2-Methyltetrahydrofuran: More than Just Another Solvent
Historical Development
Chemistry often moves ahead in fits and starts, driven by industrial needs, fresh scientific questions, and the dawning environmental consciousness. 2-Methyltetrahydrofuran (2-MeTHF), once a rarely mentioned name in the early days after its discovery, owes much of its presence today to the growing demand for greener solvents and practical, efficient alternatives in chemical manufacturing. Early production rooted itself in the mid-20th century, largely as a byproduct in the butadiene industry. For a long time, chemists gave it little attention, favoring diethyl ether and tetrahydrofuran (THF) for extraction and reaction media. As I watched the sector embrace sustainability, 2-MeTHF slowly gained favor. The growing push to move away from petroleum-based processes nudged researchers to revisit old molecules through a new lens. 2-MeTHF found its moment thanks to the shift toward using renewable resources, such as agricultural biomass, for its preparation. Looking back, I see this as a practical win for the broader effort to marry chemistry with environmental stewardship.
Product Overview
2-MeTHF steps up where traditional ethers struggle. With a five-membered ring and a solitary methyl group hanging from the second carbon, it remains stable, volatile enough for most tasks, and, importantly, capable of dissolving a range of compounds from polar to nonpolar. Its slight solubility in water chalks up another advantage, simplifying phase separations in workups. As a practical matter, chemists faced fewer safety headaches relative to peroxides compared with ordinary THF. When I switched to using 2-MeTHF during catalytic reactions, that extra safety margin became obvious–there were fewer peroxide worries, and I could run reactions across broader temperature ranges without worrying about solvent stability compromising my work.
Physical & Chemical Properties
Taking a closer look at its physical nature truly helps to understand why 2-MeTHF outpaces some rivals. It presents as a clear, colorless fluid with a characteristic ether-like smell, boiling at around 80°C and offering noticeably less water miscibility than THF. That persistent difference in polarity, stemming from the methyl group, helps in extracting organic compounds from aqueous mixtures. Beyond that, its lower tendency to form explosive peroxides stands as one of its most understated features. The oxygen atom in the ring brings solvent properties similar enough to other ethers, but the methyl group tilts the balance, showing up in everything from solubility to peroxide formation. In real lab conditions, I noticed my samples of 2-MeTHF lasted longer before needing checks for peroxide buildup. Stability in storage and utility in the flask–that balance helps labs cut waste and hazards.
Technical Specifications & Labeling
Regulation gives structure to the way chemicals travel and get stored, and solvents like 2-MeTHF fall squarely in that system. Typically, suppliers label purity, water content, and inhibitor presence. Many chemists, including myself, pay close attention to whether 2-MeTHF arrives stabilized, since unstabilized samples tend to form peroxides after prolonged storage. Labels for 2-MeTHF usually report assay by gas chromatography, with specifications often exceeding 99 percent. More companies highlight whether the solvent comes from renewable plant-based sources, a detail seeing more demand from pharmaceutical and flavor industry buyers. This information, while ordinary to some, signals real commitments to quality and responsibility. The specifics on technical sheets may vary, but the clarity in labeling helps everyone up and down the supply chain know what to expect from each batch.
Preparation Method
Modern production borrows heavily from green chemistry, using furfural derived from agricultural residues. By hydrogenating furfural, chemical plants turn renewable biomass into 2-MeTHF. This process, which replaces petrochemical methods, plugs the molecule right into the ‘circular economy’ conversation. For decades, producers relied on side reactions in butadiene processing, but as biomass conversion scaled up, the conversation about solvents and environmental cost became richer. Small-scale syntheses usually take the catalytic hydrogenation of furfural under mild pressure, which demonstrates how laboratory-scale work syncs with commercial practices. Practical chemistry needs practical feedstocks, and 2-MeTHF gives chemists a chance to move beyond oil barrels, instead using materials that farmers once considered waste.
