Understanding 2-Nitropropane: A Deep Dive Beyond the Basics
Historical Development
Interest in 2-nitropropane grew during the postwar chemical boom. Researchers chased new synthesis routes not just for the sake of discovery, but to keep pace with the rapidly evolving needs of paint, printing, and solvent industries. Early production methods looked unwieldy to the modern eye. Chemists originally mixed hot nitrogen dioxide with propane, hoping for decent yields. Quality control wasn't what it is now, so product purity often varied, but those early experiments paved the way for safer, standardized processes that stand today. I see how each generation of chemists built on hard lessons about safety and purification, navigating the often volatile world of nitration chemistry. These improvements gave 2-nitropropane a path out of the lab and into widespread industrial use, raising practical questions alongside its benefits.
Product Overview
2-nitropropane sits at a crossroads between industrial utility and safety controversy. This clear, oily liquid appears mild at a glance, given its subtle odor and ease of handling, but history shows it demands respect and caution. Many applications turn to it as a solvent, especially in formulating inks, paints, and adhesives. Its ability to dissolve both natural and synthetic resins sets it apart from more volatile or aggressive solvents. Enthusiasts for this niche chemical highlight its tunable evaporation rates and reliable performance, but these same qualities require operators to remain vigilant about air quality and ultimately workplace health.
Physical & Chemical Properties
The liquid form carries a distinct edge. Unlike flammable compounds with low boiling points, 2-nitropropane boils around 120°C, limiting vaporization at room temperature but not eliminating risk. Its density—higher than water—means spills don’t float, complicating cleanup in manufacturing environments. It mixes well with most organic solvents, a trait that buyers look for when blending specialty coatings. In terms of chemical stability, it stands up well against weak acids and bases, but reactive settings push it toward dangerous decomposition, especially if overheated. Safety data points to an explosive hazard under severe conditions. This reputation for unpredictability keeps handlers alert, and modern processors design containment strategies for worst-case scenarios.
Technical Specifications & Labeling
Clear labels can make the difference between accidents and safe, consistent output in industrial plants. Chemical suppliers provide information such as purity, moisture content, and byproduct levels directly on barrels and material safety documentation. Experienced workers confirm product quality by cross-checking lot numbers and conducting in-house tests. Few chemical intermediates draw as much scrutiny from internal auditors and regulating bodies as 2-nitropropane, considering both its health profile and its role in regulated industries. Labels must meet international standards for hazardous materials, while keeping crucial safety warnings, handling guidance, and storage recommendations at the surface. My own experience reading and interpreting these documents taught me the value of redundancy in workplace safety. Mistakes rarely happen when every bottle, drum, and delivery truck reinforces the rules.
Preparation Method
Industries prepare 2-nitropropane mainly through the vapor-phase nitration of propane. Plants use specialized reactors to control temperature and pressure, maintaining a balance that maximizes yield but reduces the formation of toxic byproducts. Operators monitor sensors to ensure nitrogen dioxide—never a benign gas—meets propane in just the right concentrations. Once the reaction finishes, cooling systems condense the liquid, and distillation towers strip away impurities. Laboratories can adapt this method on a small scale for research, but scaling up means introducing layers of control that bring engineering and chemistry together. Failures in this process teach costly lessons, given the risk of runaway reactions. Consistent output starts with training up operators and running frequent audits, especially where nitration chemistry introduces new hazards.
Chemical Reactions & Modifications
2-nitropropane doesn’t work alone. Chemists see its value as a stepping stone: it serves as a feedstock for making pharmaceuticals, dyes, and chemical intermediates with more complex structures. Reactive centers on the nitro group turn basic molecules into building blocks for larger syntheses. Reduction reactions yield amines, while further modification can open paths toward specialty chemicals. Some research groups have developed routes that use this compound to introduce nitrogen content selectively in organic molecules, bypassing some older, messier strategies. Modification steps, while straightforward on paper, call for careful temperature control and selective reagents—the nitro group can easily go the wrong way, tempting fate in an already combustible environment. Plant managers invest in lab-scale trials before scaling up to full production, and I’ve seen how these incremental steps guard against workplace accidents as much as cost overruns.
Synonyms & Product Names
Not every supplier sticks to a single name. Most technical literature refers to this solvent as 2-nitropropane, but trade sheets from across the world reveal synonyms like isonitropropane or 2-NP. Regulatory filings and hazard warnings, especially in shipping, might list its CAS number to avoid confusion. Branding sometimes injects local language, so plant workers learn to cross-reference identifiers quickly, especially during audits or safety drills. Clarity across names keeps miscommunication in check, given the dangers that come along with inconsistent labeling.
