An Editorial Look at 2-Ethylbutyric Acid: Past, Present, and Promise
Tracing the History of 2-Ethylbutyric Acid
Curiosity has always driven people to track down new chemicals, figure out their potential, and refine the ways we make and use them. 2-Ethylbutyric acid, a carboxylic acid with the formula C6H12O2, traces roots to mid-20th century organic chemistry, stepping out from the footsteps of earlier fatty acid discoveries. The specialty lies not just in its structure, with an ethyl branch adding to the traditional butyric backbone, but in how scientists, after understanding simpler acids like butyric and propionic acids, began to recognize the value of branching in creating new fragrances, flavors, and specialty intermediates. Early research was all about mapping the chemical landscape, and over time, the acid found footholds in academic labs before industry caught on and began exploring its potential.
Getting to Know the Product
2-Ethylbutyric acid stands out in the crowded world of short-chain carboxylic acids because of its distinctive smell, sometimes described as cheesy or even fruity at low concentrations. This dual nature carved places in both synthetic chemistry labs and the flavor and fragrance industry. Its uniqueness also comes from the structural twist added by the ethyl branch, changing how it interacts with enzymes and other substrates, and offering options for building more complex molecules. To anyone who has worked with acids before, opening a well-sealed sample of 2-ethylbutyric instantly leaves a mark—its pungency lingers, pointing to its volatility and readiness to react.
The Physical and Chemical Traits You Can't Ignore
Looking at this acid reveals a clear, oily liquid with a tendency to rise off the bench, spreading its intense odor around. It carries a melting point below room temperature, meaning most encounters happen with the liquid form. Its boiling range hovers around 185 to 186 degrees Celsius. With a relatively low molecular weight and decent solubility in water, alcohols, and other common solvents, it frequently appears as a handy intermediate in synthesis work. The pKa slides into the typical carboxylic acid range, so it participates actively in acid-base reactions. One thing that always stands out: the low flash point. Combined with its volatility, that requires extra care in both handling and storage.
Labeling and Specs That Matter Beyond the Paperwork
Any chemist worth their salt will agree that just reading the technical sheet doesn’t truly prepare you for dealing with 2-ethylbutyric acid. Although bottle labels detail purity levels, presence of water, residual solvents, and even possible trace amounts of related acids, the real test comes out on the bench. Labs focus heavily on confirming purity through gas chromatography, making sure contaminants can’t sneak their way into downstream reactions or products. Yet it’s easy to forget that chemicals like this call for respect at every step, not just a careful look at numbers on a spreadsheet.
Synthesis: The Realities of Preparation
Traditional routes to 2-ethylbutyric acid reflect both the resourcefulness and ingenuity of organic chemists. One reliable method starts with condensation reactions involving aldehydes and alkyl halides, bringing together building blocks through carefully controlled conditions. Another approach uses Grignard reagents to install the ethyl branch onto butyric acid derivatives—a classic in the playbook of 20th-century synthesis. Techniques have evolved to maximize yields and cut down on by-products, especially considering the strong odor and environmental impact of acid vapors. In industry, continuous optimization remains front and center, aiming for greener solvents and energy-light processes that cut down both cost and emissions.
Reactivity and Ways to Tweak the Molecule
2-Ethylbutyric acid joins an active club of carboxylic acids known for versatile chemistry. It reacts briskly with alcohols to form esters, a favorite transformation in the fragrance world, where esters offer much more palatable scents. Simple hydrogenation or halogenation at the alpha position can further expand its chemical legacy, making precursors for pharmaceuticals or specialty additives. The carboxyl group draws interest for amide couplings and salt formation, while the branched chain inspires studies into metabolite mimics and enzyme substrate specificity. In practice, handling its interconversions always brings challenges—especially dealing with volatility and controlling side reactions from impurities that tend to tag along through multi-step syntheses.
Names That Follow the Acid
Chemists and industry veterans often swap between a few monikers for 2-ethylbutyric acid. The systematic IUPAC name pops up in journals, but you’ll also encounter terms like ethylbutanoic acid, 2-ethylbutanoic acid, and sometimes older trade designations in legacy documentation. This tangle of titles sometimes complicates digital searches and regulatory paperwork, so double-checking cross-references keeps everyone on the same page during procurement or literature review.
