2-Pyrrolidone: A Closer Look at a Versatile Chemical
Historical Development
Looking into the story of 2-Pyrrolidone, it's interesting to see how one simple molecule can influence so many industries. Chemists first synthesized it in the late nineteenth century. It didn’t really make a splash until the wave of post-war chemical research got rolling. Expanding polymer science, drug synthesis, and solvent demand all played a part. Lab benches from Germany to the United States started churning it out as researchers realized its backbone shows up naturally in some alkaloids. Years later, industry started scaling up production. This wasn’t just another lab curiosity — it gradually became a staple in large-scale chemical manufacturing.
Product Overview
2-Pyrrolidone packs a punch far beyond its simple ring structure. This five-membered lactam opens possibilities in pharmaceuticals, high-performance polymers, agrochemical synthesis, and electronics. Its structure gives it a unique place among amides and lactams. Unlike bigger or more reactive molecules, 2-Pyrrolidone offers solid performance as a base building block. Folks making drugs like racetams or certain antibiotics often reach for it, drawn by its chemical stability and reliable behavior. It’s not flashy—but that reliability is exactly why it shows up in so many places.
Physical & Chemical Properties
Chemically, 2-Pyrrolidone stands out for its ability to blend polar and non-polar characteristics. It looks like a colorless to faintly yellow liquid at room temperature. Its mild but distinct odor isn’t hard to spot once you’ve worked around it. It dissolves in water and mixes easily with most organic solvents. With a melting point near 24°C and a boiling point just under 250°C, you run into it as both liquid and solid depending on storage. Its ring structure makes it a natural at hydrogen bonding, which contributes to its solvent abilities. Having spent time in a chemical lab, I can say that its low viscosity and chemical inertness makes life easier during synthetic workups where consistency counts.
Technical Specifications & Labeling
Anyone working with 2-Pyrrolidone knows purity needs to be above 99 percent for pharmaceuticals or electronics. Commercial samples usually arrive in tightly sealed containers, labeled to reflect hazard class and purity grade. Label accuracy means a lot here—a misstep can scrap an entire project or risk worker safety. Regulatory agencies treat it with care, classifying it under chemical control lists in some countries. Every batch gets documented with its chemical abstract number, lot number, and standard hazard warnings. In my experience, the best suppliers don’t cut corners on documentation, and those extra steps can save a project from an expensive setback.
Preparation Method
Producing 2-Pyrrolidone requires some careful handling. A common industrial route starts from γ-butyrolactone reacted with ammonia. Chemists run this under pressure and controlled heat, balancing reaction time against purity and yield. Over the years, tweaks to catalysts and operating conditions pushed yields higher and pollution lower. Small-scale labs can make it with simpler apparatus, but bigger plants need closed systems to control emissions and waste. Safety procedures have become more strict, reflecting both environmental expectations and workplace hazards. As a chemist, I see how cleaner synthesis routes, often driven by public pressure or regulatory changes, keep raising the bar each decade.
Chemical Reactions & Modifications
2-Pyrrolidone serves as a platform for a raft of chemical modifications. You can knock a methyl group onto it, swap around the nitrogen substituents, or open the ring for making other lactams. Its versatile ring makes nucleophilic attacks and ring-opening reactions straightforward. Researchers lean on it as a source compound for drugs such as piracetam, nootropics, and more. Polymers like polyvinylpyrrolidone rely on it as a critical monomer. These transformations show why its demand holds steady: industries can hang a lot of functional groups off this base structure. From my work, chemical flexibility always wins out in the long run, especially when the backbone remains stable through tough process conditions.
Synonyms & Product Names
Even a workhorse chemical like 2-Pyrrolidone picks up nicknames and aliases over the years. Some folks call it 2-pyrrolidinone or just pyrrolidone. In the research literature, N-pyrrolidone shows up too. As companies develop formulations or package it for specific sectors, branded names appear, each nudging at their target markets. Despite the variety, the core structure stays the same. Reading technical papers, you quickly pick up on these synonyms—missing one means missing past research or useful formulation tips tucked away under a different name.
