An Editorial Look at 3-Chloro-1,2-Propanediol: Chemical, Caution, and the Path Forward
Historical Development
Chemistry has a way of surprising the world with its twists and turns, especially in food technology and industrial manufacturing. In the story of 3-Chloro-1,2-propanediol, also known in scientific circles as 3-MCPD, the tale starts some decades ago around the mid-20th century, when food scientists hunted for efficiency and flavor. Early observations of this compound date back to studies on acid-hydrolyzed vegetable protein and soy sauces. Researchers soon realized this byproduct formed during high-temperature processing, especially under acidic conditions. Momentum grew as regulatory agencies and food safety watchdogs began sounding alarms about its unintended presence in table sauces and processed goods. By the '80s and '90s, study after study put a spotlight on 3-MCPD, pushing industries to reckon with not only its formation but the potential risks hidden in everyday condiments and foodstuffs. Looking back, the journey of awareness around this compound feels like a case study in how discovery, regulation, and science often move hand-in-hand but rarely in straight lines.
Product Overview
3-Chloro-1,2-propanediol doesn’t show up in supermarkets marketed as a product for direct consumer use; its notoriety stems from slipping in unnoticed during the production of food additives and some plastics. Industries recognize it as both an industrial intermediate and an uninvited contaminant. Its presence crops up in products that have little in common at first glance: hydrolyzed plant proteins, processed foods, paper, and even in some epoxide resins. The markets invested in plant protein isolates for food manufacturing, as well as chemical manufacturers, watch 3-MCPD levels closely, balancing between productivity and safety. While some chemists appreciate its reactivity in synthetic organic chemistry, most food technologists treat it with a sense of wariness, given regulatory and health implications.
Physical & Chemical Properties
This compound brings classic markers of a small organochlorine: a low-melting substance with miscibility in water and organic solvents. As an alcohol with chlorinated backbone, 3-Chloro-1,2-propanediol remains stable under typical storage conditions, yet it melds into various reaction streams when given the right push. Anyone who’s poured over safety data sheets in the lab knows it for its viscosity and faint odor. Beyond the lab, many folks would never guess how a clear, colorless liquid becomes a matter of food and environmental safety debate. Its boiling point, relative density, and solubility mark it as versatile both in industrial applications and, unfortunately, as a contaminant in food production lines.
Technical Specifications & Labeling
Regulations sliced through years of industry inertia, pushing for careful monitoring of 3-MCPD content, especially in substances destined for the food chain. Labels don’t often show “3-Chloro-1,2-propanediol” in consumer-facing ways, but industry documentation in Europe and Asia spells out tolerances in the low parts per billion for foods like soy sauce, hydrolyzed proteins, and infant formulas. Analytical chemists rely on sensitive methods—think gas chromatography with mass spectrometry—to quantify even the tiniest traces, as regulators rarely tolerate high levels. In manufacturing environments, handling processes attach cautionary tags, and technical sheets flag the compound’s toxicological profile. This isn’t a chemical where safety shortcuts can last long.
Preparation Method
On the production side, few processes start with an intent to create 3-MCPD outside of synthetic chemistry labs. Industrial pathways usually involve the chlorination of glycerol, under acidic conditions—a testament to the underlying lesson that manipulating one molecule can spark unintended reactions. In real-world food processing, acid hydrolysis of fats or vegetable proteins tends to create the right conditions for 3-MCPD to appear. This unintentional synthesis stands as a lesson for food and chemical manufacturers about the complexity of thermal or acidic treatments. Those studying organic synthesis, on the other hand, sometimes harness its formation intentionally, pushing for more selective and mild conditions to limit byproducts when purity is required.
Chemical Reactions & Modifications
3-MCPD doesn’t sit silently—in chemical environments, it interacts with bases, acids, and nucleophiles. It reacts further to yield chemicals like glycidol, a process that sees use in specialty synthesis but also raises eyebrows for toxicity reasons. Industries have tweaked food processes, pivoting towards enzymatic hydrolysis rather than acid hydrolysis to try and sidestep its formation. Chemical engineers and safety officers in factory settings put in work to limit exposure, often through retooled reaction conditions or by post-processing cleanup steps such as adsorption or membrane filtration. Each effort reflects a broader principle: mitigating risk at the molecular level often demands engineering, vigilance, and an honest dialogue with science’s unpredictable nature.
