2-Methylquinoline: A Chemical with Hidden Influence
Historical Development
2-Methylquinoline hasn’t grabbed the same spotlight as its parent quinoline, but it has carved a long, humble road through the history of organic chemistry. Chemists tracing the roots of quinoline derivatives often bump into the rich tapestry woven by coal tar distillation in the late 1800s. Back then, aromatic compounds like quinoline turned out to be fertile ground for new scaffolds in dye chemistry and later, for pharmaceuticals. 2-Methylquinoline emerged from those bustling lab benches thanks to eager experiments with simple methylation reactions. Even in the early 20th century, its presence did more than add another molecule to the quinoline family; it offered chemists a relatively accessible building block for further discovery. While some may see it as a niche compound, the people tinkering with nitrogen heterocycles know its history is packed with trial, error, and the occasional burst of inspiration that moved the field forward.
Product Overview
2-Methylquinoline floats around as a slightly oily liquid, familiar in research circles and industries with a taste for complexity. Its name may pop up as quinaldine—chemists have a way of getting poetic with nomenclature—but don’t let a new label fool anyone. The chemical often travels in brown glass bottles, tightly sealed, sometimes shipped under mild caution because of its moderate toxicity and pungent odor. Folks working with it see it as more than a reagent; it’s a scaffold, a precursor, or even a model compound to unravel the mysteries of quinoline-based synthesis. It never became the blockbuster chemical for mass-market products, but its fingerprint shows up in more research journals and process patents than most would guess.
Physical and Chemical Properties
Anybody who’s handled 2-methylquinoline remembers the color—clear to pale yellow. Its distinct, almost sweet, aromatic scent fits right in with other nitrogen-containing aromatics. Boiling points hover a bit over 230 degrees Celsius, and it sits solid at room temperature until about minus ten degrees Celsius. Its molecular structure brings just enough rigidity and resonance stabilization, courtesy of its fused rings, that it stands up pretty well against slow oxidation. Solubility can be tricky: it takes to most organic solvents and shrugs off water. The methyl group stuck at the second position doesn’t just nudge the boiling point a hair above simple quinoline; it gently tweaks its reactivity, making some reactions easier, some harder.
Technical Specifications and Labeling
In the real world, you can recognize a lab-savvy scientists’ bench by the neatly labeled bottles: “2-Methylquinoline (Quinaldine), C10H9N, ≥98% purity.” Labels stress its flammability risk. The safety data on those bottles tells a cautionary tale, nudging users towards gloves, goggles, and good ventilation. Purity matters quite a bit; a stray aldehyde or a poorly separated isomer can make a mess in sensitive syntheses. Reliable suppliers tie the batch specs to chromatographic fingerprints, ensuring that what you see is really what you get. The chemical’s modest shelf-life and its propensity for slow air oxidation mean you use it within a few months to dodge unwanted darkening and decomposition.
Preparation Method
Few stories about 2-methylquinoline skip past its trademark synthesis. The Skraup synthesis, originally meant for straight quinoline, found itself repurposed with tweaks that let methylated anilines take the stage. Operating anywhere from academic glassware to mid-scale industrial reactors, most routes start from aniline derivatives, blend in a dose of glycerol, and call on oxidizing acids or agents. With recent synthetic chemistry’s gentle embrace, catalytic systems—especially those built around transition metals and green solvents—have brightened up the process, inviting better yields and less environmental headache. Even so, old-school batch operations linger on, and scaling up always means juggling cost, safety, and product quality. The process doesn’t always get reinvented, but chemists keep nudging it forward, one optimization at a time.
Chemical Reactions and Modifications
2-Methylquinoline stands out not simply as a finished product, but as a starting material aching for transformation. That methyl group on the aromatic ring isn’t just window dressing; it attracts oxidative or electrophilic chemistry with a little more gusto than one might think. Researchers swap it for aldehydes under controlled conditions, stretch it out to acids, or sometimes tack on halogens to set up for cross-coupling. Countless times, a synthetic chemist has looked at a blank route map and realized the advantages of tossing quinaldine into the mix. Pharmacologists, too, have danced around the scaffold, functionalizing at the methyl or nitrogen sites with dreams of biological activity or new colorants. Each reaction, each derivative, adds another layer of possibility to this rather unassuming molecule.