Safety & Operational Standards
Working with 2-Pyrrolidone doesn’t invite the hazards of truly volatile organics, but that doesn’t mean you can skip the gloves and goggles. Its low volatility means less risk of inhalation, yet skin and eye irritation remain a risk without protection. Industrial users set up proper venting and strict supervision during transfer and reaction steps. Regulatory bodies set occupational exposure limits, though they can vary by region. Labs keep safety sheets close; training on accidental contact protocols stands as routine. Having overseen safety briefings, I always remind newcomers that “harmless” solvents can turn tricky if ignored or mishandled over long periods.
Application Area
Applications for 2-Pyrrolidone cut across sectors. In pharma, it acts as a base for nootropics, antiepileptics, and antibiotic precursors. Paint and coating manufacturers prize its solvent power, which helps to dissolve synthetic resins that resist almost everything else. Textile industries use it for dye affinity and processing. Electronics manufacturers value it for high-end cleaning processes and as a precursor for photoresists. Agrochemical synthesis leans on it for stability and reactivity when other solvents struggle. For several years, I’ve seen its role expand as researchers uncover new ways to tweak its backbone, leading to novel drugs and specialty solvents.
Research & Development
Labs worldwide continue tweaking 2-Pyrrolidone, searching for better synthesis methods and new uses. Researchers test greener catalysts or bio-based production routes, hoping to lower environmental footprints. Academic teams publish every year on new derivatives, enhanced polymer blends, and pharmaceutical leads built on the pyrrolidone backbone. Collaboration between industry and universities keeps ideas flowing, and new patents emerge at a steady pace. Experienced chemists like me know that research pushes sometimes hit slow patches, but the persistence pays off with practical answers to production bottlenecks or environmental hang-ups.
Toxicity Research
Toxicologists have dug deep into how 2-Pyrrolidone acts in living systems. Short-term exposures seldom cause serious harm in humans, but repeated skin contact causes irritation or even dermatitis. Inhalation presents less risk, but accidental ingestion leads to nausea and stomach upset. Rodent studies show no clear evidence of carcinogenicity or reproductive harm, yet researchers flag a need for continued long-term studies. Industry and regulators agree—better safe than sorry. Waste treatment and emissions controls remain high priority. I’ve seen changes ripple through facilities as each new study prompts tighter controls—nobody welcomes a surprise finding when it comes to chemical safety.
Future Prospects
The future for 2-Pyrrolidone follows a familiar path shared by many chemicals rooted in over a century of use. Demand looks steady, but new regulations and consumer expectations around green chemistry will push suppliers to find cleaner synthesis routes and more recycled starting materials. In pharmaceuticals, its backbone remains a launch-point for drug designers chasing novel therapeutics. High-end electronics and photolithography could see more custom uses. Sustainable production and waste reduction will only grow more important. Having seen industry bounce back from tougher transitions, I believe researchers and manufacturers can evolve with the times—if history teaches anything, it’s that technical challenges can unlock genuinely useful innovations.
What is 2-Pyrrolidone used for?
More Than a Chemical Name
Not everyone recognizes the name 2-pyrrolidone, but most of us rely on stuff made with its help every day. It doesn’t stand out in lab coats and goggles, though its influence shapes dozens of modern industries. This compound, with its five-membered ring, deserves more attention for everything it helps make possible.
Printing and Inks: Hidden Ingredient on Your Paper
Most home printers turn out sharp documents partly thanks to 2-pyrrolidone. Its solubility with water and organic solvents allows inkjet inks to flow smoothly through tiny nozzles, sticking well to paper fibers. People expect printer ink to stay put and not smudge, especially when handling forms or photos. That reliable ink performance often comes from this quiet helper, not just the brand name on the cartridge. In an office where printers never sleep, the right ink formula is key to keeping things moving.
Medicine Makers Count on It
Pharmaceutical workers use 2-pyrrolidone as a building block for many drugs, including antiseptic agents and some antibiotics. Its chemical flexibility allows it to blend with other ingredients, making pill coatings easier to swallow or helping injectable drugs stay stable until needed. I remember shadowing a pharmacist who showed that a drug’s effectiveness rests not only on its active ingredient, but on the helpers mixed in. Keeping patients safe requires strict quality controls, especially in drug manufacturing. This is where the experience of chemists and the track record of 2-pyrrolidone really matter.
