2-Methyl-2,4-Pentanediol: More Than a Simple Solvent
Deep Roots in Chemistry’s Story
Chemistry classes always felt less intimidating when the stories behind everyday reagents came to light. 2-Methyl-2,4-Pentanediol (sometimes called hexylene glycol) has been around for nearly a century, discovered in the formative days of organic synthesis. Its development paralleled the rapid growth of industrial organic chemistry after the First World War, as scientists needed new, adaptable solvents. The search for safer, more stable alternatives to diols like ethylene glycol drove research. Labs in Europe and North America tinkered with straight-chain glycols and branched derivatives, stumbling across this five-carbon diol with a little extra methyl twist. Its structure gave it properties different enough from propylene glycol to open new doors in manufacturing and product design.
What Sets It Apart
Hexylene glycol holds a spot on chemical shelves thanks to its balance between water and oil solubility. It does not smell as sharp as typical alcohols nor does it sting as much—a bonus for people using it all day. On a molecular level, this compound brings two hydroxyl groups, spreading across a six-carbon backbone with a methyl substitution at the second carbon. That tweak not only boosts its miscibility but also pumps up its boiling point, so it resists flashing off under moderate heat. I remember handling it in the lab and appreciating its thicker feel compared to most glycols—the viscosity speaks directly to its structure, and it made sense as soon as I poured it into the reaction flask. Its refractive index and relatively high boiling point let specialists use it in places where more volatile alcohols wouldn’t last. Still, its relatively low toxicity—especially compared to siblings like ethylene glycol—helps it slide into a lot of consumer and industrial applications without raising too many eyebrows.
Labeling and Compliance: The Everyday Reality
Any chemist or technician who works with 2-Methyl-2,4-Pentanediol ends up reading the label over and over. Regulatory bodies require specifics for labeling: chemical name, CAS number, hazard pictograms for eye irritation, and handling instructions. In my own experience, labels also remind staff of proper ventilation, spill management, and first aid. The rules are not red tape for their own sake—they’re about making sure warehouse workers, lab techs, and end users know exactly what they are dealing with. This chemical’s relatively low acute toxicity, coupled with long decades of use, means guidance is straightforward but not lax. Companies align standards with OSHA guidelines, the European REACH program, and other regulatory frameworks that evolve with new toxicity data. Delivering this information in ways that workers can understand—not just scientists—makes a real difference in daily safety.
Making It Happen: How Production Works
The paths to 2-Methyl-2,4-Pentanediol start with more basic organic feedstocks. Early industrial producers used acetone as a precursor and introduced hydrogen under pressure, a process called hydrogenation. I spent time in a pilot plant where methyl isobutyl ketone, another common precursor, joined the blend, producing higher yields through hydrogenation as well. Technicians would keep reactors at just the right conditions to avoid cracking or excessive byproduct formation. This process has not changed much because the chemistry remains straightforward, and raw materials come from petroleum refining—another reminder of the interconnectedness of industrial chemistry. Feeds, temperatures, catalysts, and recycled process streams all factor into maximizing output and minimizing waste. Downstream, fractionation and simple distillation clean up the product, because purity matters in paints, coatings, and pharmaceuticals alike.
The Power of Reactions and Modifications
This chemical’s structure opens up a lot of reactivity. The two alcohol groups act as nucleophiles, attacking acyl chlorides, isocyanates, and other activated compounds. Producers add it to resin synthesis, where its two sites for reaction help form flexible, crosslinked polymers. Years ago, in a coatings research project, I watched it serve both as a chain extender and as a plasticizer in polyurethane and epoxy networks. Its reactivity isn’t aggressive, so it doesn’t ruin formulations with runaway reaction rates. Chemists aiming for esters or ethers find it straightforward to work with as well. Modification of hexylene glycol with fatty acids can generate nonionic surfactants, trusted in cleaning agents and emulsifiers. All these reactions trace back to the same principle: nature of the functional groups dictates the range of possible outcomes.