Synonyms & Product Names
Industry documents, food safety alerts, and scientific papers throw out several names for 3-Chloro-1,2-propanediol: 3-MCPD, alpha-chlorohydrin, and glycerol alpha-monochlorohydrin. None conjure up images of dinner tables or convenience snacks, yet these names pop up frequently in regulatory discussions and toxicological databases. Synonyms serve more than just as jargon; they create clarity—or confusion—for anyone trying to trace research or safety standards across borders.
Safety & Operational Standards
Anyone working hands-on with this compound knows the importance of minimizing personal exposure. Lab protocols make clear the need for gloves, goggles, and adequate ventilation. The other side of safety emerges in regulatory policy, where food producers must validate that concentrations have been pushed down to as close to zero as science permits. Residue limits don’t stem from arbitrary choice; they reflect years of toxicology and public health debate. Researchers and regulatory authorities, especially in the EU and Asia, set most legal limits far below the levels shown to cause harm in animal models. The US Food and Drug Administration posts warnings about excessive levels, and the World Health Organization recommends benchmark tolerances, shaped by ongoing toxicology work. No one takes a cavalier approach here, given both the compound’s reactivity and its toxicity.
Application Area
Most folks rarely encounter pure 3-Chloro-1,2-propanediol outside chemical plants or research labs. It finds controlled uses as a chemical intermediate for making surfactants, resins, and cross-linking agents. Its inadvertent entry into food often becomes the bigger story, serving as a lesson on the trade-offs in industrial food science. Food technologists have chased alternative hydrolysis methods, and companies in food manufacturing have learned to tweak or overhaul legacy production lines to avoid regulatory headaches. Packaging and paper manufacturers also keep an eye on contamination and occupational safety protocols wherever chlorinated organics take shape, knowing how easily worker health concerns can turn into litigation and lost public trust.
Research & Development
The amount of research on this compound has ballooned ever since its first identification as a food contaminant. Analytical chemists consistently try to sharpen detection thresholds, moving from old colorimetric approaches to high-resolution mass spectrometry. Food scientists and process engineers keep tinkering, developing food-grade processes intended to slash the formation of 3-MCPD by shifting away from acid-catalyzed hydrolysis. Elsewhere, toxicology labs run long-term animal studies to unravel the pathways of metabolism, tissue distribution, and genotoxic risk. Collective focus from regulatory agencies, scientists, and manufacturers often highlights creative solutions—sometimes fostering safer processing aids or novel food additives as replacements for classic hydrolyzed proteins. It’s a cycle of learning, refining, and adapting, motivated by the twin goals of safety and food quality.
Toxicity Research
Toxicologists first flagged 3-Chloro-1,2-propanediol because animal studies showed triggers for several health concerns—especially kidney and reproductive effects. Rat models sounded the initial alarms, and scientists moved quickly to determine risk factors for human exposures. The chronic question remains: how little is low enough, especially across different cultures and consumption patterns? Regulatory bodies sift through reams of data to decide tolerances and enforceable limits, often aligning with recommendations from the World Health Organization. Popular press sometimes struggles with the nuances, but toxicological benchmarks rely on solid peer-reviewed animal studies. Occupational safety guidelines stay tight for chemical workers, who handle not just 3-MCPD but chemical cousins like glycidol, which add to the hazard profile. The whole journey underscores the reality that chemical contamination can warrant deep, slow work—not just a reactive ban but a methodical overhaul of research, policy, and industrial hygiene.
Future Prospects
Looking at the coming years, regulatory scrutiny around food and consumer safety only grows. Industry must invest in cleaner production technologies while food safety scientists keep developing sharper analytical and detoxification tools. No one expects demand for plant-based protein or advanced polymers to stop, but the push toward transparency and minimized contamination continues pressing technology forward. Synthetic chemists, too, explore new reactions that aim to sidestep toxic intermediates like 3-MCPD, signaling room for greener and more selective processes. Consumer advocacy shapes industry decisions almost as much as regulation; shoppers want to know more about what ends up on their plates. The best hope for meaningful progress relies not just on single breakthroughs or fines but on a mix of informed science, honest communication, and responsible manufacturing. This chemical reminds everyone how awareness, vigilance, and innovation weave together in the never-ending job of keeping food and the environment safer for everyone.
What is 3-Chloro-1,2-Propanediol used for?
What Purpose Does This Chemical Serve?