Plastics with Staying Power
Nylon sucks up a lot of 2-pyrrolidone during manufacturing. The familiar strength of nylon stockings, car parts, and fishing lines partly comes from this compound feeding the polymer chains. Nylon-4, in particular, depends on it to get the flexibility and toughness it’s known for. My uncle worked at a tire factory and used to talk about finding fiber blends that resisted wear. Learning later how much the right chemical feedstock impacts daily products adds value to every shopping trip for tools, shoes, and even outdoor sports gear.
Cleaner Surfaces, Simpler Formulas
Lots of household cleaners and industrial solvents feature 2-pyrrolidone. This chemical dissolves tough stains or lubricates metal, which means you find it behind that stubborn grease-cutting power under the sink. In electronics, circuit board cleaners often depend on it to wipe away residue. From my own experience fixing laptops, I’ve seen how removing flux or ink smears takes more than elbow grease—it calls for the right cleaner. Too harsh, and you damage parts. Too weak, and nothing happens.
What’s Next?
More people want green chemistry and safer workplaces. 2-pyrrolidone’s low toxicity and high biodegradability help address those demands. Still, strict storage rules and safe handling stay important, since any chemical can cause issues if misused. Factories can look at closed systems and better training, cutting down on waste and keeping workers healthy.
Better chemistry doesn’t just mean more effective products or smoother industrial processes. It means thinking about human health and the planet at every step—right down to the helper compounds like 2-pyrrolidone.
Is 2-Pyrrolidone safe to handle?
Looking Beyond the Labels
2-Pyrrolidone pops up in a surprising range of workplaces. People blend it into pharmaceuticals, cleaning products, and industrial fluids. It dissolves plenty of things with ease, which makes it popular in labs and factories. If you stand in a manufacturing plant, the faint odor of 2-pyrrolidone will turn up where solvents and specialty chemicals cross paths.
Safety trends don’t come out of thin air. A chemical like 2-pyrrolidone goes through plenty of scrutiny, and organizations such as the European Chemicals Agency and the U.S. Environmental Protection Agency keep a close eye on its effects. They pay attention because this compound gets mixed regularly with human hands and factory hardware.
What Science Says About Health Risks
If you poke through published safety data, the story grows clearer. Touching 2-pyrrolidone can cause skin and eye irritation, and scientists who study chemicals under a microscope found it doesn’t just evaporate and disappear without a trace. Repeated or extended exposure sends people home with red hands and sore throats. Vapors get past dust masks, lodging deep in airways and irritating the system. The Material Safety Data Sheets drive the message home: avoid breathing vapors, keep splashes off your skin, and use it only in a spot with good airflow.
Acute hazards—those that show up right away—are fairly mild. Most workers deal with irritation when spills land on the skin or if fumes hang in the air. Chronic exposure still leaves many questions because few large-scale, long-term studies cover the risks from regular low-level contact. Toxicology reports often suggest no evidence for cancer or birth defects at levels found in workplaces, but real-life safety sometimes trails behind what lab tests reveal.
In the Shop, Not Just in the Lab
My own years around solvents taught a few lessons quickly. Gloves are cheap, and eye protection feels like overkill—until a splash proves how badly your day can turn. One time, a coworker found out the hard way after a tiny leak from a valve: red skin, questions about allergic reactions, and a call to the plant nurse. His case got documented, and safety protocols improved for everyone after that. Good ventilation moved from background advice to a daily checklist item.
Accidents rarely announce themselves. A slippery patch of 2-pyrrolidone on the floor turns standard shoe soles into skates. The stuff isn’t instantly dangerous, but repeated small slips, skin contact, or inhaled fumes pile up unseen risks over the months.
Practical Safety: Solutions Rooted in Experience
Nobody likes more rules for the sake of rules, but 2-pyrrolidone sticks out because safe systems pay off. Start with basics: nitrile gloves hold up well, better than old vinyl or latex ones. Safety glasses with side shields give you an edge. Work in places where fans move air and chemical fume hoods suck up vapors. Wash up soon after exposure, even if you can’t see a mark or feel anything out of place.