Nicknames and Chemical Aliases
2-Methyl-2,4-Pentanediol appears in the literature and on product shelves under a handful of synonyms. Hexylene glycol gets used so often that it has nearly eclipsed the systematic name. Looking back over older journals, I found references to diacetone glycol and even MP-diol. In international catalogs, translations and abbreviations echo the same patterns. These names can cause confusion for newcomers, which turns cross-referencing between chemical suppliers into a necessary skill—a little like knowing the regional names for baking soda. Familiarity with its CAS number—107-41-5—cuts through ambiguity.
Guidelines and Habits That Drive Safe Handling
No matter how safe a lab or plant seems, good habits make the difference in preventing accidents. Reading the safety data sheet with care matters every time a jug gets opened. Hexylene glycol needs gloves and goggles, and even though it lacks the acute toxicity punch of solvents like methanol or toluene, skin contact and splashes will still cause irritation. Eye protection isn’t negotiable. Spills aren’t particularly dramatic thanks to its high boiling point and low vapor pressure, but cleanup needs attention. The right ventilation setup reduces vapor buildup and limits exposure. Companies set exposure limits based on long-term animal studies and rare occupational incidents, constantly updating policies as fresh research arrives. Workers themselves are often the first to spot weaknesses in safety protocols, and the companies that listen reap the benefits of fewer incidents and more engaged employees.
Where It Ends Up—And Why That Matters
This diol winds its way into paints, coatings, hydraulic fluids, and even cosmetics. In oil and gas, it appears in drilling fluids and in gas sweetening. Its spread into cosmetics, especially as a humectant and viscosity builder, raised questions about skin exposure. European regulators, always quick to respond to data, have kept a close eye on dermal toxicity while not jumping to harsh bans. I remember running trials on water-based coatings, where hexylene glycol enabled stable emulsions without relying on more expensive or less readily available glycols. Its dual solubility in water and oils bridges phases in formulations, and even personal care brands appreciate that. It can dissolve perfume ingredients, maintain texture in creams, and deter product separation over time.
The Drive for New Discoveries
Ongoing research shapes how industries use and think about 2-Methyl-2,4-Pentanediol. Universities produce fresh data on structure-property relationships, giving technical teams more insight into why certain blends behave better in low-VOC coatings. Researchers in biochemistry use it as a precipitant in protein crystallography, helping to map protein structures at atomic resolution. This surprised me when reading crystallography protocols, where I recognized the chemical not as a solvent but as a critical matrix component. Scientists keep analyzing biodegradation rates in aquatic environments, responding to growing concerns over contaminants and their impact on water quality. Manufacturers are paying more attention to reducing impurities, not strictly for regulatory box-checking, but as a way to minimize allergic reactions or off-odors in sensitive applications. More than ever, the work in these labs influences decisions in factory control rooms and boardrooms alike.
Understanding Risks—And Facing Them Squarely
Any chemical that enters consumer products must stand up to scrutiny, and hexylene glycol is no exception. Studies show mild toxicity, with the primary concern centering on skin and eye irritation after short-term, high-exposure events. Large doses can stress the kidneys or liver over time, but such exposures don’t come close to ordinary consumer use. On the workplace side, inhalation at high concentrations causes headaches and nausea, prompting sensible ventilation guidelines. Toxicologists are always chasing more detailed endpoints—long-term reproductive or developmental effects, for instance—though decades of monitoring provide reassurance. The trick lies in not relaxing standards just because current data looks benign. In my time at manufacturing plants, regular reviews of incident logs usually revealed that mistakes traced to simple lapses—wrong glove material, overlooked ventilation, assuming a mild chemical posed no hazard. Continuous small efforts keep larger problems at bay.