Synonyms and Product Names
The name “quinaldine” flows nearly as often as 2-methylquinoline among those in the trade. Dive into historic publications or chemical supplier catalogs and you’ll also trip over “alpha-methylquinoline,” “quinaldin,” or even “2-quinolylmethane.” Some dye chemists reference it simply by its Registry Number or short abbreviations, but in labs, it’s the unique CAS number that ensures nobody mixes it up with its isomer cousins. Well-defined synonyms matter, especially as newer regulations demand unmistakable identification, reducing mix-ups in multi-national shipments and research citations.
Safety and Operational Standards
Handling 2-methylquinoline isn’t for the careless. Its toxicology hasn’t been dissected to the last decimal place like some larger-volume chemicals, but the warning signs ring out clearly—a skin irritant and an inhalation hazard with modest acute toxicity. Material safety data repeats the need for chemical-resistant gloves, eye shields, and chemical fume hoods that keep escaping vapors at bay. Spills demand proper absorbent and a quick, steady cleaning hand, not just a paper towel and a shrug. Labs where this chemical makes regular appearances train their team, refresh their safety drills, and build in extra steps to minimize dermal and respiratory exposure. Environmental regulations call for tight waste disposal, as the molecule lingers and resists half-hearted biodegradation. Over the years, regulatory standards have tightened, nudging chemists to balance research priorities against a rising bar for safe handling and disposal.
Application Area
2-Methylquinoline’s biggest footprint falls in the R&D trenches of dyes, pharmaceuticals, and agrochemical discovery. While it never headlined a blockbuster drug, it plays supporting roles across candidate libraries where heterocyclic modifications open doors to better drug properties or new mechanisms of action. Dye chemists count on it to coax out beautiful, long-lasting shades for inks and pigments. The molecule finds patches of utility in analytical chemistry, especially as an intermediate for more reactive or specialized compounds. In years past, it graced the world of anti-malarials, local anesthetics, and the prepping of tonics for microscopy stains; even now, its skeleton forms a central piece of active research into antitumor and antimicrobial agents. Anyone looking to swap one group for another on an aromatic scaffold, or to push nitrogen heterocycles into new biological territory, keeps a bottle of quinaldine close at hand.
Research and Development
In the research world, 2-methylquinoline still generates real excitement. Its adaptable structure appeals to those building chemical libraries hoping to unlock new drug leads. Medicinal chemists tend to tweak a methyl group for solubility improvements, cellular uptake, or metabolic stability. The past few decades saw increases in combinatorial chemistry, and 2-methylquinoline joined the crowd of heterocyclic scaffolds that passed through countless well plates. Chemoinformatics studies, aiming to draw links between structure and activity, highlight how core modifications in quinaldine can bridge the gap between chemistry and biology. Modern academic groups, sometimes teamed up with industry, examine its role in enzyme inhibition, receptor modulation, or new dye chemistry. Each year, a few new publications and patents underscore that 2-methylquinoline stays relevant, even as trends shift and technologies change.
Toxicity Research
What the scientific world knows about the toxicity of 2-methylquinoline owes much to careful animal studies and regulatory filing. Acute toxicity registers higher than many common lab solvents, but not high enough to trigger exclusion from hands-on research—provided you respect the protocols. Studies have shown mild to moderate irritation on skin and eyes; at higher exposures, it can produce signs of central nervous system depression or mild liver stress in rodent models. Long-term impact data is sparse, yet guidelines steer professionals towards caution, emphasizing protective behavior and ongoing monitoring. The absence of widespread reports of chronic poisoning makes sense, given typical use happens in controlled environments. Still, with new research tools, understanding of its metabolic fate and biocompatibility continues to deepen, and regulations tighten in harmony.
Future Prospects
Staring down the road, 2-methylquinoline seems well-positioned for a new chapter—especially in niche research projects and as source material for greener synthesis. The march toward sustainable chemistry, biocompatible dyes, and new pharmaceuticals lifts interest in nitrogen heterocycles every year. Improved methods for selective methylation, or milder, less polluting synthesis routes, could sharpen its appeal. Trends in AI-driven drug discovery highlight molecules with a balance of stability and reactivity, and 2-methylquinoline meets that mark for a range of potential leads. Industrial scaling—always slow to shift—signals some openness to improved processes as environmental costs are factored more closely into chemical economics. As data piles up, researchers and policy makers will likely push for richer safety assessments and more transparency in the life cycle of specialty chemicals like quinaldine. Its days as a humble building block haven’t ended; the next few decades could see this old favorite branching into fields nobody would have predicted back in the days of coal tar and ink-stained glassware.
What is 2-Methylquinoline used for?