Living by Safety Rules
From personal experience, the importance of solid safety habits comes through every time I handle this acid. Its sharp odor quickly fills a poorly ventilated room, warning against taking shortcuts on PPE. Direct contact with the liquid irritates skin and eyes, and inhalation can trigger headaches or nausea in sensitive individuals. Working with local fume extraction, even when transferring small amounts, keeps exposure in check. Over the years, laboratories and manufacturers have doubled down on clear labeling, secondary containment, and accessible spill kits. Regular drills and hazard communication help smooth out issues before they become headaches, reinforcing a safety-first mindset that makes all the difference.
Where 2-Ethylbutyric Acid Shows Up
What started purely as a chemical curiosity slowly spread into a variety of application areas that touch daily life, sometimes in the background. Derivatives add body and flavor notes in the creation of synthetic cheese or fruit flavors, illustrating the odd connection between a science lab and a food plant. Perfume and fragrance producers transform the acid into esters that lend subtle nuances to designer scents. Pharmaceutically, its backbones serve as intermediates for specialty actives, especially those exploited for their branching effects, which can tweak biological activity and improve drug properties. In agricultural chemistry, researchers have looked at its utility among plant regulators and feed additives, leveraging both its metabolic and sensory qualities.
Pushing Boundaries in Research and Development
One of the things that excites chemists is how a simple molecule can keep surprising people long after first being cataloged. Researchers dive into ways to harness bio-based feedstocks, aiming to cut reliance on fossil-fuel precursors and find fermentation routes using engineered microbes. Fine-tuning the synthesis of derivatives has led to new candidate molecules for drug discovery and agricultural use, where branching often delivers improved selectivity and metabolic profiles. Collaboration among academic, industrial, and regulatory circles steers efforts toward safer processes, as well as expanding our fundamental understanding of the structure-activity relationships that this acid exemplifies.
What Science Says on Toxicity
Like many carboxylic acids, 2-ethylbutyric acid lands in a grey zone: not wildly toxic but far from inert. Acute exposure can irritate eyes, mucous membranes, and skin, so recurring training and awareness campaigns remain pivotal, especially for new lab staff. Researchers have looked at the potential for long-term harm and have found that while brief encounters pose limited risks, chronic high exposures can alter liver enzyme profiles and disrupt metabolic pathways. Animal studies reveal dose-dependent responses that regulators track closely, forming the basis for workplace exposure guidelines and consumer safety margins when using derivatives in food or fragrance applications. Transparency with up-to-date data helps build public trust, especially in regulatory review.
The Road Ahead: Prospects That Matter
Looking forward, the journey of 2-ethylbutyric acid mirrors broader shifts toward sustainability, safer workplaces, and smarter chemistry. Biobased production pathways attract funding and make headlines, serving both environmental goals and a growing demand for supply chain traceability. As researchers keep improving process efficiency and minimizing unwanted byproducts, downstream industries such as pharma, food, and specialty chemicals stand poised to benefit from cleaner, more predictable intermediates. As I see it, the molecule’s dual roots in flavor and fine chemistry give it a bright future in any domain that values creativity, rigorous safety, and social responsibility—key ingredients for pushing the chemical industry in new, meaningful directions.
What is 2-Ethylbutyric Acid used for?
Real-World Uses and Why They Matter
People don’t walk around talking about 2-ethylbutyric acid. It doesn’t pop up in everyday conversations, but it plays a role once you look behind the scenes—mainly in flavors and fragrances, sometimes in medicine. This organic compound carries a scent that’s anything but subtle—strong, sharp, almost sweaty at high concentrations, but it takes on more pleasant notes in small doses. That strange transformation gives it a special place in the lab and in manufacturing.
Flavor and Fragrance Industry
Think about the artificial flavors in candy or the complex base notes in high-end perfumes. Chemists use 2-ethylbutyric acid to enrich fruity flavors or mimic the tang in foods and drinks. Even in my own kitchen experiments with food-grade esters, a drop or two goes a long way, pushing the aroma from bland to unforgettable. It acts as a building block, not only alone but also as part of mixtures that create specific effects—a whiff of pineapple here or a bit of cheese note there.
