The Story of 2-Nonanone: Beyond a Simple Chemical
Historical Development
2-Nonanone has a story stretching back to the days when chemists first started probing the flavors and scents of plants. This molecule gained attention mostly because of its role in the smell of fruits and some flowers. Early researchers noticed its distinct, slightly fruity aroma, setting it apart from other ketones present in nature. As analytical techniques improved, labs could spot it in complex mixtures, bringing a growing awareness of its function beyond just an obscure compound. Chemists in the early twentieth century began isolating it from essential oils and then engineered routes to synthesize it, pushing the molecule from perfume curiosity to commercial relevance.
Product Overview
2-Nonanone shows up as a pale, oily liquid with a recognizable odor, often described as reminiscent of pears or blue cheese. This specific ketone, also known as methyl heptyl ketone, appears both in nature and in synthetic mixtures. Its appeal to the fragrance, flavor, and chemical industries has grown steadily as companies learned to harness its unique scent profile. In food, it mimics ripe fruit, while perfumers use subtle amounts to add complexity to blends. The compound serves as an important building block for other chemicals and flavoring agents, helping to create both simple and sophisticated end-products.
Physical & Chemical Properties
2-Nonanone’s structure grants it a nine-carbon backbone capped with a ketone group at the second position. This gives the molecule a boiling point around 191°C, placing it at a sweet spot between volatility and persistence. It dissolves readily in organic solvents such as ethanol and ether, but shows little interest in water. That pattern feels familiar in organic chemistry, where chain length and functional groups steer solubility. The molecule clings to surfaces as a thin film, ideal for evaporation in aroma applications, and resists rapid breakdown under normal conditions.
Technical Specifications & Labeling
People working with 2-Nonanone in industry follow strict standards for purity, labeling, and documentation to keep quality predictable. These rules draw from international chemical safety codes and their local interpretations. High-quality material typically ranges above 98% purity, filtered for physical contaminants and any volatile impurities. Clear labeling marks the molecule’s identity, CAS number, appropriate hazard warnings, and handling tips. Reliable suppliers stand by such specs so professionals can handle the chemical safely and consistently every time.
Preparation Methods
Traditional organic synthesis produces 2-Nonanone by combining easily accessible starting materials such as heptanone with suitable methylating agents. One common method relies on the oxidation of secondary alcohols or the careful manipulation of Grignard reagents reacting with shorter-chain esters. These multi-step syntheses take patience and experience, especially as chemists scale for commercial runs. Advances in green chemistry and catalysis have started reshaping preferred approaches, as industry demands not just efficiency but smaller environmental footprints.
Chemical Reactions & Modifications
2-Nonanone acts as a handy intermediate in organic synthesis. Its reactive carbonyl group invites nucleophilic additions and reductions, making it a useful starting point for producing secondary alcohols, amines, or even longer-chain derivatives. Labs use 2-Nonanone to explore tailored scents, alcohols, and acids that demand a stable backbone but versatile chemistry. The ketone group can undergo condensation reactions, connecting it with other building blocks for pharmaceutical or fragrance compounds.
Synonyms & Product Names
Chemists may spot 2-Nonanone listed under a variety of names, such as methyl heptyl ketone or simply Nonan-2-one. These synonyms reflect its structure and traditional nomenclature. In trade catalogues, the molecule often appears as a fragrance ingredient or intermediate, but its unique chemical identity stands out through consistent CAS registration.
Safety & Operational Standards
Any lab or factory that touches 2-Nonanone pays close attention to safety. The molecule brings a flammable vapor that demands careful ventilation and grounded containers. Operators rely on gloves, goggles, proper storage, and up-to-date Material Safety Data Sheets. Spills need swift cleanup, and inhaling concentrated fumes leads to headaches or irritation. Fire risk rises with open flames or static, so processes rely on grounded equipment and proper exhaust. Training keeps employees familiar with emergency procedures and accident prevention.
