2-Methylpentanediamine: More Than Just a Niche Chemical
Historical Development
Scientists have not always placed much emphasis on 2-methylpentanediamine, a compound that wears its straight-chain, branched structure with quiet humility. Even as industrial chemists in the 20th century churned out bulk aminated molecules for rubbers and plastics, this diamine mostly stayed behind the scenes, overshadowed by flashier siblings like hexamethylenediamine. Only with the growth of specialty polymers, advanced coatings, and performance-driven adhesives did folks start taking a closer look at what this branched diamine could bring to the table. Synthetic methodology began to mature in post-war research labs, especially as the petrochemicals boom opened doors to higher-pressure hydrogenation and selective amination. Gradually, new routes and derivatives turned early experimental curiosities into sources of value for end users demanding a blend of toughness, flexibility, and chemical resistance.
Product Overview
No single application defines 2-methylpentanediamine. Instead, what stands out is its sheer versatility. Producers and users learned to appreciate how a single methyl group tweak could start to change polymer performance, setting it apart from straight-chained analogues. In comparison to common diamines, this compound appeals to engineers aiming to dial in flexibility or durability, depending on the system. While others bank on commodity pricing and huge volumes, this molecule carved its niche by solving headaches in epoxies, polyamides, and curing systems that hit a performance wall with more basic building blocks. For industries looking to move from standard solutions to something tailored, it offers a way to leap over old limitations, whether that means lower viscosity, altered thermal behavior, or other property control knobs.
Physical & Chemical Properties
The shift of a methyl group from the backbone does more than just show off the power of organic chemistry. Painstaking work on the lab bench demonstrated its tangible impact on melting points, boiling points, and solubility profiles, not to mention a unique flavor of reactivity. In its pure form, 2-methylpentanediamine takes shape as a clear to slightly yellowish liquid with a sharp, characteristic odor—no getting around the fact that most aliphatic diamines pack a punch to the nose. It mixes well with water and most common organic solvents, a trait that helps drive its appeal in resin modification and crosslinking. There’s no sidestepping its strong basicity, which lets it take part in a wide range of reactions from amidation to curing, giving it a practical range that otherwise would not come so easily from more straightforward diamines.
Technical Specifications & Labeling
Labels reflect more than just regulatory requirements; they give end users a sense of what to expect. Folks handling 2-methylpentanediamine learn quickly that even small impurities—straight-chain analogs, unreacted amines, or residual water—can throw off final performance. Producers aiming for high-value applications find themselves chasing down purity levels above 98 percent, with strict limits on color, amine value, and water content. GHS labeling and hazard statements don’t just tick boxes for compliance but also highlight the care needed for safe handling, especially since diamines can be tough on skin and eyes. Being proactive about lot traceability, storage temperature, and shelf-life makes it easier to stay out of trouble in both the plant and the lab.
Preparation Method
Not every approach works for everyone. The current go-to methods usually trace back to reductive amination or hydrogenation routes, using a blend of catalysts and reaction conditions dialed in to balance yield with purity. Selectively amining intermediates, often derived from petrochemical feedstocks like methylpentanone, calls for precise control of temperature and pressure. Industrial players that set up these processes find themselves running the gauntlet from small-scale glassware all the way to continuous systems handling hundreds of kilos at a shot. Reaction tuning, coupled with solvent selection and purification steps like distillation or extraction, marks the difference between a cost-effective process and a chemistry headache that just eats into the bottom line.
Chemical Reactions & Modifications
Once in the bottle, 2-methylpentanediamine does not stay idle for long. Its combination of both terminal amine groups and mid-chain branching lines it up for a host of modifications, from classical condensation reactions to co-monomer selection in engineered polyamides. In resin chemistry, it plays a central role in setting crosslink density and imparting unique thermal behavior, opening doors for new types of adhesives that shrug off heat and humidity. Strecker syntheses, Michael additions, and epoxy cure formulations often single out this molecule when a little extra flexibility—or a change in mechanical modulus—would tip performance in the right direction.
Synonyms & Product Names
Chemists don’t always settle for a single name. Depending on supplier, catalog, or industry jargon, the compound often appears as 2-methyl-1,5-pentanediamine or 1,5-diamino-2-methylpentane. Some resin producers and specialty catalogues give it in-house names with letter-and-number codes, especially when tailoring derivatives for niche applications. That can cause plenty of headaches for newcomers, especially in markets where imports or local transliterations muddy the waters. Staying up-to-date on IUPAC conventions and supplier SDS documentation cuts down on mix-ups.