Safety & Operational Standards
No shortcut in handling 2-nitropropane really exists. Workers should wear gloves, splash-proof goggles, and the right breathing protection since inhaling even a small amount can trigger headaches or nausea. Production lines vent gasses through scrubbers, while enclosed filling systems keep vapors from drifting across work areas. Fire crews keep foam extinguishers in reach, anticipating the risk of ignition during transfer or mixing. Inspections look for leakage at joints and storage containers get grounded to avoid static-induced sparks. Regulatory bodies like OSHA and their global counterparts enforce strict exposure limits based on years of occupational health studies. These standards stem from real incidents where oversight or faulty equipment led to worker illness. I’ve walked through plants where triple-checking valves and following lockout-tagout protocols isn’t just a formality but a culture, driven by history and hard-won experience.
Application Area
Printing presses, adhesives factories, and coatings manufacturers rely extensively on 2-nitropropane for its ability to dissolve, carry, and help form films. Flexible ink production, for example, hinges on quick-drying solvents that don’t sacrifice print clarity or shelf life. Paint formulators favor its performance when seeking smooth, bubble-free finishes in automotive or industrial applications. In the realm of chemical synthesis, this compound helps introduce nitro groups into more valuable intermediates, broadening the toolkit for research and commercial production. At each junction, stakeholders must measure performance against cost, safety, and downstream health implications, and often weigh these factors differently depending on whether regulation, environmental impact, or customer demand sets the pace.
Research & Development
Innovation in the world of 2-nitropropane never stands still. Companies and research labs invest in greener synthesis methods, aiming to lower the carbon footprint and reduce hazardous byproducts. Some teams push for process intensification, squeezing more yield from less energy and smaller reactors. Others focus on downstream chemistry: finding ways to convert this intermediate into pharmaceuticals or specialty materials without the chronic exposure risks that have dogged this compound since its discovery. Analytical labs design new real-time monitoring techniques, using sensors to catch leaks or off-spec batches before they reach workers or the environment. My own encounters with plant R&D confirm the tension between speed and safety, as decision-makers try to keep competitive while respecting both regulatory scrutiny and staff wellbeing.
Toxicity Research
Studying 2-nitropropane’s health effects changed the way industry treats not just this compound, but volatile solvents as a whole. Chronic exposure in poorly ventilated workplaces links directly to liver damage and potential carcinogenicity. Decades of occupational health data, much of it initially overlooked or underreported, forced regulators and plant managers to reassess acceptable workplace conditions. Today’s studies use advanced methods to measure airborne concentration at a molecular level, charting cumulative doses and correlating them with changes in employee health. Animal studies flag up systemic toxicity at lower doses than many comparable solvents, making real-world research essential for updating standards. Management culture in chemical plants has shifted from hiding these risks to training workers about exposure pathways, medical surveillance, and the importance of reporting any symptoms.
Future Prospects
Debate continues about the long-term role of 2-nitropropane. On one side, demand persists in mature industries that prioritize reliability, especially where substitutes fail on performance or economics. On the other, rising awareness about environmental health drives companies to hunt for less toxic, greener alternatives. Researchers experiment with reformulations that blend renewable solvents or radically new reaction media, while engineers rework plants for closed-loop operations. Lawsuits and health studies keep pressure on both suppliers and end-users to justify continued reliance on this compound, sparking creative approaches to measurement, containment, and emission control. Looking ahead, 2-nitropropane might see its strongest use as a controlled intermediate in specialist synthesis, with mainstream applications migrating toward options that promise less risk to people and environment alike. If anything, the legacy of this complex solvent lies in the hard lessons absorbed through decades of both progress and missteps, setting the stage for safer, smarter chemical handling in the years to come.
What is 2-Nitropropane commonly used for?
The Stuff Behind the Label
Walk into any workshop where paints or varnishes are mixed, and you might catch a whiff of something sharp in the air. There's a good chance 2-nitropropane plays a role. Over the years, this chemical found its way into paint thinners, inks, and the coatings used for everything from cars to furniture. It's rarely something you see in a pure bottle on a shelf, but it's snuck into hundreds of everyday products.