Chemical Reactions & Modifications
Good solvents do more than just dissolve compounds; sometimes, they become participants or seats for clever reactions. 2-MeTHF, as an ether, avoids most strong acid chemistry due to its susceptibility to ring cleavage. At the same time, its resilience against many bases proves useful when running organometallic coupling or reduction reactions. Comparatively, its lower boiling point creates chances for solvent swaps by distillation, and its relative inertness means the solvent doesn’t often interfere with catalysts or exotic intermediates. Synthetic chemists saw immediate benefits–Grignard and organolithium reagents thrive in 2-MeTHF, because it stabilizes reactive intermediates via coordination with the oxygen atom. As new reactions entered the literature, more labs recognized that switching from THF to 2-MeTHF usually meant better yields, cleaner workups, and fewer batch failures. Side reactions remain limited, and most users note higher selectivity in certain asymmetric transformations. Those are practical wins, not theoretical footnotes.
Synonyms & Product Names
The chemical trades under plenty of names, reflecting its spread through different sectors and regulatory databases. 2-Methyltetrahydrofuran, 2-MeTHF, methyl tetrahydrofurane, and furanidine all show up on bottle labels in research labs and warehouses. Legacy names and IUPAC conventions share space with newer, more marketable monikers in the age of green chemistry advocacy. For anyone navigating international sourcing or multi-lingual paperwork, keeping track of these synonyms prevents expensive mistakes, since substitutes are not always interchangeable. In academic writing, 2-MeTHF has become the shorthand, streamlining communication and clarifying intent whenever reaction protocols specify its use.
Safety & Operational Standards
Working safely with 2-MeTHF, like all ethers, takes a disciplined approach. The solvent remains flammable and forms explosive mixtures with air. Exposure limits in workplace settings come from government bodies, placing safe-use thresholds well below concentrations that would cause irritation. In the lab, I’ve watched strict controls pay off. Grounded equipment, flame-proof storage, and frequent air monitoring eliminate most operational risks. Disposal rules remain strict, owing to its potential to bioaccumulate and environmental persistence if allowed into waterways. Commercial operations, drug research labs, and even academic teaching spaces all face the same need for regular solvent checks, fire control protocols, and staff training. These routines sound tedious, but over time, they reduce real hazards and send fewer workers to the emergency room. Reporting spills, segregating waste, and always checking for peroxides are not optional extras—they are what keeps labs in business and people safe.
Application Area
The practical reach of 2-MeTHF stretches across pharmaceuticals, flavors, adhesives, and lithium battery manufacturing. Most notable is its uptake in active pharmaceutical ingredient (API) synthesis, where its low peroxide formation, eco-friendly sourcing, and favorable solvent partitioning give it clear edges. Battery researchers and electronics manufacturers lean toward 2-MeTHF for its capacity to solvate advanced electrolytes and stabilize polymer matrices. Even the food industry pays attention, since flavor compound extraction and refinement often benefits from its organic-aqueous partition behavior. As a chemist, using 2-MeTHF in complex natural product isolation turned tedious batch separations into straightforward extractions, trimming hours off my work without sacrificing sample quality. As sustainable chemicals gain favor, the spread of 2-MeTHF has only grown, and the pattern looks unlikely to reverse.
Research & Development
Research into 2-MeTHF keeps breaking new ground, from process intensification in continuous flow chemistry to improved recycling protocols aimed at waste minimization. Chemical engineering teams work on refining biomass conversion, seeking higher atom economy and even tighter integration between agricultural feedstock suppliers and chemical producers. Universities around the globe experiment with new catalytic cycles, often reporting higher selectivity and yield from reactions launched in 2-MeTHF compared to older solvents. In synthetic methodology, recent years saw the emergence of lithium-mediated cross-coupling that benefited directly from the solvent’s unique solvation qualities. This ongoing investment means the best uses of 2-MeTHF are probably still ahead of us, as regulatory and environmental pressures keep driving researchers toward even cleaner, safer chemistry. I’ve watched teams bridge the gap between prototype and production, relying on 2-MeTHF to ensure that transition remains both feasible and cost-effective.