Looking Ahead: Adapting to Changing Standards
Demand for safer, environmentally friendlier chemicals keeps shifting the landscape for specialties like 2-Methyl-2,4-Pentanediol. There’s a push for renewable feedstocks, and some suppliers have started experimenting with bio-based routes, although most of the current volume still ties back to petrochemicals. Scrutiny of effluent quality is rising—not only for regulatory reasons but also as part of companies' voluntary commitments. In sectors like coatings, the trend leans toward water-borne, low-emission chemistries, so solvents and additives must meet tighter VOC criteria. Green chemistry principles steer research teams to design modifications or find alternatives that balance performance, safety, and environmental impact. For everyday users, it means clearer labels, smarter packaging, and more accessible safety information. Years from now, legacy chemicals like hexylene glycol may face tougher scrutiny, especially if better-performing or safer replacements come along, yet its adaptability will buy more time on the shelf. The compound’s journey from early industrial curiosity to a linchpin of modern manufacturing tells a story about chemistry’s ability to evolve—never standing still, always moving forward one experiment, one regulation, and one innovation at a time.
What is 2-Methyl-2,4-Pentanediol commonly used for?
What Makes 2-Methyl-2,4-Pentanediol Special
Walk through any research lab or industrial plant dealing with chemistry, and you’ll hear 2-Methyl-2,4-Pentanediol (often called MPD or Hexylene Glycol) mentioned in all sorts of contexts. This colorless liquid, known for both its chemical stability and ability to mix with nearly everything, pops up in more products and processes than most folks realize.
Role in Industrial Chemistry
MPD finds steady work as a solvent. Manufacturers count on it to dissolve dyes, resins, paints, and lacquers. The pharmacy world uses it to blend ingredients efficiently during drug production, increasing shelf life and keeping medications stable. MPD’s low toxicity draws attention compared to other industrial alcohols, so firms laying out health and safety priorities lean toward it whenever they can.
Everyday Products and Cosmetics
I’ve seen MPD show up often in consumer products. Think shaving cream, lotion, makeup, and hair gel. Its knack for locking in moisture and adding glide makes grooming easier and skin less cranky. You will spot it near the top of many ingredient lists for personal care items because it helps keep formulations smooth. Makers of skincare gear say MPD lets creams and serums stay silky instead of clumping up, a real win for both companies and customers.
Protein Crystallography: Behind the Science Curtain
Those of us who’ve poked around structural biology know just how vital MPD is for crystallizing proteins. Scientists add it to solutions to coax proteins to form strong, well-defined crystals. Without it, many labs would run into dead ends trying to solve protein structures by X-ray diffraction. The benefit here touches more than just curious researchers. Understanding protein shapes at the atomic level accelerates drug discovery and innovation in medicine.
Use in Coolants and Industrial Fluids
Industrial cooling systems rely on MPD, too. It keeps fluids flowing freely, staves off rust, and helps dyes mix without separating out. Brake fluids and hydraulic systems in vehicles use it to prevent jamming and corrosion, keeping machines working longer and safer. Whenever there’s a need for safety, reliability, or both—MPD steps up without fuss.
Safety First—Why Care?
While MPD lands on the safer end for industrial chemicals, it still demands respect. Prolonged or repeated skin contact causes irritation, especially in workplaces where gloves might be skipped for convenience. Proper ventilation and gear matter. Honest conversations about chemical handling go further than the bland “handle with care.” Regulatory agencies in the US and Europe have set limits for worker exposure, which hasn’t always happened with chemicals from earlier decades. That shift saves lives and health in the long run.
Paths Forward
Innovation means change, and chemistry is always on the lookout for solvents and additives with even fewer drawbacks. Some labs and companies have started testing greener alternatives and tighter recycling of solvents. By sharing data on safety incidents and working with environmental health experts, the chemical industry has a shot at keeping both workers and the environment safer, all while keeping countless products on the shelf.
Is 2-Methyl-2,4-Pentanediol toxic or hazardous to health?
