3-Aminopropanol: Reflecting on a Quiet Chemical Powerhouse
Looking Back: Historical Development and Use
3-Aminopropanol doesn’t often make headlines, though its journey in chemistry circles stretches back close to a century. Its creation came amid a wave of interest in amino alcohols, a family of compounds that bridged ideas in synthetic organic chemistry and practical industrial use during the early and mid-20th century. Researchers and industrial chemists looking for flexible molecular building blocks focused on 3-aminopropanol. Its molecular simplicity—one amino group paired with a primary alcohol—was appealing because it let them test diverse reactions and applications. Over time, patents started referencing it as a tool or intermediate, particularly as the chemical sector ballooned in scale and ambition after World War II. Today, no blockbuster stories attach to its name, but you’ll spot aminopropanol quietly playing its part in synthesis labs and capacities that rarely shine in mainstream science media.
Understanding 3-Aminopropanol: Product Overview
3-Aminopropanol stands out as both a versatile chemical and something of a laboratory workhorse. It is known by other names like 1-Amino-3-propanol or β-Amino-1-propanol, but those all point to the same three-carbon chain adorned with an amino group and a hydroxyl. Researchers depend on it not because it dazzles with unique properties but because it gets jobs done. In the real world, tucked away on warehouse shelves or laboratory benches, it rarely comes labeled for mass-market appeal. The world of chemical intermediates moves without much public fanfare, though it underpins huge portions of drug development, pesticides, and polymers.
Physical & Chemical Properties: Why the Details Matter
Understanding a chemical’s guts—its melting point, boiling point, appearance—means more than reciting a datasheet. 3-Aminopropanol shows a combination of water solubility, a relatively low boiling point around 213°C, and an easy-to-handle, syrupy liquid form at room temperature. You’ll find it colorless and with a slight ammonia-like odor. These details matter on the plant floor and in the lab, because a substance you can mix with water opens doors that greasy or insoluble compounds shut. The presence of both an amino and an alcohol group gives this molecule two distinct handles for further reaction—sort of like a Swiss Army knife for organic synthesis. Its pH in solution moves towards the alkaline side but isn’t as caustic as you’d find with a straight amine; that kind of reliability keeps lab techs and process engineers coming back.
Technical Specs & Labeling in Practice
Chemicals like this demand respect for both safety and standardization. Most industrial or laboratory suppliers provide 3-aminopropanol at purities ranging from 98% to 99%, usually because high purity translates to fewer headaches for downstream reactions. Labels might mention water content and trace amounts of related compounds; the industry takes this seriously, especially when the final use touches food, pharmaceuticals, or household goods. Regulatory bodies drive much of this rigor, with details like CAS number (156-87-6) tying together global supply chains and legal tracking. Scientists have learned to keep an eye on labeling since an error can ripple straight through a manufacturing process and turn reliability into recall.
Preparation: Peeking Into the Back Room
Old-school routes for making 3-aminopropanol started with 3-chloropropanol or 3-bromopropanol, using ammonia for substitution. These workhorse reactions sound straightforward, but yields and purity depend on how well a chemist balances temperature and pressure, or minimizes byproducts. Alternative routes—like catalytic aminolysis or hydrogenation of acrylonitrile derivatives—have been tried, especially as demands for cleaner, greener chemistry have gained traction. The choice of route shapes the environmental and economic footprint of a chemical. In smaller-scale labs, where speed and convenience trump price, chemists might switch things up based on what reagents they have on hand. One lesson is clear: even so-called “commodity” chemicals reward creativity when you duck into the nitty-gritty of synthetic routes.
Reactivity and Modification: Not Just a Bystander
3-Aminopropanol stands out for its double functionality, which opens a toolbox of reactions. The hydroxyl group lets it swing into action for etherification, esterification, or even ring-forming steps. The amino side steps up for acylation, alkylation, or condensation with carbonyl compounds—or as a nucleophile in polymerizations. Real innovation often comes from chemists who see these reactivity patterns and try something new: linking bioactive fragments, building water-soluble drug conjugates, or fine-tuning surfactants for cleaner industrial processing. Its ability to seamlessly mesh with acids, anhydrides, epoxides, and alkyl halides means it pulls weight far beyond its atomic count. It’s a great example of how even small molecules can punch above their weight in smart hands.
