2-Methylbutane: A Deep Dive into an Everyday Laboratory Hydrocarbon
Historical Development
Decades ago, chemists started looking for hydrocarbons that could solve real workbench problems. 2-Methylbutane, better known by some as isopentane, quietly carved out a place in labs across the world. In the mid-20th century, as organic chemistry matured into an industrial power, folks needed simple, volatile organics to wash, crystallize, or extract their ever-growing family of chemicals. Production methods improved as refinery throughput soared, helping 2-methylbutane shed its status as an afterthought from petroleum cracking and gain real recognition. As chemical companies expanded, 2-methylbutane showed up in freezers, glassware, solvents, and as a building block for making even stranger molecules. It filled gaps no single other hydrocarbon could quite manage with the same balance of volatility, ease of handling, and predictability.
Product Overview
Not flashy, not rare — 2-methylbutane stands out for its reliability. This colorless liquid boils at just above room temperature, making it great for low-temp work. Many scientists have turned to it for its quick evaporation and minimal residue. You can find this hydrocarbon in nearly every chemical supplier’s catalog under both common and systematic names. It’s a go-to material both in undergraduate teaching labs and professional research settings. It does a better job than pentane when specific freezing properties are needed, giving it the edge for niche jobs. The experience of opening a cold bottle of isopentane in a lab, feeling its trademark fumes, is something most chemists recognize from years past to now.
Physical & Chemical Properties
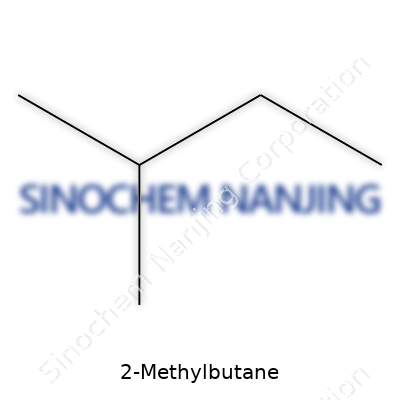
2-Methylbutane’s molecular formula is C5H12. It packs five carbon atoms with branching at the second spot, a feature easy to spot in its stick model or skeletal representation. Its boiling point hovers around 27–28°C — so even in a modestly warm room, it tries to escape the bottle. Density sits at less than water, and it feels light in the hand. Its volatility makes it an excellent solvent for quick cooling, especially if you’ve ever had to snap-freeze tissue in the lab. The liquid forms a clear interface with water, reminding experimenters why hydrocarbons don’t just mix and disappear. Technically, it doesn’t offer much in the way of exotic chemistry, and this predictability is its charm.
Technical Specifications & Labeling
Every bottle or can shipped comes with the same simple declarations — purity, CAS number (78-78-4 for those who track chemicals by number), warnings about flammability, and often a purity rating that’ll make or break a lab’s day. Most 2-methylbutane is delivered with purity above 98%, sometimes as high as 99.5% for special cases. Labeling laws make sure users know the fire risks and the importance of good ventilation. You don’t see too many special storage instructions apart from keeping the container closed, protected from sparks, and out of direct sunlight. In my own work, a faded sticker on a metal can sat as a reminder that, no matter how mundane, proper labeling keeps everyone in the lab honest and safe.
Preparation Method
Refinery streams with a mix of branched and unbranched alkanes serve as the source. Manufacturers use distillation to separate out 2-methylbutane from similar hydrocarbons. It doesn't call for unusual catalysts, reactions, or complex plants; the process relies on the differences between boiling points and other basic physical properties. This method scales up easily, and as a result, large quantities hit the market yearly. Some older texts mention laboratory syntheses — reduction of certain ketones, rearrangement reactions — but in practice, the bulk supply always comes down to distillation efficiency.
Chemical Reactions & Modifications
2-Methylbutane is as inert as you’d expect for a saturated hydrocarbon. It resists acids and bases, dodges nucleophiles, and stays quiet unless forced to react under extreme conditions. Still, chlorination and other radical halogenation reactions will swap out hydrogens, leading to a family of more reactive derivatives. Many chemists, myself included, see it mostly as a solvent or cooling agent, not a reactant. If curiosity wins, free-radical substitution yields products that chemists can chase further, but most labs look elsewhere for creative synthesis. Over the years, attempts to make value-added chemicals using 2-methylbutane as a scaffold turned up little demand, so its main use remains outside the limelight of advanced organic transformations.