Managers who listen to their team learn more quickly than those who assume the book covers everything. A safety briefing feels like wasted time until the day you’re thankful for it. In shops where I saw good habits stick, spills got cleaned up fast and new employees learned from workers who wore their gear. Simple checklists and regular reminders made a difference. When everyone knows what they’re handling, they protect both themselves and others.
Why It Matters
2-Pyrrolidone won’t cause a disaster by itself, but careless habits add up. Real-world safety goes beyond the label and the chemical registry. People who respect the risk end up avoiding headaches—both real and metaphorical. In the right hands, with a good eye on the basics, 2-pyrrolidone stays a helpful tool and doesn’t turn into tomorrow’s safety story.
What is the chemical formula of 2-Pyrrolidone?
Getting to Know 2-Pyrrolidone
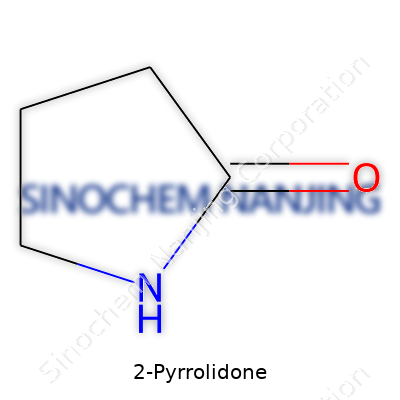
2-Pyrrolidone shows up in more places than most people realize. Its chemical formula—C4H7NO—reflects a quiet complexity: four carbons, seven hydrogens, a nitrogen, and a single oxygen atom. This blend of atoms forms a five-membered ring, kind of like a tiny molecular bracelet. That structure brings more to the table than just a catchy formula.
Why Understanding Chemical Structure Matters
For anyone reading a food package or searching for safe cleaning products, these chemical names might just blur together. Still, the makeup of a molecule like 2-Pyrrolidone shapes how it works in medicine, industry, and even daily living. Because of its polar nature, 2-Pyrrolidone dissolves both in water and organic solutions. In the lab, that makes it a versatile solvent. Science classrooms love it for this reason, and manufacturers use its unique properties when making polymers, pharmaceuticals, and even inks for your home printer.
Personal Experience and Broader Impacts
During my time working with research chemicals, I grew to appreciate how each atom twists and turns a molecule’s abilities. C4H7NO isn’t just textbook knowledge. Back in the day, while testing biodegradable plastics, 2-Pyrrolidone offered a starting point for N-vinylpyrrolidone—another mouthful, but an item on ingredient lists that signals safer products compared to old petrochemical standards. These connections mean something, especially for families looking out for environmental impacts and health.
Industry Uses: From Pharma to The Everyday
Pharmaceutical labs synthesize medicines from 2-Pyrrolidone because it reacts cleanly and predictably. A lot of the pain relievers and antibiotics on shelves rely on blocks built with the help of this molecule. Its role stretches beyond pills. Inkjet printers keep homes and schools running, and many rely on water-soluble ink bases made with 2-Pyrrolidone. Even in agriculture, safer pesticide formulations often include this stuff, since it helps active ingredients get where they belong without lingering in the soil.
Health and Safety: Clear Information Matters
With more chemicals touching our lives, transparency remains key. Knowing C4H7NO by its actual chemical formula helps researchers and public health experts track its effects and avoid confusion with other, more harmful compounds. Consumers can make more informed choices seeing a straightforward label on a product. Exposure to small amounts of 2-Pyrrolidone poses low risk, supported by decades of safety data, yet ongoing review ensures no surprises for those who use these products every day.
What Can Be Improved
Perhaps clearer labeling and more accessible fact sheets would help consumers and workers alike. Universities and manufacturers could join up to provide community classes about basic chemical safety and applications. Regulators might encourage regular updates on new research, making sure the public stays in the loop as products change. In my own experience, the best results come when experts, watchdog groups, and average people get honest about what chemicals do, where they show up, and how to handle them safely.
How should 2-Pyrrolidone be stored?