Everyday Contact With Chemicals Isn’t Rare
Lots of folks hear the names of industrial chemicals and wonder if their lives are in danger just because those substances exist. 2-Methyl-2,4-Pentanediol, which sometimes turns up in laboratories or factories as “hexylene glycol,” shows up in many places—paints, coatings, brake fluids, inks, and even some cleaning products. The fact that a person runs across this chemical in certain jobs or industries sparks questions about safety. With a background in chemical handling and workplace safety training, I’ve seen what happens when dangerous chemicals go unchecked. The key to protecting health comes down to knowing ways a material can hurt people, plus some common sense habits.
Acute Health Risks: Skin, Eyes, and Inhalation
2-Methyl-2,4-pentanediol brings hazards that someone might overlook at first glance. Direct contact irritates skin. If a splash lands in eyes, pain definitely follows. Repeated skin exposure sometimes leads to dermatitis, and accidental swallowing triggers issues like headaches or drowsiness. Now, breathing in its vapors—especially somewhere stuffy—can cause throat and nose irritation. At really high levels, someone might feel dizzy or act groggy. I remember having to flush a coworker’s eyes at a shop because a poorly sealed container splashed right as she twisted off the cap. Incidents like that show the value of basic protections—gloves, goggles, proper ventilation.
Toxicity: The Bigger Picture
This chemical often raises fewer red flags compared to stuff like toluene or formaldehyde. The National Library of Medicine rates it as having “low acute toxicity.” For an adult to get sick enough from swallowing this substance, the dose would need to be quite high—well above the trace amounts anyone might accidentally come across through skin or air. Animal studies suggest that one-off exposure isn’t likely to cause chronic injury. But “low toxicity” never means zero risk. Long-term effects after years of constant exposure aren’t entirely ruled out, though current science hasn’t clearly linked this glycol to cancer or birth defects.
Environmental Concerns and Responsible Use
Some folks worry about what happens if 2-methyl-2,4-pentanediol spills into water or drains. Environmental agencies list it as a water contaminant with moderate persistence, meaning it doesn’t vanish overnight. Aquatic life can struggle if concentrations build up. No one wants an accidental release into a stream behind the warehouse, and I’ve seen the panic that follows even a minor spill. Proper disposal—never pouring it down a drain—and getting staff trained keeps both people and wildlife safer.
Staying Safe With Practical Steps
Workplaces usually rely on classic safety measures. Material Safety Data Sheets break down exact risks and recommend gloves, eye protection, and regular air flow. Anyone pouring, mixing, or cleaning with the chemical learns to wash their hands, limit any skin exposure, and keep food away from work areas. At one lab, careful labeling and a clear cleaning station made all the difference after a bottle tipped over—fast cleanups mean less risk for the next person on shift.
What People Should Watch For
Most people in daily life won’t bump into this chemical at all. Professionals who work with hexylene glycol get training and gear, which brings down risk. If anyone starts to feel burning skin, watery eyes, or a nagging cough in places where industrial chemicals are in use, air quality and proper handling practices need attention. Nobody wants a workday cut short by a mishap that simple equipment could have prevented.
Building Industry Trust
Chemical concerns never fully vanish, but facts and regular safety training keep risks in check. Studies back up the conclusion that 2-methyl-2,4-pentanediol rarely causes major health problems if used with care. When industries communicate about hazards honestly and make safety a daily routine, people feel more secure in the spaces where they work and live.
What are the physical and chemical properties of 2-Methyl-2,4-Pentanediol?
A Closer Look at Structure and Physical Aspects
2-Methyl-2,4-pentanediol often shows up in labs, manufacturing, and even home workshops. Folks sometimes call it hexylene glycol. This colorless liquid sports a faint, almost sweet odor. In daily use, its thick, syrupy consistency stands out. Even in a hot room, it refuses to boil off easily, sticking around because its boiling point climbs above 200°C. Add it to cold weather, and it struggles to turn solid before -50°C pops up. It laughs at freezing, and its ability to hug water molecules opens plenty of doors in industry and science.