There’s also a health angle: the FDA has marked 2-ethylbutyric acid as generally recognized as safe (GRAS) in tiny amounts, so food technologists include it to round out profiles without the risks that follow other chemicals. Safety data supports this—low acute toxicity, low volatility, minimal risk of chronic exposure with proper use. Oversight stays tight, but the industry knows the boundaries and sticks with protocols backed by science rather than just gut feeling.
Pharmaceutical Relevance
Even though most people never spot it on an ingredients list, pharmaceutical companies investigate 2-ethylbutyric acid for more specialized roles. The compound can crop up as a precursor for creating active drug molecules. For instance, researchers use it to help stabilize intermediates during medicinal chemistry syntheses. Certain studies focus on its connection with inherited metabolic disorders, especially in the context of how the body breaks down amino acids, though that’s mostly on the diagnostic side.
Challenges and Solutions
Problems usually crop up in odor management and safe handling. Just a little spill and the smell can take over a space. Having worked in labs, I know that ventilation and sealed storage aren't optional—they're essential. Responsible disposal prevents leaks into the water supply. Strict protocols mean less exposure, fewer complaints, and better safety outcomes for workers.
Some corners of the chemical industry eye greener production techniques, aiming to make 2-ethylbutyric acid from renewable sources. Companies now experiment with bio-based fermentation instead of oil-derived feedstocks. This keeps supply chains less vulnerable and trims environmental impact. Advancements here depend on careful investment, strong regulation, and keeping an eye on cost—nobody wants the price to creep up just because a “greener” label replaces effective chemistry.
Why This All Matters
The demand for nuanced flavors, reproducible fragrances, and reliable drug raw materials drives interest in compounds like 2-ethylbutyric acid. I’ve seen firsthand how something that starts as a sharp-smelling oddball in a vial can bring a product together in ways the average consumer might never trace back to a lab or factory. This chemical stands as a reminder that the little-known details often matter most, whether they shape candy, perfume, or a new therapy.
What is the chemical formula and structure of 2-Ethylbutyric Acid?
What 2-Ethylbutyric Acid Looks Like on Paper
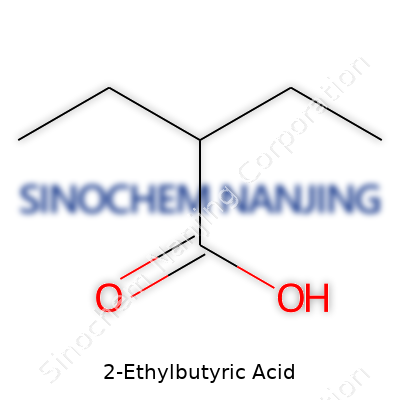
2-Ethylbutyric acid brings together a straightforward chemical formula: C6H12O2. That puts it in the family of carboxylic acids, which means it sports a carboxyl group (-COOH) on one end. Its backbone comes from a six-carbon chain where a two-carbon branch (ethyl group) hops onto the second carbon of a butyric acid structure. This layout leads to a zig-zag of carbon atoms. The structure's real identity comes from the carboxylic acid end and the extra ethyl branch, both influencing how the molecule behaves.
Drawing Out the Structure
If you picture it, the layout gives you something like this: CH3-CH(C2H5)-CH2-COOH. The carboxyl group anchors one end (the -COOH), a two-carbon side chain sticks out from the second carbon, and the rest form that straight butyric skeleton. That branching breaks up the symmetry, nothing fancy, but enough to set its profile apart from plain butyric acid.
Real-World Relevance Beyond the Lab Bench
Chemistry books don’t always make clear why tiny changes in molecular structure matter, but with 2-ethylbutyric acid the impact extends into many corners of daily life. As someone who spent years in a food chemistry lab, I’ve seen this acid hold its own in the world of flavors and scents. It lends a pungent, sometimes cheesy aroma to some cheeses and processed foods. Even tiny tweaks to a molecule’s setup change the taste and smell, which guides how food makers develop new products or improve old ones.
On the industrial side, this acid takes part in building more complex chemicals, plastics, and pharmaceuticals. Understanding its behavior pays off when designing better adhesives or tuning drug delivery. Out in the field, farmers and feed companies check on this acid and others like it to track silage fermentation, which feeds into animal health and farm economics.