Synonyms & Product Names
Names in chemistry often tangle up newcomers, but this one goes by several: 3-Aminopropanol, 1-Amino-3-hydroxypropane, β-Amino-1-propanol, or even 3-Hydroxypropylamine. Search engines and regulatory lists all tip to the same molecule, though suppliers may sell it with slightly tweaked branding or mention if they’ve used a salt form for ease of handling. At the end of the day, tracking all these aliases reminds workers and researchers to double-check paperwork before mixing, since the alphabet soup of synonyms can trip up even seasoned veterans. It pays to trust but verify.
Safety & Operational Standards: Keeping the Shop Running
Working with chemicals carries risk, and 3-aminopropanol is no exception. Direct skin contact can cause irritation, and inhaling its vapors over time isn’t great for anyone’s lungs or eyes. In the lab, gloves and goggles take priority. Proper ventilation remains non-negotiable, especially as higher temperatures or open vessels can send vapors into the air. Storage standards call for cool, dry places, away from acids and oxidizers. Spill management comes down to absorbing small releases with noncombustible material. These steps aren’t just bureaucratic—they’re part of a safety culture built on real-world experience and, sometimes, hard lessons from the past. Hazards don’t only threaten workers: downstream effects—from leaks or improper disposal—can affect water supplies or animal health. Tough safety standards exist because cutting corners here means rolling the dice with people’s lives.
Applications: Where 3-Aminopropanol Earns Its Keep
Though rarely in the spotlight, 3-aminopropanol stays in regular use across a surprising spread of industries. Drug companies lean on it for making molecules meant to fight infections or control pain. It serves as a linker in antihistamines, or as a building block in more complex specialty chemicals. In agriculture, it helps shape certain herbicides and fungicides, especially in places where water solubility and biological compatibility matter. Polymer chemists appreciate its bifunctional nature, which lets it tweak the flexibility or stickiness of finished plastics or coatings. It even surfaces in cosmetic formulation or as a pH adjuster in industrial cleaning fluids, thanks to its agreeable reactivity and low persistence in the environment. Its work behind the scenes keeps supply chains humming, even if the public rarely hears the details.
Research & Development: Pushing Beyond the Routine
Every so often, restless minds in chemical research look for new ways to stretch the boundaries of familiar molecules. In the case of 3-aminopropanol, recent years have seen work on greener synthetic routes—packing more crust into each atom of carbon, or sidestepping hazardous intermediate steps. Some labs test its use in drug discovery, where hydrophilic linkers can help shuttle active ingredients through tight biological spaces. Others focus on deriving new surfactants that combine just the right balance of cleaning power and biodegradability. The field’s shifting interest in “sustainable chemistry” adds an extra wrinkle, as regulations and consumer demand push research toward safer, less toxic products built from simple, flexible molecules like aminopropanols.
Toxicity Research: Keeping an Eye on Long-Term Impact
Work on toxicity takes a cautious approach, since what starts as mild skin or eye irritation in controlled studies may shift over years of workplace or environmental exposure. Acute effects often show up quickly, as the skin tingles or eyes sting, but chronic exposure remains a research target. Studies in lab animals, where data exists, generally indicate low to moderate toxicity, with effects most prominent at high doses or careless handling. This reality keeps regulators attentive and shapes safe use guidelines. Researchers keep pushing for more data on breakdown products, in case small fragments from 3-aminopropanol turn out to add unexpected risk to water, soil, or food chains.
Future Prospects: Staying Relevant in a Changing World
The chemical trade changes as new constraints emerge on waste, toxicity, and efficiency. 3-Aminopropanol, like many workhorse intermediates, must adapt or lose relevance. Chemists focusing on renewable feedstocks and energy savings look for ways to keep using its versatile structure while minimizing byproducts and hazards. Future wins may come where 3-aminopropanol finds a niche in new materials, especially those designed for advanced medicine, environmental cleanup, or next-generation polymers. Its simple structure gives it a flexibility that’s likely to keep it in play, even as the next wave of regulatory or environmental thinking raises the bar for what makes a chemical worth making. The story of 3-aminopropanol isn’t glamorous, but it underlines the slow, steady progress in the chemical sciences, where reliability, safety, and reactivity still win the day.
What is 3-Aminopropanol used for?
More Than Just A Lab Chemical
Anyone who’s worked in chemistry has probably handled a bottle of 3-Aminopropanol at some point, but rarely stops to think about how far its reach extends beyond the lab bench. This compound’s usefulness goes well beyond its funny smell and clear appearance. It blends into many corners of manufacturing and production, driving innovation in unexpected ways.