Toxicity Research
Toxicologists keep a watchful eye on 2-MeTHF, keenly aware of the problems that shadow many ethers. Acute toxicity remains low, with animal studies showing relatively high LD50 values. Still, long-term inhalation studies point to neurological and liver effects at sustained high exposure. Careful risk assessments underwrite its use in drug manufacturing, where even minute traces left in pharmaceuticals can trigger scrutiny. Lab technicians, well-trained and equipped with ventilation and personal protective equipment, seldom approach dose thresholds that would cause harm. Even so, safety data sheets and national chemical inventories flag the need for closed handling and regular exposure monitoring. Environmental fate studies show moderate persistence, prompting regulations on waste management and emissions. From my own experience, consistent safety drills and prompt reporting of spills or exposures keep headaches rare, while ensuring occupational health standards remain more than just paperwork.
Future Prospects
2-MeTHF sits at a crossroads where industrial necessity and green ambitions meet. As energy costs change and pressure mounts to keep toxic run-off out of water and soil, solvents made from renewable feedstocks will attract even more attention. Policy support for biomass and ‘green chemistry’ only underlines this trend. The next few years are likely to see expanded process integration, combining on-site production of 2-MeTHF from local biomass, with fewer transportation miles and associated emissions. Innovations in continuous reactor design promise improved yields and easier solvent recovery, all while using less energy per kilogram produced. Enhanced analytical tools will make detection and removal of trace impurities more efficient, letting pharmaceutical and battery sectors further tighten quality control. For people like me, who blend research with process improvement, the path forward with 2-MeTHF means looking for smarter, safer, and greener ways to keep progress ticking without piling up environmental debt for the next generation.
What is 2-Methyltetrahydrofuran used for?
Hands-On Perspective: What Makes 2-Methyltetrahydrofuran Stand Out
Anyone who’s spent time in a chemistry lab recognizes the usual suspects like ethanol and acetone. But there’s a growing shift toward options that do the same job, but with bonus perks. That’s where 2-methyltetrahydrofuran, or 2-MeTHF, comes in. This solvent looks unassuming, yet it’s changing approaches in both research and manufacturing.
A Greener Choice for Laboratories
My experience working with solvents began years back, and I remember the sharp smells, thick gloves, and warnings stacked up around standard ether and dichloromethane. 2-MeTHF brought something new. It’s derived from renewable resources, including corncobs and bagasse, making it a path toward greener chemistry. Bio-based solvents aren’t a cure-all, but every switch helps. Fresh research from the Green Chemistry Institute highlights that 2-MeTHF reduces environmental impact compared to petroleum-derived solvents. This feature alone puts it on the radar for labs aiming to make a real dent in their waste and carbon use.
Efficiency on the Bench: What Happens in Practice
Solvent choice shapes every step in an experiment. Organolithium reactions used to be a hassle since ethers like THF work but break down in air. 2-MeTHF lasts longer, especially as temperatures creep up. It resists forming peroxides, so I spend less time prepping, and more time getting things done. Reactions that use Grignard reagents or lithium aluminum hydride go smoother in 2-MeTHF—fewer surprises and more reliable yields. This matters day-to-day, when you just want to see the chemistry, not the cleanup.
Role in Industry: Not Just for the Lab Coat Crowd
Large companies look for ways to cut risk and improve workplace safety. Traditional ethers evaporate rapidly, sparking concerns about fires and hazardous vapors. 2-MeTHF boils at a slightly higher temperature and offers less volatility, trimming down those risks. In pharmaceuticals, the drive to swap out toxic or environmentally stubborn solvents grows each year. 2-MeTHF fits right into drug synthesis pipelines, whether for making antibiotics, cancer therapies, or everyday generics. The Chemical Abstracts Service and journals from the American Chemical Society point out its growing role in pilot plants and full-scale production lines.