Application Area
2-Nonanone benefits a wide range of sectors. Its most recognizable use sits in flavors and fragrances, where even small doses create a punchy, distinctive note. Food manufacturers rely on it to mimic the smell and flavor of blue cheese, some fruits, and select bakery products. Perfumers blend it into formulations calling for an unexpected twist or natural undertones, taking advantage of its pleasant but noticeable aroma. The chemical works as a solvent in analytical chemistry and sometimes serves as a reference standard. Some pest control companies explore its power to confuse insect olfactory systems, providing groundbreaking greener options to traditional pesticides.
Research & Development
Research keeps pushing 2-Nonanone into new territory. Universities and start-ups examine its value for medical testing where volatile organic markers help diagnose disease. Other groups investigate its presence as a component in plant-fungi interactions, adding insight into ecosystem chemistry. There’s active work on bio-based synthesis routes to shrink carbon footprints and new formulations in luxury perfumes that rely on its nuanced flavor. Analytical chemists develop faster, cheaper routes for detecting trace levels in food, soil, and water—supporting both quality control and regulatory compliance.
Toxicity Research
Toxicologists keep a close eye on molecules like 2-Nonanone. Studies indicate moderate acute toxicity with high exposure, leading to irritation of eyes, throat, and central nervous system upset. Regulatory bodies set workplace exposure limits to protect workers handling the chemical. Long-term data remain limited, but animal studies have not shown strong evidence of carcinogenic effects when used as directed. Careful handling and best practices in storage and ventilation help reduce risks in real-world settings. Consumer products using trace levels follow stringent food safety and fragrance guidelines.
Future Prospects
2-Nonanone’s future looks bright for those willing to innovate. Industrial players work hard to streamline greener production routes, using renewables and catalysts to cut waste. The search for sustainable solvents and next-generation fragrance molecules has only intensified, pushing this versatile ketone into the spotlight. Pest management researchers continue testing its powers for insect control, chasing the promise of selective, eco-friendly repellents in agriculture and urban settings. Meanwhile, scientists mapping air quality and food safety contribute fresh data that allow regulators and users to keep up with evolving risks. It’s that blend of utility, novelty, and obligation to safety that keeps 2-Nonanone on the radar, and it doesn’t seem likely to disappear from innovation pipelines anytime soon.
What is 2-Nonanone used for?
Where Everyday Life Meets Science
Walk down the cleaning aisle, cut into a banana, or stroll through a garden after the rain and you’re picking up hints of chemicals that rarely get named. 2-Nonanone is one of those hidden characters. This molecule shows up where scent and flavor really matter, nudging changes behind the scenes rather than stealing the spotlight.
Fragrance and Flavor: More Than a Pleasant Note
Food scientists reach for 2-Nonanone when they want to dial up the creamy, fruity notes in things like strawberries, tomatoes, and cheese. It’s listed as a “flavoring agent,” but that undersells the role it plays every day. Trace amounts in bananas or blue cheese give those foods their irresistible, natural smell. Its subtlety tricks our brains into thinking, “Yes, this is the real thing.” Studies show the profile 2-Nonanone brings can make processed foods feel fresher or higher quality, which explains why food companies watch production quality so closely.
Perfumers, always searching for the next evocative top note, use 2-Nonanone to add delicate, almost coconut-like hints to a scent blend. Most people won’t notice the chemical itself, but they’ll feel the lift it gives a perfume’s opening.
Chemical Tools for Farms and Fields
Beyond the kitchen or dressing room, 2-Nonanone holds a surprising role in pest control. Some research points to its potential as a safe, plant-derived alternative to harsher synthetic pesticides. Tomato plants, for example, naturally release 2-Nonanone when caterpillars attack. Certain insects, like rootworm larvae, find it discouraging or repellent. Farmers and scientists think about these traits when figuring out how to protect crops with fewer side effects for the environment or people.
I grew up in a rural county where so many neighbors worked in agriculture. Reducing dependence on classic insecticides has always been a shared hope, for both health and cost reasons. Whether 2-Nonanone can reliably scale up enough as an organic deterrent is still under study, but it’s a promising area for those tired of chemical overload in our soil and water.
