3-Heptanone: History, Challenges, and the Road Ahead
Historical Development
Long before 3-Heptanone ever found its way into today’s industrial or research labs, curious chemists stumbled across it in the slow, methodical search for patterns in organic molecules. Back in the days when organic chemistry hinged on smell and flame tests more than GC-MS machines, folks learned to recognize it by the subtle banana or fruity scent it gives off. Over the decades, as analytical tools evolved, those faint hints of identification grew into solid knowledge about 3-Heptanone’s structure and possible behavior. Each improvement in glassware or detection technology helped pin down just how this molecule fits into the web of family structures called aliphatic ketones. By recognizing its natural presence in some foods and connecting that with precise lab synthesis, the gap between raw nature and industrial production narrowed, letting specialists harness it in controlled settings.
Product Overview
At its core, 3-Heptanone is an aliphatic ketone: basically, a seven-carbon backbone with the carbonyl group stamped on carbon number three. Its chemical formula, C7H14O, can sound intimidating unless you’ve mucked around with similar molecules—then it just looks like an unassuming cousin to more popular ketones. It tends to show up as a colorless liquid with a subtly sweet, fruity odor, something that hints at its occasional cameo in natural flavors and fragrances.
Physical & Chemical Properties
Folks working in the lab know that properties aren’t just numbers on a sheet. For 3-Heptanone, boiling at around 146°C puts it squarely in the range where standard glassware handles it without drama. Its vapor pressure sits high enough to make it noticeable, but not so volatile that you lose half your batch on a warm afternoon. The compound’s solubility tips toward nonpolar solvents, so water isn’t much of a friend here. As with most straight-chain ketones, it won’t surprise anyone with its reactivity, but neglecting proper storage means it’ll seep its peculiar smell through a room faster than one expects.
Technical Specifications & Labeling
Storage rules come not from bureaucracy but from chemistry’s hard lessons. 3-Heptanone needs a tight-sealed container, shielded from direct sunlight, and cool, dry conditions. Properly labeled drums or bottles spell out the chemical’s identity, concentration, and the relevant safety symbols, alerting handlers to its flammability and the need for basic PPE like gloves and goggles. Regulatory frameworks in most countries classify it as a flammable liquid. That’s more than belt-and-suspenders; it prevents costly accidents and long, complicated insurance claims.
Preparation Method
Making 3-Heptanone never felt quite like baking cookies, but the reactions draw from the same logic: start with the right ingredients, know your chemistry, mind the conditions. One practical route uses oxidation, starting from 3-Heptanol or through cleavage of more complex precursors—usually in the presence of controlled oxidizing agents that won’t chew up half your product. The process often benefits from reflux setups, using mild catalysts to keep side reactions in check. For those who’ve done their share of organic syntheses, the protocol lines up with other mild ketone formations, but the choice of starting alcohols or longer-chain hydrocarbons can make a world of difference in yield and cleanliness.
Chemical Reactions & Modifications
3-Heptanone stands ready for a handful of classic organic reactions. It undergoes nucleophilic addition at the carbonyl, just as textbook chemistry promises. Reductions deliver back the corresponding alcohol, while more robust conditions allow it to serve as a decent substrate in condensation reactions. Acetals form with the right alcohols and acids, and halogenation goes smoothly if someone aims for further derivatization. As with so many mid-weight ketones, it delivers decent, reliable behavior in both analytical and synthetic contexts.
Synonyms & Product Names
Anyone working in chemical supply or industry recognizes 3-Heptanone by a slew of names, depending on tradition or region. “Heptan-3-one” pops up on many technical labels; “ethyl butyl ketone” or “propyl propyl ketone” show up in older literature. Even within the same supply house, documentation varies, so cross-checking synonyms keeps confusion low and experiment results clear.