Why It’s So Popular in Industry
Chemistry is about function, and 2-nitropropane doesn't get fame for no reason. As a solvent, it has a knack for making other compounds dissolve quickly. If you've ever wondered why printing inks dry as smoothly as they do, 2-nitropropane could be part of the answer. Presses running day and night depend on chemicals that help ink flow and stick, and this one checks those boxes for printers across the world.
It also steps up in coatings and lacquer thinner used on wood and metal. I remember refinishing an old wooden chair and checking the label on the paint stripper—there it was, tucked into a list of ingredients. The stuff helps break down sticky resins, making the process less of a slog.
The Science Beyond Cleaning Up Messes
There's more to 2-nitropropane than stripping paint. Chemical manufacturers use it when making other compounds. Take drugs and dyes—the kinds used to brighten up clothes or improve medical treatments. Industries rely on this chemical because it speeds up reactions. It’s about efficiency; the right solvent means less energy burned and higher yields.
Producers of specialty adhesives and sealants lean heavily on solvents like this, especially where quick drying and clarity matter. My time working in a packaging plant showed me just how much gets poured, measured, and blended behind the scenes—not just for function but for making processes run like clockwork.
Safety Questions That Demand Answers
Pulling back the curtain, the hazards stick out. As much as 2-nitropropane helps, it comes with baggage. Health agencies flagged it for its links to cancer from inhalation or skin contact. OSHA put it on the short list of chemicals that demand respect. Breathing it long-term is bad news, and factories using it need real ventilation and strict spill controls.
Old habits—like pouring waste down the drain or tossing leftover solvent—don’t fly anymore. Regulations landed for good reason, and companies stepped up with closed systems and better filters. I saw coworkers suit up like it was an operating room, just for mixing paint. Gloves, goggles, and respirators aren’t optional. They’re the rule.
Rethinking the Approach
The future sits at a crossroads with 2-nitropropane. Some manufacturers moved to safer or greener substitutes, even if it meant sacrificing a bit of speed or shine. Bio-based solvents grew in popularity, especially where consumer health grabbed the spotlight. Companies investing in research churn out alternatives year after year.
Industry keeps looking for the right balance—something that won’t slow production or hit the bottom line, but still cuts the risks. The journey isn’t quick or simple, but with growing pressure from both workers and consumers, the need to upgrade old processes feels stronger than ever.
Is 2-Nitropropane hazardous to health?
Getting Acquainted With 2-Nitropropane
2-Nitropropane often shows up in paints, coatings, and some specialty printing processes. Its use stretches back decades. Most people outside chemical or manufacturing industries don’t think about it, but it comes with a pretty clear warning label. Based on what’s known, breathing in this chemical—or even getting it on bare skin—risks more than just discomfort.
Health Risks Backed By Research
Scientists have studied this chemical closely since the 1970s. Results haven’t been comforting. Inhalation brings headaches, nausea, dizziness, and eye or skin irritation for pretty much anyone who works near it. Animal studies show something even worse: consistent exposure leads to liver damage and, in some studies, cancer. The U.S. Environmental Protection Agency classified 2-Nitropropane as a probable human carcinogen. That should make anyone—worker or employer—pay attention.
I spent several summers working inside small machine shops. Even with gloves and fans running, chemical exposure always hovered at the back of my mind. I remember guys coming home with that strong chemical smell on their clothes, talking about splitting headaches or burning skin. Lots of them laughed it off, but as research keeps piling up, it feels foolish to ignore clear signs.
Exposure in the Workplace
People on factory floors, especially where solvents mix with paints and inks, face the biggest risk. Some older buildings still haven’t switched away from this chemical in their processes. Proper ventilation lowers exposure, but plenty of shops turn off those systems to keep warm in winter, or try to save on electric bills. That's how accidents start. The Occupational Safety and Health Administration (OSHA) in the U.S. sets a strict exposure limit for this chemical, capping it at 25 parts per million over an eight-hour shift. If you can smell a strong chemical odor, chances are good that something’s off.
Consumer Dangers and the Broader Picture
Most people won’t find bottles of 2-Nitropropane around the house. Still, anyone using imported paints, varnishes, or ink-based products should check labels. Consumer safety data sheets tell the story in a few lines: avoid breathing vapors, use outdoors or in well-ventilated areas, and never let it contact skin too long. This isn’t fearmongering—it’s advice written in response to years of incident reports.
