2-Heptanone: Not Just Another Industrial Compound
Digging Into the History of 2-Heptanone
Most people don’t spend their days thinking about compounds like 2-heptanone. Still, it shows up in more corners of our world than we realize. Chemists began studying it in the early days of organic chemistry, as industry pressed for better solvents and fragrances. Over a century ago, researchers first isolated 2-heptanone from natural oils, surprised to find its scent both light and sharp. It earned a quiet spot among solvents and chemical intermediates, never making front page news like aspirin, but quietly shaping industries from food flavoring to pest control.
A Straightforward Look at Its Physical and Chemical Quirks
2-Heptanone stands out for its clear, colorless liquid form. Its scent, a cross between bananas and sharp cheese, cannot go unnoticed even at low concentrations. With a boiling point close to 151°C and modest water solubility, this molecule finds its sweet spot between volatile and stable, which makes it useful on the shop floor and in research labs. It won’t stick around like heavy oils, but it won’t bolt for the air the moment you open a bottle. The ketone group at its second carbon spot shapes its reactivity, attracting nucleophiles and serving as a springboard for more complex molecules.
Technical Specifications and Labeling Speak Volumes
When you crack open a container of 2-heptanone, you rely on labeling to know what you’re working with. Suppliers list purity, usually north of 98%, since impurities change how the molecule behaves in scent or reaction. Labels flag flammability and vapor risk, with safety data pointing to the need for adequate ventilation and protection from open flames. Industries and labs keep a close eye on specifications, since deviations—even minor ones—can alter outcomes in flavor chemistry or materials science work.
How People Make 2-Heptanone and Push Its Boundaries
The preparation of 2-heptanone relies on oxidation methods. Traditionally, chemists would oxidize 2-heptanol or, in more modern settings, work with catalytic dehydrogenation of heptanal or heptanoic acid. This route offers a steady yield and meets the scale of demand seen in manufacturing today. Curious researchers keep their eyes open for greener methods, aiming to cut out hazardous reagents. Biotechnological routes, using engineered microbes, attract attention by promising to make the process cleaner and safer, although cost and efficiency keep chemical synthesis in the lead for now.
Chemical Reactions and the Search for Novel Conversions
If you hand 2-heptanone to a synthetic chemist, they’ll look to its reactive carbonyl group for creative possibilities. This group, doubly bonded to oxygen, sets the stage for additions, condensations, and reductions. The compound features in syntheses of more complex flavors, pharmaceuticals, and even specialty materials. There’s research into modifying it for use in pheromone mimics—since certain insects produce 2-heptanone naturally as a warning signal. This chemical’s reactivity drives innovations in both academic and applied chemistry circles.
Names Matter: Decoding the Labels of 2-Heptanone
In catalogs and research papers, 2-heptanone might turn up as methyl n-amyl ketone, heptan-2-one, or ethyl butyl ketone. Tracing these names isn’t just academic—mislabeling can lead to confusion in handling, purchasing, or regulatory review. Producers and labs rely on strict nomenclature, not simply to follow rules, but to avoid costly errors or safety lapses. Naming also helps track its natural and synthetic sources, from blue cheese to control of bee behavior.
Practical Concerns: Safety and Operations in Everyday Use
Anyone working with 2-heptanone needs to respect its hazards. The speed at which it evaporates calls for real containment, not just cracked windows or casual fume hoods. Skin contact, inhalation, or eye exposure can spark irritation. Fire risk runs high, especially in poorly ventilated spaces. Safety protocols—gloves, goggles, no open flames—shouldn’t feel optional. Reports of industrial accidents remind us that small lapses in respect for basic standards can lead to big problems. Each worker’s grip on these practices spells the difference between safe, productive work and emergency room visits.