Safety & Operational Standards
Protective gear matters whenever handling this stuff. Anyone who has worked with small-molecule diamines learns quickly that gloves, goggles, and a well-ventilated hood aren’t just suggestions—they’re basic survival, given the irritant effects. Even modest skin exposure can lead to dermatitis over time, and splashes put eyes at risk. Strong odors make fume control non-negotiable. Plants and labs draw on established protocols, from spill procedures to first-aid plans, and typically reinforce these during onboarding and safety audits. Regulatory oversight, both local and cross-border, keeps a close watch on storage volumes and transport container specifications, since leaks or releases don’t just endanger workers but can also trigger wider environmental headaches. Reinforced secondary containment and regular container inspections go a long way in preventing accidents.
Application Area
The sweet spot for 2-methylpentanediamine remains systems where a balance of flexibility, chemical resistance, and thermal stability make or break the final product. Epoxy resin formulators reach for it when they want to produce adhesives and coatings able to take a beating in automotive and construction settings. Polyamide engineers value it for nylon variants that need a bend without loss of toughness, such as cable protectors, under-hood parts, or wear-resistant industrial components. In the world of specialty chemicals, formulators lean on its profile when designing hardeners, corrosion-resistant paints, biocompatible materials for medical devices, or even additives for lubricants. I’ve seen it bridge gaps in performance that no other single additive covers, especially as sustainability and lower-tox approaches limit the viable choices for demanding applications.
Research & Development
University labs and corporate R&D outfits continue to push this molecule into new spaces, especially as green chemistry gains steam and conventional amines fall under greater regulatory pressure. The structural features of 2-methylpentanediamine make it a strong candidate for designing monomers that sidestep legacy challenges around shelf stability, low-temperature flexibility, or environmental fate. Next-gen formulations look beyond just durability, asking for better processability, tuneable mechanical strength, or even improved recyclability—traits that demand a rethinking of which diamines make it into the mix. Developments in bio-based and renewable feedstocks could soon reshape how this molecule is sourced, which would open the door to lower-carbon materials for high-profile markets from automotive to consumer electronics.
Toxicity Research
Understanding the toxicology of 2-methylpentanediamine means digging past the raw LD50 numbers and getting into real-world exposure scenarios. While acute toxicity registers as moderate compared to aromatic amines, chronic effects remain under close study. Short-term skin or eye contact easily causes irritation, and ingestion or inhalation at high levels prompts a search for emergency response. Regulatory agencies and corporate risk groups are watching for long-haul effects, especially since some branched amines may disrupt biological membranes and enzyme systems in sensitive species. Animal studies, coupled with sophisticated cell assays, paint a picture that supports strict exposure limits and thorough personal protective equipment, especially for those handling bulk quantities or working in closed processor environments. Environmental management teams also track wastewater outflows and air handling systems for trace releases.
Future Prospects
The next decade will likely see 2-methylpentanediamine moving out from behind the curtain, especially as traditional chemistries run up against regulatory and sustainability obstacles. Forward-thinking engineers are beginning to view this molecule as more than a tweak on legacy systems, instead seeing it as a platform for high-performance, cleaner materials that broaden the toolkit for everything from green construction to automotive light-weighting. Investment in renewable feedstock processes promises to lower the environmental footprint, reducing dependency on fossil-derived intermediates. Collaborative R&D between academia, industry, and regulators could lead to safer process designs, closed-loop recycling, and stronger integration with bio-based materials supply chains. As the world demands longer lifespans, recyclability, and better protection from harsh environments, 2-methylpentanediamine’s blend of structure and reactivity holds promise for engineers and chemists with an eye toward the future, not just in the lab but on the factory floor and in the products people use every day.
What is 2-Methylpentanediamine used for?
What Exactly Is 2-Methylpentanediamine?
2-Methylpentanediamine sounds like a mouthful, but the reality behind the chemistry is more straightforward. This compound belongs to the family of diamines, organic molecules holding two amine groups. It features a six-carbon backbone, with a methyl group adding some structure. These characteristics help it “click” with other chemicals, making it valuable for specific industrial purposes.
A Behind-the-Scenes Key Player in Epoxy Resins
Folks working in adhesives and coatings know that resins form the backbone of tons of modern products. 2-Methylpentanediamine gets used as a hardener for epoxy systems. The story here is worth telling: Epoxy adhesives, those durable glues used in planes, cars, wind turbines, and construction gear, rely on hardeners to develop real strength. Without this stuff, the resin never forms the rock-solid networks that hold up under stress. In my years hanging around hardware stores and job sites, nobody gets excited over resin chemistry, but without the right hardener, half those “miracle fixes” would be soggy goop.