Safety & Operational Standards
Years spent in the lab teach patience about safety. 3-Heptanone’s flammability brings an extra level of attention, especially when working around open heat sources or in poorly ventilated spaces. Splashing, spills, and vapors can irritate eyes and skin, maintaining the need for gloves, eye protection, and proper fume hoods. Fire extinguishers—class B rated—always stand close when handling even moderate amounts. Ventilation becomes not just a recommendation but a requirement, as vapor accumulation poses both health and combustion hazards.
Application Area
In a world obsessed with technical advances, it’s often flavors and fragrances that quietly drive demand for 3-Heptanone. Food scientists bring it on board to recreate subtle fruit or cheese notes; fragrance developers craft it into perfumes and colognes. Outside the flavor house, its predictable reactivity makes it valuable as a starting material or solvent in organic synthesis, and specialty lab work taps its properties for chromatography and chemical separations. While not a blockbuster chemical, it earns its keep across several sectors that thrive on precision and reliability.
Research & Development
Efforts to broaden the uses or streamline the production of 3-Heptanone keep drawing researchers back. By tweaking catalysts and fine-tuning starting substrates, chemists look to raise yields and lower environmental impact. Biotechnological approaches also enter the conversation, as engineered yeast or bacteria offer greener, potentially cheaper synthesis, shifting production from petrochemical routes to bio-based platforms. Analytical chemists continue exploring its behavior, both as a standard and a matrix in developing new detection methods, which has ripple effects for quality assurance in food and pharmaceuticals.
Toxicity Research
Toxicologists turn a skeptical eye toward any industrial compound, and 3-Heptanone is no exception. Animal studies show that, at moderate inhalation or ingestion levels, it leads to mild irritation but not the systemic organ damage seen with some related solvents. Long-term, high-dose exposures have raised concerns over central nervous system effects—headaches, dizziness, and nausea in occupational settings remain well-documented. Occupational limits set by regulatory bodies reflect these results, and chronic exposure guidelines keep lab and factory floor staff from crossing into dangerous territory.
Future Prospects
The next decade stands to see change in how 3-Heptanone is produced, handled, and valued. Green chemistry methods will likely replace some traditional oxidation routes, prompted by both cost and sustainability mandates. Industry insiders keep one eye on flavor trends and the other on regulatory shifts, knowing consumer and environmental pressures don’t wait for consensus. Biomedical research could open unforeseen uses, as the molecule’s moderate volatility and structural similarity to some pheromones spark curiosity. Continued research on toxicity ensures that safety standards adapt to new findings, and as emerging applications in biomimicry or synthetic biology bloom, folks in the lab will keep adjusting protocols and standards with each new breakthrough.
What is 3-Heptanone used for?
What Is 3-Heptanone?
3-Heptanone stands out as a colorless liquid with a pleasant scent, reminding me of waxy fruit candies or mild cheese. Chemists know it by its formula C7H14O. Often, people don’t realize how chemical compounds shape daily experiences, even if the name sounds far from home. Like the lingering taste of artificial banana, 3-Heptanone hides behind familiar flavors and smells.
Perfume and Flavors: The Outspoken Role
Take a look inside perfume bottles or flavored snacks, and you’ll spot clever chemistry. 3-Heptanone has a fruity, slightly nutty aroma, making it a go-to choice for perfumers searching for softness and nuance. Its subtle scent doesn’t overpower; it adds a little roundness or brightness to a mix. Food makers tap into its profile as well. It pops up as a flavoring agent in products trying to mimic or boost fruit notes, especially in sweets and baked treats. Sometimes, it even offers an extra layer to cheese-flavored snacks.
Industrial Uses: Beyond Scents and Flavors
Factories need solvents that dissolve grease, resins, oils, and other sticky problems. 3-Heptanone proves useful here. Its chemical structure gives it the right touch—strong enough to dissolve oily messes, gentle enough for delicate processes. Paints and cleaners benefit from its versatility, getting smoother finishes or better cleaning power. Printing inks also draw from it, as its volatility helps inks dry more cleanly, leaving crisp lines on everything from packaging to magazines.