Chemicals That Shape Industries
Spend a bit of time in any industrial lab, and quinoline derivatives crop up more than most folks expect. The compound 2-Methylquinoline, sometimes called quinaldine, isn’t just a lab curiosity. Its story runs through many real-world applications, turning up in unexpected places ranging from pharmaceuticals to dyes and even in some of the chemistry underpinning day-to-day life.
Building Block for Medicines
Chemistry researchers, both in academic institutes and big pharmaceutical companies, often rely on 2-Methylquinoline as a starting point for drug design. Many antimalarial, antibacterial, and anticancer drugs trace their chemical ancestry to quinoline rings. 2-Methylquinoline offers chemists a stable backbone that's easy to build on. This foundation leads to the creation of drugs that help real people facing tough diseases. You’ll find quinaldine derivatives making a difference in the fight against malaria, where the structure helps block the lifecycle of parasites inside the body.
Spotting Color in Dyes and Pigments
Quinaldine works well in certain dye-making processes. Synthetic dyes based on quinoline provide rich color that's tough to fade or wash out. Textile manufacturers, especially those producing bright and lasting colors for fabric, turn to 2-Methylquinoline derivatives. Paper and ink producers join this group, depending on the molecule for new colors that stick even under harsh conditions.
Photography and Light Technology
Traditional photography, before the digital age, leaned on chemistry. Quinaldine-based compounds played a part when it came to making sensitive emulsions used in film. Photographers and lab technicians depended on the reactions of quinoline derivatives to produce distinct images. Even now, some specialty films and research areas still call for these compounds.
Flavor and Fragrance Chemistry
You might not expect to find 2-Methylquinoline around anything edible, but chemistry has a way of surprising people. Flavors and fragrances often use trace levels of complex molecules to round out their profile. Some cigarette brands, for instance, use quinaldine as a flavor additive to tweak taste. Its aroma, earthy and slightly sweet, shows how chemistry moves from the lab to the senses.
Environmental and Lab Uses
Beyond production lines, 2-Methylquinoline crops up in environmental science. Scientists tracing pollution in rivers and lakes often detect quinoline derivatives. They help map industrial spill sources and trace contamination. In the lab, 2-Methylquinoline acts as both a reagent and an intermediate, helping form the building blocks for other technical chemicals.
Concerns and Safer Solutions
Whenever chemicals shape multiple industries, questions about safety and the environment follow. 2-Methylquinoline is no different. Some of its uses lead to emissions or waste that needs careful handling. Research keeps pushing for greener synthesis routes and better waste treatment. Chemists explore catalyst-driven processes and bio-based starting materials to cut down on pollution and resource use. Governments and watchdog groups track the chemical’s footprint, calling for tougher oversight in industries that use or make it in large amounts.
Real-World Impact
The chemistry of 2-Methylquinoline shows how one molecule reaches into medicine, color, taste, and even the guts of industry itself. Connecting the dots between science, production, health, and the environment always takes deep expertise and real-world experience. Safer practices, ethical choices, and continued research help ensure this compound keeps serving benefits without adding to environmental risk.
What are the physical and chemical properties of 2-Methylquinoline?
Getting to Know the Basics
2-Methylquinoline, often found in labs as a light yellow oil, owes most of its identity to that methyl group attached to the quinoline ring. Just one small alteration through methylation changes both the way it acts and what it can do in the real world. The chemistry behind 2-methylquinoline feels straightforward, yet its potential can surprise those who work with it closely.
Physical Properties That Make a Difference
This compound tends to catch attention because of its appearance: an oily yellow liquid, sometimes drifting into faint pale straw shades. It usually gives off a characteristic, slightly pungent odor that is tough to miss if you have handled these in a lab. Its boiling point sits near 238°C, higher than simple hydrocarbons, thanks to its aromatic ring, which gives extra stability and strength to the molecule. Melting point drops off quickly, around -1°C, so it rarely appears as a solid unless temperatures head below typical room conditions. Its density lingers near 1.1 g/cm³—heavier than water, lighter than many other organics.
What stands out from my own time in the lab: the solubility. Water barely budges it; only the smallest amount might dissolve. On the other hand, organic solvents like chloroform, ether, or ethanol draw it right in. That selective solubility gives researchers several options in extraction or purification. Each step can be planned knowing that it’s easy to keep water away or recover this component from an organic phase, making it a favorite choice when you need to separate mixtures smoothly.