This particular amine doesn’t just do the basic job. Because of its molecular shape, it often helps produce materials with solid chemical resistance and toughness. These features matter for building bridges, sticking moldings onto concrete, and even patching up water pipes. Engineers want products that shrug off chemicals and bad weather, and 2-Methylpentanediamine helps make that happen.
Rising Demand from the Polyurethane Market
Polyurethane foams pop up in mattresses, insulation panels, shoes, and even refrigerators. 2-Methylpentanediamine turns up in specialty isocyanate curing systems. Polyurethanes work on a similar principle as epoxies: they need something to launch the reaction that “locks” everything together. This diamine plays a role where fine-tuning the foam’s hardness or flexibility matters. You might sleep every night on something made possible by a chemical you’ve never heard of, and that’s no exaggeration. Reports from markets like Asia and North America suggest increased demand here, driven by consumer trends in comfort and energy efficiency.
Supporting Niche Applications Across Industries
Beyond the obvious uses, 2-Methylpentanediamine joins the chemical toolkit for making corrosion inhibitors and certain pharmaceuticals. Some research points to specialty intermediates for agrochemicals—without this ingredient, a few innovative products wouldn’t leave the lab. Having worked with teams that troubleshoot plant problems, specialty diamines sometimes deliver the exact performance boost process engineers need. Not everything scales up from a textbook reaction, so an unusual ingredient like 2-Methylpentanediamine can save a project.
Safe Use and Looking Ahead
Industrial chemicals demand respect. 2-Methylpentanediamine can be a skin and eye irritant, so anyone handling it needs gloves, goggles, and a good ventilation system. Companies monitor potential environmental impact and workplace exposure closely, guided by health and safety rules set by groups like OSHA and the European Chemicals Agency. Decades ago, some factories didn’t care about these risks—today’s best practices protect workers and communities way better.
Room for Innovation and Improvement
There’s always a push to lower emissions and replace hazardous substances, even in specialty areas. Chemists keep searching for even safer, outperforming alternatives. Green chemistry principles inspire new approaches, focusing on less waste and reduced toxicity. Researchers can sometimes tweak formulations using biobased or recycled starting materials, although performance and price remain critical for commercial choices.
2-Methylpentanediamine might not show up in everyday conversation, but the world leans on it in ways that touch all of us. Whether you’re driving to work, relaxing at home, or stepping onto a hospital floor, you’re never too far from the silent strength that specialized chemistry provides.
What is the chemical formula of 2-Methylpentanediamine?
Understanding the Building Blocks
Chemistry shapes daily routines, from the plastics in household items to the coatings on cars. Getting down to the basics, a compound like 2-Methylpentanediamine packs value not just in its name but also in its formula. It’s C6H16N2. On the face of it, that’s six carbons, sixteen hydrogens, and two nitrogens. I’ve seen how a simple formula like this can open doors to multiple applications, especially in fields like polymer manufacturing, water treatment, and pharmaceuticals.
The Importance of Structure in Real-World Uses
Structurally, 2-Methylpentanediamine combines a straight chain with a methyl group branching off the second carbon. Chemists appreciate this setup for what it brings to the table: more versatility during synthesis and a bit of a challenge during purification. Coming from my own time in the lab, I can say that even a minor change in structure often shakes up physical properties like melting point, solubility, and stability. This particular molecule, because of its amine groups, acts as a useful bridge in reactions where basicity and reactivity drive the show. Factories turn to it for making resins and hardeners, where it serves as a cross-linker giving products extra durability.
Quality, Safety, and Environmental Impact
Manufacturers focus their attention on compounds like 2-Methylpentanediamine not only for performance but also for safety. The nitrogen atoms introduce certain hazards—think of strong odors or the risk of skin irritation. I remember how strict our safety protocols got any time diamines showed up in shipments. Some colleagues faced irritation just from airborne particles. So, using proper PPE and ventilation cuts down on risks. Thinking about the environment, the trend toward greener processes isn’t lost on this chemical either. Achieving the right formula is just a starting point. Disposal and production both require serious attention, keeping water streams and workers safe from amines leaking into the ecosystem.