Health and Safety—An Underdog Issue
Anyone who spends time around carboxylic acids knows they aren't always friendly. 2-Ethylbutyric acid can irritate eyes, skin, or the respiratory system, and it’s important to follow basic laboratory practice—ventilation, gloves, maybe even goggles if things get messy. Spirited chemical storage keeps surprises to a minimum. Most exposures come through industrial or research work, so public risk stays low, but keeping safety data up to date always deserves attention.
Digging into Solutions for Safer Chemistry
Safer handling grows from experience and thoughtful communication. Labs that train staff to respect volatile acids prevent most accidents. It just takes someone – chemist, food technician, or maintenance worker – sharing practical know-how and where things can go sideways. In wider industry, switching to closed systems reduces vapor exposure and accidental releases. Updating labeling, ventilation, spill kits, and regular audits all cut down harm for workers, communities, and the environment.
The Takeaway from a Simple Formula
A small detail like an ethyl branch converts a basic chemical into something with unique value and challenges. For the science crowd, exploring how 2-ethylbutyric acid fits into everything from cheese-making to pharmaceuticals opens doors to smarter, safer uses. Anyone curious about food or chemistry benefits from keeping an eye on both the structure and broader value of even the smallest molecules.
How should 2-Ethylbutyric Acid be stored and handled safely?
Understanding 2-Ethylbutyric Acid in the Real World
Anyone who works with chemicals like 2-Ethylbutyric Acid knows how strong smells and corrosive burns quickly turn an ordinary day sideways. This organic compound doesn’t forgive mistakes. Even a whiff leaves memories that linger. Along with causing skin or eye irritation, it belongs on the list of substances that never get a free pass in the lab or warehouse. In a busy workspace, extra care isn’t just about ticking off safety boxes; it’s about keeping your coworkers healthy and your context tight.
Storing Without Worries
Glass bottles or stainless-steel containers line the shelves of most chemical rooms for a reason. Over time, I’ve seen plastics weep and deform from acids weaker than this one. Roof leaks or humidity sneak in, corroding containers and causing real headaches. Best practice—keep 2-Ethylbutyric Acid sealed up, tight-fitting caps in cool, well-ventilated spaces. Don’t let the temperature swing. Direct sunlight on an old bottle speeds up degradation, turning a simple storage shelf into a chemical incident.
Never set storage next to food or office supplies. Cross-contamination sounds like a distant problem until you see what a spilled acid bottle can do to a shared lunch fridge.
The Gear That Makes Work Safe
Nitrile gloves have saved my hands from burning more times than I care to admit. Not all gloves work—latex and thin vinyl offer little protection. Lab coats, goggles, and sometimes even face shields offer protection from splashes. The acid’s oily texture makes drops linger against skin. Forgetting safety glasses once leaves a real mark, as even tiny droplets can cause severe pain.
A lot of folks want to cut corners, skipping the heavy gloves or thinking a quick pour won’t splash. That kind of thinking leads straight to the safety shower, and every emergency wash teaches more than a year’s worth of lectures.
Dealing With Spills and Fumes
Ventilation goes beyond opening windows. Fume hoods or extraction fans pull acidic vapors out of the workspace before headaches and coughing start. Don’t keep the acid near anything alkaline or flammable—these reactions snowball fast, causing more damage than most expect.
Small spills happen, but dry sand or soda ash mops it up without a fuss. Don’t mop with water or basic cleaning supplies, since you want to neutralize, not just spread the acid. Always dispose of the waste according to hazardous guidelines—no pouring down the drain.
Training Matters More Than Rules
No safety manual alone keeps people out of the emergency room. Regular training does more than any checklist. Walk through how to check containers for leaks, practice spill response drills, and don’t skip over eyewash and safety shower locations. I’ve watched new team members memorize steps quickly after hands-on practice. Turnover in labs happens, and knowledge gaps open up. Keep everyone in the loop, not just managers.
By focusing on these down-to-earth habits, people stay healthy and workspaces hum along without drama. Simple, clear routines beat scary warning labels every single time.
What are the potential health hazards of 2-Ethylbutyric Acid?