Where 2-Heptanone Leaves Its Mark: Applications in the Real World
Ask around and you’ll find 2-heptanone in some surprising places. In food and beverage industries, it boosts flavors. At low concentrations, it cuts through bland notes in cheese and baked goods, punches up the aroma of certain distilled spirits, and flavors confections. Perfume makers leverage its scent for fruity, creamy undertones. Its solvent power unlocks sticky resins, helps extract flavors and fragrances, and fits into coatings and cleaning solutions. Maybe most surprising is its role in pest control—this molecule features in the natural alarm signals of honeybees and rodents. Researchers tinker with it in hopes of controlling unwanted insect behavior or signaling breed health in apiaries.
Research and Development: Where Curiosity Drives Change
The journey of 2-heptanone doesn’t stop at industry use. Scientists devote time to understanding how animals and microbes make and use it, forming a bridge between chemistry and biology. There’s work underway refining detection methods, since monitoring tiny amounts can pinpoint food spoilage or airborne hazards. Biochemists probe its interactions with protein targets in insects, looking for new angles in pest control. Meanwhile, material scientists consider its solvent properties for greener extraction or cleaning regimens. The pursuit of higher yield synthesis, lower waste, and new function keeps 2-heptanone at the intersection of academic curiosity and practical demand.
Toxicity: What the Evidence Really Says
Any chemical that crosses from lab to food to factory floor has to answer tough questions about safety. Toxicity tests on 2-heptanone show moderate acute toxicity, with studies in rodents highlighting respiratory and nervous system effects at high exposures. Workers handling gallons of it daily face the most risk, so occupational standards—air monitoring, exposure limits, and emergency protocols—remain critical. Regulatory bodies like the EPA and ECHA examine new data for long-term risks, especially as synthesis volumes climb. The food world keeps concentrations low, well beneath safety thresholds. Ongoing toxicology studies aim to catch subtle or chronic effects, although so far, evidence supports safe use within set limits.
Looking Forward: The Road Not Yet Taken
As sustainability pressure tightens on all chemical manufacturing, 2-heptanone’s future hangs on cleaner production and smarter use. Biotech advances could shift its synthesis from petrochemicals to microbial fermentation, keeping dangerous byproducts out of the process and cutting down carbon footprints. In pest management, this one molecule might unlock eco-friendlier tactics, reducing reliance on older, harsher pesticides. Better detectors and safer formulations can stretch its presence from the lab bench to industrial vats. For manufacturers and researchers, the challenge lies in keeping its quirky properties while meeting tougher environmental and safety expectations. Progress demands openness to new science and accountability for impacts from field to factory.
What is 2-Heptanone used for?
Everyday Value Beyond the Lab
Most folks haven’t heard of 2-heptanone, but this odd-sounding molecule connects the world of chemistry with our everyday lives. I remember the sharp whiff of bananas in an old bakery, only to learn much later that a chemical like 2-heptanone can give off a similar aroma. The scent isn’t just for show—companies use it to flavor foods and fragrances. So whenever someone bites into candy with that creamy, fruity top note, odds are they’re enjoying a hint of what 2-heptanone brings to the table.
Role in Flavors and Fragrances
Chefs and perfumers seek out 2-heptanone for its pleasant, nutty scent. This means your vanilla-flavored ice cream or your favorite baked treat might carry a small dose. It works well because it’s strong enough without hitting you over the head. It’s also used in perfumes, where it gives depth to floral or fruity blends. Beyond food and perfume, certain tobacco blends get a boost from it, adding subtle complexity to the flavor.
Looking at the Science
2-Heptanone isn't just about nice smells. Scientists find it in bee venom. Researchers have shown bees release this molecule as a signaling tool—a kind of alarm. In some studies, 2-heptanone worked like an anesthesia against pests that bother bees. Beekeepers and scientists keep an eye on this, hoping to find a humane pesticide inspired by nature, rather than broad-acting chemicals that stress soil and water.
Spotlight on Health and Personal Care
This molecule isn’t limited to what we eat or put on our skin. It pops up in health care, too. Some medical researchers use it to detect diseases through breath analysis. For example, doctors studying children with certain genetic disorders noticed higher levels of 2-heptanone in urine samples, pointing to possible diagnostic clues. It offers a window into how our bodies break down fats, giving early warning signs for a handful of rare illnesses.