Practical Solutions That Protect Health
Reducing health risks starts with awareness. Factories swapping out 2-Nitropropane for safer chemicals report fewer workplace health complaints. It’s not always easy—new formulas cost more, or don’t work quite the same—but if keeping workers healthy means shutting down sick days or worse, companies who jump ahead stand to gain in the long run.
Strict enforcement of proper handling goes a long way. Good exhaust fans, modern personal protective gear, and regular air monitoring can turn a dangerous workplace into a safer one. Employees who speak up about broken fans or faulty gloves should be heard, not brushed aside. Regulatory bodies need to keep checking up, running surprise inspections, and updating limits as new science comes in. That’s how real change happens, both on the factory floor and beyond.
Looking Ahead
Ignoring warnings from scientists and health organizations rarely ends well. The risks tied to 2-Nitropropane don’t go away with time. Industry and regulators both carry the responsibility to make smarter choices—not just when accidents happen, but every day. For workers, knowing the facts means staying safer on the job. For bosses and lawmakers, putting health above short-term savings offers longer-lasting value.
What are the storage requirements for 2-Nitropropane?
Understanding the Risks and Realities
Storing chemicals like 2-Nitropropane calls for a solid grip on safety. This liquid stirs up memories of handling containers in industrial labs. One spill, one whiff in a room with bad airflow—there's no ignoring how quickly a routine job can push workers into emergency mode. That yellowish solvent, valued by paint and ink makers, doesn't announce danger with a flare. Instead, it lingers. The risk sits in its easy flammability, its toxic fumes, its stubbornness when mixed with dampness or heat. OSHA’s standards and the data in every Safety Data Sheet aren't optional reading—they're guardrails, often shaped by hard lessons.
Fire and Explosion: The Real Enemies
Years of working near chemical depots teach the value of separation. Mixing 2-Nitropropane with sparks, open flames, or hot surroundings invites fire or worse, an outright explosion. Its flash point sits alarmingly low, around 38°C (100°F). Storage takes place far from ignition sources, in rooms with robust ventilation and something stronger than luck: well-maintained, explosion-proof lighting and switches. I remember a near-miss at a small coatings plant—one forgotten pilot light threatened an entire shipment. That drill through a wall for gas lines suddenly became a ticking bomb until someone noticed in time. A glovebox is useless if fire skips across the workbench.
Choosing the Right Containers and Rooms
At street level, chemical drums often rust or suffer dents. For 2-Nitropropane, only containers made from compatible steel or HDPE keep the substance sealed and stable. Tight-fitting lids and reliable gaskets make the difference—one leaky cap blows apart the myth of "minor" exposures. Storage in tightly sealed, labeled drums wards off moisture—contact with water, acids, or oxidizers must never happen. Shelving and pallets look unremarkable until a heavy drum knocks loose from a crowded rack. Spacing steers clear of stacking mistakes and barrels toppling onto each other.
Air Quality, Spills, and Real-World Precautions
Ask any long-time facility manager—the scent of nitro compounds in the air triggers brisk action. Airflow proves as crucial as any lock. Good mechanical ventilation, directed outside, draws away vapors. Without it, even a small leak can turn a break room next door into a zone for headaches, nausea, or something much worse. Spill kits with absorbent pads, gloves, and goggles sit close by. A weekly drill or walk-through, boring as it may sound, quickly exposes forgotten cleanup supplies or cracked containers. Containment curbs under storage racks make sure a leak never turns a workplace into a hazardous wading pool.
Training and Responsibility
Hands-on training wins out over any wall poster. Workers should recognize every hazard label, know the evacuation plan without thinking. More than a checklist, this means clear labeling of all drums and contact information for emergency responders taped where eyes find it fast. Reacting to exposure—whether eye splash, skin contact, or inhaled vapor—demands ready access to eyewash stations and showers. OSHA fines matter less than real injuries that keep breadwinners from their jobs. Responsible managers rotate stock, keep clear records, and toss expired or deteriorating material. Regulatory compliance tracks habits, not just rules.
Moving Forward: Investing in Safer Practices
No shortcut or wishful thinking protects against the hazards of 2-Nitropropane. Investment in proper storage, training, and emergency readiness sets apart shops that thrive from those that fumble through audits. People remember close calls more than successful routine days. Every barrel stored with care means one less story about mishap and harm. In hazardous work, safety culture is built from gritty attention to detail, not assumptions about tomorrow always being like today.