How the Chemistry Unfolds
The methyl group attached at position 2 tweaks the reactivity in subtle ways. Working with quinolines, the ring structure already holds up well to heat and light. Add in that methyl at position 2, and the rate at which substitutions occur at nearby positions changes compared to plain quinoline. For example, electrophilic aromatic substitution takes a different path; the substituent can either slow down or speed up reactions, depending on what's attacking the ring.
Combustion happens readily in air. Lab safety data sheets stress its flammability, and it’s not just lip service. Spills near open flames have ended experiments early, so proper containment is wise. Reactions with strong oxidizing agents should stay closely watched; exothermic surprises aren’t unheard of among methylated aromatics.
Why These Properties Matter
Over the years, studies have shown how methylquinolines serve as bases for dyes, pharmaceuticals, and other specialty chemicals. The foundation starts with how well 2-methylquinoline can withstand processing or sit through harsh reactions without decomposing. Resistance to light and air extends shelf life, which helps manufacturers and researchers cut down on loss and waste. As a chemical intermediate, its reactivity profile means scientists get a stable starting point with just enough “give” for creative transformations. That’s part of why researchers in medicinal chemistry and material science keep returning to this building block.
Potential for Smarter Approaches
Plenty of work still focuses on greener synthesis and better safety practices. Organic solvents, while effective, often bring their own drawbacks. My own experience has shown newer solvent systems can reduce emissions and make waste easier to handle—sometimes swapping in bio-derived solvents lets you get the same separation without the environmental baggage. Devising safer storage protocols and standardizing handling practices keeps not just researchers, but everyone along the supply chain, better protected.
2-Methylquinoline may seem simple at first glance, but day-to-day experience with it brings a real appreciation for how small changes in structure create wide-ranging impacts on behavior and usefulness. From boiling points to reactivity, the details matter—and knowing them helps open up new avenues in research and industry alike.
Is 2-Methylquinoline hazardous or toxic?
Chasing Clarity in Chemical Safety
People tend to glaze over when chemicals come up, but the questions matter, especially with compounds like 2-methylquinoline. You run into this molecule in labs, in industrial settings, and sometimes as a by-product during different manufacturing processes. Many don’t realize how exposure to such substances could affect workers, or even nearby communities. I’ve seen crews wear gloves and masks around the stuff — not just because it smells odd, but because nobody wants to take chances with their health.
Digging Into the Science
Start with the basics. 2-methylquinoline, part of the quinoline chemical family, shows up as a pale yellow liquid. Studies point to this compound being toxic if swallowed, inhaled, or if it soaks into your skin in significant amounts. According to resources like the European Chemicals Agency and the National Center for Biotechnology Information, exposure can lead to irritation in the eyes, nose, throat, or lungs. High doses, like those in certain animal studies, caused some rats to grow tumors, raising eyebrows about its possible cancer risk in humans too. Those are findings that stick with you.
Long-Term Concerns
Even at lower concentrations, repeated or ongoing exposure can start to add up. Many solvents and aromatic compounds carry longer-term risks for things like liver toxicity or central nervous system effects. 2-methylquinoline fits into this group. Just because a worker isn’t coughing today doesn’t mean nothing’s happening in the background. Regulatory agencies keep a close watch; the U.S. Environmental Protection Agency notes gaps in research on chronic effects, so nobody’s rushing to call it safe. There’s a lesson in that — gaps in science shouldn’t translate to gaps in protection.
What’s Actually at Risk
It's tempting to shrug this off as someone else’s worry, but industry moves fast. Sometimes, spills happen or someone skips a step with personal protective equipment. That leaves room for exposure, especially if a plant doesn’t have proper ventilation or training in place. I’ve watched otherwise careful folks get complacent when things get busy. In those moments, even a small spill of 2-methylquinoline can become a health scare. If the chemical soaks into work clothes or pools on a surface, people can absorb more than they realize.
A Safer Path Forward
There’s no magic bullet, but every facility handling this material needs a clear plan. That means proper labeling, using fume hoods, and regular safety drills. Newer engineering controls — like enclosed systems or improved drainage — can keep exposure way down. Don’t overlook the basics: gloves, goggles, and clean-up kits should be easy to access, and everyone should know how to use them. Updated safety data sheets don’t collect dust; they become living documents that workers actually use.
Education matters just as much as gear. I’ve seen how short, regular training sessions give everyone more confidence and fewer surprises on the job. Let’s face it: chemicals like 2-methylquinoline aren’t going away, so the best answer lies in solid science, real-world experience, and a little bit of openness about what we still don’t know. Being proactive with these systems not only keeps people safe, but helps build trust with regulators and the public who want to know what’s drifting through the air beyond the factory fence.