Synonyms & Product Names
The naming story for this compound is a classic in organic chemistry. Textbooks call it 2-methylbutane; industry catalogs nearly always list "isopentane." Walk down the hallway in most chemistry departments and mention either — people will know you mean the same stuff. Other less frequent names include methylbutane, but those don’t have the clarity of proper IUPAC or accepted common names. Such overlapping naming sometimes confuses newer students, but common usage tends to default to "isopentane" in the United States, especially in labs stockpiling solvents or refrigerants. Several brands supply the compound, rarely under house names, because purity and consistency, not branding, matter more to researchers.
Safety & Operational Standards
Using 2-methylbutane requires respect for volatility and fire risk. Safety sheets never hesitate to put flammability in bold, forcing everyone to store it in explosion-proof cabinets and crack windows or turn on fume hoods before opening the container. I’ve watched hands move a little slower and more carefully when this liquid comes out; one spark and things go sideways fast. Occupational rules make sense — keep away from heat, flames, and static discharge. Accidental inhalation can lead to headaches or even a trip to the emergency room if exposure gets out of hand. The liquid feels cold on bare skin because of that rapid evaporation, and gloves stay non-negotiable. Any workspace using 2-methylbutane keeps spill kits and fire extinguishers close at hand for good reason.
Application Area
The world of biology leans hard on 2-methylbutane for flash freezing delicate tissues, a tradition that stretches back through countless histology manuals. Researchers preparing brain or muscle slices for microscopy count on this hydrocarbon because it cools rapidly down to temperatures that preserve structure but doesn’t form the ice crystals that wreck cells. In industrial chemistry, it sometimes steps in as a blowing agent for foams, although other hydrocarbons shoulder much of that work these days. The solvent power of 2-methylbutane carries value in extraction protocols, especially when unwanted residues from heavier organics mess with results. Some applications live on in specialty refrigeration systems and as calibration standards for gas chromatography. Every bottle echoes decades of daily, trusted use in both science and industry.
Research & Development
The research frontier for 2-methylbutane looks a little quiet—few dramatic innovations, more careful tweaks. Advances focus on making refinery separation cleaner or on adapting the cold-chain logistics for biobanking and clinical tissue handling. The occasional paper appears on using 2-methylbutane in polymer chemistry or materials science, always hunting for a small performance edge. As scientists dig deeper into biological tissue preservation, some start looking at blends or mixing protocols involving 2-methylbutane to tease out just the right cooling curve. In my own experience, the real value comes from watching students and technicians consistently get sharp, reliable results — not from headline-grabbing breakthroughs.
Toxicity Research
2-Methylbutane doesn’t show the acute or chronic toxicity of aromatic hydrocarbons or heavy industrial solvents, but the dangers are far from trivial. Research and regulatory reviews agree that inhaling high concentrations knocks out central nervous system function, leading to dizziness, loss of coordination, and, at peak exposure, unconsciousness. Chronic occupational studies lean on older records, pointing more toward good ventilation and engineering controls than urgent health scares. Animal studies rarely connect moderate 2-methylbutane exposure to carcinogenic effects. Still, anyone pouring, measuring, or storing this hydrocarbon should stick to best practices, including using personal protective equipment and staying mindful of the vapor’s ability to displace air in poorly ventilated spaces.
Future Prospects
Bluntly, 2-methylbutane won’t headline a new era in chemistry. Its future sits in the continued trust of scientists and technicians who count on it every day. Areas like tissue preservation and extraction science look stable; demand grows where life sciences, particularly biobanking and precision diagnostics, need more refined workflows. The constant push for safer, more sustainable solvents sits as the only real competitor. Some new cold-chain protocols might replace or supplement 2-methylbutane, but its blend of price, performance, and availability keeps it in steady demand. I haven’t seen eager young researchers turning it into biofuels or green plastics, but no one underestimates substances that have anchored laboratory procedures for generations. The small role 2-methylbutane plays in research and industry leaves a big mark on reliability, safety, and outcomes — a reminder that humble molecules often support the most important breakthroughs behind the scenes.
What is 2-Methylbutane used for?
What Does 2-Methylbutane Do?