Cleaning, Industry, and Beyond
Outside the beauty and medical scene, factories lean on 2-heptanone for cleaning up grease and oil. It breaks down industrial residue without causing as much environmental harm as some older solvents. Paint companies use it to get consistent drying times and a smoother finish. Shoe factories and leatherworkers also make use of it to help glue and dye materials.
Keeping It Safe
Anyone who’s ever worked in a lab knows safety matters. 2-Heptanone, while useful, can irritate the skin and eyes in concentrated form. That means manufacturers and workers wear gloves and good ventilation keeps fumes at bay. The U.S. Environmental Protection Agency keeps a watchful eye, setting limits for workplace exposure. Responsible companies act on science—protecting both their workers and their end customers.
Opportunities and Alternatives
As more shoppers look for safer, cleaner, and eco-friendly products, the chemical industry faces new demands. Substituting milder, plant-based alternatives where possible leads to less risk and waste. Yet, 2-heptanone has carved out a place because of its effectiveness at low concentrations. Researchers still search for even less allergenic or more sustainable ways to get the same results, especially in food applications and pest control. By blending tradition with new insight, everyday chemistry can keep getting safer and smarter.
Is 2-Heptanone safe to handle?
Understanding 2-Heptanone
2-Heptanone has carved out a spot in all sorts of industries, from flavors and fragrances to scientific labs and even pest control. You might come across it in artificial banana flavoring, or find it included in certain industrial cleaning formulas. The question keeps surfacing inside safety training rooms and research meetings: is working around 2-Heptanone a risk?
What Science Shows
The safety data on 2-Heptanone runs deep. According to the U.S. National Library of Medicine, inhalation of high concentrations in an unventilated space can bring on headaches, dizziness, or nausea. At lower levels—like those found in workplace settings that follow good standards—it usually isn’t linked with severe reactions. Still, symptoms of overexposure make it clear how important it is to limit direct, prolonged contact.
People often overlook that 2-Heptanone has a low evaporation point and produces vapors easily, especially during warmer months or in tight indoor quarters. Even those who keep a careful distance can end up inhaling fumes or letting it touch the skin by accident. I’ve handled plenty of industrial solvents in my work, and the biggest lessons I’ve learned usually happen after a minor mistake—the missed glove, the hurried splash during cleanup. A careless routine can turn a mild irritant into tomorrow’s health problem.
Regulations and Limits
Agencies like OSHA and NIOSH have set exposure limits for solvents with similar properties, though 2-Heptanone doesn’t have its own specific permissible exposure limit. Some European agencies give it an occupational exposure guideline of about 5 ppm for an 8-hour shift. Risk managers in responsible workplaces read these numbers as starting points. They tighten air monitoring procedures and build in extra protection for anyone handling raw chemicals.
For families and workers alike, stories of exposure usually come from poorly ventilated areas. Warehouse workers and cleaning staff sometimes skip protective equipment. Because 2-Heptanone’s strong but sweet odor masks how quickly it evaporates, people can forget they’re breathing it in until symptoms show up. The compound can cause mild skin irritation and eye discomfort at higher doses; gloves and goggles should stay within arm’s reach.
Reducing Hazards and Building Safer Habits
Dealing with 2-Heptanone safely doesn’t require an expert degree—just consistent habits. Anyone working with open containers or mixing equipment needs well-fitted gloves, splash goggles, and steady air circulation (at minimum, a decent exhaust fan). I always advise a “don’t trust your nose” approach: clear, mechanical ventilation runs throughout the day, not just for big spills.
Training makes the single biggest difference. New hires often hesitate to speak up about safety issues around solvents, so employers who check air monitors, enforce glove use, and let people report headaches or eye irritation without penalty cut problems down before they grow. Material Safety Data Sheets might seem dense, but those two pages on recommended protection gear make short work of confusion for workers and managers alike.