The density lands right around 0.92 g/cm³, showing it's a bit lighter than water. Hexylene glycol doesn't mix evenly with every liquid, but water, alcohols, and many common organic solvents welcome it. Try adding it to petroleum-based products or some strong acids, and you’ll often spot separation or hazy mixtures. That's worth remembering as anyone working with paint thinners or cleaning solutions can tell you.
Straightforward Chemistry in Work and Play
Its chemical shape looks simple enough—two hydroxyl groups hooked to a five-carbon backbone with an extra methyl group off to the side. This extra arm gives the molecule a bulkier shape than plain old glycols. Because it has two alcohol groups, its reactions show up in chemistry lessons and manufacturing tricks. It serves as a mild stabilizer, a surfactant, and sometimes, a solvent that helps stubborn compounds dissolve in solutions. As a result, many workers handling paints, coatings, and even cosmetics come across this stuff regularly.
You’ll notice its flash point—about 106°C—lands much higher than typical organic solvents like acetone or ethanol. In real-world use, that brings safety. It doesn’t burst into flame at a whiff of heat. Still, breathing high-vapor concentrations in closed-up rooms can bother the lungs and eyes. Wearing gloves and keeping ventilation on go a long way. Repeated contact with skin sometimes dries things out, so anyone mixing large batches learns to protect their hands.
Why it Matters: From Factories to Households
Factories lean on this glycol for more than just mixing things together. Take paints and coatings. The compound keeps pigments and binders in the right consistency, stretching shelf life on store shelves. In cosmetics, 2-methyl-2,4-pentanediol adds slip and smoothness to lotions and creams. You’ll sometimes see it listed in ingredient panels on personal care products—turn over a bottle of liquid makeup or skin cream, and there it is.
Its wide use in antifreeze and hydraulic fluid stands as proof of its performance. Even folks working in agriculture might see hexylene glycol as an ingredient in some pesticide formulas, thanks to its power to blend water and oil-based ingredients.
Tackling Chronic Risks: Safe Handling and Better Choices
Long years working with chemicals teach plenty of respect for safe storage and handling. Any chemical, given time and exposure, can surprise you. Although data so far points to low acute toxicity in usual volumes, chronic overexposure tells a different story. Accidental spills or poor ventilation sometimes send workers home early with headaches or dizziness. Training, fresh airflow, and careful labeling do more than meet rules—they keep people out of trouble.
Some companies dig into green chemistry for replacements that break down safely. Bio-based alternatives now slip into some products. Still, industries that need reliable, affordable performance turn to 2-methyl-2,4-pentanediol, and they’ll likely stick with it while the search for safer, renewable options continues. Making more informed choices starts with good science and experience in the real world.
How should 2-Methyl-2,4-Pentanediol be stored and handled safely?
Understanding the Real Risks
2-Methyl-2,4-pentanediol shows up frequently in industrial settings. Anyone who’s handled it for more than a week recognizes the slight sharpness of its scent. It’s easy to grow comfortable around this compound, given its clear, syrupy look. Complacency creeps in, but this material, like many chemicals, asks for respect and a dose of practical caution.
Ventilation and Storage Considerations
Ventilation deserves real attention. It’s tempting to stash chemicals in available storage rooms, but small spaces without fresh airflow don’t cut it. Every time I saw someone store 2-methyl-2,4-pentanediol in a cramped corner, the risk of inhaling too much vapor increased. This compound gives off vapor that builds up if left unchecked. Chemical-safe cabinets with clear labels belong far away from heat, sparks, or sunlight.
Many people use steel drums or high-density polyethylene containers, choosing tight, unbroken seals. Air and moisture turn the storage scene into a mess, not just for safety but also for chemical stability. At my last job, one container leaked due to a lousy cap and soft flooring soaked up the spill, making cleanup costlier and more difficult. If you ever notice a sticky ring under a container, that’s your warning sign—deal with it right away.
