2-Hydroxypropanoic Acid: Beyond the Lab Bench
From Fermentation Vats to Global Markets: A Historical Perspective on 2-Hydroxypropanoic Acid
People have been making and using 2-Hydroxypropanoic Acid, better known as lactic acid, for centuries, long before anybody stuck a fancy label on it. My grandmother always kept a pickle jar in the pantry, and the signature tang in those vegetables came thanks to lactic acid. This compound emerged through simple fermentation, a process humans learned to harness long before we understood the molecules doing the work. In the late 19th century, chemists like Louis Pasteur gave the process more scientific footing. These days, industry leans heavily on lactic acid for food preservation, pharmaceuticals, bioplastics, and cosmetics, but every bottle, tube, or tub owes its existence to those first gurgling crocks of fermented cabbage and milk. People saw the value of lactic acid long before chemists figured out the specifics.
Understanding the Product: What Sets Lactic Acid Apart
Lactic acid looks unassuming—a clear to yellowish liquid that doesn’t exactly announce its importance. You can buy it in strengths from diluted solutions up to concentrated forms. What makes it remarkable is its biocompatibility. Unlike synthetic chemicals that linger or cause trouble in the environment, lactic acid decomposes naturally. Industry uses both racemic and optically pure versions, with L(+) lactic acid forming the basis for many food and pharma products due to its natural occurrence in the body and safer metabolic fate. The industry’s shift toward greener processes emphasizes lactic acid’s place in the circular economy. People have learned that a product rooted in natural cycles fits into a future that’s hungry for sustainable solutions.
Physical and Chemical Properties Shape Its Roles
Lactic acid brings a unique set of physical and chemical properties that push it into so many corners of daily life. With a melting point just below room temperature and a boiling point around 122°C at low pressure, it’s not a material you find in a crystal dish. What matters most is its ability to dissolve in water, alcohol, and some organic solvents, and its moderate acidity—strong enough to adjust pH in foods, mild enough to go on skin. Its hygroscopic nature means it loves moisture, which boosts its value as a humectant in cosmetics while posing storage headaches. Lactic acid tends to racemize in solution, but careful production steers purity for pharma or food-grade requirements. Every property—solubility, acidity, reactivity—decides where it shines or stumbles.
Labeling and Technical Specifications Matter Outside the Lab
Nobody grabs a bottle of lactic acid at the store without pausing to check the label, especially in regulated industries. Labels carry purity data, sources (fermentation or synthetic), and concentration to make sure users know what they’re working with. In food and supplements, manufacturers must state whether the source is plant-based or animal-derived, since lactic acid can come from both. Lab workers eye the enantiomeric ratio, which points to how biologically active a batch will be. Regulatory bodies outline technical grades for industrial, food, cosmetic, and pharmaceutical uses, and market trends increasingly favor lactic acid produced through GMO-free microbial processes. As demand for transparency grows, so does the need for clear, accurate labels and specifications.
Making Lactic Acid: Old and New School Approaches
Crafting lactic acid can get high-tech, but the heart of the process relies on simple living organisms. The classic route involves fermentation, using bacteria like Lactobacillus in a sugar-rich broth. Bakeries, dairy farms, and breweries all generate waste streams ideal for fermentation. Technicians control temperature, pH, and feedstock closely to optimize yield and purity. Synthetic methods use petrochemical precursors but compete with bio-based processes on cost and sustainability. As biorefining technology improves, fermentation pulls ahead for environmental reasons and product traceability. No matter the method, the process influences purity, toxin level, and byproduct profile, shaping where lactic acid ends up in the marketplace.
Chemical Reactions and Modifications Expand Its Reach
Chemistry classrooms may introduce lactic acid as a simple alpha-hydroxy acid, but industry looks beyond the textbook for reactions that change its character. Lactic acid forms esters when reacted with alcohols, opening up uses in solvents, flavors, and fragrances. Through condensation, lactic acid links into long chains to create polylactic acid (PLA), a biodegradable plastic that now crops up in packaging and medical devices. These polymerization reactions rely on high-purity monomers and precise conditions. Lactic acid also acts as a mild reducing agent and serves as a chiral building block in pharmaceutical synthesis. Each reaction unlocks fresh avenues for application, making lactic acid a molecular workhorse.