Its Role in Everyday Products
3-Aminopropanol steps up as a building block for other chemicals, especially in the creation of surfactants—the stuff that lets detergents lift greasy stains out of jeans and off countertops. These surfactants owe their cleaning power to the specialized molecules crafted from compounds like this one. It also shows up behind the scenes in certain pharmaceutical processes. Drug makers often rely on flexible molecules to tweak how a medicine works or absorbs in the body, and 3-Aminopropanol offers that flexibility thanks to both its alcohol and amine groups.
Not Just for Scientists
Outside the obvious chemistry world, industries making cosmetics, textiles, and plastics keep 3-Aminopropanol in steady demand. One example that sticks out comes from working in a small family-run cleaning supply business. Our “professional strength” carpet cleaner used amine-based surfactants that cleaned stubborn traffic lanes and pet stains from berber carpet. No one buying a jug thought about the chemistry, but it’s what kept their house looking new. The compound slips into the manufacturing chain for personal care items—from shampoos to lotions—because it helps other ingredients mix and blend smoothly.
Health and Environmental Viewpoints
Any chemical used so widely brings up understandable questions about health and safety. 3-Aminopropanol can irritate skin and eyes if handled without care. Factories follow safety guidelines to protect workers’ health, though accidents are possible. Wash stations, goggles, strong ventilation—these all act as simple defenses that keep people safe. On the environmental side, this chemical doesn’t build up in the environment under normal conditions, and it breaks down in wastewater plants. Still, as someone who has toured industrial chemical facilities, the margin for error tightens near waterways or crowded neighborhoods.
Greater pushback from communities near factories sometimes forces companies to reevaluate old practices. For example, larger companies that manufacture amine-based substances now invest in “green chemistry” upgrades, adjusting synthetic steps to cut down dangerous byproducts. They also use closed-loop production systems to keep emissions low. These investments protect both the neighborhood and the business.
Why Responsible Use Matters
The real lesson from seeing chemical supply chains up close is that convenience and cost push companies to pick ingredients like 3-Aminopropanol. Choosing safer production methods and holding manufacturers accountable gives everyone—consumers and workers—some peace of mind. As a society, making the effort to use these compounds responsibly matters because invisible risks exist as soon as chemicals leave the lab shelf and start mixing into daily routines. By staying curious and demanding better answers about what’s in everyday products, everyone can help encourage safer, cleaner uses of compounds that touch so many lives.
What is the chemical formula of 3-Aminopropanol?
What Sets 3-Aminopropanol Apart
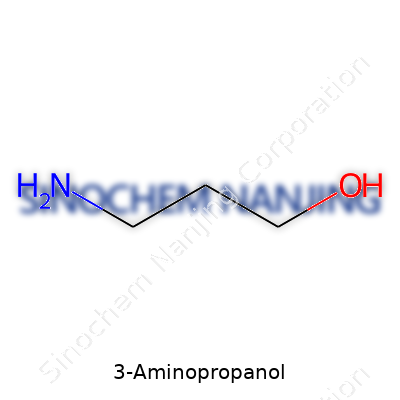
Step into the world of organic chemistry, and names like 3-aminopropanol start to look a lot less intimidating once you break them down. I took my first organic chemistry class convinced that everything would boil down to big, complicated molecules. Turns out, compounds like 3-aminopropanol are about as straightforward as you get. Its chemical formula, C3H9NO, spells out exactly what you’re dealing with: three carbons, nine hydrogens, one nitrogen, and one oxygen. That tells you a lot at a glance.
Everyday Uses Start with the Basics
Think about all those humdrum things we use daily—shampoos, cleaning products, the adhesives that keep things together. Compounds like 3-aminopropanol fit right into the mix. It carries both an amine group and an alcohol group on a three-carbon chain, so it connects the worlds of water-friendly reactions and the chemistry that brings plastics or pharmaceuticals to life.
From my own experience working in a biochemistry lab, the importance of understanding functional groups has never felt trivial. That lone nitrogen opens a door to reactions with acids, and the oxygen in the alcohol group brings solubility, both fundamental for building more complex molecules. Companies use this chemistry not just for curiosity, but for making safer, smarter, or more sustainable products.