2-Methylbutane, known as isopentane, deserves a closer look. This chemical doesn’t pop up in the news, but it gets people’s attention in industries that touch all our lives. You’ll find it in labs, gas stations, and factories, quietly doing work that keeps things running smoothly. Its low boiling point and flammability make it stand out, but the uses go deeper than most folks realize.
The Lab Connection
Research labs keep isopentane close by because it cools samples in a flash. In histology, researchers dip biopsy samples into super-cold isopentane to freeze them almost instantly. This trick keeps tissue structures sharp for microscope studies. Back in college, we froze plant petals for a project—the details we saw in those slides stuck with me years later. Without a solvent like isopentane, that kind of detail would have melted away. Labs count on it for consistent and fast results when other chemicals fall short.
Fuel and Refining Workhorse
Fuel makers care about isopentane for another reason: it helps get gasoline to the right level of volatility. Blending in the right amount lets engines start even in cold climates and cuts down on knocking. Energy sector data from the U.S. Energy Information Administration points out that isopentane is one of the lighter hydrocarbons boiled out of crude oil during refining. You can read a lot about fuel blends, but what’s clear is this chemical helps tailor gasoline to real driving conditions. Having used different fuel grades from summer to winter, I’ve noticed how engine performance shifts, and isopentane often sits behind those improvements.
Expanding Foams and Packing Goods
Take a closer look at the foam around electronics shipments or inside a new refrigerator. Many of those products get their cushion because isopentane helps expand polystyrene and polyurethane foams. Its fast-evaporating nature creates those little pockets of air in the material. As a consumer, I appreciate products arriving intact, but there’s another side to this story. Manufacturers have faced pressure to move toward chemicals with a lighter touch on the environment, and there’s a quiet transition toward less volatile blowing agents. Still, isopentane keeps hanging around because it gets the job done.
Environmental Footprint and Future Questions
Any chemical making its way through the fuel supply chain and into the air brings questions. Isopentane can contribute to the formation of ground-level ozone if released outdoors, as highlighted by the Environmental Protection Agency. Workers handling it run safety risks since it fumes easily and burns fast. Factories have to stick to strict ventilation and storage measures—corners just can’t be cut here. The same goes for disposal: waste streams must be carefully managed to protect water and air.
Looking for Solutions
The push for greener chemistry can’t be ignored. Research teams and companies have started trialing alternative blowing agents and working to capture volatile losses at refineries. For smaller labs, ideas like recapturing evaporating solvents or switching to less hazardous chemicals are gradually gaining steam. Regulatory groups press for tighter emissions standards. For those in the field, clear protocols save lives and money over time.
2-Methylbutane may seem technical, but it’s woven into daily routines, industry shifts, and evolving safety standards. The next time someone opens a foam-packed box, fills up a car in the winter, or reviews a medical biopsy, chances are isopentane played a silent role in making it possible.
Is 2-Methylbutane hazardous or flammable?
Why 2-Methylbutane Deserves Respect in the Lab and Beyond
People working in labs, classrooms, or certain industries cross paths with chemicals daily. 2-Methylbutane, commonly called iso-pentane, stands out as one of those volatile compounds with a reputation for catching fire without much warning. I’ve handled this liquid more than once during biology work involving freezing samples. A small spill on the bench got me nervous, not because of its odor, but because the safety data kept ringing in my head—this stuff lights up fast.
A Flashpoint Below Room Temperature
Look at its properties and you start to understand the fuss. 2-Methylbutane has a flashpoint of around -49°C. That’s much colder than your freezer can get, but it means any open container or spilled liquid in regular temperatures releases fumes ready to ignite. Vapors can hang around and drift toward a flame or spark, whether that spark comes from static, a light switch, or a heat source on the next bench over. All it takes is the right mixture with air, and you might get a fire big enough to set off alarms or worse.
The Explosion Factor in Everyday Scenarios
Anyone who’s opened an old can of paint thinner or gasoline understands the peculiar fear that comes with volatile liquids. 2-Methylbutane belongs in the same camp, only it evaporates even faster. I remember a summer internship where a careless moment—forgetting to cap a flask—resulted in fumes that triggered the lab ventilation system. If things had gone differently, it could have meant burns or worse.