Names Tell Their Own Story: Synonyms and Product Branding
It’s easy to get tripped up by a compound that wanders under so many aliases. In science, it goes by 2-Hydroxypropanoic Acid or α-hydroxypropionic acid. Food technologists call it E270 on ingredient lists. In consumer skincare, the front label just says “lactic acid.” Other industries refer to it as milk acid, sour milk acid, or by proprietary blend names in ingredient decks. Every name signals a sector and sometimes a story: lactic acid can come from corn, sugar beet, whey, or chemical synthesis, and branding reflects that choice. Being aware of the context keeps the terminology straight and avoids confusion at the shelf or in the lab.
Keeping People Safe: Best Practices and Industry Standards
Any chemical can cause trouble without the right respect, and lactic acid is no exception. Skin creams use it in low concentrations, but anyone handling concentrated forms knows they need goggles and gloves. Food-grade production follows rigid standards to keep contaminants out, with batch records tracking every step. Industrial settings watch for spills due to floor hazards—lactic acid can make surfaces slippery and damage some types of flooring. Workers stick to protocols for dilution, neutralization, and proper ventilation, especially in facilities where acid vapors build up. International standards spell out handling, transport, and disposal, and reputable manufacturers give clear instructions so users stay safe and regulations get met.
Where Lactic Acid Goes to Work: Real-World Applications
Few chemicals slide so easily between industries as lactic acid does. At breakfast, it’s hiding in yogurt or bread, preserving freshness and taste. Hospitals rely on its derivatives for biodegradable sutures, drug delivery carriers, and even wound-care solutions. In plastics, lactic acid starred in the rise of compostable PLA materials—forks, cups, film wraps—that tackle the disposable plastics problem millions of people contend with every day. Personal care shelves fill up with lotions, exfoliants, and cleansers using lactic acid for mild, effective skin renewal. Industrial water treatment taps into its buffering properties. Each industry discovers fresh uses as the world leans into biobased solutions, giving lactic acid a front-row seat in sustainable innovation.
New Directions: Research and Development Advances
Research teams across the world see lactic acid as more than just a commodity. Laboratories chase new microorganisms to boost fermentation yields or thrive on unconventional feedstocks like agricultural waste, keeping one eye on cost and the other on environmental impact. Polymer scientists shape PLA properties for medical implants or high-strength packaging that breaks down naturally. Synthetic chemists engineer lactic acid derivatives with custom-tailored characteristics, such as modified acidity or improved compatibility in drug formulations. Environmental health experts keep close tabs on lifecycle analysis to make sure biobased solutions genuinely shrink carbon footprints in the long run. Funding and intellectual curiosity keep the field moving forward, as lactic acid’s reputation as an old workhorse mixes with its promise as a modern marvel.
Digging into Toxicity and Human Health
Lactic acid earns its spot in food and skincare in large part due to a strong track record with safety, but nobody working with chemicals should take that too lightly. Lactic acid can still irritate skin and mucous membranes in concentrated form and poses a bigger risk if inhaled or ingested in high doses. Studies show most people tolerate its low concentrations well, and the body has efficient pathways to metabolize and clear lactic acid. Regulatory agencies like the FDA and EFSA grade it as generally recognized as safe (GRAS) for use in foods and pharmaceuticals, but researchers remain vigilant for potential allergenic or cumulative effects, especially in novel delivery methods or combinations. Chronic exposure in industrial settings rarely turns dangerous when safety procedures are followed, but good training and clear labels do much to keep workplace incidents rare.
Looking Ahead: Where Lactic Acid Can Go From Here
People want solutions that don’t just work but also tread lightly on the planet. Lactic acid ticks both boxes—rooted in fermentation, fully biodegradable, and fit for new green technologies. The prospects look brightest in new bioplastics, advanced medical devices, and sustainable food preservation systems. Researchers explore specialty polymers, high-performance blends, and bio-based resins with the building block of lactic acid. Demand will keep rising as economies demand more circular production, and policymakers push for tougher rules on waste and emissions. Lactic acid’s flexibility ensures it won’t disappear anytime soon, and its long journey from pickle jars to research labs keeps gathering speed.
What is 2-Hydroxypropanoic Acid used for?
A Familiar Name in Everyday Life
Most people know 2-hydroxypropanoic acid by its more common name, lactic acid. Long before it appeared on ingredient labels or in science classrooms, folks recognized its punchy taste in sour milk and pickled vegetables. Lactic acid exists at the crossroads of food, health, and industry, even if most shoppers don’t notice it on nutritional tables or cleaning supplies.