Potential in Pharmaceuticals
Another pocket of interest sits in pharmaceutical research. 3-Heptanone’s structure is handy as a base for making other complex molecules. Lab workers see it as a building block in the design of pain relievers and antimicrobial compounds. While it doesn’t act alone in medicine, its presence speeds up or smooths out crucial steps along the way. Innovation in this field never comes easy, so every reliable starting point gets credit.
Safety and Environmental Notes
Experience shows there’s no good reason for anyone to splash 3-Heptanone around the house. It gives off fumes that, if inhaled too much, irritate the nose and eyes. Gloves and proper ventilation solve those problems at work. Regulatory systems in many countries set strict exposure limits, protecting workers and the environment. Waste treatment at factories breaks it down safely, reducing risk to rivers or farmland. There’s a shared responsibility here—chemical makers, regulators, and everyday folks benefit when everyone insists on strict safety standards.
Room for Improvement
One big challenge comes from making this compound efficiently and cleanly. Some manufacturers use petroleum routes, which tie production to fossil fuels and greenhouse gas emissions. Green chemistry points the way forward, looking for bio-based sources or clever recycling within factories. Scientists worldwide are racing to develop new synthesis methods with fewer byproducts and lower energy use. Every improvement counts, since even small molecules like 3-Heptanone show up across so many industries.
What is the chemical formula of 3-Heptanone?
Understanding 3-Heptanone’s Makeup
The landscape of organic chemistry throws out plenty of formulas, but 3-heptanone carries its own straightforward fingerprint: C7H14O. Seven carbons, fourteen hydrogens, and a single oxygen atom tie together to make this molecule. At its core, 3-heptanone is part of the ketone family, which plays a supporting role in everything from flavorings to lab research. The “3” points right to where the ketone group shows up—a carbonyl planted on the third carbon of the chain.
The Workhorse in Everyday Chemistry
Walk through a food science facility or a fragrance lab and you’ll spot why these formulas matter. Nobody gets fired up about chemical formulas during dinner, but 3-heptanone goes behind the scenes in gingerbread, blue cheese, and perfumes to give them their bite or distinct aroma. This molecule also pops up in solvents for fats, oils, and waxes. So, understanding its structure isn’t just for science textbooks—it links to things people use, eat, and smell every day.
Why Knowing the Formula Matters
Every chemical formula tells a story about reactivity, safety, and even human health. The combination in C7H14O steers how 3-heptanone interacts in the body and the environment. This ketone passes through the liver and can also show up in breath, linked to fat metabolism—especially in people following ketogenic diets or experiencing certain health conditions. That forensic angle drives researchers to know its composition inside and out.
If someone mishandles a bottle in the lab, that oxygen atom means volatility—3-heptanone can evaporate quickly, leading to exposure. People working with this ketone need clear facts for safe handling. According to toxicology data, it isn’t acutely toxic at low levels, but breathing in too much can bring on dizziness or irritation. With this knowledge, safety guidelines encourage good ventilation and minimized direct contact.
Balancing Industry, Safety, and Research
Regulators and manufacturers expect clarity about each ingredient they use. Proper labeling with molecular formulas gives transparency to downstream users, whether pharmaceutical companies, food technologists, or cleaning product designers. When regulations on volatile organic compounds (VOCs) or food additives change, knowing exactly what sits in the bottle keeps companies ahead of compliance headaches. Traceability comes from accurate chemical formulas, not guesswork.
Having handled a few esters and ketones in my own lab days, the temptation sometimes rises to cut corners on documentation or safety data. A missing oxygen or swapped number in C7H14O means working with the wrong substance or, worse, risking an unwanted reaction. Sharing clear data with coworkers and students saved us more than once, from quality control issues or accidental exposure.
Room for Improvement and Cleaner Science
Looking at the environmental side, 3-heptanone brings up big questions. As industries aim for greener chemistry, monitoring where molecules like this end up matters. Labs continue to research bio-based pathways to synthesize it, cutting out petrochemicals altogether. Keeping formulas transparent enables smarter design—engineers and chemists pick the right molecules for safer, cleaner products.