Why Accuracy in Chemical Identity Matters
Get the formula wrong, and you invite mix-ups that go beyond embarrassment. The right arrangement of atoms means you can predict reactivity, safety, and suitability for a product. I once heard a story about a formulation chemist who mistook a similar-sounding compound for another. That slip-up didn’t just halt production—it led to weeks of compliance testing, extra costs, and sleepless nights.
In regulated industries, even small details take on big importance. The stakes ramp up in pharmaceuticals or food processing. Regulatory bodies like the FDA don’t have patience for careless mistakes, and neither should we. Publishing clear, trustworthy chemical information builds confidence, sure, but it also prevents disasters.
Fact Check: C3H9NO
The chemical formula for 3-aminopropanol, C3H9NO, lines up with its structure: a three-carbon backbone, a terminal amine group (NH2) on one end, and a hydroxyl group (OH) on the other. This simple arrangement supports all sorts of downstream chemical reactions, from polymer synthesis to surfactant production. Publications such as the Merck Index and PubChem confirm this formula.
Looking Forward: Solutions for Stronger Chemical Communication
Even in a world full of data, human error or lack of clarity keeps popping up. Fact-checking sources before recording or sharing chemical structures helps foster better habits. It pays to review safety data sheets, cross-reference with respected chemical databases, and invest in ongoing chemical literacy, not just for researchers but also for anyone working in formulation or compliance.
In classrooms, open-source chemistry databases and hands-on demonstrations build the kind of practical familiarity that sticks far longer than rote memorization. The more comfortable we are with formulas like C3H9NO, the less chance small mistakes become costly setbacks.
Is 3-Aminopropanol hazardous or toxic?
Looking at Everyday Chemical Safety
My earliest job out of college landed me inside a chemical warehouse. Shelves lined with barrels, each with names that were hard to pronounce, and every container screamed hazard warnings. One of the compounds, 3-aminopropanol, didn’t look any different from a bucket of water, but looks in chemistry almost never tell the full story.
The Real Chemical Story
3-Aminopropanol brings both promise and challenges to labs and factories. On a practical note, workers use this ingredient for surfactant production, textile processing, and as an intermediate in pharmaceuticals. Its popularity comes from being a handy building block, not some fringe chemical only seen in textbooks.
Hazard isn’t about publicity—it’s about what ends up in your hands, lungs, or eyes. Here’s what science tells us:
- Skin and eye contact with 3-aminopropanol leads to irritation, redness, maybe even burns if you spill a concentrated solution.
- Breathing in a mist or vapor causes sore throat or coughing. Prolonged exposure, especially in cramped workshops lacking ventilation, triggers headaches, dizziness, or other symptoms.
- According to PubChem and the European Chemicals Agency, the oral toxicity rate for rats sits around 2 g per kg—meaning accidental ingestion by people poses a real threat if the chemical’s not stored right.
The Value of Clear Training
Most accidents involving chemicals like 3-aminopropanol don’t trace back to some shocking toxicity rating. They usually trace back to sloppy storage, aging gloves, or casual attitudes. My own experience handling chemicals taught me: Proper labeling, protective gear, and knowing exactly what you’re working with make all the difference.
Labels flag eye and skin damage risk. Gloves and face shields become as natural as seatbelts in this line of work. People outside labs—maintenance workers, janitors, even delivery drivers—get left out of the conversation, but they cross paths with chemical containers every day. Training needs to extend past lab technicians, reaching everyone whose hands roll those drums across a warehouse floor.
Potential Solutions: Cutting Hazards Down to Size
The ultimate hazard comes down to exposure and management, not the raw scorecard from a data sheet. Several real-world steps reduce risk:
- Keep containers tightly closed. This sounds simple until one sees how many leaks start from careless caps.
- Ventilation matters. I learned it best in a tiny, stuffy storeroom with no open window. Even a basic fan helps keep vapor from building up.
- Showering attention on workplace communication—posting clear hazard summaries, showing what burns look like, fixing damaged eyewash stations—saves time and health in the long run.
- Rotating containers out of storage before labels fade or caps crack helps prevent surprise spills.
So, is 3-aminopropanol hazardous? Definitely, with careless handling. In the right hands, with sharp eyes and simple barriers in place, it fits into the daily toolkit without causing alarm. Real safety lasts when everybody, not just the scientists, knows the risks and looks out for one another.
How should 3-Aminopropanol be stored?