3-Chloro-1,2-Propanediol, known to chemists by its snappier name 3-MCPD, often turns up in the world of industrial chemistry. Few folks outside food science circles ever talk about it, but it comes up more often than you’d expect, especially when the conversation shifts to food safety or the byproducts lurking in everyday products.
The Chemistry in the Food Chain
You’ll most often hear about 3-MCPD because of something as ordinary as soy sauce. For decades, manufacturers have used hydrochloric acid to speed up the processing of soy proteins. This process, called acid hydrolysis, gets flavors out faster but leaves behind trace levels of 3-MCPD. Think about that for a second—many of us have poured soy sauce over rice without knowing about these chemical ghosts. Scientists found out that 3-MCPD can pose health risks, especially if you’re exposed to high levels for years.
Beyond the Dinner Table
Food isn’t the only place this chemical shows up. Manufacturers use 3-Chloro-1,2-Propanediol as an intermediate in the production of epichlorohydrin, which then turns up in resins, coatings, and certain types of plastics. It helps build things like epoxy resins, which coat everything from cans to wind turbines. Having worked in an engineering lab, I saw how common these materials can be in construction and automotive products. Places where durability and chemical resistance matter often rely on this chemistry.
3-MCPD also sneaks into personal care products and cosmetics. Surfactants, built with chemicals like this, help blend products and make soaps feel right in your hands. In one project I joined in college, surfactant formulation came down to striking a balance between cleaning power and skin sensitivity. Even a tiny change in the starting chemistry can throw off the whole thing.
The Problem with Exposure
Researchers link high 3-MCPD intake to health problems in animal studies. The World Health Organization and European Food Safety Authority have both flagged this chemical for its potential to disrupt kidney function and raise cancer risks in lab animals. Most countries set strict limits for how much can appear in food. Nobody wants to find out years later that trace chemicals have added up to actual harm.
Food regulators urge companies to fix their processes. Slow, natural fermentation cuts down 3-MCPD levels, so some food companies have turned away from the shortcut of acid hydrolysis. The story always sticks with me—a return to slower, traditional approaches can sometimes protect public health in ways that shortcuts never do.
Looking for Better Pathways
3-Chloro-1,2-Propanediol remains useful—synthetic resin supply chains depend on it—but its risks can't be ignored. Manufacturers have tried to develop cleaner processing steps or find safer alternatives, both for food flavoring and industrial use. More funding flows into greener chemistry every year, and research teams keep testing plant-based feedstocks and new types of catalysis. Consumer demand for cleaner products forces companies to adapt, instead of clinging to business as usual.
Researchers, regulators, and industry professionals all play a part in pushing for safer chemicals. In my own work, even little adjustments—changing how a lab runs extraction, rethinking safety checks—have made a difference. Transparency about chemical risks empowers everyone, from scientists to dinner table cooks, because nobody likes surprises in their sauce or on their skin.
Is 3-Chloro-1,2-Propanediol hazardous to health?
Understanding Where 3-Chloro-1,2-Propanediol Shows Up
Small things in life can pack a punch. 3-Chloro-1,2-propanediol (3-MCPD) proves that point. This chemical often forms during food processing, especially in foods that get heated with salt and fat—think soy sauce, processed vegetable oils, and even certain baby formulas. A lot of people who've grown up dipping sushi in soy sauce or eating bakery products containing margarine have probably come across it, whether they knew it or not.
Why 3-MCPD Gets Spotlight in Health Circles
My first time hearing about 3-MCPD came from food safety warnings. Reports started trickling out of Europe nearly twenty years ago, with scientists finding surprisingly high levels in common foods. The big question humming in the background was whether people should worry about it. Studies with rats showed that eating enough of this stuff damaged kidneys, messed with male fertility, and even kicked off the formation of tumors. Real damage—not just numbers on lab reports. Even though people aren’t rats, the chemical's ability to cause DNA changes in tests means we can't shrug it off.
What the Experts Say
Human bodies handle chemicals differently than lab animals, but some risks cross species lines. Regulatory agencies like the European Food Safety Authority (EFSA) and the World Health Organization (WHO) take worries about 3-MCPD seriously. The EFSA in 2016 set what they call a "tolerable daily intake," capping long-term intake at 2 micrograms for every kilo of body weight. This limit isn't about zero risk; it's about how much the body can deal with over a lifetime without showing major health problems. It doesn’t take a PhD to figure out that most people don’t weigh servings of soy sauce or processed snacks to see what’s in them—so real exposure can get tricky.