Data backs up this experience. The National Institute for Occupational Safety and Health (NIOSH) classifies 2-Methylbutane as a “Highly Flammable Liquid and Vapor.” Even pouring it slowly creates an invisible vapor cloud. The autoignition temperature runs at about 420°C, which means even a hot plate left on unattended could become a hazard. It doesn’t take much: a leaky container, static from a synthetic lab coat—these little details make all the difference in preventing fires.
Health Effects and What the Signs Feel Like
Aside from fire, 2-Methylbutane brings health risks when inhaled. Breathing in the vapor can lead to headaches, dizziness, and even nausea. If a space isn’t well ventilated, high exposure knocks out your coordination before you realize it. I’ve seen researchers underestimate the power of a chemical hood, thinking short exposure wouldn’t matter. A few minutes later, they looked pale and needed air.
Reducing Risks, One Habit at a Time
Respecting 2-Methylbutane starts with the basics: proper storage in flammable liquid cabinets, good ventilation, and using flameproof containers. Always grounding metal items before pouring prevents static sparks, and double-checking lids after each use becomes second nature over time. Regular training keeps the risks front-of-mind. Sharing real-life close calls in safety meetings does more good than a dozen posters on the wall.
Moving Forward with Safer Practices
Everyone likes to believe “it won’t happen to me,” but handling 2-Methylbutane proves that a moment’s carelessness can lead to disaster. Understanding what the liquid wants to do—spread, evaporate, ignite—means staying one step ahead of the game. Anyone working with volatile chemicals benefits from learning about flammability, no matter how routine the task may feel. Awareness and habits save lives, not just compliance with rules.
What is the chemical formula of 2-Methylbutane?
Getting to Know the Molecule
2-Methylbutane, which some chemists call isopentane, sits among the family of hydrocarbons known as alkanes. If you write it out on paper, the chemical formula pops out as C5H12. Just looking at the formula, you can already picture a simple, unadorned structure: five carbon atoms carrying twelve hydrogens, no double bonds, rings, or oxygen in sight. Still, 2-methylbutane doesn’t just exist to pad out a page in an organic chemistry textbook. Plenty of folks use it, even though they might not realize it.
Why This Formula Matters in the Lab and Industry
Back during undergraduate years, the name isopentane often came up in the organic chemistry lab. Many folks got their first taste of it as a solvent—nonpolar, easily handled if you know what you're doing, and useful for cooling baths thanks to its low boiling point. The formula C5H12 might seem forgettable, but recognizing the difference between 2-methylbutane and its straight-chain cousin n-pentane lets researchers avoid headaches with unexpected results. Try dissolving something polar in it, and you'll learn fast why picking the right alkane matters.
The story doesn’t end in the lab. Large-scale producers keep close tabs on isomer content for gasoline blends. 2-Methylbutane, given its higher octane rating compared to some straight-chain relatives, commands a place in fuel engineering. Fracturing and reforming refineries work daily with molecules like this for efficiency and emissions concerns. Misjudging the details can ripple out into vehicle performance and environmental factors. Hard data from energy research backs this up: branched alkanes contribute to engine antiknock properties, making a small molecule, like 2-methylbutane, influential on the big stage.
Health, Safety, and Environment
No one wants a nasty surprise in the workplace or classroom. 2-Methylbutane comes with hazards. Low flash point means it can light up with just a stray spark. Inhaling too much vapor leads to dizziness and headache, something most chemists learn to avoid early. Regulatory bodies like OSHA and NIOSH publish safety sheets spelling out limits and guidelines, ensuring workers understand the risks. The molecular simplicity doesn’t make it harmless.
Uncontrolled release into the environment stirs trouble, especially for air quality. High volatility means it evaporates in seconds. It adds to ground-level ozone formation, part of the bigger problem with volatile organic compounds (VOCs) and urban smog. Air quality studies routinely flag C5 hydrocarbons like 2-methylbutane as significant contributors in traffic-heavy cities.
Solutions Rooted in Knowledge and Practice
Good habits go a long way in handling compounds such as this. Everyone who uses 2-methylbutane should know the material’s properties—starting from its structure up. That means basic chemical literacy before anything goes near a flask. Industry players rely on strict monitoring of emissions and workplace exposure, using ventilated hoods and proper containment. Regular audits by environmental agencies keep practices up to par. At the university and in manufacturing, safety drills and clear labeling continue to prove their worth, reducing incidents and health risks.