Looking Forward
Researchers keep an eye on chemical exposure trends and encourage industry groups to update safety guidelines as new data rolls in. Safer handling means less guesswork and fewer trips to the doctor for blurry vision or sore throats. With consistent attention to best practices, 2-Heptanone can stay useful without putting health at risk.
What are the physical properties of 2-Heptanone?
What Sets 2-Heptanone Apart
2-Heptanone, often called methyl n-amyl ketone, looks like a clear, colorless liquid. The smell hits you quick—sweet, almost banana-like. That alone turns heads in both the fragrance and flavor industries. More than just a pretty scent, it boils at about 151°C and has a melting point down near -35°C, pointing to stability and resilience you want from a solvent or chemical intermediate. Its density sits around 0.81 g/cm3 at room temperature, sliding it into the lighter side among ketones.
Looking Through a Practical Lens
Many solvents create issues around flammability, and 2-heptanone isn’t shy here. Its flash point sits roughly 44°C, which asks manufacturers and handlers to respect ventilation and temperature controls. Workers in labs, paint shops, or perfumeries benefit from knowing this fact like they’d remember an emergency exit route—keep the space cool and watch for open flames.
Water solubility matters once spills enter the picture. 2-heptanone only mixes about 4 g per liter in water, so in the event of a spill, most of it sticks to surfaces or floats instead of dissolving away. This boosts the need for absorbent pads or containment rather than expecting the nearest drain to handle cleanup. I’ve watched coworkers burn time with solvents that refuse to mix—certainly leaves an impression about choosing tools and cleaning up safely.
The vapor pressure of 2-heptanone (around 5 mmHg at 20°C) suggests it doesn’t evaporate immediately, but given enough time or heat, it’ll fill a room. That carries risks for indoor air quality. It’s impossible to ignore the pressure to invest in fume hoods and protective gear if your daily grind involves regular exposure. Jobs shouldn’t cost you your health, so attention should fall on both the chemical and the way it enters the work environment.
Why These Properties Matter
Flammability, low water solubility, and its modest vapor pressure define not just storage and transportation, but also insurance premiums and regulatory attention. In factories or research spaces, these numbers dictate how safely work flows. Regulations like OSHA’s hazardous chemical rulebook draw hard lines and keep everyone honest. Thanks to chemical accidents in poorly ventilated shops, safety officers have pushed for tighter caps, better gloves, and improved air monitoring.
2-heptanone’s physical state and vapor tendencies shape its uses in more ways than labels suggest. It acts as an animal repellent in some settings and carries flavors in others. When you get the properties wrong—say by ignoring its volatility—your product’s shelf stability or your staff’s safety takes the hit. Open containers in a hot warehouse could mean more wasted product, or in worst cases, a safety report nobody wanted.
Pushing for Smarter Solutions
Stronger training goes hand in hand with material safety data sheets. Many shops could use more hands-on spill control practice—those “what-if” moments turn into muscle memory. Tech advances keep building better sensor systems, offering early-warning on vapor buildup. Biodegradable absorbents and smarter packaging designs help prevent leaks from spiraling into full-blown emergencies. In the end, physical property details save money, minimize risks, and keep both workers and products safer.
Understanding what 2-heptanone does outside a textbook, in busy, imperfect spaces, draws a line between safe practices and costly mistakes. For anyone handling it, putting these values into real-world perspective matters more than the numbers themselves.
How should 2-Heptanone be stored?
Taking Chemical Safety Seriously
Commercial labs and factories usually work with a slew of different chemicals, but few attract attention like 2-Heptanone. Smelling a bit like bananas and often showing up in everything from flavor manufacturing to industrial cleaning, this solvent brings a few quirks to the table—some that catch folks off guard if they store it wrong.
What’s Tricky About 2-Heptanone?