Problems With Food Processing and Transparency
Food manufacturers don’t include 3-MCPD levels on labels. Instead, it comes down to trust—trust that regulators check products before they hit shelves. But even trusted foods can catch us off guard. Some years ago, certain soy sauces made headlines for exceeding limits set by food safety bodies, jarring anyone who thought “traditional” automatically meant “safe.” In my own kitchen, grabbing a different brand or reading up on production methods became less about snobbery and more about reducing risks that pile up quietly, meal after meal.
What Works to Cut Down the Risk
No need to panic and toss every bottle in the pantry. Avoiding major sources—mainly those processed in ways known to produce 3-MCPD—makes a difference. Buying soy sauce labeled “naturally brewed” instead of “acid-hydrolyzed,” going easy on processed baked goods, and keeping an eye out for announcements from trusted health authorities can limit exposure in daily life. Manufacturers can also cut levels by dialing back harsh processing methods or switching up ingredients. Eating less processed food, in general, reduces risk—not just from this one chemical, but a whole catalog of additives that creep into processed diets.
Where We Go From Here
The science keeps moving, and new testing methods are better at catching risky levels of 3-MCPD. Still, no one wants to roll the dice on their health because of an unpronounceable ingredient they never asked for. Pushing for tighter regulations, clearer transparency, and food choices that rely less on heavy processing brings peace of mind. Food should feel good, not become a guessing game for hidden chemicals.
What are the storage requirements for 3-Chloro-1,2-Propanediol?
Real Risks Wrapped in a Small Bottle
Step into any medium-sized chemical supply room and someone will tell you about that one container of 3-Chloro-1,2-Propanediol that leaked, spreading an acrid scent that stings the nose. This chemical isn’t something to treat lightly. Even though small vials don’t look menacing, handling errors can lead to real safety slips. The substance has been flagged for its potential health hazards, including toxicity and possible links to cancer if regular exposure happens. That puts the burden on both suppliers and users to treat storage seriously—not as a box to check, but as non-negotiable daily habit.
Stable Conditions: A Matter of Safety
This compound doesn’t thrive under sunlight or heat. Temperatures above room level can speed up decomposition, throw off vapor, or damage the integrity of the bottle holding it. A stable, cool, well-ventilated spot guards against the volatile nature of this liquid. In my experience, colleagues who let storage spaces get hot in summer invite bigger hazards, from increased vapor pressure to higher odds of a spill reaching a flash point.
Moisture and Isolation Questions
Moisture isn’t just a nuisance here. Water seeping into an opened or poorly sealed bottle can trigger unwanted chemical reactions. Keeping caps sealed tight saves more than just the chemical itself—it reduces hazards and stops accidental mixing. Far too often, carelessness leads to bottles grouped on shelves next to acids, alcohols, or oxidizers. Mixing these families encourages risk and could result in a runaway reaction if a leak occurs. Smart storage plans place each family of chemicals on separate, clearly labeled shelving, away from direct sunlight or water sources.
Legal and Ethical Responsibility
Nobody gets a pass just because their operation is small. Governments build legal codes around substances like 3-Chloro-1,2-Propanediol for a reason. The European Chemicals Agency and OSHA both list it for careful control with solid records of incidents. Fines hit hard, but the bigger shock comes when staff get sick or an accident surfaces in local headlines. Workers deserve protection from unseen hazards. In my time maintaining a research stockroom, labels faded over years and new techs missed warnings. Regular inventory reviews and relabeling maintain clarity and keep safety procedures effective.
Practical Steps Work Best
Handling this compound safely means using chemical-resistant gloves and goggles without compromise, even if handling only takes seconds. Always keep a spill kit and eye-wash station close. Regular staff training keeps even experienced hands from growing careless. The best run labs and warehouses post detailed storage instructions right near shelving and review protocol every time a new stock arrives. A clean-up log in plain sight and a waste container specifically for contaminated gloves and wrappers reduce long-term hazard. Taking these practical, hands-on steps keeps 3-Chloro-1,2-Propanediol locked down, and keeps the people who work with it healthy and protected.
How should 3-Chloro-1,2-Propanediol be handled safely?