Industrial Cleaners and Safety Concerns
Factories making solvents and cleaning products take a different approach. 2-Nonanone dissolves oils and waxes without the aggression of some traditional cleaners. It can cut through stubborn grime on machinery or windows, which I’ve seen first-hand in old industrial kitchens. Workers tend to favor products that use less skin-irritating chemicals, and 2-Nonanone lands low on the skin allergy risk charts compared with many solvents.
Still, working safely with volatile organic compounds always deserves attention. The National Institute for Occupational Safety and Health (NIOSH) lists exposure limits to help protect those in manufacturing plants. No chemical comes risk-free, and consistent training and personal protective equipment stay important, no matter how mild the molecule sounds on paper.
Finding Solutions Through Awareness
It’s easy to ignore a name like 2-Nonanone, which slides into products quietly. Paying attention to these lesser-known chemicals, though, lies at the heart of more sustainable industry and safer consumption. Companies can take it further by disclosing not just the “active ingredient” but the subtle helpers behind better food, fewer pests, and less toxic cleaning formulas.
Is 2-Nonanone safe to handle?
What Is 2-Nonanone?
2-Nonanone pops up a lot in discussions about flavors and scents. It smells a little like fruit, shows up in some cheeses, and contributes to that earthy undertone in certain plants. Manufacturers like it in perfumes and cleaning products, and it even lands in the world of pest control since some bugs don’t appreciate its aroma.
Personal Experience Meets The Facts
My first real introduction to 2-Nonanone happened during a stint in a flavor chemistry lab. I learned to respect anything bottled with a lab label and a warning symbol. 2-Nonanone isn’t as notorious as benzene or toluene, but it's not something to splash around for fun either. Touching, inhaling, or swallowing it, according to the European Chemicals Agency and U.S. EPA, can cause trouble. Skin can feel irritated, and no one enjoys a strong chemical whiff up their nose.
A Safety Data Sheet lists advice you don’t want to ignore: work in a place with good air movement, use gloves that chemical suppliers trust, and put goggles over your eyes. Handling it with a sense of “this is just like vinegar” risks a lesson you won’t forget. Many people underestimate mild-smelling chemicals, but “pleasant” doesn’t mean “harmless."
How Harmful Is It If Exposed?
No one expects to get sick from one splash or small sniff, but it's easy to collect small exposures over time. 2-Nonanone can irritate lungs and eyes. If someone pours it without enough ventilation, headaches and dizziness can surprise them. That's a sign to rethink how chemicals are used at home or work.
Reports show industrial workers sometimes face issues by ignoring ventilation or forgoing gloves. Cleaning spills with bare hands feels quicker, but skin absorbs more than people realize. If someone handles chemicals every day, the risks stack up fast. Training makes a difference—schools and workplaces that actually walk people through Chemical Hygiene Plans usually see fewer accidents.
Strategies for Reduced Risk
The biggest step for anyone handling 2-Nonanone: treat it like it's more than a bottle of perfume. Labs run best with fume hoods, and even the most careful technician wears gloves. At home, anyone using a product with 2-Nonanone needs open windows and a spot far from kids and pets. Most cleaning and fragrance bottles lock away chemicals for a reason, so popping off safety caps should get attention.
Educating people makes a real difference. If someone doesn't understand the risks, they might skip important steps. Training is more than a box to check—it becomes a habit that protects hands, lungs, and brains. Looking at labels and following basic rules works better than risking a poison control call.
Disposal matters too. Dumping leftover chemicals in the sink or trash threatens water and soil. Community hazardous waste programs exist for a reason. I’ve seen towns unite to sponsor safe cleanups, and the turnout proves that education and access change habits.
Room for Improvement
Companies and governments both play a big part in chemical safety. Clear labeling, public access to safety data, and ongoing worker education pull a lot of weight. People need real-life stories and facts, not just warnings in tiny print. A better system means fewer emergencies and healthier communities. Those who respect chemicals like 2-Nonanone set an example worth following.