Why The Correct Formula Is Critical
Accuracy in chemical formulas prevents mishaps both minor and major. Any chemist entering the lab understands that a typo in the formula can mean a failed batch or even a dangerous byproduct. In drug manufacturing, getting the formula wrong could have consequences on patient safety. Speaking from handling regulatory paperwork, compliance officers check and recheck the formula before approval. Two extra hydrogens or carbons and you’re not just risking discarded material—you’re risking credibility and public trust.
Solutions: Precision and Knowledge Sharing
To tackle these challenges, the industry benefits from rigorous training and up-to-date databases. New chemists get hands-on exposure through guided synthesis and analytic tests—IR, NMR, and MS all help confirm this compound’s identity. Ongoing workshops keep seasoned workers alert to best practices for handling compounds that might pose health or environmental concerns.
Looking ahead, digital tools and automation ramp up safety and accuracy. Real-time data checks, transparent supply chains, and cooperative sharing of safety experiences all contribute to a safer, more reliable chemical industry. This keeps 2-Methylpentanediamine—and every other compound—valued not only for its function, but also for the careful hands that handle it.
Is 2-Methylpentanediamine hazardous to health?
Understanding 2-Methylpentanediamine
2-Methylpentanediamine turns up in industrial settings as a chemical used to make polymers, coatings, and adhesives. The compound doesn't get much attention at home, but people who work in factories or labs making plastics or specialty chemicals might see it in action. In my own years dabbling in various chemical plants, I saw thousands of drums filled with things nobody talks about on the news — 2-Methylpentanediamine fits that bill.
Routes of Exposure Matter
Skin contact, inhalation, or accidental spills put workers at risk. I’ve watched operators handle drums without gloves, and sometimes masks get forgotten during a long shift. The National Institute for Occupational Safety and Health (NIOSH) data points out eye, nose, and throat irritation from exposure to similar diamines. If you don’t wear the right protection, splashes could lead to skin burns or blisters, and fumes might irritate the airways. Chemical companies are careful now, but history teaches us that rules only work if people follow them.
Scientific Evidence and Toxicology
It pays to look at reliable data. The European Chemicals Agency classifies 2-Methylpentanediamine as hazardous, warning about corrosive effects on skin and eyes. Animal studies with closely related compounds showed lung and kidney effects after prolonged exposure. Humans experience redness, swelling, pain, or burning after direct skin or eye contact. Breathing in vapors brings coughing or chest discomfort — long shifts in tight spaces can make this worse.
High doses might even cause systemic toxicity, based on case reports from medical journals. A story sticks out from a training in Louisiana: a worker rushed into a spill without protection, thinking urgency mattered more than safety. The aftermath left long-term scars and breathing trouble. These aren’t rare events; they’re the backbone of calls for tighter safety measures.
Lessons from Chemical Handling
No chemical acts alone. Poor storage, old containers, leaky pumps, or shortcuts in transferring raw materials all add up. In my experience, near-misses never make headlines, but every factory has them. Supervisors keep safety sheets handy, yet plenty of workers rely on word-of-mouth. Consistent training, real-time monitoring of air quality, and health checkups could stop injuries before they begin. PPE (Personal Protective Equipment) only helps when people actually use it, and management checks on supplies instead of waiting for an accident.
Looking Forward with Responsibility
The chemical industry knows how to reduce risks. Automation keeps hands away from danger zones, better ventilation lowers inhalation risk, and well-labeled containers mean nobody’s confused mid-shift. Even so, enforcing rules takes vigilance, not just paperwork. The Occupational Safety and Health Administration (OSHA) recommends clear signage, thorough incident tracking, and medical surveillance for those working with hazardous amines.
Beyond factories, local medical teams should get a heads-up on symptoms tied to chemical exposure in their communities. A few basic questions about chemical work at intake can bring at-risk workers help sooner. As new research appears, it’s up to manufacturers and regulators to keep limits current. No one wants to learn safety lessons the hard way.
What are the storage conditions for 2-Methylpentanediamine?
Why Proper Storage Matters
Ask anyone working in a chemical lab about storing diamines, and you’ll hear a range of stories. Some will mention ruined vials and the clean-up that follows a spill. Others will talk about solvents turning yellow after contact with air or failing syntheses from using old, contaminated samples. Once, I found out the hard way that keeping 2-Methylpentanediamine out on a busy bench led to strange smells, ruined project material, and annoyed colleagues. Keeping storage practices simple and safe plays a huge role in daily lab life.