Everything comes back to the basics: knowing the chemical formula and sharing it clearly. With C7H14O, it’s not just about memorizing a set of atoms, but ensuring human health, sustainability, and trustworthy research stay at the front of the line.
Is 3-Heptanone hazardous or toxic?
Getting Familiar with 3-Heptanone
3-Heptanone pops up in chemical supply catalogs and research papers every now and then. Known as a simple ketone, people use it for making fragrances, flavoring agents, and sometimes in lab work. Its smell is sweet and fruity—some say it reminds them of raspberries or apricots. The tricky part with chemicals like this? Even a fruity smell can hide risk if you don’t know what’s inside the bottle.
Questions About Safety
Hazard isn’t always stamped in bright red letters. You have to dig through data sheets and toxicology reports to get the full story. For 3-Heptanone, official documents like the Safety Data Sheet (SDS) get straight to the facts. If you get it on your skin or in your eyes, irritation can happen. Inhaling the vapor isn’t pleasant either—dizziness or headaches might follow in a space without good air flow. Anyone who has worked with solvents or volatile chemicals for a few years knows that even mild side effects add up if ignored.
Diving Into the Facts
So far, no evidence links 3-Heptanone directly to cancer or life-threatening organ damage. Still, the U.S. National Library of Medicine highlights short-term toxicity. Exposure above recommended limits often makes people feel lightheaded or sick to their stomachs. If you get a face full of it, your eyes might sting, or your nose could start running. Research in laboratory animals shows low acute toxicity, but “low” in the world of chemicals doesn’t mean “harmless.”
Recent workplace studies and real-world accidents remind us of another lesson: Even chemicals considered “low hazard” on paper become dangerous through careless handling or leaks in storage. I remember a chemist in a university lab forgetting about ventilation. The room filled up with fumes from a similar solvent, and cleanup turned into a long day with everyone coughing. It’s not just a matter of reading the label—the environment, equipment, and training all matter.
Protecting Ourselves and Others
Personal experience and workplace rules have taught me the basics: open windows, wear gloves, keep goggles handy. With 3-Heptanone, these simple steps work well, since it evaporates easily and irritates on contact. Factories and labs use air filters, emergency showers, and fume hoods, but even in smaller spaces, running a fan or keeping containers closed makes a difference.
Some people might wonder about the risk if 3-Heptanone gets into food or the environment. The FDA allows tiny amounts of synthetic flavors, including 3-Heptanone, in snacks, candies, and beverages. The World Health Organization and FDA both review studies before giving such approvals. The amounts used in food are much lower than in a manufacturer’s drum or a laboratory flask, so relative safety improves with small doses. Still, spills near water sources or large-scale factory leaks would create different problems, potentially harming wildlife or fish.
What the Future Needs
Every year, researchers dig deeper into cases of repeated exposure and long-term risks. Smarter labeling, regular training, and better record-keeping on chemical use provide real-world safeguards. Reaching out to local poison control or occupational health bodies always makes sense after exposure—better a false alarm than a missed risk. Responsible production and thoughtful use keep workplace hazards at bay, and spreading awareness through open conversation helps keep both workers and communities safer.
How should 3-Heptanone be stored?
Understanding 3-Heptanone’s Personality
3-Heptanone, a clear liquid with a sweet, fruity scent, often makes its way into labs and production facilities. Like other ketones, this chemical doesn’t play well with heat, flames, or sunlight. Getting careless around it invites trouble, and ignoring best practices often ends in spills, or worse, health problems. Breathing its vapors brings on headaches or nausea, and skin exposure leaves irritation. I’ve seen more than one lab tech regret skipping out on careful storage, only to learn the hard way.