Risks and Solutions: Keeping Perspective
Nothing comes without downsides. 2-MeTHF combusts at high concentrations, and overuse can narrow biodiversity if its raw resources aren’t managed well. People often wonder if swapping one solvent for another just pushes the problem around. So, I look for producers with transparent supply chains, and I push for regulations that set standards both for product safety and environmental safeguards. The EPA offers guidance but could go further. Wider adoption of closed-loop systems, where solvents cycle through purification and reuse, addresses a lot of these worries head on.
The Path Forward
The bigger picture shows a steady move away from chemicals that put health and the planet at risk for convenience. By picking 2-MeTHF, researchers and manufacturers back a system prioritizing safety, efficiency, and sustainability. That sets a tone for others to follow, and shakes up old habits in labs and factories worldwide.
Is 2-Methyltetrahydrofuran safe to handle?
Understanding 2-Methyltetrahydrofuran
2-Methyltetrahydrofuran, often called 2-MeTHF, turns up as a solvent in plenty of chemistry labs. Its reputation as an eco-friendlier cousin to regular tetrahydrofuran (THF) tempts scientists and technicians to use it for reactions and extractions. Out in the field, people like myself have handled it in glass beakers under the fume hood, trusting the safety data sheets and our own experience to keep things straightforward. The truth is, this compound is flammable—really flammable. Its vapor can sneak into the air, reaching sources of ignition that might not seem dangerous at first glance. Flammability is just the start; inhalation poses a real risk, especially when ventilation falls short of perfect.
Physical Risks: Fire and Fumes
Anyone who’s run a reaction late into the night knows the sharp, almost sweet scent that jumps out when 2-MeTHF spills. Those vapors aren’t just unpleasant—they hit the central nervous system. The National Institute for Occupational Safety and Health (NIOSH) has placed limits around workplace exposure, recognizing how dizziness and nausea come fast in a closed space. Add the potential for flash fires, and the risks become more than theoretical. I once witnessed a grad student use too much solvent outside a hood, only to see it catch fire after sliding near a hot plate. No one got hurt, but the lesson stuck: complacency brings real danger.
Toxicity and Long-Term Exposure
Acute exposure affects focus and breathing. If 2-MeTHF soaks into skin or splashes near the eyes, irritation hits almost immediately. Toxicity studies on animals suggest liver and kidney stress when intake climbs, but data on chronic exposure in humans still needs a close look. From my years among cautious chemists, I’ve seen most people limit skin contact using nitrile gloves and shielding arms and faces. Still, accidents happen; hospitals see their share of chemical eye injuries and rashes from sloppy handling.
Comparing Safer Alternatives
Some chemists tout 2-MeTHF as safer than THF due to lower peroxide formation and better biodegradability. That sounds promising. Still, its flammability and vapor danger haven’t vanished—just shifted. Professional organizations, including the American Chemical Society, recommend double-checking ventilation and never assuming a “green” solvent takes care of all hazards on its own. Substituting with 2-MeTHF doesn’t change the core need for discipline in solvent storage, fire suppression, and emergency response.
Practical Steps for Handling
Approved chemical hoods make a huge difference. In my lab days, every bottle sat in a clearly labeled flammables cabinet, away from heat and electrical equipment. Training mattered as much as hardware—colleagues quizzing each other on fire extinguisher location, running spill drills, and keeping eye wash stations clear. Double-gloving helped with big volumes, and goggles always came down before opening a cap.
In the end, no chemical becomes truly “safe” by reputation alone. 2-Methyltetrahydrofuran brings both green chemistry appeal and real hazards to the bench. Listening to experience, trusting the literature, and taking precautions—those small habits prevent serious incidents. Safety builds from how people act, not just what they handle.
What are the physical properties of 2-Methyltetrahydrofuran?