All these routines rest on the same foundation: understanding what the chemical formula C5H12 represents. Far from a trivia question, it guides safe handling, shapes product formulations, and drives regulatory action. So, recognizing 2-methylbutane’s identity and importance gets much easier once you look past the numbers.
How should 2-Methylbutane be stored?
Why 2-Methylbutane Matters and How Handling Gets Overlooked
Plenty of researchers bump into 2-Methylbutane, also called isopentane, especially in life sciences. Cryopreservation, histology, and sample transport routines rely on it for its ultra-low freezing point. Handling such a volatile solvent often seems straightforward, but I remember a colleague in grad school ignoring basic rules and sparking a minor scare—just because nobody talked much about proper storage. A little extra care saves headaches, injuries, and wasted research.
Risks You Don't Want in Your Lab
2-Methylbutane evaporates at room temperature, catches fire easily, and forms explosive mixtures with air. It can irritate skin, eyes, and lungs, and in a tight space, the fumes linger longer than most expect. The National Institute for Occupational Safety and Health flags it as “immediately dangerous to life or health” at high concentrations. A careless spill or a cracked bottle means one spark brings a world of trouble. Add in the fact that this chemical moves fast from liquid to vapor, and a few minutes of inattention can fill a poorly ventilated room with enough fumes to set off alarms or trials.
Secure Storage: Go Beyond the Bare Minimum
My early lab days taught me shortcuts always catch up with you. Sticking isopentane in a random fridge, for instance, means you’re risking a fire unless every part of the appliance is “explosion-proof.” Many common lab refrigerators spark when they cycle, and that spark plus isopentane vapor could wreck hundreds of hours of work—or more. Industry safety guidelines and OSHA both agree: use approved flammable liquid storage refrigerators or freezers, never household models.
Some labs slack off and store 2-Methylbutane on open shelves. All it takes is a bottle tipping onto a hot plate, and suddenly you’ve got smoke and shouting instead of calm data collection. Locking flammable liquid cabinets, made of double-walled steel with plenty of ventilation, put a real barrier between accidental ignition and disaster. Think of it as protecting not just yourself, but the years of research walking in and out of the room.
Practical Tips: Build Habits, Not Just Labels
Lab culture plays a huge role. I once worked somewhere that used color-coded labels and ran regular training refreshers. No one eyed the extra step when restocking chemicals; nobody left bottles in odd places, either. It built a habit—handling gloves, tightly capped bottles, and the right spill kits on hand. Those small steps matter.
Ventilation counts too. Even with all other precautions, a good chemical fume hood or exhaust system keeps people breathing easy. Treat the storage area as a high-alert zone—no eating, no drinking, and absolutely no open flames or heaters nearby.
How to Raise the Bar on Safety—Without Added Expense
A flammable chemical cabinet does cost money, but skimping runs bigger risks. If budgets run tight, sharing safety equipment across research groups, pooling chemical orders, and setting clear policies all help. Labs don’t need to throw out the basics for fancy tech—small checklists for handling, storage, and cleanup guide new team members and keep old ones honest. Universities, companies, and even smaller clinics often underestimate how far training and simple reminders go.
The real win comes from everyone knowing why each step matters, not just moving bottles from shelf to fridge. Take advice from people with scars, not just manuals: one accident can echo for years. Store 2-Methylbutane with respect, keep your lab organized, and you’ll get answers from your research—not your insurance company.
What are the safety precautions when handling 2-Methylbutane?
The Hazards Lurking in the Lab
2-Methylbutane, known in some labs as isopentane, brings some big risks with its low flash point and easy evaporation. The liquid catches fire at just room temperature. Anyone who's knocked over a container or seen an unexpected flame leap up knows what this means. Solvents that vaporize quickly, like 2-methylbutane, don’t let you get comfortable. Vapors travel far before finding an ignition source, so the risk spills out of the beaker and across the room in a heartbeat.
What My Years in Chemical Labs Have Taught Me
Over the years, I’ve seen a lot of careless mistakes with volatile solvents. Simple gloves, a lab coat, and goggles go a long way, but rushing or cutting corners ups the odds of disaster. Nitrile gloves—never bare hands—keep exposure down. I’ve watched skin absorb splashed solvent before. I never want that to happen again. A good fume hood can’t be skipped with this one, either. The vapors get rough on the lungs, and any spark, even from static, sets off trouble.