Background on 2-Ethylbutyric Acid
2-Ethylbutyric acid rarely pops up in everyday conversations, but this chemical gets used across different industries. It appears in flavors, fragrances, and sometimes in the making of plastics and resins. I’ve seen cases where it shows up in the workplace or slips into the environment through improper disposal. Though most folks don’t come across pure 2-ethylbutyric acid at home, those working around manufacturing or testing labs could find themselves exposed in higher concentrations.
Short-Term Health Hazards
If you've ever caught a whiff of it, you'll know that 2-ethylbutyric acid has a sharp, unpleasant smell. Short bursts of exposure can irritate the nose, eyes, and throat. Think about the burning feeling you get with some harsh cleaning products — only this time, your body sends up warning signs fast. The eyes begin to sting, and your throat turns scratchy. Skin contact leads to redness or a rash, especially without protection.
Breathing in larger amounts ramps up the discomfort. Workers can get headaches, dizziness, or start to feel queasy. Sometimes these symptoms can be passed off as just a bad day, but if people keep working near this stuff, the risks start stacking up. I’ve read about cases where repeated, short-term exposures led to chronic symptoms, making it tough for some workers to pinpoint the actual cause.
Long-Term Health Impacts
The real issues start to show over time. Ongoing contact with 2-ethylbutyric acid can lead to long-term problems with breathing. The lungs might start feeling tight, or people might notice a nagging cough. Some research suggests potential organ toxicity, particularly affecting the liver and kidneys, when individuals experience regular or high exposures. This sort of damage doesn’t show up overnight. It creeps in, often going unnoticed until symptoms become severe. Animal studies count as early warning signs here — researchers have reported changes to organ function after prolonged exposure.
I’ve also seen how constant skin contact produces persistent dermatitis. For folks who already have sensitive skin or underlying allergies, 2-ethylbutyric acid can make things worse. It attacks the natural barriers of the skin and leads to chronic conditions that sometimes need medical help to manage.
Environmental Concerns
2-Ethylbutyric acid not only affects individual workers but can also slip through cracks and leak into water or soil. Once it enters the environment, it can impact aquatic life since many small species lack the natural defenses needed to handle strong chemicals. These effects ripple up the food chain, causing larger impacts that reach well beyond the original source of release.
Improving Safety and Reducing Hazards
Workplaces have a responsibility to address exposure risks. Good ventilation in storage and handling zones knocks down concentrations in the air. Personal protective equipment, including gloves and eye gear, helps create a reliable frontline defense. Training workers to spot symptoms early keeps problems from snowballing.
For people who might use products containing 2-ethylbutyric acid at home, it pays to keep an eye on labels, especially for imported goods or industrial cleaners. Disposing of chemicals through proper channels matters, too. Local waste programs usually provide safer options to avoid pollution and protect water supplies.
Staying informed about safety data sheets and updated research gives everyone — from manufacturers to everyday consumers — a solid footing when it comes to managing risks tied to chemicals like 2-ethylbutyric acid.
Where can I purchase 2-Ethylbutyric Acid and is it available in different grades?
Locating 2-Ethylbutyric Acid for Purchase
A search for 2-Ethylbutyric Acid usually begins with specialty chemical companies or laboratory suppliers. This isn’t one of the chemicals you pick up at a local hardware store or pharmacy. These compounds often come from companies that have experience supplying research, industrial, or even pharmaceutical materials. Some of the trusted names in the business—Sigma-Aldrich, Thermo Fisher Scientific, Tokyo Chemical Industry—offer clear listings for chemicals like this. Representatives don’t just move product; they also help answer questions about quality, batch traceability, and shipment documentation.
Each supplier asks for details about your intended use. If you don’t have a license, or aren’t affiliated with a lab, sales teams may turn you away. Regulations matter—right down to purchase permits and storage requirements. I learned early on that these measures safeguard both buyer and supplier, especially since some acids can become hazardous if handled without training or proper gear. If a supplier doesn’t ask clarifying questions, or ships without verifying your identity, that’s a red flag.
Grades of 2-Ethylbutyric Acid
Grades reflect the purity or the intended use of a compound. Most chemicals like 2-Ethylbutyric Acid come in more than one grade, such as laboratory, reagent, or industrial. The highest purities often serve research or pharmaceutical development. The next step down—technical or industrial grade—shows up in large-scale manufacturing or flavor industries.