What is the chemical formula of 2-Nitropropane?
Getting Straight to the Facts
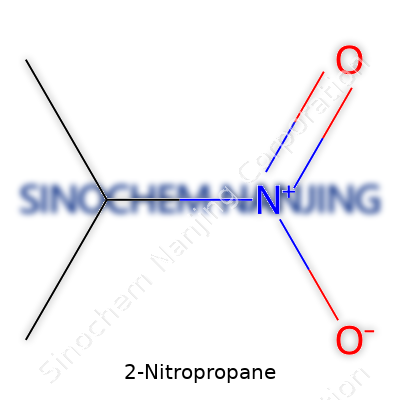
2-Nitropropane doesn’t spark debate in most living rooms, but its formula—C3H7NO2—crops up everywhere chemicals matter: labs, factories, and even courtrooms handling hazardous spills. This stuff isn’t just a string of symbols on a chalkboard. We’re talking about a compound commonly used as a solvent, especially in paints, inks, and other materials. Its makeup means business both for industry and public safety, so let’s break down why knowing this formula and understanding the stuff behind it is anything but trivial.
Breaking Down C3H7NO2
Here’s what you see: three carbons, seven hydrogens, one nitrogen, and two oxygens. That nitro group—NO2—links up with a propane backbone. In practice, that arrangement sets the stage for some serious reactivity. Over the years, I’ve watched engineers use 2-nitropropane to dissolve or thin out stubborn resins where basic water or alcohols just don’t cut it. Its chemical structure gives it a foot in the door where less aggressive solvents fail.
The Real Impact—and the Risks
Chemistry doesn’t operate in a vacuum. Solvents like 2-nitropropane boosted industry’s capabilities as far back as the 1950s, but the story doesn’t end there. Toxicological data gathered over decades flags this compound as a likely carcinogen. The U.S. Environmental Protection Agency (EPA) and International Agency for Research on Cancer both issued warnings after occupational studies and animal tests revealed increased tumor risks with high-level, long-term exposure.
Having worked with environmental health experts, I know how that black-and-white formula on the page translates into real repercussions. Factory workers painting or cleaning with solutions containing 2-nitropropane risk breathing in more than just fumes. Symptoms like headaches or nausea come quickly; the bigger threat lurks in the background, with heightened risk for liver and kidney issues. There’s a reason strict exposure limits exist—these don’t come from scare tactics but from direct observation and hard data.
Solutions Aren’t Optional
Knowing the formula lets people assess risk and spot alternatives. C3H7NO2 is a must-know for industrial hygiene plans and product bans. In my time supporting environmental audits, safer options often come up. Companies aim for “green chemistry” where possible, swapping 2-nitropropane out for less hazardous choices like water-based solvents or methyl soyate. Even if a process absolutely requires something as aggressive as 2-nitropropane, mitigation plans can include better ventilation, sealed containers, and personal protective equipment.
Fact is, people can’t treat chemicals as mysteries. Workers benefit when labels, training, and research stay up-to-date, and when managers genuinely care about handling, storage, and disposal. Keeping tabs on chemical formulas doesn’t just fill out material safety data sheets—it draws a line between business as usual and public health disaster.
Trust in Good Information
The formula for 2-nitropropane isn’t trivia. It triggers regulations, safety checks, and science-backed discussions about alternatives. Anyone responsible for using, handling, or disposing of this chemical saves money and lives by respecting what those eight little letters and numbers mean. And that’s why chemistry matters to all of us—even if we never crack open a textbook outside of high school.
How should 2-Nitropropane spills be handled?
The Mess We’re Dealing With
It only takes a whiff of 2-nitropropane to know you’re tangled up with a big problem. Anybody who’s seen a chemical accident knows safety often hangs on the slimmest thread. 2-nitropropane brings more than a headache: it’s toxic, flammable, and doesn’t wait around for slow decision-making. In my years working in industrial safety, quick knowledge and teamwork have saved more skins than anything found in a textbook.
Big mistakes crop up when folks underestimate what they can’t see. This stuff seeps into soil fast and vaporizes into the air. It doesn’t just sit in puddles waiting for cleanup crews. A spill on the shop floor isn’t like spilled milk—open windows won’t clear it, and mops actually make it worse by spreading oily residues and kicking up fumes. The solvent-like odor might seem minor, but it signals real danger for lungs and long-term health. There’s also the fire risk, which never lets up if any spark comes near.