Before You Stash That Drum: Why Storage Really Matters
I’ve spent years watching people handle chemicals and, too often, it’s the small stuff that trips us up. Everyone focuses on big spills or fires, but sometimes overlooking basic storage practices leads to waste, accidents, or ruined product. With 2-Pyrrolidone, a common enough solvent and intermediate in the pharma and electronics industries, a bit of care up front makes all the difference.
The Role of Containers and Temperatures
Storing 2-Pyrrolidone starts with the right container. Manufacturers typically supply it in sealed steel, HDPE, or glass containers. These choices help keep moisture out and discourage any unwanted reactions. I’ve seen open-mouthed jugs used in a few shops, and they don’t do any favors for anyone dealing with contamination or evaporation.
The biggest concern is water. 2-Pyrrolidone soaks up moisture from the air. Once that happens, the quality suffers. The shelf life shortens, there’s a higher risk of microbial growth, and in sensitive applications, your whole batch might wind up unusable. Keep it tightly sealed between each use — even a small gap adds up over time and opens the door to problems.
Room Temperature Isn’t Always Safe Enough
Walk into a storage room in mid-summer and sometimes you’ll feel the warmth before you see the barrels. 2-Pyrrolidone, though not a volatile solvent like acetone or ether, still suffers from prolonged exposure to heat. Ideally, storage stays below 25°C (77°F). Warm conditions encourage slow breakdown, and some impurities form faster. If the location hits higher temperatures, product quality drops, and odors or color changes become more likely.
I once saw a warehouse storing solvents right by a sunny window, and half those drums cooked from the inside out. Even if unopened, 2-Pyrrolidone won’t stay fresh in a hot space. Finding a cool, shaded spot or — for larger volumes — a climate-controlled warehouse saves a lot of money and trouble. Don’t store it near furnaces, heaters, or anywhere heat sources might surprise you.
Keep It Away from Trouble
No chemical exists in a vacuum. Accidental mixing happens. 2-Pyrrolidone doesn’t react violently with most common materials, but strong oxidizers or acids can cause a mess, and sometimes a hazard. It’s safer to keep 2-Pyrrolidone away from such chemicals. If it leaks and finds its way into incompatible drums, shelf reactions follow, sometimes dangerous ones. Physical separation goes a long way — at least a few meters or, in tight spaces, good barrier management prevents mistakes even on busy days.
Don’t Forget Labeling and Documentation
Proper labeling stays non-negotiable, especially when multiple clear liquids sit side by side. Labels fade, so check now and then. Keeping updated logs helps trace any quality issue back to its source and supports overall safety. Failing to account for shelf life or shipment dates turns into confusion in production, and that’s where the biggest errors start. Many businesses now use digital inventory, making real-time tracking something anyone can manage.
Simple Solutions Make Big Impacts
Biggest changes start with small choices: keep containers sealed, choose proper storage temperature, separate from other chemicals, and double-check labels. For any company or lab using 2-Pyrrolidone, these steps cut down on cost, reduce risk, and help protect everyone involved. Watching out for one chemical helps build habits that transfer to safer handling everywhere else.
What are the physical properties of 2-Pyrrolidone?
Straightforward Profile of 2-Pyrrolidone
2-Pyrrolidone doesn’t get much limelight outside labs and manufacturing plants, but anyone who’s handled solvents, plastics, or pharmaceuticals should care about how it behaves. This chemical usually appears as a colorless liquid, although anyone giving it a whiff will notice its faint, but distinctive, odor that brings to mind ammonia or a stuffy storeroom. It feels slick between your fingers (not that touching pure solvents is a great idea). With a melting point near 25°C, you’ll see it go from solid to liquid right around room temperature. If your workbench is a couple degrees cooler, you might spot some crystals forming in the bottle.
What Temperature Reveals
Boiling comes into play a few lab tasks down the line. 2-Pyrrolidone boils at about 245°C, which puts it in a different league from water or ethanol. That’s one reason it sticks around in high-heat reactions when other solvents would vanish. It also means folks using it in polymer production systems have a wider processing window, less chance of product evaporating out of nowhere, and fewer headaches with handling equipment.