Anyone who’s spent time in a storage room with chemicals knows the difference a few degrees can make. With 2-Heptanone, the flash point is about 40°C (104°F)—meaning it can catch fire under not-so-extreme conditions. Add volatility to the mix, and it doesn’t take much for vapors to build if containers don’t seal well or the climate gets too warm. The stuff irritates eyes and skin, and inhaling high levels for too long can leave you dizzy or with a headache. Even though it drifts out of containers quickly, the dangers don’t drift away.
Safe Spaces: No Lazy Shortcuts
Years back, I watched a co-worker stick a drum of solvent near a heater, figuring a day or two didn’t matter. That mistake nearly lit up the entire storage bay. Rule number one with 2-Heptanone: give it a home away from sources of heat and sparks. Stay far from furnaces, open flame, or any machinery that could throw off a hot spark or two.
Strong ventilation proves its worth every day. Without it, fumes can sneak up quickly and concentrate in unexpected corners. I’ve seen too many old storerooms with one window, no fan, and everyone wondering why things smell spicy. Good air movement blows vapors away and shrinks the danger zone. Most modern labs use metal safety cabinets that keep vapors in check and resist fire. If a spill hits a hot surface, you want to be sure neighboring drums won’t follow.
Keeping Things Labeled and Locked
Mislabeling brings confusion at the worst possible moments. I’ve visited shops where half the containers looked the same color, marked with smeared ink or half a label. With chemicals like this, clear marking and easy-to-read warning stickers beat fancy safety posters on the break room wall. It’s all about keeping the work area honest and helping out the new employee who can’t tell methyl ethyl ketone from the solvent they’re supposed to mop up.
Approved containers matter. Polyethylene works, but metal drums with good closures keep vapor losses down. You don’t want to walk into a storage area and realize a greasy pool has collected under loose caps. Locking up drums after use prevents curious hands—from kids at home or night-shift staff in a warehouse—from mistaking the stuff for something harmless. I’ve run safety checks for years and find simple, sturdy locks keep stories of accidental exposure out of the company’s safety log.
Planning for the “What If” Moments
No single warehouse has perfect conditions all year long. Electrical storms, broken AC, even a dropped barrel changes the whole risk profile. Fire extinguishers and clean-up kits near the entry help catch spills fast. Absorbent pads, gloves, and a reliable respirator don’t just look good during inspections—they give regular staff a fighting chance if something goes sideways.
Anyone using or storing 2-Heptanone should know the basics: keep it cool, keep it away from sparks, seal it up tight, and check labels every season. Cutting corners in storage means inviting a problem you can’t walk away from. Smart storage helps everyone stick around for next year’s safety drill.
What are the hazards and first aid measures for 2-Heptanone exposure?
Why 2-Heptanone Matters in the Workplace
Workers in certain factories and laboratories handle chemicals like 2-Heptanone every day. This solvent has a sharp banana-like scent and often shows up in cleaning agents, glues, and oils. Companies lean on it for its quick evaporation and greasy stain-busting power. When you spend time around warehouses, machine shops, or chemical plants, you notice there isn't always a strong focus on the subtle risks of these everyday materials. I’ve seen folks wipe chemical splatter off their skin and go right back to work, not realising the hidden cost.
Breathing and Skin Hazards
Breathing in 2-Heptanone vapors can irritate your nose, throat, and lungs. Prolonged exposure makes headaches and dizziness more likely, and if the ventilation’s poor, folks can wind up woozy or nauseous. People underestimate how quickly vapors build up, especially in smaller rooms that lack airflow. Some may even start to lose coordination or feel confused. Studies have shown that constant low-level exposure increases the risk of nerve problems over time.
Skin and eye contact shouldn't get brushed aside, either. Spilling 2-Heptanone on bare skin can cause redness or a burning sensation. Eyes react even faster, with stinging and watering that can linger for hours. In one warehouse I worked at, a sudden splash during cleanup sent a co-worker to the emergency room because he rubbed his face before rinsing his hands. These stories repeat themselves because folks rush or skip safety gear like gloves and goggles.