A Look at Its Appearance and Behavior
You pour a glass of water and it's clear, odorless, and benign. 2-Methyltetrahydrofuran, on the other hand, tells a different story the moment you open its container. This solvent brings an ether-like smell—sharp, slightly sweet, not unlike a whiff from an old chemistry lab. Sitting on the countertop, it shows off its clear, nearly colorless look. The liquid flows thin, almost eager to move from pipette to beaker. At room temperature, nothing about it seems off, but there’s a volatility that suggests the need for caution.
Handling Volatility and Flammability
People who’ve worked with flammable solvents know that the flash point changes everything. This one flashes around -11°C, which means it starts giving off enough vapor to ignite at temperatures below freezing. From experience, if you’re working with a solvent like this, open flames and sparks stay far from the scene. You learn quickly: ventilate the lab well, and cap containers fast. The vapor, lighter than air, rises quickly. If there’s a leak, it hangs in the air, making headaches or dizziness possible if you stay exposed too long.
Boiling and Freezing Put to the Test
In laboratories, boiling points guide decisions. With 2-Methyltetrahydrofuran, you hit 80°C before it starts to boil, which falls in the same zone as other lightweight solvents. Get too cold and it stays liquid down to around -136°C. This range lets chemists work in cold climates or at low temperatures without fear of a frozen, unusable solvent on the bench.
Solubility—What Mixes and What Doesn’t
Ask those who work in synthesis: solvents that play nice with water offer certain conveniences. 2-Methyltetrahydrofuran steps up. It dissolves in water better than many ethers, making it easy to separate water out after a reaction. It blends fully with most common organic solvents. That translates to flexibility—mixing, extracting, running columns, or pulling off tough separations—this liquid allows a cleaner, easier path in the lab.
Density Matters in Every Pour
Pouring a solvent into a measuring cylinder, you notice how quickly it overflows—density can catch you off guard. With a density of about 0.86 g/cm³—less than water, more than lighter hydrocarbons—2-Methyltetrahydrofuran floats atop water. During workups, it’s a straightforward layer on top, preventing confusion that sometimes arises with denser solvents.
Environmental and Health Considerations
As someone who’s spent late nights in crowded research spaces, the health and environmental impact of chemicals never stays out of mind. Compared to some older ethers like THF, 2-Methyltetrahydrofuran poses a slightly lower risk of forming explosive peroxides, though regular monitoring is still vital. Green chemistry circles often vouch for it, since it’s produced from renewable resources like hemicellulose. That sustainable edge means using it feels less like making a compromise between performance and responsibility.
Moving Toward Safer, More Sustainable Use
No solvent brings a silver bullet to safety and sustainability, but 2-methyltetrahydrofuran comes close. Limiting inhalation, storing in cool, well-ventilated spaces, and keeping it out of fire’s way shape good habits. Labs benefit from shifting away from petroleum and seeking out alternatives made from biomass. For students and professionals, that’s a wakeup call—look past the old standbys and pick solvents that protect both people and planet, without giving up results.
How should 2-Methyltetrahydrofuran be stored?
Real Hazards, Real Solutions
Growing up around a small auto shop in my neighborhood, I watched plenty of containers come and go, most of them carrying things a lot less volatile than 2-methyltetrahydrofuran. Even back then, folks took special care with solvents and fuels, most shipped in metal cans with big warning labels. Today, we know that 2-methyltetrahydrofuran falls into that “please don’t wing it” column. It burns easily, evaporates fast, and if left neglected, starts to form peroxides—the kind of thing you really don’t want building up near heat or light. So, thinking this stuff can be tossed in with general warehouse goods misses the point. Strict routines, grounded in chemistry and a healthy respect for bad luck, go a long way in keeping workplaces and homes safe.
Understanding What You’re Dealing With
To get it right, a person has to know what makes this solvent tricky. 2-Methyltetrahydrofuran’s low boiling point means it wants to escape as vapor, so open containers or leaky lids let more fumes build up than you’d expect at room temperature. Those fumes linger, hanging around until a careless spark turns them into a blast. It’s also prone to grabbing oxygen from the air over several months, quietly producing peroxides that wait for someone to shake the bottle or pour it out, causing surprise explosions.
