2-Ethylbutylamine: The Substance Behind Science and Industry
A Glimpse at the Journey
Walking through the timeline of organic chemistry, it’s odd how little spotlight shines on amines like 2-ethylbutylamine. Early researchers in the mid-20th century mapped simple alkylamines with little fanfare, mostly because the world cared more about the big breakthroughs, the obvious game-changers. Still, every layered synthesis, every refinement in amine isolation, owes something to the likes of 2-ethylbutylamine. As the field pivoted toward complex pharmaceuticals and high-performance materials, such "simple" molecules often acted as silent partners, offering up the backbone for various tailored derivatives. It's these foundational blocks—and the creativity scientists bring when working with them—that keep the modern lab running.
From Formula to Warehouse: A Profile
2-Ethylbutylamine doesn’t turn heads on paper: an alkylamine with the formula C6H15N, a basic chain structure with a bit of a branch. It looks like thousands of others, but in the sea of molecular variety, even small differences can open new paths. This compound shows up as a clear, colorless to light yellow liquid, usually carrying the sharp, fishy scent familiar to many amines. Storage often happens in drums or sealed containers, since it decides to evaporate quickly if left uncapped. From paint hardeners to organic syntheses, this molecule does a surprising amount of heavy lifting once researchers and manufacturers get ahold of it.
Beneath the Surface: Physical and Chemical Choices
2-Ethylbutylamine boils somewhere between 115 to 117 °C, sliding into the liquid phase at room temperature. It mixes well with water and plenty of organic solvents, which makes it handy for reactions and blending in both research and industrial settings. As a primary amine, it’s ready to participate in all the classic amine reactions. The slightly branched alkyl chain means shifts in reactivity compared to the straight-chained butylamine, offering subtle handles for chemists designing functional materials or specialty intermediates. One can’t ignore its basic nature; people rely on the characteristic reactivity with acids, isocyanates, and halides when plotting out syntheses.
Technical Labels and Identity Crisis
Nobody likes confusion on a lab bench, so 2-ethylbutylamine usually arrives correctly labeled, sometimes also called 2-ethyl-1-butanamine or 2-ethylbutan-1-amine. These alternate names trace back to the ways chemists choose to count carbon atoms on the chain. The labeling shows compliance with chemical inventory rules—people often double-check CAS numbers and notify about amine content due to the potential for toxicological effects.
Getting There: Preparation and Practice
Creating 2-ethylbutylamine draws from classic amination techniques. The route often starts with 2-ethylbutyl halides or alcohol derivatives, typically undergoing substitution with ammonia or amination with a reducing agent. Reductive amination, using 2-ethylbutanal and ammonia with a reducing touch, streamlines the process. There’s skill in purification, since side reactions easily introduce other amines or unwanted byproducts. Operators adjust temperature, solvent, and reagent ratios to maximize yield while minimizing waste—a testament to both chemistry’s constraints and the persistence of the human workforce to improve on old recipes.
Pushing the Chemistry: Reactions and Possibilities
On the bench, 2-ethylbutylamine acts as a nucleophile, attacking electrophiles ranging from alkyl halides to activated carbonyls. It reacts with acids to form salts useful in further synthesis or separation schemes. Functional group protection uses its amine group, allowing selective transformations without scrambling the rest of the molecule. Modification, often by acylation or sulfonation, has bred new intermediates for agrochemicals and pharmaceuticals. Research groups sometimes build longer chains from this base, looking for new surfactants or specialty polymers. The way this amine slots into multi-step syntheses keeps surprising even seasoned chemists—sometimes it’s all about the right branching on a chain.
What’s in a Name?
Every scientist knows the pain of searching databases for a compound under its alternate name. The synonyms pile up: 2-ethylbutan-1-amine, 2-ethyl-1-butylamine, sometimes simply "ethylbutylamine." Databases like PubChem, Reaxys, or Chemical Abstracts Services track all these, aiding researchers who cross-check data from synthesis planning to toxicity studies, and ensuring the right bottle lands on the right lab shelf. The accuracy in naming prevents batch errors and helps meet compliance—and in a regulated environment, one bad label can spell disaster.