Pick the Right Container
Glass bottles with tight, non-reactive lids work well. Avoid plastic if possible, since ketones have a bad habit of eating away at some polymers, turning what seemed safe into a leaky mess. Steel with a good seal also does fine—just don’t forget to label everything clearly. Overlook that label, and sooner or later, someone grabs the wrong bottle.
Control Temperature and Light
Warm rooms or spots near equipment pump up the risk—vapors build up, increasing pressure in containers and making accidents much more likely. Always stash 3-Heptanone in cool, dry spots, far from direct sunlight. Dedicated chemical cabinets, especially those that ventilate well, bring peace of mind. Storing this compound at room temperature makes sense, but chasing after efficiency sometimes leads people to stick chemicals in unplanned places. Those corner shelves near windows or radiators quickly become hazards.
Keep Away from Incompatible Substances
I once watched a small spill of 3-Heptanone mix with oxidizers on a bench and cause a nasty reaction. Strong acids, alkalis, and oxidizers need separate homes. Even if storage space runs low, crowding different chemicals together turns minor spills into emergencies. Keeping 3-Heptanone apart from ignition sources and reactive chemicals cuts out accidents before they happen. Stubborn habits, like stacking bottles where it’s convenient, don’t mix with chemical safety—an organized shelf saves a lot of apologies and paperwork.
Ventilation Can’t Be Overlooked
Good airflow in storage areas helps stop vapor build-up. Don’t just rely on air conditioners or regular fans. Fume hoods and purpose-built cabinets with exhaust bring down the risk. I’ve seen labs that put all their faith in a cracked window and paid for it when fumes gathered indoors. Standards set by agencies such as OSHA and the CDC stress proper ventilation for a reason—long-term exposure to 3-Heptanone’s vapors brings more harm than most realize at first.
Personal Safety and Training
Anyone handling chemicals, whether daily or once a month, benefits from proper training. Hands-on demonstrations, not just paperwork, leave stronger memories. Lab coats, gloves made of materials like nitrile, and splash-proof goggles help, but knowing why each step matters, even more so. Sharing horror stories or examples from the news during these sessions gets attention and changes habits.
Planning for Spills and Disposal
Spill kits stocked with absorbents, neutralizing agents, and disposal bags should never gather dust. Too many facilities treat cleanup as an afterthought, hoping no accidents happen. Used 3-Heptanone or contaminated gear belongs in special waste containers, not down the drain. Environmental agencies chase down unauthorized dumping, and fines can hit hard. Responsible disposal keeps neighborhoods cleaner and reputations safe.
What are the physical properties of 3-Heptanone?
Why 3-Heptanone Matters
Many people work with chemicals every day, whether brewing up flavors for food, tracking pollutants in the air, or tinkering in the lab. Some substances show up more often than others, and 3-Heptanone tends to pop up in all sorts of scientific conversations. It has a sharp, fruity aroma that makes it useful not just in flavors and fragrances, but also in research settings. Getting a handle on its physical properties often helps scientists and technologists make smarter choices, especially when safety and efficiency matter.
What You Notice First: Appearance and Smell
Anyone who opens a bottle of 3-Heptanone can pick up on its distinct scent. The clear, colorless liquid looks like water at first glance, but it pours with a different kind of weight and leaves behind a slightly oily texture. That strong, banana-like smell isn’t just a curious detail—it alerts handlers to its presence, which makes spills and leaks easier to identify before problems get out of hand.
Boiling, Melting, and Getting Around the Lab
3-Heptanone boils at about 151 degrees Celsius (304 degrees Fahrenheit). That puts it squarely in the middle range for ketones. Its melting point drops to -35 degrees Celsius (-31 degrees Fahrenheit), so it stays liquid in all but the coldest freezers. In the lab, those numbers matter because handling and storage rules often shift as temperature changes. A chemical that stays liquid at ordinary room temperatures can be weighed, measured, and mixed using standard glassware and tools.
