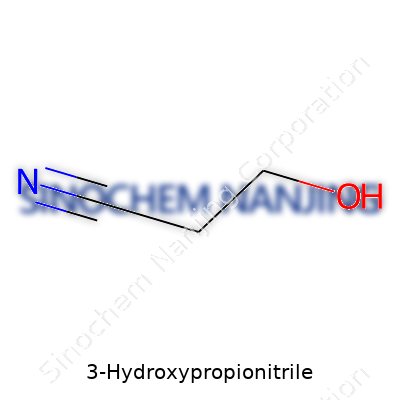
| Names | |
| Preferred IUPAC name | 3-hydroxypropanenitrile |
| Other names |
Acrylic nitrile alcohol 3-Hydroxypropanenitrile β-Hydroxypropionitrile Hydroxyacetonitrile 3-HPN |
| Pronunciation | /ˌθriː-haɪˌdrɒksi-prəˈpɒnɪtraɪl/ |
| Identifiers | |
| CAS Number | “627-16-9” |
| 3D model (JSmol) | ``` 3D Structure;06384;3-Hydroxypropionitrile;CC(O)C#N ``` |
| Beilstein Reference | 1209226 |
| ChEBI | CHEBI:43060 |
| ChEMBL | CHEMBL16516 |
| ChemSpider | 14374 |
| DrugBank | DB03766 |
| ECHA InfoCard | ECHA InfoCard: 100.003.159 |
| EC Number | 200-876-6 |
| Gmelin Reference | Gmelin Reference: 83681 |
| KEGG | C00789 |
| MeSH | D011984 |
| PubChem CID | 7926 |
| RTECS number | UC7350000 |
| UNII | U2371G7TJ1 |
| UN number | UN number: "UN 2647 |
| CompTox Dashboard (EPA) | DTXSID3049244 |
| Properties | |
| Chemical formula | C3H5NO |
| Molar mass | 71.08 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | almond-like |
| Density | 1.026 g/mL at 25 °C (lit.) |
| Solubility in water | miscible |
| log P | -0.64 |
| Vapor pressure | 0.67 mmHg (at 25 °C) |
| Acidity (pKa) | 14.06 |
| Basicity (pKb) | pKb = 4.06 |
| Magnetic susceptibility (χ) | -34.4·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.363 |
| Viscosity | 0.848 cP (25°C) |
| Dipole moment | 3.58 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 105.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -78.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -391.8 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS07 |
| Pictograms | GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H312, H332, H319 |
| Precautionary statements | P210, P261, P280, P303+P361+P353, P304+P340, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 108°C |
| Autoignition temperature | 444°C |
| Explosive limits | 4.3%–20% |
| Lethal dose or concentration | LD50 oral rat 119 mg/kg |
| LD50 (median dose) | LD50 (median dose): 118 mg/kg (rat, oral) |
| NIOSH | RN0159400 |
| PEL (Permissible) | PEL: No OSHA PEL listed |
| REL (Recommended) | REL: 3.5 mg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Acetonitrile Acrylonitrile Propionitrile 3-Hydroxypropionic acid |