In the Kitchen and on the Plate
Fermenting foods brings more than flavor. Dairy products like yogurt and kefir survive thanks to lactic acid. The distinctive tang in kimchi or sourdough bread owes everything to the activities of lactic acid bacteria. It keeps pickles crisp and fresh, and those same properties make it valuable for food preservation. Acting as a natural acidulant, it maintains a proper pH and slows down the growth of less desirable microbes. With more people leaning into fermented foods for gut health, the role of this acid keeps growing, both in home kitchens and on factory lines.
Health and Medicine Use
Human bodies don’t just bump into lactic acid at the dinner table. Muscles rely on it during bursts of effort, swapping oxygen for glucose when exercise ramps up. That burning sensation during a sprint? Lactic acid playing its part. In healthcare, lactic acid appears in topical creams and ointments, especially for dry or rough skin. Doctors sometimes use it for tests to analyze blood flow and tissue health.
Industry Beyond Food
Lactic acid gets busy outside of food and medicine too. In cleaning products, it works as both a descaler and an antibacterial agent, making it a popular choice for eco-friendly cleaners. The biodegradable nature of lactic acid gives it an edge over harsher chemicals, meeting the needs of households looking for safer cleaning methods.
Polylactic acid plastics, made from lactic acid, now show up in everything from biodegradable cups to medical implants. Since these plastics break down in composting conditions, they look more attractive to industries worried about plastic waste. That switch over to bioplastics doesn’t solve everything, but it marks a big step away from petroleum-based materials that persist in landfills for decades.
What Needs to Change
The benefits bring challenges. Lactic acid production draws mostly from corn and sugarcane, raising questions about resource use, land competition, and the impact on food supplies. Running factories on crops pushes up demand for monocultures, which can strip soil and hurt biodiversity. The plastics industry is growing fast, but unless cities invest in the right waste management and composting systems, “green” plastics end up in the same old dumps as regular ones.
Farmers, manufacturers, and local governments can work together to change sourcing and disposal habits. Crop rotation, organic practices, and sourcing from more sustainable plants like cassava or agricultural byproducts could help. Building up composting facilities in urban areas could keep bioplastics out of landfills and close the loop on waste. Consumers, too, can look past labels and ask retailers where their products come from, and how to properly dispose of them.
Looking Ahead
2-hydroxypropanoic acid isn’t going anywhere. Its trail leads through home kitchens, hospitals, and recycling centers. People depend on it in ways that go unnoticed—until someone checks a label or trades in a single-use plastic cup for a compostable one. Paying attention to its journey from farm to factory to breakfast table helps everyone understand the risks and rewards buried in a simple acid with a complicated role.
Is 2-Hydroxypropanoic Acid safe for human consumption?
Understanding What It Is
Most people might not recognize the name 2-hydroxypropanoic acid right away, but it shows up on ingredient lists more often than you’d guess. This compound is better known as lactic acid. It pops up in yogurt, sauerkraut, sourdough, pickles, and even some processed foods. Lactic acid emerges naturally as part of fermentation—a process humans have trusted for centuries to preserve food and add flavor.
Natural Roles and Body Chemistry
Lactic acid isn’t just a food additive. Human muscles produce it during exercise. That burning feeling after sprinting comes in part from lactic acid, which shifts around as the body tries to keep up with demands for energy. Most folks can handle lactic acid in reasonable amounts; it cycles through the bloodstream and gets broken down by the liver.
Regulations and Food Safety
Food safety groups around the world treat lactic acid as a low-risk food additive. The U.S. Food and Drug Administration labels lactic acid as “generally recognized as safe” for use in food. Europe’s food safety authority and other regulatory bodies have similar positions. These organizations set limits based on solid research and a history of safe use.
In the food industry, lactic acid does more than just tweak flavor. It works as a pH adjuster, a preservative, and sometimes as a curing agent in meats. I’ve bought yogurt and cheese at the grocery store plenty of times, and lactic acid doesn’t raise any eyebrows among shoppers in my experience. Just tasting a spoonful of sauerkraut gives a hint of its tangy work.
People with Special Conditions
Some rare medical conditions change the picture. Those with certain metabolic disorders might not process lactic acid as efficiently, and doctors warn against high intake in these cases. In healthy people, the kidneys and liver keep levels balanced without any trouble. Lactic acid doesn’t linger or build up from regular eating—our bodies know how to handle it.