What Works in the Real World
From years spent in workshops, I’ve seen good habits save more money and grief than any fancy equipment. Start with tight-fitting, chemical-resistant caps—not the brittle plastic kind but heavier-duty options with real seals. Containers need to stand up to a drop and shrug off drips. Keep them away from sunlight—tuck them in a flammables cabinet, the kind you can lock. Those cabinets should stay in cool, dry spots. Keep them far from electrical panels, heating vents, or anything that sparks. Many labs add visual reminders or color-coded tape, making sure new hires spot hazards before they even reach the shelf.
Lessons from Lab Fires and Spills
Lab reports and fire department records give plenty of proof: letting vapors build up or letting the bottle sit for a year without checks invites trouble. One report from a university lab showed an old bottle explode during routine handling because peroxide crystals had formed at the neck. A simple check with peroxide test strips would have caught it, as would a regular rotation out of old stock. Workers should check labels, test for peroxides if a container has been around more than six months, and write the opening date with a marker. Keep small amounts on hand, ordering only what will get used in a month or two.
Steps Toward Safer Practices
Rules mean little if folks ignore them. Plenty of companies run safety drills, encourage reporting of near misses, and make sure fire extinguishers and spill kits are close by. Fire extinguishers rated for chemical fires belong near storage areas, along with grounding wires to avoid static sparks during dispensing. Employees deserve real training—videos and hands-on practice, not just a stack of guidelines nobody reads. Emergency contacts and safety data sheets should hang close, not stuffed in a drawer.
Taking Responsibility Beyond the Label
Storing 2-methyltetrahydrofuran safely isn’t just a legal hoop to jump through. It’s showing respect for people and property, learning from close calls of others, and building habits that keep everyone safe. Better storage keeps workers healthy and businesses open, and prevents a quick job from becoming tomorrow’s headline for the wrong reason.
What are the applications of 2-Methyltetrahydrofuran in industry?
Greener Solvent in the Chemical World
2-Methyltetrahydrofuran—often shortened to 2-MeTHF—stands out in chemical manufacturing these days. Years of hazardous solvent use left a big mark, so seeing labs and production lines move toward safer options feels like real progress. This compound comes from renewable resources, mostly agricultural waste, which keeps it out of the oil-based category. By swapping it in for traditional choices like tetrahydrofuran or toluene, companies cut down on toxic byproducts and lessen blowback to workers and the environment.
People have seen classic solvents catch fire too easily or wreck air quality inside the plant. 2-MeTHF has a higher boiling point than its predecessor, giving chemists extra control during reactions and keeping evaporation losses in check. The safety record gets a lift, too, with fewer health scares from handling fumes. These points have led major pharmaceutical manufacturers—including several companies listed in recent American Chemical Society reports—to replace the old standbys with this newer alternative.
Batteries and Electrochemical Cells
Energy storage pushes materials to their limits. Right now, researchers use 2-MeTHF in lithium-ion battery electrolytes. It stands up well during repeated charge and discharge cycles, meaning your phone or car battery will last a bit longer with less risk for short-circuiting. The solvent’s performance at low temperatures brings out more power during winter, and its low viscosity helps ions zip through the cell. Better performance translates to longer running times and real savings for electric cars, where every mile counts. Start-ups working on next-gen energy storage see 2-MeTHF as a crucial part of making batteries safer, more efficient, and less harmful after disposal.
The Growing Field of Biomass Processing
People working to break up tough plant matter reach for 2-MeTHF because it helps separate sugars and lignin without turning valuable products to mush. Unlike harsher solvents, this one keeps key molecules intact, allowing for better yields of bio-based fuels and chemicals. Most renewable jet fuel and bio-based plastics rely on this sort of processing, so improvements here ripple out across many green-tech companies. Major advances may come not just from new feedstocks but from smart solvent choices that cut energy use in each step.