Getting to Know 3-Aminopropanol
If you have ever worked in a lab or around industrial chemicals, 3-Aminopropanol rings a bell. It serves as a building block in a wide range of products, from pharmaceuticals to surfactants. Yet for all its usefulness, this chemical demands respect, especially in storage. My time working in a university lab showed me that one slip, a careless move, or an overlooked container could spark problems that ripple through both safety and research progress.
Hazards Lurking in the Bottle
3-Aminopropanol isn’t a casual substance. We’re dealing with an organic compound that’s irritating to eyes and skin, and it packs enough punch to affect breathing if you don’t handle it right. The manufacturers outline its risks for a reason. Accidents trace back to poor storage far more often than folks admit. A bottle left near sunlight, a leaky cap, or cross-contamination serves as an accident in the making. The label’s fine print isn’t there to be ignored.
How I Learned to Store it Right
Early in my career, an improperly sealed container left a sharp, unpleasant odor lingering in the air and caused headaches for half my team. Since then, I stick to sealing bottles tight and labeling every container with the date received and opened. Storing it requires a cool, dry area, away from sunlight, away from sources of ignition, and in a spot with solid ventilation. Simple metal shelves can corrode, so I stick to chemically resistant cabinets for amines like this.
Why Container Choice Matters
3-Aminopropanol gnaws away at ordinary plastic or metal over time. I always choose containers recommended by the manufacturer, usually high-density polyethylene, glass, or other compatible plastics. Containers with tight-fitting lids prove vital; one poorly closed cap did more harm than any mismeasurement during experiments. I never decant into unmarked glassware, because ambiguity brings danger.
Climate Control Is More Than Comfort
The storage room’s temperature stays below 30°C, and humidity stays low. Moisture invites mold and can react with chemicals stored nearby. In one shared lab, a colleague’s careless placement allowed a leak, which mixed with a cleaning supply and sent the custodian home with a rash. Interviews with chemical safety officers back up my experience: temperature and humidity controls aren’t overkill. They prevent reactions, corrosion, and instability.
Don’t Ignore the Label or the Law
I once saw a poorly labeled bottle put us at risk of a reportable incident during an inspection. It sounds simple, but accurate labeling with hazard warnings and container dates kept us off OSHA’s naughty list and protected newcomers on the team. Safe storage goes hand-in-hand with compliance—throwing a bottle on a shelf and walking away invites headaches, or much worse.
Cleanliness Buys Peace of Mind
I wipe down storage spaces every month. Residue builds up, bottles stick, then labels fall off and secrets get kept until someone pays the price. Every workplace benefits from a simple log, regularly checked for leaks or warped containers. Fire extinguishers and spill kits nearby take care of panic moments. Even if you never need them, knowing they’re close keeps everyone focused.
Building Safer Habits Together
3-Aminopropanol rewards careful respect. A cool, dry spot away from sunlight, proper containers, clear labeling, and steady housekeeping shape a chemist’s best safety net. Personal vigilance grows from shared stories and real lessons, not just training slides.
What are the safety precautions when handling 3-Aminopropanol?
Understanding the Risks
3-Aminopropanol isn’t just a strange name found on chemical drums. This clear, colorless liquid shows up in labs and manufacturing, often used to make surfactants or as an intermediate in other chemical processes. Though it might seem harmless, it can irritate skin, eyes, and even your lungs if you aren’t careful. Many folks, myself included, learn fast that a tiny splash can turn into a burning feeling or lingering cough.
Personal Protective Gear Gets the Basics Done
Using gloves is non-negotiable. Nitrile or neoprene gloves keep direct contact at bay, letting you work without feeling that sting or itch. Forgetting eye protection runs the risk of chemical burns. Safety goggles with splash guards do more than comfort—they block out the painful outcome of even a brief exposure. Lab coats and closed shoes put up another line of defense. Covering up with long sleeves makes sure you don’t spend the week nursing a skin rash.
Proper Ventilation Really Matters
Breathing in chemical fumes hardly ever seems urgent—until it’s too late. Fume hoods aren’t just for show, and their constant hum means clean, moving air that sweeps away vapors before you notice them. On stuffy days in the lab, that fan noise becomes reassuring. Even a brief exposure has been shown to trigger throat and lung irritation. Relying on local exhaust and not just open windows or fans offers much better protection.