What are the physical properties of 2-Nonanone?
Stepping Into the World of 2-Nonanone
2-Nonanone shows up as a colorless liquid with a scent that brings to mind green bananas or even blue cheese. It’s found in a bunch of plants and foods, including tomatoes and certain cheeses. In my own experiences working in food science labs, opening a vial of this compound can take over a whole room with its aroma. These properties don’t just entertain the nose; they shape how 2-Nonanone gets used in flavors, fragrances, and even pest control.
Key Physical Properties
The boiling point of 2-Nonanone lands around 191 to 193 degrees Celsius. This means it doesn’t disappear into the air as quickly as lighter ketones. In real life, that means a sample left open on a bench will be around for a while before it evaporates completely. This quality makes it handy for scent products or substances that need a steady release over time.
It carries a density close to 0.82 grams per cubic centimeter. If you’ve ever measured liquids in a laboratory, you know that this feels lighter than water but heavier than most solvents found in your regular paint thinner. Pouring it gives you a sense of something a bit slick, a little less runny than alcohol but by no means syrupy.
The melting point sits between -18 and -22 degrees Celsius. It means 2-Nonanone stays liquid under most storage and transport conditions. Even in a cold storage warehouse, you’d find this substance flowing just fine. That comes with a practical advantage in flavor houses and chemistry labs: no need for elaborate heating equipment to get the bottle ready to pour.
2-Nonanone is only slightly soluble in water, with a solubility of around 0.6 grams per 100 milliliters at room temperature. So if you drop some into a glass of water, you’ll see it float and form droplets rather than mix in completely. But it blends easily with alcohol, ether, and many oils, something that’s key if you’re working in perfumery or food science where blending into fatty or oily bases is part of the job.
Why These Properties Matter
Physical properties like boiling and melting point influence how 2-Nonanone is stored and shipped. Strong scent and low solubility in water also shape how this compound works in products that have to survive on a shelf for months. In the field of food science, managing volatility and layering flavors is all about balancing evaporation rates and persistence of aroma. In pest control, a compound that lingers in the air without dissolving away in rain expands its reach.
These properties also come with risks. A low flash point close to 68 degrees Celsius means there’s a fire hazard in hot environments. Personal safety needs attention—nitrile gloves and good ventilation are a must in any lab setting, not just in a chemical plant. If you spill 2-Nonanone, its smell will linger and become obvious, making cleanup pressing from both a safety and comfort perspective.
Practical Solutions for Uses and Handling
Anyone storing or using 2-Nonanone can cut down on risks by using flame-proof storage and well-sealed containers. Its low solubility in water makes spill response easier; simple absorbent materials work well, unlike with more water-soluble compounds that seep into every crevice. In work environments I’ve been a part of, labeling and prompt cleanup matter just as much as fancy engineering controls.
The need for personal protective equipment and fume extraction isn’t academic. Even brief exposure to vapors can lead to headaches and nausea. That’s why limitations on exposure in workspace air currents have roots in both personal stories and regulatory science. Keeping staff trained, and supplying simple items like goggles and gloves, builds a safer workplace together with smart engineering controls.
How is 2-Nonanone stored and shipped?
Why Storing 2-Nonanone Safely Isn’t Optional
Anyone who’s spent time around industrial chemicals knows things like 2-Nonanone get nowhere close to a kitchen cabinet. This clear liquid, with its faint fruity odor, ends up in flavorings and fragrances, but also lands on chemical hazard lists for a reason. Storing it means facing real risks: spill hazards, fumes that hit the nose hard, and the threat of fire if the wrong spark shows up. 2-Nonanone can ignite more easily than many folks realize—its flash point is around 66°C (151°F), which sits squarely in the “careful now” zone.
Drums or steel containers keep the liquid locked away from sunlight and heat. Not every depot out there pays attention to humidity and temperature, but with 2-Nonanone, letting things slide could invite accidents. Proper ventilation goes a long way too. Years back, I visited a plant that put their solvent drums behind a fenced-off shed, but skipped venting it. The odors built up quick. Some workers got headaches, others felt woozy. No one wants an “incident” report blamed on something simple like opening a window.