Water Relationships and Solubility Tricks
I still remember a spill on a lab bench, how fast it dissolved the dye we were testing: 2-Pyrrolidone blends perfectly with water. It also loves to mix with common organic solvents—alcohols, chloroform, and similar chemicals. People working with drug delivery systems or special coatings pay close attention here, since its blend-ability (as some old-timers put it) allows for stable mixes, clean dispersions, and fine-tuned product formulation.
Density and Flow
You’ll notice 2-Pyrrolidone pours slower than plain water. Weighing in at about 1.1 grams per cubic centimeter at standard conditions, it’s denser than you’d expect. Handling drums or bulk containers demands care, especially for operations scaling up from bench to pilot plant, where a surprise in mass calculations can lead to costly missteps. This also plays into how it flows through pipes and pumps—valuable to anyone setting up a process in a chemical plant.
Viscosity, Polarity, and Getting Down to Work
The viscosity of 2-Pyrrolidone sits in the Goldilocks zone: thicker than water, much thinner than glycerol. This property comes up when making adhesives or spinning synthetic fibers because it affects how evenly materials spread or draw. Its polar nature lets it disrupt stubborn mixtures and makes it useful in specialty solvents and intermediates. Those making pharmaceuticals rely on this trait for dissolving complex drug ingredients, controlling release times, and boosting absorption.
Practical Safety and Storage
2-Pyrrolidone can absorb water from the air, which changes how it acts in some applications. Anyone storing it for months learns to keep the caps tight and containers dry to maintain its reliability. Direct skin contact isn’t wise, since it readily soaks through and brings along whatever it’s dissolved. Proper gloves and ventilation gear do the trick. In shipping or warehouse settings, labeling and careful handling avoid needless health risks.
Connecting Chemical Properties With Everyday Use
Knowing these practical details has real value: pharmaceutical techs, chemists making specialty plastics, and ink formulators sidestep plenty of headaches by respecting how 2-Pyrrolidone melts, dissolves, flows, and reacts to water. It’s easy to overlook physical properties until a batch fails or equipment clogs. Reliable information and hands-on experience keep the wheels turning in the right direction.
| Names | |
| Preferred IUPAC name | Pyrrolidin-2-one |
| Other names |
1,2-Pyrrolidone Pyrrolidin-2-one Butyrolactam Gamma-aminobutyric acid lactam 2-Oxopyrrolidine |
| Pronunciation | /paɪˈrɒlɪˌdoʊn/ |
| Identifiers | |
| CAS Number | 616-45-5 |
| 3D model (JSmol) | `/data/structures/3d/2Pyrrolidone.jmol` |
| Beilstein Reference | 58338 |
| ChEBI | CHEBI:18849 |
| ChEMBL | CHEMBL14258 |
| ChemSpider | 797 |
| DrugBank | DB04616 |
| ECHA InfoCard | 02bb6b823d-41a8-4f37-9442-81a760fd2793 |
| EC Number | 210-483-1 |
| Gmelin Reference | 8827 |
| KEGG | C01785 |
| MeSH | D011693 |
| PubChem CID | 6989 |
| RTECS number | UY4375000 |
| UNII | 95F0U2S440 |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | 2-Pyrrolidone CompTox Dashboard (EPA): "DTXSID4044361 |
| Properties | |
| Chemical formula | C4H7NO |
| Molar mass | 85.11 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Faint amine-like |
| Density | 1.116 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | -0.85 |
| Vapor pressure | 0.029 hPa (20 °C) |
| Acidity (pKa) | 20.5 |
| Basicity (pKb) | -1.1 |
| Magnetic susceptibility (χ) | -7.48×10⁻⁷ |
| Refractive index (nD) | 1.483 |
| Viscosity | 34.7 mPa·s (25 °C) |
| Dipole moment | 4.09 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 138.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -443.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2212.7 kJ/mol |
| Pharmacology | |
| ATC code | N01AX10 |
| Hazards | |
| Main hazards | Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, Warning |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319 |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 87°C (closed cup) |
| Autoignition temperature | 315 °C |
| Explosive limits | 3% - 38% |
| Lethal dose or concentration | LD50 Oral Rat 6500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 6500 mg/kg |
| NIOSH | PYR50000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
N-Methyl-2-pyrrolidone Pyrrolidine Gamma-Butyrolactone Succinimide |