Fine Chemicals and Flavor Industry
Nobody likes a headache after walking through a flavoring plant. 2-Methyltetrahydrofuran scores points for lower smell and easy cleanup. Makers of spices, fragrances, and natural extracts pay close attention to trace chemicals in their processes. Because 2-MeTHF leaves less residue, it works well in selective extractions. Recent reviews in industry journals mention improved yields and purer end products with fewer steps. That’s less waste and less downtime for cleaning, which saves money and lets quality stay high.
Looking Forward: A Matter of Scale and Access
While this solvent benefits a lot of industries, price and supply can slow things down. Right now, many producers stick with tried-and-true options because they’re cheap and everywhere. If more plants start converting agricultural byproducts to 2-MeTHF at scale, supply should meet demand and prices will drop. Regulation may play a role as countries set new environmental standards. To keep projects on track, managers and chemists need training on handling this new solvent, and researchers need to keep testing it in tough, real-world jobs.
With the right focus on safety, accessibility, and economic incentives, more companies should find it easier to put 2-MeTHF to work. This shows how smarter resource use and cleaner chemistry can help whole industries take a practical step forward.
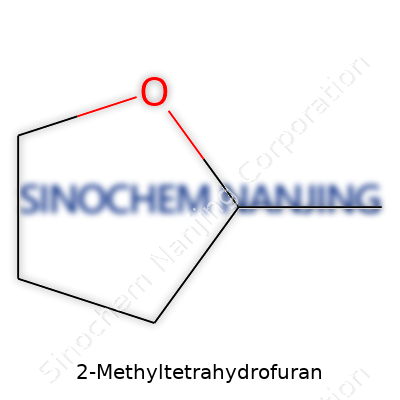
| Names | |
| Preferred IUPAC name | 2-Methyloxolane |
| Other names |
MeTHF 2-Methyl-THF 2-Methyltetrahydrofuran 2-Methyloxolane 2-Methyloxolane 2-Methyltetrahydro-2H-furan |
| Pronunciation | /tuːˈmɛθ.ɪl.tɛ.trə.haɪ.drəˈfjʊə.ræn/ |
| Identifiers | |
| CAS Number | 96-47-9 |
| Beilstein Reference | 1282067 |
| ChEBI | CHEBI:41411 |
| ChEMBL | CHEMBL3306517 |
| ChemSpider | 8728 |
| DrugBank | DB14083 |
| ECHA InfoCard | 200-766-1 |
| EC Number | 201-763-7 |
| Gmelin Reference | 113328 |
| KEGG | C06545 |
| MeSH | D058289 |
| PubChem CID | 13590 |
| RTECS number | LU5950000 |
| UNII | Z6SY8AMJ2A |
| UN number | UN2366 |
| Properties | |
| Chemical formula | C5H10O |
| Molar mass | 86.13 g/mol |
| Appearance | Colorless liquid |
| Odor | ether-like |
| Density | 0.86 g/mL |
| Solubility in water | 14 g/100 mL (20 °C) |
| log P | 0.35 |
| Vapor pressure | 53.4 mmHg (20 °C) |
| Acidity (pKa) | 28.8 |
| Basicity (pKb) | 15.73 |
| Magnetic susceptibility (χ) | -53.8·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.407 |
| Viscosity | 0.46 mPa·s (at 25 °C) |
| Dipole moment | 1.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 165.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -320.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3547.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302, H315, H319, H336, H402 |
| Precautionary statements | P210, P243, P233, P240, P241, P242, P264, P303+P361+P353, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | -11 °C |
| Autoignition temperature | 215 °C |
| Explosive limits | 1.5–12% |
| Lethal dose or concentration | LD50 Oral Rat 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 2000 mg/kg |
| NIOSH | NA |
| PEL (Permissible) | 200 ppm |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | 2000 ppm |
| Related compounds | |
| Related compounds |
Tetrahydrofuran Diethyl ether Furan Tetrahydropyran |