Loose Drums and Loose Ends: What’s Wrong With Shortcuts?
The cost of shaving a corner on containment makes headlines every year. Leaky barrels, weak seals, or stacking too high—these ordinary mistakes spill chemicals into soil and water, and nobody wants that on their watch. A recent EPA summary went after several warehouses that let solvents creep into the ground. The fines topped six figures. Cutting costs can cost a small business everything.
Standard steel drums with proper labeling and fully sealed tops stand up to the journey better than plastic options. They don’t leak as easily and hold up against bumps and scrapes. Any time a shipper ignores secondary containment—those catch-basins that sit under drums—they’re hoping for luck. In my warehouse days, losing an hour to mop an inch of solvent off the floor taught me fast: double-check seals, keep containers upright, and treat every spill like a five-alarm blaze, even if the odor barely lingers.
Shipping: More Than “Just Load and Go”
Trucking chemicals like 2-Nonanone comes with “HazMat” labels for a reason. A real transit plan means using trained drivers, not the neighbor’s pickup. DOT regulations require clear signage, documentation, and regular checks. Some companies still try shortcuts, hoping the odds land their way. Regulations don’t serve as red tape, they offer basic protection. Out on a July highway, a steel drum in the wrong spot on a flatbed can heat up, pop a bung, and spill. Even a minor fender-bender becomes national news when chemicals get loose.
Between warehouse and loading dock, communication keeps mistakes in check. Drivers who know the risks, warehouse workers who treat every drum as hazardous, and folks who read the shipping paperwork—all reduce the chances of disaster. Early in my career, I saw a team catch a mislabeled drum before it left for a food plant. That one check may have saved more than hourly wages; it could’ve protected the health of hundreds down the supply chain.
Solutions Everyone Can Live With
Tight training, proper containers, and full transparency help everyone sleep better at night. Investing in emergency kits and keeping clear spill control processes gives teams an edge. A solid system not only reduces chemical waste, it keeps people safe, keeps fines away, and keeps 2-Nonanone on the right side of the warehouse wall—headed where it’s meant to go, not loose on the ground.
Good stewardship writes its own story; slip-ups make the headlines. Keeping 2-Nonanone where it belongs takes more patience than muscle, and rewards those who treat safety as an everyday value.
What industries commonly use 2-Nonanone?
Fragrances: More Than Meets the Nose
Step inside a perfume lab or a candle-making factory, and you’ll bump into 2-Nonanone right away. This compound offers a dry, fruity scent that helps round out fragrances. People want smells that tug at memory and comfort. Sometimes that's a hint of fruit, maybe a trace of earth. 2-Nonanone nails those notes, finding its place in both high-end perfumes and the air freshener hanging from your rear-view mirror. I’ve met folks in fragrance development who lean on its reliability—it won’t overpower the blend, but it adds something warm and familiar. Even cleaning products quietly benefit, smoothing out harsh chemical smells in floor cleaners and sprays.
Flavor Industry: A Quiet Backbone
Move over to the world of food flavoring, and you’ll see 2-Nonanone popping up in tiny but important amounts. It’s added to foods for that subtle blue cheese tang, a touch of earthy green in potato chips, or extra complexity in a dairy-flavored treat. Food scientists study this molecule closely because it’s approved as a flavoring agent. Their job centers on making processed foods taste a bit more like the real thing, and small doses of 2-Nonanone help trick the brain just enough. There's always a careful eye on safety; regulatory bodies like the FDA and EFSA have signed off on it, but flavorists don’t push their luck and stick to amounts far below reported thresholds.
Pesticides: Keeping Crops Safe
Head over to agriculture, and 2-Nonanone fills a different role. This compound pops up in natural pest control products, where it acts as an insect repellent. Researchers have found that bugs back away from it, so it’s been embraced by growers looking to avoid the heavier touch of synthetic chemicals. Some organic farms turn to it as a less toxic choice, aiming for enough crop protection without coating fields in nastier substances. From my conversations with folks in sustainable farming, it’s clear that having extra tools like this makes life a lot easier when fighting pests that don’t quit.