Practicing Safe Science
2-Ethylbutylamine isn’t especially notorious, but one sniff tells you to treat it with caution. Exposure can cause skin and eye irritation, while inhalation might trigger headaches or nausea. Labs use gloves, fume hoods, and eye shields not out of paranoia, but through years of learned safety—anyone who’s accidentally splashed amines knows how fast their effects set in. Transport and storage guidelines, sometimes set by local and national agencies, require tight lids, clear labels, and secure handling. As with many chemicals, spills mean both environmental and health issues, and cleanup teams rely on absorbent pads and neutralizing agents to limit risk.
Out in the World: Applying the Compound
Plenty of fields draw on chemicals that sound esoteric to outsiders, and 2-ethylbutylamine is no exception. Its most common uses stem from its ability to act as a precursor, forming building blocks for pharmaceuticals, pesticides, and specialty coatings. With fine-tuned amine functionality, it gets pulled into polymer science, turning up as a modifier in epoxy hardeners or as an intermediate in resin production. The agricultural industry sometimes uses derivatives as herbicides or pest control aides. Drug-makers experiment with it in the search for new therapeutic agents—small alkylamines remain valuable components when optimizing biological activity. Some technical applications rely on amines for corrosion inhibition or complex formation; here, subtle structural changes in the side chain can shift effectiveness dramatically.
Peering Ahead: Research and Development
These days, research doesn’t settle for minor modifications. Labs hunt for novel amine derivatives with improved properties—better solubility, reduced toxicity, or added specificity in catalysis or binding. Modern combinatorial chemistry and automated reaction platforms lean on molecules like 2-ethylbutylamine, since their branching opens doors closed to straight chain amines. Synthetic chemists count on the amine group’s reactivity to test new catalytic cycles, greener production routes, or more selective pharmaceutical transformations. Advances in separation science, such as chiral chromatography, depend on precisely tailored amines. This hands-on tinkering, the kind you see in research notes covered in scribbles and crossed-out ideas, pushes 2-ethylbutylamine beyond its reputation as "just another amine."
Toxicity: Knowing the Limits
Some amines turn up in scary headlines, but toxicologists rate 2-ethylbutylamine as moderately hazardous—handle it with care, but don’t expect catastrophe from a momentary encounter. Long-term exposure or ingestion raises concerns about organ damage, respiratory effects, or sensitization. Research into breakdown products, metabolic fate, and bioaccumulation continues. Regulatory reviews flag the importance of monitoring workplace air and wastewater for any trace contaminants. Those who have worked directly with alkylamines often advocate for regular refresher training and investment in better ventilation, a lesson learned from preventable accidents and near-misses in less regulated times.
Looking Down the Road: Where Next?
If history teaches anything, it’s that amines like 2-ethylbutylamine evolve with the industries using them. There’s always a demand for reliable intermediates, but the future calls for smarter, safer, greener chemistry. Researchers probe biosynthetic routes or look for recycling-friendly processes, aiming to cut waste and energy use. In pharmaceuticals, the hunt for next-generation drugs means even old "workhorse" amines get their chance to shine as new building blocks. Tackling toxicity will require not just new handling protocols but innovations in downstream processing and waste management. Chemical manufacturing, already under the microscope for its environmental footprint, will press for products that combine performance with sustainability—maybe leading to bio-based versions or novel derivatives of 2-ethylbutylamine. A simple molecule, yet one that keeps inviting new questions, offering new answers, and shaping the work of anyone invested in the future of chemical science.
What is 2-Ethylbutylamine used for?
Chemicals that Shape Modern Life
Understanding chemicals like 2-ethylbutylamine sheds light on how so many everyday products come together. I’ve seen the impact of specialty amines in everything from crop protection to next-generation materials. 2-Ethylbutylamine stands out as one of those building blocks that keeps winding up behind the scenes in several crucial industries.
Pharmaceutical Pathways
The right chemical backbone makes or breaks a new treatment. In lab projects I’ve worked on, amines like this one push reactions along and become part of complex molecules. Medicinal chemists use 2-ethylbutylamine to start or adjust synthetic pathways, and it fits well during the creation of pharmaceutical intermediates. Beyond being a reactant, it can end up as part of the side chains in drugs. This helps improve how a medicine moves through the body or how long it stays active. Research papers confirm its presence in exploring painkillers and antiviral compounds, giving chemists more flexibility with design and function.