Small children and infants, whose metabolism works differently, need a closer look. Medical formulas and infant foods sometimes avoid lactic acid to be safe, following pediatric recommendations. Most adults, though, face little risk with ordinary amounts in food.
Industrial and Cosmetic Uses
Lactic acid also surfaces in non-food products. Some skin creams and peels include it for gentle exfoliation. On the production line, strict laws separate food-grade lactic acid from that intended for cosmetics or cleaning products. Food-grade lactic acid must stay pure and free from harmful contaminants, which strict oversight helps guarantee.
Looking at the Bigger Picture
People worry about chemical names and unfamiliar additives, but lactic acid stands out as a familiar and trustworthy ingredient. Research over many decades backs up its safety in foods. To keep things safe, consumers should stick to eating foods as intended and follow any medical advice if they have health concerns. Washing produce, reading labels, and buying from reputable stores help cut down on risk—not just for lactic acid, but for all food additives.
Trusted sources like the FDA, EFSA, and registered dietitians offer clear recommendations backed by solid evidence. As with anything in life, moderation and informed choices matter most.
What is the shelf life of 2-Hydroxypropanoic Acid?
Understanding 2-Hydroxypropanoic Acid
2-Hydroxypropanoic acid goes by the name lactic acid. It shows up in food, cosmetics, pharmaceuticals, and even the textile industry. Folks in food manufacturing use it for acidity regulation and flavor. Skincare companies add it to products for exfoliation. Chemists reach for it because it’s biodegradable, affordable, and easy to work with. No matter the application, lactic acid faces one big question—how long does it stay effective?
Shelf Life: Not Just a Number on a Label
Lactic acid won’t last forever. Its shelf life depends on a few key things: purity, packaging, climate, and concentration. As someone who has dabbled in both lab work and food prep, I’ve learned a few truths. Acid solutions—especially those made at high purity—tend to hold up well if kept away from sunlight and sealed tight. So much comes down to storage. Leave a bottle of lactic acid in a hot, humid room, and it'll degrade a lot faster than if you tucked it away in a dark, cool cupboard.
Manufacturers commonly recommend using lactic acid within two years of production. This figure assumes a standard plastic or glass container, stored at around 20-25°C, shielded from moisture and direct light. After about two years, purity starts to slip, and organic acids can break down or react with container materials. Once you crack open a new bottle, air speeds up these changes, so don’t ignore expiration dates.
Real-World Risks of Ignoring Shelf Life
Using spoiled lactic acid risks more than just weak results. For instance, in food production, old acid could create off-flavors or even unsafe products. Take it from someone who once overlooked expiry dates during a fermentation experiment—one whiff was enough to empty the lab. In pharmaceuticals or skincare, degraded lactic acid could lose effectiveness or irritate sensitive skin.
There’s also a big cost angle. No company enjoys tossing out expired inventory. But keeping outdated acid on the shelf just creates more headaches down the line—product recalls, customer complaints, and wasted time tracing back problems.
Solutions: Storing and Monitoring for Maximum Value
It’s not rocket science. Good storage stretches shelf life, often beyond labeled dates. Store lactic acid in a well-sealed, chemical-resistant container. Keep that container in a cool, dark spot—think of the way you store good wine. Avoid temperature swings, and don’t let humidity into the mix. For big operations, some labs track acid quality with regular titration or pH tests, just to be sure nothing sneaks past.
My advice always circles back to basic housekeeping. If you’re handling industrial or bulk quantities, rotate inventory using a “first in, first out” approach. Clearly mark containers with open dates. For high-stakes batches, keep small reserve samples frozen as controls, just in case you need to check purity later on.
Why It’s Worth Focusing on Shelf Life
It might seem simple, but paying attention to the shelf life of lactic acid guards the quality of everything downstream. Food tastes fresher, cosmetics perform as intended, and chemical processes run without nasty surprises. That attention to detail builds trust—between producers, regulators, and the people who depend on those products every day. No one gets there by chance; strong habits and a few extra steps keep lactic acid safe and useful till the very last drop.
How should 2-Hydroxypropanoic Acid be stored?