Laboratory Uses: Getting to the Core
Every once in a while, chemists need markers or standards—compounds that show up reliably and clearly. 2-Nonanone stands out as a reference in chromatography work. In labs, I’ve seen analysts reach for it when checking instrument performance, running checks in forensic, food safety, or environmental labs. Without these common compounds, the data would start to wobble, and results might not hold up under scrutiny.
Potential Challenges and The Path Forward
With all these uses, you’d expect high demand. Getting 2-Nonanone to market consistently has real challenges. Sourcing it from petroleum or synthetically-building it can turn expensive, especially as industries look to green up their operations. Some startups hunt for ways to make it through fermentation or extraction from plant waste—hoping for both lower cost and cleaner credentials. Lab safety also matters. Inhalation or skin contact brings risks, so workplaces must train their teams and keep exposure to a minimum.
People want products that smell nice, taste familiar, and harm less. 2-Nonanone plays a small but vital role in making those things happen, proving that even obscure-sounding chemicals carry weight far beyond their odd names.
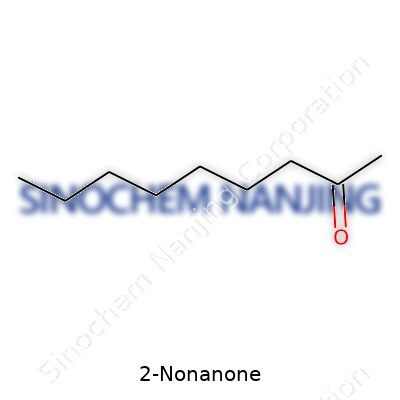
| Names | |
| Preferred IUPAC name | Nonan-2-one |
| Other names |
Methyl heptyl ketone Heptanoyl ethide |
| Pronunciation | /tuː-noʊˈneɪ.oʊn/ |
| Identifiers | |
| CAS Number | 821-55-6 |
| Beilstein Reference | 1381666 |
| ChEBI | CHEBI:37640 |
| ChEMBL | CHEMBL15836 |
| ChemSpider | 6826 |
| DrugBank | DB02168 |
| ECHA InfoCard | 02a1c6a9-44e5-47e6-9bbf-9d6b3e6c02ba |
| EC Number | 203-937-5 |
| Gmelin Reference | 7879 |
| KEGG | C06504 |
| MeSH | D009648 |
| PubChem CID | 8103 |
| RTECS number | SA9100000 |
| UNII | G67I7N67OB |
| UN number | UN 1993 |
| Properties | |
| Chemical formula | C9H18O |
| Molar mass | Molar mass: 142.24 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity; floral; herbaceous; green |
| Density | 0.819 g/mL at 25 °C (lit.) |
| Solubility in water | 1.56 g/L (20 °C) |
| log P | 3.2 |
| Vapor pressure | 0.286 mmHg (25°C) |
| Acidity (pKa) | pKa ≈ 20. |
| Basicity (pKb) | pKb = 15.2 |
| Magnetic susceptibility (χ) | -7.11 × 10⁻⁶ |
| Refractive index (nD) | 1.417 |
| Viscosity | 2.60 mPa·s (25 °C) |
| Dipole moment | 2.87 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 328.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -320.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5645.8 kJ/mol |
| Pharmacology | |
| ATC code | N01AX13 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319, H336 |
| Precautionary statements | P210, P264, P280, P301+P312, P305+P351+P338, P337+P313, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 102°C |
| Autoignition temperature | 198 °C |
| Explosive limits | 1.2% - 7.6% |
| Lethal dose or concentration | LD50 oral rat 3200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 3200 mg/kg |
| NIOSH | NIOSH: RN3675000 |
| PEL (Permissible) | 100 ppm |
| IDLH (Immediate danger) | 500 ppm |