Good Habits That Cut Risk
Set up glassware or open bottles only where the air moves well. Don’t try to pour or aliquot outside a hood just for convenience. That’s how spills and dangerous vapor clouds sneak up. Store every bottle in a flammables cabinet, away from sunlight or heat vents. Even during busy hours, take a few seconds to clean up drops or splashes with absorbent pads and seal them in proper waste bags right away.
I recall a friend leaving an open vial on a bench just for a lunch break. He came back to a headache and dizziness. That reminds me how quickly vapors spread. Keep containers closed every time. Even small mistakes build up if repeated enough times.
Seeking Safer Alternatives
Some researchers have switched to less volatile alternatives when possible. In my own team, we sometimes substitute with less flammable solvents when the protocol allows. For tissue freezing, alternatives exist, but if 2-methylbutane remains necessary, it helps to limit how much stays on the bench at any time.
Emergency Steps and Training
Every lab needs an eyewash station, a working fire extinguisher, and a spill kit visible and ready to use. I’ve helped run practice drills, and they make a real difference. People react smarter and faster if training feels like muscle memory. Anyone handling this chemical should know exactly where the nearest exits and alarms sit.
For accidental spills, alert labmates right away and evacuate if fumes get heavy. Never mop up by hand—absorb with spill pads, ventilate, and let trained staff handle fumes and disposal. Medical help should be called for any splash in the eye or on skin.
Final Word from Experience
Working with 2-methylbutane isn’t about bravado; it’s about staying sharp and sticking to old-school caution. Following the right steps helps everyone get home safe at the end of the day. No shortcut outstrips steady, careful habits and respect for what such chemicals can do.
| Names | |
| Preferred IUPAC name | 2-Methylbutane |
| Other names |
Isopentane Isoamyl hydride 2-Methylbutan |
| Pronunciation | /tuː ˈmɛθ.ɪl.bjuː.teɪn/ |
| Identifiers | |
| CAS Number | 78-78-4 |
| 3D model (JSmol) | `3Dmol.js?commonbio&gebb&modeldata=16423` |
| Beilstein Reference | 1720236 |
| ChEBI | CHEBI:30362 |
| ChEMBL | CHEMBL142299 |
| ChemSpider | 5957 |
| DrugBank | DB01999 |
| ECHA InfoCard | 100.007.696 |
| EC Number | 201-142-8 |
| Gmelin Reference | 164648 |
| KEGG | C06587 |
| MeSH | D008955 |
| PubChem CID | 6560 |
| RTECS number | NL7700000 |
| UNII | J3J8GIF8YF |
| UN number | UN1265 |
| Properties | |
| Chemical formula | C5H12 |
| Molar mass | 72.15 g/mol |
| Appearance | Colorless liquid |
| Odor | gasoline-like |
| Density | 0.62 g/mL |
| Solubility in water | Very slightly soluble |
| log P | 2.8 |
| Vapor pressure | 53.3 kPa (at 20 °C) |
| Acidity (pKa) | 48 |
| Magnetic susceptibility (χ) | χ = -7.03·10⁻⁹ |
| Refractive index (nD) | 1.352 |
| Viscosity | 0.00349 cP |
| Dipole moment | 0.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 282.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -134.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3509.5 kJ/mol |
| Pharmacology | |
| ATC code | VO43A07 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H304, H336, H411 |
| Precautionary statements | P210, P261, P304+P340, P312, P403 |
| NFPA 704 (fire diamond) | 1-4-0 |
| Flash point | -60 °C |
| Autoignition temperature | 245 °C |
| Explosive limits | 1.2–8.4% |
| Lethal dose or concentration | LD50 Oral Rat 15,800 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 5870 mg/kg |
| NIOSH | NIOSH: NM 2975000 |
| PEL (Permissible) | 1000 ppm |
| REL (Recommended) | REL: 1900 mg/m3 |
| IDLH (Immediate danger) | 8000 ppm |
| Related compounds | |
| Related compounds |
n-pentane cyclopentane isopentane neopentane 2,2-dimethylpropane |