The Basics: Keep it Cool and Airtight
2-Methylpentanediamine needs to stay in a cool, dry place, away from direct sunlight or heat sources. I keep my supply in a chemical refrigerator, separate from food or drink, and away from acids. Heat speeds up unwanted reactions or evaporation, so storing it away from radiators or sunlight really matters. Humid shelving invites moisture in, and moisture spells trouble for most amines.
Handling the Odor and Vapors
Most diamines, 2-Methylpentanediamine included, have a sharp, ammonia-like odor. Without good ventilation, the smell gets everywhere—and exposure isn’t healthy. If you work somewhere with poor airflow, vapors linger and can irritate eyes and lungs. I always store the bottle tightly closed and keep it in a vented cabinet designed specifically for amines and other organics. Occupational Safety and Health Administration (OSHA) guidelines highlight the importance of avoiding inhalation and minimizing exposure to fumes, especially in poorly ventilated rooms.
Container Choices Make a Difference
Over the years, I’ve learned never to use ordinary containers for things like 2-Methylpentanediamine. Sealing it in high-density polyethylene (HDPE) or glass with strong caps works best. Some plastics degrade over time from chemical contact, and leaky lids spell disaster. Keeping the original label and hazard markings on the bottle helps with quick identification and avoids accidental mix-ups. In my experience, a missing label in a busy storage room slows everyone down and raises safety concerns.
Fire Risks: Prevention Beats Cleanup
Flammable warnings on bottles are there for a reason. 2-Methylpentanediamine catches fire easily around open flames or sparks. Storing it far from ignition sources and flammable solids pays off in the long run. Fire codes ask for flammable chemicals to stay inside flame-retardant cabinets. Once I walked into a lab where an otherwise diligent team stored all their amines right next to an old toaster oven. Just one accident could have turned a regular day into an emergency.
Minimizing Waste and Spills
It’s tempting to stock up, but keeping only what’s needed helps reduce headaches when old chemicals turn bad or containers fail. I check bottles for leaks, corrosion, or crystalline residue and transfer any suspect material to safer containers, labeling and dating everything along the way. Regular checks, good housekeeping, and keeping materials sealed when not in use protect both the lab crew and the environment. If a spill happens, the right absorbents, good ventilation, and speedy cleanup lower potential health and safety risks—all things very real to anyone who’s scrubbed residue from concrete or lab tile.
Building Better Storage Habits
Most of the time, careful storage isn’t about fancy systems. It’s about double-checking lids, keeping containers in the right place, and sharing best practices with your colleagues. Simple measures—like using vented flammable cabinets, checking labels, and teaching proper spill response—make sure small mistakes don’t become big problems. Sharing lessons learned goes a long way in keeping everyone safe.
How should 2-Methylpentanediamine be handled safely?
Real Dangers Behind the Science
Most people outside a lab haven’t heard of 2-Methylpentanediamine, but anyone working with industrial amines understands the risks. This clear liquid holds more than a chemical formula; it can irritate skin, eyes, and lungs. Sloppy handling once led to chemical burns on my colleague’s wrist, enough for that lesson to stick. Too often, overconfidence in routine leads to shortcuts. You can’t see or smell every risk, so consistent habits matter.
Personal Protection Isn’t Optional
A good pair of chemical-resistant gloves and safety glasses shield more than everyday hazards. I use nitrile gloves because latex just won’t hold up if you get a splash. Open-toe shoes have no place in a lab. Working one afternoon, a quick slip sent droplets over a neighbor’s shoes. She ended up with red, burning skin. The right shoes and a simple lab coat or apron would have avoided that pain. Splash-proof goggles are just as important—standard glasses don’t cover the sides.
Ventilation Makes a Difference
A single whiff of 2-Methylpentanediamine leaves a bite in your throat and nose. Without decent ventilation, that discomfort turns into serious breathing problems. One facility I visited ran their synthesis in a closed bay, using fume hoods connected to the outside with HEPA and charcoal filtering. You don’t need a space-age system for safety, but local exhaust like a chemical fume hood at minimum pulls vapors away before they cause harm.
Spill Response: Planning Beats Panic
Spills never come with advance warning. Years ago, a broken beaker on a bench sent process engineers running for the safety shower. Quick thinking with absorbent pads, neutralizer, and proper disposal meant no long-term impacts. Regular drills and clear spill kits should sit close to all workstations. Emergency eyewash stations do their job only if you know where to find them, and they aren’t blocked by storage boxes.