Why Tools and Training Matter
I’ve watched good people wearing nothing but gloves try to sweep up far nastier spills, thinking they were “protected enough.” That attitude leads straight to ruined boots and visits to the ER. Local fire departments often don’t keep enough gear for volatile organic spills like this, either. Workers and emergency teams need full-body suits, chemical-resistant boots, and air respirators. I remember one warehouse drill where someone used their shirt as a mask. That shortcut meant they inhaled toxins anyway, proof that improvising only invites trouble.
A proper spill kit has absorbent pads built for chemicals and neutralizing agents—never just sawdust or sand. Keep a few foam barriers on hand to stop the flow and keep run-off out of drains. Picture what happens if this stuff hits a storm drain on a rainy day; you’ve just delivered toxins downstream where neighbors draw drinking water. Containment needs to happen right away, before calling outside help.
It Takes Local Know-How
Not every neighborhood fire crew knows how to tackle industrial chemical spills. Community response plans usually get built without input from the folks actually facing chemical dangers every week. Facility managers should insist on drills with local responders, walking them through plant layouts and chemical storage routines. These “tabletop” rehearsals spot gaps in gear, training, and communication before the sirens blare for real.
One time, I watched a local team freeze up at the sight of strange drums without labeling. Even the best response stalls if the labeling is a mystery or the layout hasn’t been shared. Keeping accurate, accessible chemical inventories helps emergency crews work faster so accidents don’t spiral into headlines and lawsuits.
Common Sense and Follow-Through
Chemical spill handling shouldn’t rely on luck or guesswork—especially not with something as touchy as 2-nitropropane. Clear protocols, tough training, and coordination with local emergency crews build safer workplaces. Good practices might cost a bit more in time and supplies, but they stop serious injuries and fines in the long run. That’s a trade worth making every time.
| Names | |
| Preferred IUPAC name | 2-nitropropan-2-ol |
| Other names |
2-Nitropropane 2-NP Isopropyl nitrite |
| Pronunciation | /tuːˈnɪtrəʊˌprəʊpeɪn/ |
| Identifiers | |
| CAS Number | 79-46-9 |
| Beilstein Reference | 1209247 |
| ChEBI | CHEBI:18107 |
| ChEMBL | CHEMBL133684 |
| ChemSpider | 7277 |
| DrugBank | DB01937 |
| ECHA InfoCard | 03b8f1d1-d2bc-46cf-8b08-bfd5e2cc7844 |
| EC Number | 200-209-1 |
| Gmelin Reference | Gmelin 844 |
| KEGG | C01723 |
| MeSH | D009607 |
| PubChem CID | 6362 |
| RTECS number | TZ0875000 |
| UNII | VV2AV7309J |
| UN number | UN1608 |
| Properties | |
| Chemical formula | C3H7NO2 |
| Molar mass | 89.09 g/mol |
| Appearance | Colorless liquid |
| Odor | Sweet odor |
| Density | 0.992 g/mL at 25 °C (lit.) |
| Solubility in water | 5.5 g/100 mL (20 °C) |
| log P | 0.89 |
| Vapor pressure | 3.45 kPa (at 20 °C) |
| Acidity (pKa) | 13.6 |
| Basicity (pKb) | 11.30 |
| Magnetic susceptibility (χ) | `-9.86×10⁻⁶ cm³/mol` |
| Refractive index (nD) | 1.389 |
| Viscosity | 1.69 mPa·s (25 °C) |
| Dipole moment | 4.23 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 129.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -56.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1562.8 kJ/mol |
| Pharmacology | |
| ATC code | VO20 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H225, H304, H311, H319, H332, H351 |
| Precautionary statements | P210, P261, P280, P304+P340, P312, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-Nitropropane: 2-4-0 |
| Flash point | 77 °C (closed cup) |
| Autoignition temperature | 424 °C |
| Explosive limits | Explosive limits: 2.1–11% |
| Lethal dose or concentration | LD50 oral rat 960 mg/kg |
| LD50 (median dose) | Rat oral LD50: 940 mg/kg |
| NIOSH | NIOSH: TZ6300000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2-Nitropropane: 25 ppm (90 mg/m³) |
| REL (Recommended) | 25 ppm |
| IDLH (Immediate danger) | 40 ppm |
| Related compounds | |
| Related compounds |
Nitromethane Nitroethane 1-Nitropropane 2-Nitrobutane Nitrobenzene |