Handling Spills and Splashes
I’ve seen a lot of minor spills handled with panic and paper towels. Never a good strategy. For 3-Aminopropanol, use absorbent pads or granules designed for chemicals. Wipe up and then wash down the area with plenty of water. If skin contact happens, washing off fast with soap and cool water makes a huge difference—rushing saves skin and comfort. Any splash in the eye calls for at least 15 minutes of steady rinsing at the eyewash station. Forgetting to do this can turn a minor accident into a trip to urgent care.
Labeling and Storage Goes a Long Way
Hazards increase whenever containers get reused without labels, or storage areas mix incompatible chemicals. Keeping 3-Aminopropanol in tightly sealed, clearly marked bottles reduces mistakes. Store away from oxidizers and acids. I’ve seen damage from leaky bottles and forgotten reagents—prevention beats cleanup every time. Make sure containers stay upright, with secure lids. Regularly checking for leaks means fewer headaches.
Training and Emergency Prep Save Lives
Even after years working around chemicals, I never skip training updates. Knowing where the nearest eyewash, shower, and spill kit are saves valuable seconds. Current safety data sheets outline steps for accidental exposure or fire. Practicing those emergency drills once in a while—actually pulling out the spill kit or running through where help is—pays off during real problems. Relying on memory or luck usually backfires.
Simple Habits Protect Health
Washing hands before lunch and after handling 3-Aminopropanol keeps invisible traces off skin and out of your mouth. Never eat, drink, or use phones in the work area: one slip-up can cause illness. I’ve watched colleagues skip these steps and pay for it. Following these routines keeps bodies healthy, and works better than any warning label ever printed.
Why These Steps Matter
Not everyone walks into a lab with experience, but anyone can learn the value of solid safety habits. A single mistake costs time, money, and sometimes well-being. Taking precautions doesn’t mean working in fear—it means building the kind of workplace where nobody gets hurt and everybody goes home healthy. That’s the mark of a skilled worker who knows the risks are real but manageable.
| Names | |
| Preferred IUPAC name | 3-Aminopropan-1-ol |
| Other names |
1-Amino-3-propanol 3-Hydroxypropylamine 1,3-Propanolamine Aminopropanol-3 β-Amino-1-propanol |
| Pronunciation | /ˈæmɪnoʊ.proʊˈpæn.ɒl/ |
| Identifiers | |
| CAS Number | 156-87-6 |
| 3D model (JSmol) | `3d_jmol="C(CN)CO"` |
| Beilstein Reference | 605928 |
| ChEBI | CHEBI:17149 |
| ChEMBL | CHEMBL152553 |
| ChemSpider | 10160 |
| DrugBank | DB03765 |
| ECHA InfoCard | 03e2249b-42bf-480d-89e5-3089b46f6d54 |
| EC Number | 200-709-2 |
| Gmelin Reference | 5544 |
| KEGG | C02931 |
| MeSH | D016582 |
| PubChem CID | 10760 |
| RTECS number | UE8575000 |
| UNII | N3RRA6AT1F |
| UN number | UN2672 |
| Properties | |
| Chemical formula | C3H9NO |
| Molar mass | 75.11 g/mol |
| Appearance | Colorless liquid |
| Odor | Ammonia-like |
| Density | 0.975 g/mL at 25 °C(lit.) |
| Solubility in water | Miscible |
| log P | -0.67 |
| Vapor pressure | 0.0065 mmHg (25 °C) |
| Acidity (pKa) | 15.0 |
| Basicity (pKb) | 5.10 |
| Magnetic susceptibility (χ) | -6.40×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.453 |
| Viscosity | 20 cP (25°C) |
| Dipole moment | 1.4148 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 120.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –246.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3356 kJ/mol |
| Pharmacology | |
| ATC code | C04AB10 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes severe skin burns and eye damage. Causes serious eye damage. Harmful if inhaled. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302 + H312 + H332, H319 |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | Flash point: 96°C |
| Autoignition temperature | 405°C |
| Explosive limits | Explosive limits: 2.1–12% |
| Lethal dose or concentration | LD50 oral rat 2,150 mg/kg |
| LD50 (median dose) | 1,900 mg/kg (rat, oral) |
| NIOSH | SN1750000 |
| PEL (Permissible) | PEL for 3-Aminopropanol: Not established |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
2-Aminoethanol 3-Aminopropionic acid 1-Aminopropan-2-ol Diethanolamine |