Certified labs demand detailed certificates of analysis. Engineers and process techs in manufacturing want to see stable composition and clear physical properties. Lower grades sometimes harbor minor impurities, so buyers need honest communication with the supplier. For specialized needs, custom synthesis may enter the picture. This route seems expensive, but it’s effective for projects where off-the-shelf products don’t deliver. Once, a colleague needed an ultra-pure acid for a chromatography project; every supplier pushed their “analytical” grade, but only one could provide impurity breakdowns down to fractions of a percent.
Why Grade and Source Matter
The consequences of using the wrong grade look subtle on paper, but those details cascade into real-world problems. In research, even a small contaminant can tip a reaction or skew data. Industrial-scale applications might not demand analytical-grade acid, but erratic purity means inconsistent end products. Customers trust that a manufacturer stands behind their process.
Traceability didn’t always get this much attention. Industry leaders and regulators watched high-profile contamination crises—some involving pharmaceuticals, others with food additives. Public trust only returns when businesses adopt solid quality control practices and keep open lines with chemical suppliers. I once saw an entire process line shut down for weeks due to an impurity traced back to a batch of raw acid. That sort of loss teaches a company the value of sourcing and solid documentation.
What to Look for in a Supplier
A good supplier answers questions about purity, batch origin, handling, and even ethical sourcing. I always ask for safety data sheets and double-check import restrictions, especially for chemicals that raise export concern in certain countries. Real experience matters; if a supplier dodges questions or can’t show compliance paperwork, it’s time to look elsewhere.
Better suppliers train their support staff to work with scientists, engineers, and even regulatory officers. They offer support if shipping gets tangled or if you need to troubleshoot downstream effects.
Making Safe and Informed Choices
2-Ethylbutyric Acid is accessible if you know where to look and meet requirements for its intended use. Every step in the purchase—checking grades, reading through documentation, verifying storage and transport—fits into a bigger playbook of safety and responsibility. That’s the only way to keep projects moving and teams safe.
| Names | |
| Preferred IUPAC name | 3-Methylpentanoic acid |
| Other names |
2-Ethylbutanoic acid Ethylbutyric acid 2-Ethylbutanoate 2-Ethylbuttersäure |
| Pronunciation | /tuː ˌɪθ.əlˈbjuː.tɪr.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 88-09-5 |
| 3D model (JSmol) | `/AAAACCC1CCC(C)CC(=O)O1` |
| Beilstein Reference | 1209072 |
| ChEBI | CHEBI:30897 |
| ChEMBL | CHEMBL50604 |
| ChemSpider | 11110 |
| DrugBank | DB04132 |
| ECHA InfoCard | ECHA InfoCard: 100.009.580 |
| EC Number | EC 211-209-6 |
| Gmelin Reference | 151188 |
| KEGG | C05975 |
| MeSH | D017766 |
| PubChem CID | 10756 |
| RTECS number | EL6470000 |
| UNII | 99Y147NV4O |
| UN number | UN2529 |
| Properties | |
| Chemical formula | C6H12O2 |
| Molar mass | 116.16 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | pungent; cheesy; sweaty |
| Density | 0.923 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | 0.97 |
| Vapor pressure | 0.16 mmHg (25°C) |
| Acidity (pKa) | 4.88 |
| Basicity (pKb) | 10.73 |
| Magnetic susceptibility (χ) | -58.4 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.414 |
| Viscosity | 2.15 mPa·s (25 °C) |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 212.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -467.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3346.7 kJ/mol |
| Pharmacology | |
| ATC code | A16AX11 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H314 |
| Precautionary statements | P264, P280, P301+P330+P331, P303+P361+P353, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 2-2-0-W |
| Flash point | 143 °C |
| Autoignition temperature | 434 °C |
| Explosive limits | Explosive limits: 1.1–6.1% |
| Lethal dose or concentration | LD50 oral rat 2040 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1800 mg/kg |
| NIOSH | EW3675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2-Ethylbutyric Acid: Not established |
| REL (Recommended) | 25 mg/L |
| IDLH (Immediate danger) | IDLH: 300 ppm |
| Related compounds | |
| Related compounds |
Butyric acid 2-Methylbutyric acid Isobutyric acid 3-Methylbutyric acid |