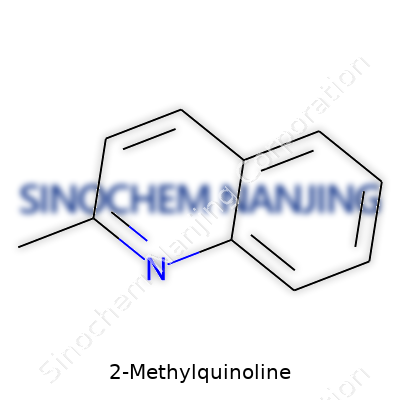
What is the molecular formula and structure of 2-Methylquinoline?
Molecular Blueprint: Why 2-Methylquinoline Stands Out
2-Methylquinoline shows up in labs and chemical plants for good reason. With the molecular formula C10H9N, it’s clear that there’s more here than just a bunch of carbon atoms strung together. The story behind this molecule goes deeper than numbers. Sticking a methyl group onto a quinoline backbone pulls in new possibilities, shifting both the physical behavior and reactivity of the molecule in ways that anyone working with dyes, pharmaceuticals, or chemical synthesis should care about.
Structural Sense: Building Blocks with Impact
Look at the skeleton of 2-Methylquinoline: two fused aromatic rings, one benzene and one pyridine. The pyridine part, with its nitrogen, tweaks the electron flow and draws the eye of any chemist interested in what can be added or swapped out next. The methyl group at the “2 position” doesn’t just round out the name, it changes the map of the molecule—impacting boiling point, solubility, and where chemical reactions take place. If you picture the structure, you'll see a methyl tag jutting off right next to the nitrogen atom. This layout drives the molecule's performance in reactions and helps pinpoint conditions where it thrives or fails.
The Stakes: Why Structure and Formula Matter
In my own experience with heterocyclic chemistry, placing a methyl group can steer a pathway or block one entirely. For the companies working to fine-tune pharmaceuticals, this isn’t academic. Small changes to molecular layout mean the difference between a potent drug and an ineffective one. 2-Methylquinoline, thanks to its structure, unlocks a set of reactions that plain quinoline just can’t match. You see it pop up as a starting point for making antimalarial agents, antimicrobial compounds, and even specialized dyes. Researchers rely on consistent molecular blueprints to predict performance, avoid hazards, and cut down on pricey trial and error.
Scientific Scrutiny and Data You Can Trust
The backbone of chemical safety and effectiveness rests on transparency and proof. Data about 2-Methylquinoline comes from direct measurements like NMR, X-ray crystallography, and elemental analysis. Reputable databases, such as PubChem and ChemSpider, back up the published molecular formula and structure. Peer-reviewed articles in journals like Journal of Organic Chemistry document these properties. Anyone who’s worked at a bench knows the value of trustworthy data—errors in molecular diagrams or formulas could ruin an entire project or create dangerous situations in a lab. With synthetic chemistry, relying on confirmed structures helps avoid waste, protects workers, and ensures the final products do what they’re supposed to.
Moving Ahead: Checking and Using Core Information
For educators and students, knowing the correct molecular formula and the finer points of structure sets a foundation for deeper learning. Industrial and pharmaceutical chemists take that information and use it to derive new molecules, run targeted experiments, and address challenges in disease treatment or material design. The key is always blending solid fundamental facts with practical applications.
Concrete Solutions for Better Outcomes
Regular training sessions on interpreting structural diagrams, coupled with real-world examples of why these details matter, can boost knowledge and safety in both academic and industrial settings. Clear labeling and up-to-date documentation, drawn from trusted sources, stop mistakes before they start. In my career, the most valuable insights have come from environments where everyone takes time to double-check chemical identities and talk through the consequences of even minor changes at the molecular level. It’s hard work, but it makes for science that works—reliably, safely, and with real impact.
How should 2-Methylquinoline be stored and handled?
What Makes 2-Methylquinoline Demanding in Storage?
Anyone who has spent time in a lab knows there’s no room for shortcuts with chemicals like 2-Methylquinoline. With its strong, pungent odor and liquid state, it has the knack for escaping loose caps and sniffing out the tiniest crack in a seal. I’ve opened containers that left stains on both the air and my gloves for hours. Because it’s flammable and toxic if inhaled or swallowed, complacency doesn’t work here. This isn’t a chemical you leave sitting out by the coffee pot.