Recognizing the Risks
3-Chloro-1,2-Propanediol, sometimes called 3-MCPD, shows up most often as a by-product in food processing and industrial production. Scientists consider it a probable human carcinogen. After digging into a few research papers over the years, I started to pay close attention to substances like this. Accidental spills or careless exposure put workers and the environment at risk, especially in tightly enclosed workspaces or during chemical handling.
Personal Protective Gear Makes a Real Difference
Gloves and goggles serve as the first line of defense. Nitrile gloves do a good job blocking skin contact, and chemical splash goggles shield the eyes. Aprons made of resistant materials add another layer of protection. I remember working in a plant in my early career where colleagues often cut corners. Several landed at the nurse’s station with rashes, all from skipping gloves while handling solvents. For a substance like 3-Chloro-1,2-Propanediol, direct skin contact could go beyond irritation—long-term risks matter even more.
Good Ventilation Can’t Be Overlooked
Proper air flow cuts down inhalation hazards. Whenever ventilation gets ignored, vapors build up. Think of it like cooking in a closed-off kitchen: steam saturates the air, making it hard to breathe. Fume hoods, exhaust systems, and even simple portable fans help disperse gases or vapors. Some workplaces upgrade to air-purifying respirators if airborne concentrations reach higher-than-safe levels.
Storage: Labeling and Isolation
Containers holding 3-Chloro-1,2-Propanediol should stay sealed and clearly marked. Locking chemicals in cabinets away from food, flame, and incompatible substances lowers dangerous mixing or cross-contamination risks. Staff should receive training so no one accidentally reaches for the wrong container or forgets to replace a cap. A friend once stored chemicals in mismatched jars, and the confusion led to a spill requiring a full evacuation. Simple habits save hours of crisis—and a lot of money.
Emergency Preparedness Saves Lives
If someone is exposed, quick response counts. Emergency showers and eyewash stations need to be close at hand, especially in production areas. Safety data sheets, (commonly called SDS), describe precise first aid steps for 3-Chloro-1,2-Propanediol. From my own work, I learned drills make the difference. Practicing spill responses helps new and experienced staff move without hesitation under pressure.
Disposal Isn’t an Afterthought
Pouring leftover chemicals down the drain creates bigger problems. Waste specialists collect and destroy hazardous substances in line with local regulations. Years ago, careless dumping in a small lab contaminated the water supply for days. After that, everyone on the team paid much more attention to disposal bins and scheduled pickups.
Working Together for Safer Outcomes
Everyone on site carries a piece of the responsibility for chemical safety. Open communication, ongoing education, and reliable procedures shape a workplace culture where risks drop and health comes first. Watching out for each other matters—sometimes it’s a reminder to grab gloves, other times it’s knowing how to call in a spill clean-up team without hesitation.
What is the chemical structure of 3-Chloro-1,2-Propanediol?
Getting to Know 3-Chloro-1,2-Propanediol
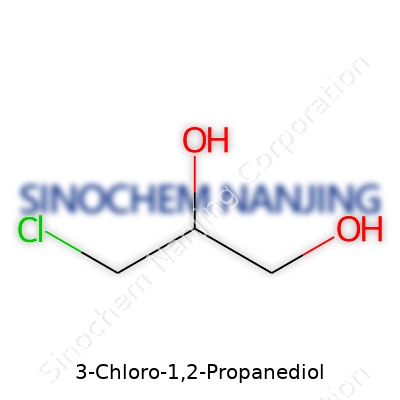
3-Chloro-1,2-Propanediol — often written as 3-MCPD in scientific papers — holds a clear yet complex chemical identity. Its structure sits at the crossroads of organic chemistry and health debates. At its core, this molecule features three carbon atoms arranged in a row, forming the molecule’s backbone. Attached to carbons one and two are hydroxyl (–OH) groups, which classify it as a diol. The third carbon binds with a chlorine atom, the group that gives the compound its concerning reputation.
Laying it out in plain molecular terms, 3-Chloro-1,2-Propanediol stands for C3H7ClO2. Its structural formula looks like HO–CH2–CHOH–CH2Cl. One hydroxyl group attaches to the first carbon, another to the second, and the third carbon catches the chlorine. Each shift in these attachments changes the molecule’s behavior dramatically.