This compound evaporates faster than water, but it lingers longer than some lighter solvents. Its vapor pressure sits at around 4 mmHg at 20 degrees Celsius. So, in an open dish, you’ll catch its scent after a while, but it won’t vanish instantly. This moderate volatility makes it safer to use indoors compared to highly volatile solvents.
Mixing In and Breaking Down: Solubility
You can shake 3-Heptanone with water and find only a tiny amount blending in—about 4 grams for every liter of water at room temperature. Dump the same liquid into ethanol or diethyl ether, and it mixes easily. This limited water solubility helps designers pick solvents for separation or extraction in chemistry projects or flavor blending. People with experience in the field keep an eye on these numbers because mistaking one solvent for another can ruin a batch or even cause a safety problem.
Handling and Safety: Density and Flammability
A liter of 3-Heptanone tips the scale at about 0.82 kg, lighter than water but not feather-light. This matters for pumps, pipes, and any setup where it needs to move from one tank or lab bench to another. Its flash point sits around 45 degrees Celsius (113 degrees Fahrenheit). The risk of fire isn’t as high as gasoline, but it needs storage away from open flames or sparks. Having worked with volatile solvents before, I know how quickly a forgotten open container can turn into an emergency. Safe ventilation remains a must, and chemical-resistant gloves and goggles come standard.
Piecing Together the Bigger Picture
Practically, the physical details of 3-Heptanone shape its use in food science, air quality research, and chemical manufacturing. Its handling straightforward but not risk-free, so every detail—down to vapor pressure or flash point—plays a role in keeping labs and factories safe. By understanding these physical features, people can sidestep mistakes and turn a niche chemical into a valuable, reliable part of their work.
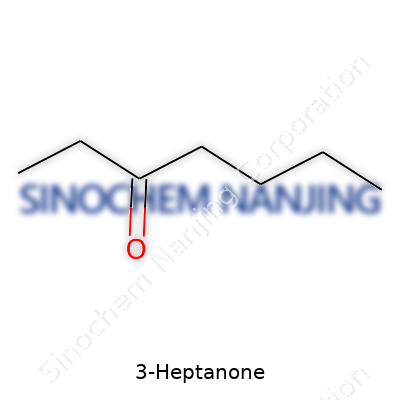
| Names | |
| Preferred IUPAC name | Heptan-3-one |
| Other names |
Heptan-3-one Ethyl butyl ketone |
| Pronunciation | /ˈθriː ˈhɛptəˌnoʊn/ |
| Identifiers | |
| Beilstein Reference | 1209689 |
| ChEBI | CHEBI:48079 |
| ChEMBL | CHEMBL14581 |
| ChemSpider | 7436 |
| DrugBank | DB02170 |
| ECHA InfoCard | EC Number: 210-037-7 |
| EC Number | 203-689-4 |
| Gmelin Reference | 7979 |
| KEGG | C05808 |
| MeSH | D006101 |
| PubChem CID | 8093 |
| RTECS number | MK7520000 |
| UNII | KL08LQ7Z2N |
| UN number | UN2713 |
| Properties | |
| Chemical formula | C7H14O |
| Molar mass | 114.19 g/mol |
| Appearance | Colorless liquid |
| Odor | banana |
| Density | 0.817 g/mL at 25 °C (lit.) |
| Solubility in water | 5.7 g/L (20 °C) |
| log P | 2.0 |
| Vapor pressure | 0.934 mmHg (at 25 °C) |
| Acidity (pKa) | pKa ≈ 20 |
| Basicity (pKb) | pKb = 15.25 |
| Magnetic susceptibility (χ) | -7.44 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.412 |
| Viscosity | 2.38 mPa·s (at 25 °C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 314.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -260.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4180.4 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 45 °C |
| Autoignition temperature | 435 °C |
| Explosive limits | 1.2–7% |
| Lethal dose or concentration | LD50 oral rat 1670 mg/kg |
| NIOSH | MV6075000 |
| REL (Recommended) | 50 |
| IDLH (Immediate danger) | 750 ppm |