First Aid Steps for 2-Heptanone Exposure
Quick action always beats waiting to see if discomfort fades. If vapors make someone dizzy or short of breath, help that person step outside or move to an area with fresh air. If breathing doesn’t improve fast, don’t hesitate to seek emergency medical help. Sometimes people try to tough it out and pay for it later with days of headaches or fatigue.
If liquid 2-Heptanone hits the skin, wash the area with soap and running water for at least fifteen minutes. Don’t bother with wipes or sanitizer—plain water removes more residue. If clothes get soaked, take them off right away and put them in a plastic bag so no one else gets exposed. For splashes to the eyes, hold eyelids open under running water for a good fifteen minutes, no matter how much it stings. Skipping this step can mean serious long-term damage, something I’ve seen in workers who thought a quick rinse was “good enough”.
Reducing Risks at the Source
The smartest workplaces build these first aid lessons into day-to-day routines. Nobody stays perfectly careful all the time, so regular reminders about using gloves, goggles, and proper ventilation make a dent in accident rates. Safety data sheets belong on walls at eye level, not stuffed in dusty binders. Having eyewash stations and clearly marked sinks within reach saves valuable time. I remember companies where supervisors drilled emergency response steps every month—those extra minutes of practice really mattered during real spills.
No one wants to see co-workers get hurt from something preventable. Making the risks obvious, planning for quick responses, and keeping protective equipment handy protect everyone who shares the space. These aren’t abstract goals—they’re steps that make people go home safe every day.
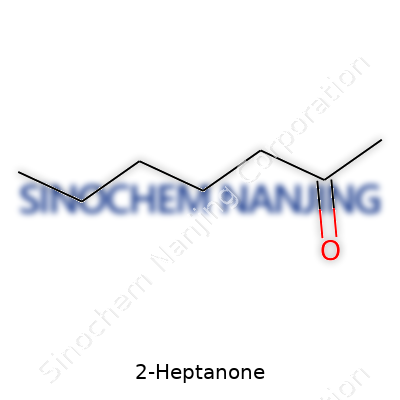
| Names | |
| Preferred IUPAC name | Heptan-2-one |
| Other names |
Heptan-2-one Methyl amyl ketone MAK Sek-Heptylketon Ethyl butyl ketone |
| Pronunciation | /ˈtuːˈhɛptəˌnoʊn/ |
| Identifiers | |
| CAS Number | 110-43-0 |
| Beilstein Reference | Beilstein Reference: "1209286 |
| ChEBI | CHEBI:28484 |
| ChEMBL | CHEMBL142361 |
| ChemSpider | 7715 |
| DrugBank | DB02171 |
| ECHA InfoCard | ECHA InfoCard: 100.003.339 |
| EC Number | 01-2119471307-38-XXXX |
| Gmelin Reference | 805252 |
| KEGG | C01579 |
| MeSH | D006419 |
| PubChem CID | 7004 |
| RTECS number | MJ5075000 |
| UNII | C18F9O6SLN |
| UN number | UN 1230 |
| Properties | |
| Chemical formula | C7H14O |
| Molar mass | 114.19 g/mol |
| Appearance | Colorless liquid with a fruity odor |
| Odor | banana |
| Density | 0.817 g/mL at 25 °C |
| Solubility in water | 5.7 g/L (20 °C) |
| log P | 1.98 |
| Vapor pressure | 0.93 kPa (at 20 °C) |
| Acidity (pKa) | 20.00 |
| Basicity (pKb) | pKb ≈ 7.66 |
| Magnetic susceptibility (χ) | -7.61 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.414 |
| Viscosity | 0.657 mPa·s (20 °C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -277.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4176.7 kJ/mol |
| Pharmacology | |
| ATC code | N01AX10 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P312, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 41 °C |
| Autoignition temperature | 435 °C |
| Explosive limits | 1.5–7.0% |
| Lethal dose or concentration | LD50 oral rat 1600 mg/kg |
| LD50 (median dose) | 730 mg/kg (rat, oral) |
| NIOSH | MH3540000 |
| PEL (Permissible) | 200 ppm |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | 500 ppm |