Why Structure Matters: Experiences from Food Safety
Years of working within the food chemistry space taught me that a slight tweak in a molecule’s structure can turn a harmless substance into a health risk. With 3-Chloro-1,2-Propanediol, those two hydroxyl groups push its reputation toward alcohol-like solubility. The chlorine helps it slip into processed foods, especially after high-temperature treatments such as refining vegetable oils or making soy sauce. The isomeric form and exact placement of those groups feed directly into the pathways the compound follows inside the body, impacting both absorption and toxicity.
Health Concerns Tie Back to the Chemical Blueprint
Data from health authorities point to concerns about this compound’s potential to damage DNA. 3-Chloro-1,2-Propanediol has cropped up in laboratory studies as a possible carcinogen. Regulatory agencies like the European Food Safety Authority examined results in rodents and set strict limits on its presence in food products. From direct observation, food factories struggle to keep levels low, as the slightest shift in the processing line — a hotter steam-injection step or an imprecise catalyst — can push chlorine toward binding with glycerol, sparking 3-MCPD formation.
My experience working in analytical labs showed that the molecule’s functional groups make it easy to detect using modern chromatography methods. That helped drive accountability for food producers but also revealed the stark frequency of its occurrence, even in otherwise meticulously produced foods. These tools let us peer into the invisible chemistry of what many people eat daily.
Pursuing Safer Chemistry: Practical Paths Forward
Addressing the presence of 3-Chloro-1,2-Propanediol in foods means understanding how molecules assemble themselves. Process engineers continue adjusting techniques to lower temperatures or swap out chlorine-containing inputs. Food companies now track the starting materials much more closely. In my work, replacing acidic catalysts or changing the water content during processing pushed the numbers down. Sharing data between labs and across borders encourages smarter recipes and clear labeling, reflecting a broader communal effort. No shortcut beats the patient work of understanding which step in manufacturing causes tiny chemical shifts — proof that small structural details have big ripple effects in the real world.
| Names | |
| Preferred IUPAC name | 2-Chloropropane-1,3-diol |
| Other names |
α-Chlorohydrin Glyceryl α-chlorohydrin Monochlorohydrin 1,2-Dihydroxy-3-chloropropane 3-Chloropropane-1,2-diol |
| Pronunciation | /θriː-ˈklɔːroʊ-waɪn-tuː-proʊˈpeɪn-daɪˌɒl/ |
| Identifiers | |
| CAS Number | 96-24-2 |
| 3D model (JSmol) | 3D model (JSmol) string for **3-Chloro-1,2-Propanediol**: ``` CC(O)CCl ``` *(This is the SMILES string for 3-Chloro-1,2-Propanediol.)* |
| Beilstein Reference | 1720801 |
| ChEBI | CHEBI:17141 |
| ChEMBL | CHEMBL49877 |
| ChemSpider | 7957 |
| DrugBank | DB02702 |
| ECHA InfoCard | 03a05eaf-a769-4846-b88a-4ad58a46b111 |
| EC Number | 200-449-4 |
| Gmelin Reference | 8226 |
| KEGG | C06576 |
| MeSH | D08.750.099.700.800 |
| PubChem CID | 7902 |
| RTECS number | TY3150000 |
| UNII | V57679Q8YI |
| UN number | UN2689 |
| Properties | |
| Chemical formula | C3H7ClO2 |
| Molar mass | 110.54 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Odorless |
| Density | 1.32 g/cm3 |
| Solubility in water | miscible |
| log P | -1.2 |
| Vapor pressure | 0.0133 hPa (20 °C) |
| Acidity (pKa) | 13.06 |
| Magnetic susceptibility (χ) | -62.3·10⁻⁶ cm³/mol |
| Refractive index (nD) | n20/D 1.454 |
| Viscosity | 28 mPa·s (20 °C) |
| Dipole moment | 2.69 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 117.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -446.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1048.6 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, suspected of causing cancer, causes skin and eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H341, H350 |
| Precautionary statements | P210, P280, P305+P351+P338, P309+P311 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 108°C |
| Autoignition temperature | 300 °C |
| Lethal dose or concentration | LD50 oral rat 152 mg/kg |
| LD50 (median dose) | LD50 (median dose): 260 mg/kg (oral, rat) |
| NIOSH | KK3150000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Chloro-1,2-Propanediol: Not established |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | 30 ppm |
| Related compounds | |
| Related compounds |
Epichlorohydrin Glycerol 1,2-Propanediol 1,3-Dichloro-2-propanol Chlorohydrin |