Understanding the Stuff Beneath the Lab Coat
2-Hydroxypropanoic acid, better recognized as lactic acid, shows up everywhere from food preservation to cosmetic creams and even heavy-duty industrial cleaners. In my early days working in a university lab, I saw firsthand what happens when storage rules get tossed aside: labels peeled, tops left loose, and the faint, tangy smell in the air. Maybe it sounds like a small thing, but the truth is, how you store chemicals impacts health, product quality, and even business costs.
Why Proper Storage Makes a Difference
Most folks think lactic acid sits pretty among safe chemicals. That’s misleading. It still punches above its weight. Its strong odor, tendency to corrode, and knack for irritating skin make it a hazard if left forgotten or stored with incompatible materials. The U.S. Occupational Safety and Health Administration spells out the risk – too much exposure, especially in poorly ventilated spaces, can trigger headaches, sore throats, or worse. This isn’t about scaring people. It's about owning up to what a few drops on the floor or a leak in a shelf can mean in a real workplace.
Getting Storage Right in Real Life
Water makes lactic acid both useful and tricky. It pulls in extra moisture from the air, which means caps have to stay tight. If you skip the seal, you get watered-down acid that won’t behave the same in a formula, whether you’re working in a food lab or brewing beer. Once, I watched a batch of yogurt curdle wrong because the starter ingredient had absorbed just enough humidity from an unsealed bottle. Nobody likes to throw away a full tank of spoiled product.
Storing this acid calls for low temperatures and darkness. Shelves in cool rooms or refrigerators work best. Sitting bottles in a sunny window accelerates breakdown. The material's flammability isn’t a daily worry, yet it reacts with strong bases, oxidizers, and metals. Tossing lactic acid behind an old can of bleach or next to sodium hydroxide becomes a recipe for shelf fires or chemical clouds. Clear labeling and organizing are anything but busywork. In my experience, a little up-front time spent sorting means fewer emergencies later.
Materials Matter in Storage
Don’t rely on every bottle you find. Certain plastics, like HDPE, and glass make the safest containers. Metal caps rust and can trigger odd reactions. Rubber doesn’t hold up. Using sturdy, compatible containers pays dividends. If your storage room has a spill tray or secondary containment, you’re already a step ahead, catching leaks before they reach floors or drains.
Solutions for Everyday Workspaces
It sounds routine, but investing in tight-sealing containers, creating regular inventory checks, and training staff connects storage guidance with actual daily routines. I remember a warehouse switch from generic shelves to designated chemical racks. Workers cut down on cross-contamination and confusion. Documentation—logbooks and digital inventory—keeps teams alert to expiry dates and helps with audits. Ventilation reduces inhalation risks; even a cheap exhaust fan beats stale, recirculated air.
A Culture of Safety Isn’t Overkill
Clear instructions, well-organized storage, and frequent checks deliver returns that matter: fewer accidents, consistently safe products, and peace of mind for everyone in the building. From factories to small labs, safe chemical storage builds trust—among teams, with regulators, and for the customers who expect clean, reliable results.
What are the potential side effects of using 2-Hydroxypropanoic Acid?
Understanding 2-Hydroxypropanoic Acid
2-Hydroxypropanoic Acid, often called lactic acid, shows up in places most people don’t expect—yogurt containers, cosmetic shelves, even medical offices. It’s got a reputation for being both useful and mild, but like anything used in products we eat or rub on our skin, it can give us trouble if handled carelessly or misunderstood.
Skin and Eye Irritation
People running cosmetic experiments in their own bathrooms might notice some irritation after using products loaded with lactic acid. Redness, itching, or a stinging sensation pop up as the most common complaints. High concentrations, like the peels professionals use, crank up the risk. Accidentally splashing it in the eyes can bring serious pain, watery eyes, and blurry vision—problems nobody wants while trying to chase a glow.
Digestive Disturbances
Swallowing lactic acid on purpose happens through certain supplements or processed foods. Most folks never notice anything. But anyone going overboard could run into stomach pain, nausea, or worse, diarrhea. The body only needs so much help; force-feeding it with concentrated acids doesn’t push health forward. Gut sensitivity varies, and extra caution pays off, especially for people already dealing with digestive disorders.
Allergic Reactions and Sensitivities
The skin doesn’t always announce its aversions right away. People might develop hives or worsening eczema after using creams with lactic acid. I’ve seen friends swear off a whole cosmetic line because one bottle kickstarted a grainy, inflamed rash. Patch tests may seem old-fashioned, but they save a lot of time, money, and frustration for people with sensitive skin.