Labeling and Storage: Small Steps Prevent Chaos
Mistaken identity in the lab—grabbing the wrong bottle—turns a regular shift into a near miss. I mark every container right as it arrives, using chemical-resistant labels and clear hazard pictograms. Storing 2-Methylpentanediamine away from acids and incompatible agents reduces the risk of unexpected reactions or fires. Good storage involves sealed containers, out of direct sunlight, at a stable temperature.
Training: The Real Line Between Safe and Sorry
People remember stories, not lectures. Sharing real-life incidents—like the glove breach or that time someone’s mask slipped—helps new staff see why each step matters. Regular check-ins, walkthroughs, and refresher sessions keep everyone sharp and invested. Safety is a team sport. Ignoring training is a shortcut to accidents.
Steps Toward Safer Workplaces
Easy access to up-to-date safety data sheets keeps answers nearby for all workers. Reviewing ventilation equipment and storing first aid supplies where everyone can find them closes the gap between assumption and action. Having emergency contact numbers posted by phones, strict rules on eating or drinking in chemical areas, and a culture that rewards smart choices, not just speed, make a real difference.
Putting Safety into Practice
No single rule keeps every workplace safe, but habits formed around protective gear, ventilation, labeling, training, and spill response shape an environment where people feel prepared. Responsible chemical handling isn’t just about following law or policy; it’s about going home healthy after each shift. Those choices pay off every day—ask anyone who’s gotten it wrong once.
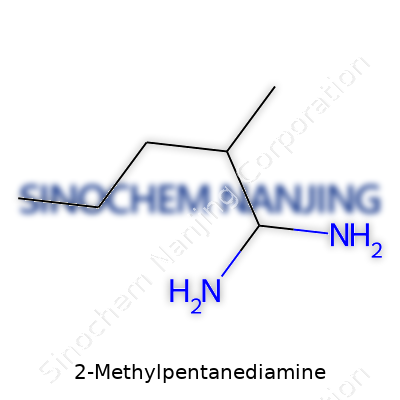
| Names | |
| Preferred IUPAC name | 2-Methylpentane-1,5-diamine |
| Other names |
2-MPD 2-Methyl-1,5-pentanediamine Pentamethylenediamine, 2-methyl- 2-Methylpentane-1,5-diamine |
| Pronunciation | /tuː ˌmɛθəlˌpɛnˈteɪndɪəˌmiːn/ |
| Identifiers | |
| CAS Number | 3077-12-1 |
| Beilstein Reference | 81863 |
| ChEBI | CHEBI:139607 |
| ChEMBL | CHEMBL3306537 |
| ChemSpider | 177408 |
| DrugBank | DB13907 |
| ECHA InfoCard | 41e731a1-c8e2-4243-83fa-e3a1ae16f701 |
| EC Number | 620-965-2 |
| Gmelin Reference | 162302 |
| KEGG | C19206 |
| MeSH | C014388 |
| PubChem CID | 14259 |
| RTECS number | SY8575000 |
| UNII | Q4E8D8K42Q |
| UN number | 2734 |
| CompTox Dashboard (EPA) | DTXSID4057518 |
| Properties | |
| Chemical formula | C6H16N2 |
| Molar mass | 116.20 g/mol |
| Appearance | Colorless to light yellow transparent liquid |
| Odor | ammonia-like |
| Density | 0.805 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | -0.38 |
| Vapor pressure | 0.34 mmHg (25°C) |
| Acidity (pKa) | 10.7 |
| Basicity (pKb) | pKb = 3.34 |
| Magnetic susceptibility (χ) | -52.0e-6 cm³/mol |
| Refractive index (nD) | 1.4277 |
| Viscosity | 3.4 mPa·s at 25 °C |
| Dipole moment | 2.31 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -59.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3976.7 kJ/mol |
| Hazards | |
| GHS labelling | **GHS labelling for 2-Methylpentanediamine:** ``` GHS07, GHS05 ``` |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314 |
| Precautionary statements | Precautionary statements: P261, P264, P280, P301+P312, P330, P304+P340, P305+P351+P338, P312, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1 |
| Flash point | 70°C |
| Autoignition temperature | 245 °C |
| Explosive limits | 2.0-11% (V) |
| Lethal dose or concentration | LD50 oral rat 1690 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1690 mg/kg |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Methylpentanediamine: Not established |
| REL (Recommended) | 200 mg/L |
| IDLH (Immediate danger) | IDLH: 15 ppm |
| Related compounds | |
| Related compounds |
1,2-Diaminopropane Hexamethylenediamine 2-Methyl-1,5-pentanediamine |