Temperature and Fire Risk
Room temperature storage works as long as direct heat stays out of the equation. Cranking up the heat in winter doesn’t justify letting the storage area get too warm. The flash point isn’t sky-high, so storing away from flames, electrical sockets, or machinery that runs hot matters. Actual workplace fires often start with lazy storage habits rather than dramatic accidents.
Handling: Gloves and Goggles Aren’t Optional
A splash in the eye or on your skin brings burning and irritation. Chemical-resistant gloves don’t just look good for inspectors—they save people from hours of irritation. Goggles and lab coats protect eyes and skin; adding a face shield sometimes helps, especially if pouring from bulk containers. If you work in a spot where splashes are possible, quick access to eyewash stations and showers can stop a minor mistake from turning ugly.
Spill Response
Even careful hands occasionally cause spills. In my experience, there’s no shame in grabbing spill kits at the first sign of trouble. Absorbent pads, neutralizers, and scoopers should live in the same room or a few steps away. Trying to improvise with paper towels or rags just spreads contamination and increases risk. Never use sawdust or similar flammable materials—they only add fuel to the fire, literally.
Disposal and Housekeeping
Nobody enjoys paperwork, but tracking containers, dates, and degradation is smart practice. Clearing out almost-empty or old bottles keeps your storage areas safer and less cluttered. Hazmat crews always thank us for keeping the area tidy and correctly documented, which speeds up disposal. Don’t pour leftovers down the drain. Instead, use licensed services to respect both regulations and your environment.
Training and Culture Matter Most
I’ve seen new hires skip over safety sheets because they trust their instincts. No amount of equipment substitutes for regular, thorough training. Supervisors need to set the tone—real feedback, live demonstrations, and friendly reminders do more good than warning posters alone. Culture changes slowly, but a steady drip of smart habits pays off across months and years.
Can 2-Methyl-2,4-Pentanediol be mixed with water or other solvents?
Understanding 2-Methyl-2,4-Pentanediol
Most people working in labs or chemical plants come across a lot of solvents, but not every solvent is treated the same way. 2-Methyl-2,4-pentanediol, sometimes called hexylene glycol, gets attention for its unique set of properties. It shows up in everything from industrial processing to cosmetics. A big question is, how does it interact with water and other solvents?
Mixing with Water: What Really Happens?
In my experience, hexylene glycol goes straight into water without putting up a fight. There’s no noticeable separating or swirling—unlike with certain oils that sit on top or refuse to blend in. Chemically, it holds alcohol groups that form hydrogen bonds easily, so it grabs onto water molecules instead of pushing them away. That tendency means people get a handy tool for making all sorts of mixtures where water and oil both need a friend. Technical data backs up this claim, too: the solubility sits near perfect under room temperature conditions. This property matters for anyone working with paints, coatings, or cosmetics, where flowing, stable mixtures keep everything on track.
Blending with Other Solvents
Not every project wants to use water, especially in industries dealing with industrial lubricants, cleaners, or resins. I’ve watched this compound smoothly mix with ethanol, acetone, and other alcohol-based or ketone-based solvents. That flexibility makes it something of a "universal friend" in solvent circles. It can act as a carrier, softener, or stabilizer, depending on where it lands. Of course, petroleum hydrocarbons—those heavy, oil-based solvents—bring a different story. Mixing can get patchy. You get microdroplets, not a perfectly smooth blend. Lower polarity solvents just don’t attract the glycol like water or ethanol do.
Why These Mixing Qualities Matter
If mixing acts up in the lab or on the factory floor, results get out of hand. Poor blending means uneven coatings, clumpy lotions, or faulty cleaning solutions. With 2-methyl-2,4-pentanediol, the predictability with water or alcohol solvents builds trust. People working with experimental formulations can reach for this substance, knowing it’ll behave. That kind of reliability saves both time and frustration.