What the Data Says about Safe Storage
Manufacturers list its flash point as around 108°C, which seems high, but labs rarely run that hot. The real issue—fumes and accidental ignition. Storing it in a tightly closed amber glass bottle away from direct sunlight helps. Heat, sparks, or static can set it off. A chemical storage fridge—one without exposed electrical parts inside—offers peace of mind. Lighting and temperature swings should be kept in check. Forget makeshift shelving or leaving it close to acids and oxidizers. This chemical won’t forgive sloppy cohabitation in cabinets.
Handling Mistakes Cost More than Embarrassment
A splash is never "just a splash" with this compound. I’ve seen seasoned chemists realize too late that just a few drops on the skin means irritation and a stink that clings for days. Splash goggles, gloves made of nitrile, and a full lab coat cut down the accidents. Keep the work in a fume hood—with its sharp, irritating aroma, even a few moments outside control can make breathing harsh. I once forgot this step and ended up woozy. Safety showers and eyewash stations within reach save the day when spills or splashes happen.
Spills and Disposal: No Excuse for Guesswork
Trying to mop up a chemical spill with a handful of tissues or a dry rag guarantees trouble. For 2-Methylquinoline, using a spill kit with absorbents specifically made for organic liquids matters. Every lab should have the right kit on hand. Bags, rags, and gloves used in cleanup must get sealed in designated hazardous waste bins. Down the drain or in the trash isn’t an option, as local environmental laws take this seriously. I’ve watched colleagues fret about a spill, because one quick sweep with the wrong gear polluted an entire workbench for months.
Training and Documentation—Not Just Paperwork
I used to skip past the Safety Data Sheet, figuring experience would fill the gaps. That’s a short-sighted approach. Proper labeling in plain language, easy-to-spot hazard symbols, and up-to-date documentation mean everyone working nearby can react without hesitation. Sharing lessons and running drills for spills or exposures once every few months builds team reflexes. When information stays locked in a filing cabinet or stored away in someone’s memory, even minor accidents can mutate into emergencies fast.
Where Teams Shine and Systems Matter
Chemicals like 2-Methylquinoline highlight the gap between a safe lab and a disaster waiting to happen. Individual vigilance counts, but a culture of looking out for one another cements real safety. I’ve noticed that labs with eye-level signage, clean bench spaces, and regular disposal routines have fewer stories of scares and near misses. Handling this compound the right way blends personal accountability with clear policies and teamwork, and this approach helps avoid both injury and downtime.
| Names | |
| Preferred IUPAC name | Quinolin-2-ylmethane |
| Other names |
Quinaldine 2-Methylchinolin 2-Methyl-quinoline α-Methylquinoline |
| Pronunciation | /tuːˈmɛθɪl.kwɪnəˌliːn/ |
| Identifiers | |
| CAS Number | 91-63-4 |
| Beilstein Reference | 120825 |
| ChEBI | CHEBI:34597 |
| ChEMBL | CHEMBL19039 |
| ChemSpider | 131769 |
| DrugBank | DB08542 |
| ECHA InfoCard | ECHA InfoCard: 100.012.682 |
| EC Number | 2.3.1.86 |
| Gmelin Reference | 9566 |
| KEGG | C01919 |
| MeSH | D015952 |
| PubChem CID | 7007 |
| RTECS number | GN8225000 |
| UNII | S0H2FQ6997 |
| UN number | UN2656 |
| Properties | |
| Chemical formula | C10H9N |
| Molar mass | 143.19 g/mol |
| Appearance | Yellow crystalline solid |
| Odor | amine-like |
| Density | 1.006 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.7 |
| Vapor pressure | 0.0781 mmHg (25°C) |
| Acidity (pKa) | 5.10 |
| Basicity (pKb) | pKb = 9.54 |
| Magnetic susceptibility (χ) | -64.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.629 |
| Viscosity | 1.540 cP (25°C) |
| Dipole moment | 2.23 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 206.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 160.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5231.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332 |
| Precautionary statements | Precautionary statements: P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-1-0-Alk |
| Flash point | 113 °C |
| Autoignition temperature | 480°C |
| Explosive limits | Explosive limits: 1.3–7.8% |
| Lethal dose or concentration | LD50 oral rat 670 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 670 mg/kg |
| NIOSH | N7442 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 mg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Quinoline 4-Methylquinoline 8-Methylquinoline 2-Phenylquinoline Isoquinoline |