Damage from Overuse in Medical Settings
Doctors use concentrated lactic acid solutions to remove warts or treat other tough skin issues. Piling it on too thickly or ignoring a doctor’s directions can damage healthy tissue around the treatment site, causing wounds that take weeks to heal. Infection risk always pops up whenever skin barriers break. No one deserves a bigger problem than the one they tried fixing.
Lactic Acidosis in Rare Health Conditions
The body normally handles lactic acid like a champ. Certain rare conditions disturb this balance. People with kidney or liver disorders, or rare metabolic conditions, sometimes build up too much lactic acid—a situation called lactic acidosis. It’s a medical emergency, not a side effect from dabbing a little cream, but those with health problems need close supervision before using supplements or treatments high in lactic acid.
Facts and Responsible Use
Regulators like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency watch over products mixing lactic acid, setting limits for safe amounts. Looking at ingredient lists, following directions, and staying far from do-it-yourself treatments with industrial-strength products helps prevent trouble. Even “natural” acids demanded respect.
Solutions and Safer Practices
People get the best results from any acid—lactic included—by starting slow and watching their body’s response. Reading, patch testing, and seeking medical advice for chronic skin or health issues turn a risky adventure into a smart routine. Bringing any health professional into the conversation, especially for vulnerable groups or children, stops problems before they start. Instead of chasing trends, steady, sensible steps protect health without sacrificing results.
| Names | |
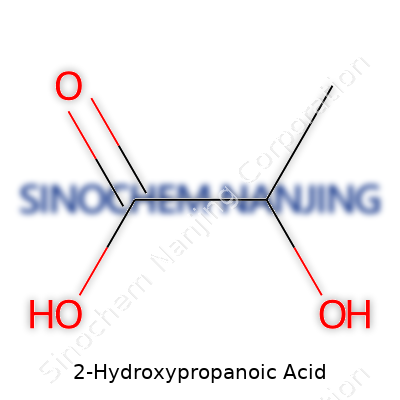
| Preferred IUPAC name | 2-Hydroxypropanoic acid |
| Other names |
Lactic acid Milk acid 2-Hydroxypropionic acid Alpha-hydroxypropionic acid Propanoic acid, 2-hydroxy- |
| Pronunciation | /tuː haɪˌdrɒk.si prəˈpeɪ.nɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 79-33-4 |
| Beilstein Reference | 4210112 |
| ChEBI | CHEBI:422 |
| ChEMBL | CHEMBL50 |
| ChemSpider | 503 |
| DrugBank | DB00190 |
| ECHA InfoCard | 03-2119471830-50-0000 |
| EC Number | 2.3.1.8 |
| Gmelin Reference | 14215 |
| KEGG | C00186 |
| MeSH | D001322 |
| PubChem CID | 612 |
| RTECS number | OG9800000 |
| UNII | J1Q1A21L5K |
| UN number | UN1819 |
| CompTox Dashboard (EPA) | DTXSID9020816 |
| Properties | |
| Chemical formula | C3H6O3 |
| Molar mass | 90.08 g/mol |
| Appearance | Colorless to yellowish, syrupy liquid or solid |
| Odor | Odorless |
| Density | 1.206 g/cm³ |
| Solubility in water | Miscible |
| log P | -0.72 |
| Vapor pressure | 0.08 mmHg (20°C) |
| Acidity (pKa) | 3.86 |
| Basicity (pKb) | 12.62 |
| Magnetic susceptibility (χ) | -12.3 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.435 |
| Viscosity | 14.6 mPa·s (20 °C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 86.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -694.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1342.6 kJ/mol |
| Pharmacology | |
| ATC code | A16AX01 |
| Hazards | |
| Main hazards | Causes serious eye damage. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05 |
| Signal word | Warning |
| Hazard statements | Hazard statements: "H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P330, P337+P313 |
| NFPA 704 (fire diamond) | 2,0,0 |
| Flash point | 53 °C |
| Autoignition temperature | 410 °C |
| Explosive limits | Explosive limits: 4–17.5% |
| Lethal dose or concentration | LD50 (oral, rat): 3730 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 3730 mg/kg |
| NIOSH | WZ2620000 |
| PEL (Permissible) | 50 ppm |
| REL (Recommended) | 25 mg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Lactic acid Pyruvic acid Malic acid Citric acid Glycolic acid |