In the field, I’ve noticed that temperature shifts affect mixing performance. Hotter environments push the solubility further—sometimes great, but sometimes triggering reactions or separation you don’t want. So, paying attention to work conditions remains necessary for successful applications. Safety sheets also flag that high concentrations should not get close to flames or strong oxidizers, reducing avoidable risk for facility crews and lab staff.
Supporting Better Practices and Finding Solutions
Easy mixing isn’t just about convenience—it points to safer, more efficient processes. Instead of using stabilizers, thickeners, or heavy surfactants, industries can streamline by picking solvents like hexylene glycol. That approach can cut waste, lower energy use, and reduce the number of ingredients people need to track or control.
On the regulatory side, meeting standards for purity, toxicity, and residue matters a lot. Asking suppliers for clear documentation, batch testing results, and storage guidelines keeps everybody safe. Getting more eyes on the latest safety research helps anticipate problems or find better alternatives if push comes to shove.
Reliable mixing with water and a host of solvents gives 2-methyl-2,4-pentanediol a place in many chemists' and manufacturers' toolkits. With the right care and up-to-date information, it supports smooth operations across a broad set of industries.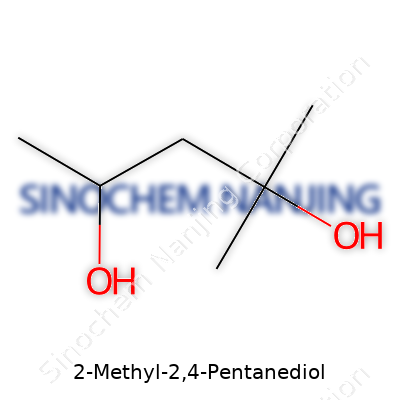
| Names | |
| Preferred IUPAC name | 2-methylpentane-2,4-diol |
| Other names |
Hexylene glycol 2-Methylpentane-2,4-diol 2,4-Pentanediol, 2-methyl- Diolane Dimethyl carbinol carbinol MPD |
| Pronunciation | /tuː ˈmɛθɪl ˈpɛnteɪnˌdaɪɒl/ |
| Identifiers | |
| CAS Number | 107-41-5 |
| Beilstein Reference | 1209222 |
| ChEBI | CHEBI:131555 |
| ChEMBL | CHEMBL1566 |
| ChemSpider | 7177 |
| DrugBank | DB04128 |
| ECHA InfoCard | 03d3462a-e8b3-4e2e-9ffa-1a1bfa6eb5e2 |
| EC Number | 203-489-0 |
| Gmelin Reference | 83734 |
| KEGG | C06501 |
| MeSH | D052438 |
| PubChem CID | 8129 |
| RTECS number | SA5950000 |
| UNII | NS50X3T6WM |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID0022923 |
| Properties | |
| Chemical formula | C6H14O2 |
| Molar mass | 134.22 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 0.913 g/mL |
| Solubility in water | Miscible |
| log P | 0.7 |
| Vapor pressure | 0.04 mmHg (20°C) |
| Acidity (pKa) | 15.2 |
| Basicity (pKb) | 6.03 |
| Magnetic susceptibility (χ) | -7.6×10⁻⁶ |
| Refractive index (nD) | 1.431 |
| Viscosity | 34.8 mPa·s (25 °C) |
| Dipole moment | 2.89 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 270.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -481.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3906.8 kJ/mol |
| Hazards | |
| Main hazards | Causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P261, P264, P271, P273, P305+P351+P338, P304+P340, P312 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 93 °C |
| Autoignition temperature | 400°C |
| Lethal dose or concentration | LD50 oral rat 3692 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 3692 mg/kg |
| NIOSH | SD3150000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) for 2-Methyl-2,4-Pentanediol is "10 ppm (skin), 45 mg/m3 (skin)". |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
1,2-Hexanediol 2-Ethyl-1,3-hexanediol 1,3-Butanediol 2-Butyl-2-ethyl-1,3-propanediol |