Agrochemical Synthesis
Modern crop protection calls for molecules that ward off pests without causing extra harm. Agriculture relies on a steady stream of intermediates, and this amine finds work in herbicides and pesticides. Rising global food demand keeps pushing farmers to use smarter solutions, and the specialty amine sector keeps answering that call. I’ve heard industry chemists mention using 2-ethylbutylamine for making selective herbicides—the type to target weeds without hurting crops. Innovation in agrochemicals continues to depend on reliable, high-quality amine supplies.
Everyday Materials and Industrial Uses
Industrial coatings, dyes, and plastics all have a more complex chemistry than people imagine. Chemical firms need amines to help shape formulations or trigger polymerization. 2-Ethylbutylamine’s branched structure offers a specific kind of reactivity, so resin and adhesive production lines count on it for making sturdy bonds and boosting product stability. Factories use it to initiate reactions in polymers found in paints, hard plastics, and specialty adhesives. I’ve seen patent filings that mention its use for rubber curing agents and corrosion inhibitors in metalworking fluids. The chemical industry recognizes its performance in situations where durability keeps things running smoothly.
Why Handling and Traceability Matter
Safety should always top the list. If you’ve worked around chemical processes, you know how easily small mistakes multiply. 2-Ethylbutylamine has toxic properties—direct exposure or breathing in vapors may cause immediate health issues. Workplaces must always use protective gear, exhaust systems, and strict spill protocols. The European Chemicals Agency tracks its handling closely, and this push for traceability helps reduce long-term risks. Responsible companies report incidents and follow established transportation methods to avoid leaks and exposure. This focus on stewardship means that workers, communities, and downstream users gain some peace of mind about the chemicals entering the supply chain.
Supply and Future of Amines
Global shifts like new medical needs or rising food production continue to drive amine demand. Plants in China, Europe, and North America keep ramping up delivery. Still, future supply could run into issues if producers ignore worker training or environmental protections. Smart companies collaborate with regulators to develop safer substitutes where possible and invest in greener routes—like using renewable feedstocks or minimizing waste. I’ve seen effective partnerships produce substitutions that cut hazards without sacrificing product quality.
What is the chemical formula of 2-Ethylbutylamine?
Diving Into Organic Chemistry’s Building Blocks
Organic chemistry often feels like a maze of confusing names and far-off scientific jargon. Yet for many who work in laboratories, teach, or rely on medical and industrial products, deciphering chemical structures becomes as ordinary as reading a street map. Take 2-ethylbutylamine for instance. This compound might sound intimidating, but its formula—C6H15N—breaks down into a simple arrangement of carbon, hydrogen, and nitrogen. Not just numbers and letters on a page, these symbols form the backbone of a compound with practical uses and safety considerations that count in both research and manufacturing settings.
What Sets 2-Ethylbutylamine Apart?
Picture a butyl group, a staple four-carbon chain in organic chemistry, and attach an ethyl group to its second carbon. Then swap a hydrogen with an amine group (NH2). That's the skeleton behind C6H15N. Locating the amine on the right carbon changes its chemistry. Compared to regular butylamine, 2-ethylbutylamine behaves a bit differently during reactions and when mixed with other chemicals, mainly due to the added branch on its chain. It’s small tweaks like these that change how a compound interacts with the world—from solubility in water, to volatility in open air, to how it fits in pharmaceutical recipes.
Why Knowing the Formula Matters
Clarity about structure means safer labs and better science. I’ve known more than one lab partner who mistook compounds that looked alike at first glance. That accident can lead to wasted samples or worse, harm through unexpected reactions. Using the exact formula C6H15N for 2-ethylbutylamine lets chemists order the right substance, workers prepare safe storage conditions, and environmental experts plan disposal that respects local regulations. It’s not just about crossing your 't’s and dotting your 'i’s on a lab sheet; it’s about keeping hands, eyes, and lungs out of harm's way.
Real-World Applications and Challenges
2-Ethylbutylamine finds a home in labs where new drugs or agrochemicals get their start. Its unique skeleton sometimes fits in synthetic pathways where other amines don’t cut it. Still, every step from delivery to disposal needs special attention. Amines have a reputation for irritating skin and lungs; they can even corrode some storage materials if you don’t choose wisely. Mistakes with labeling or mixing can trigger regulatory fines or endanger workers. In my experience, chemical supply rooms often benefit from double-checking both formulas and chemical names before anything comes off a truck—or worse, gets opened without proper gear nearby.
Building Knowledge, Strengthening Safety
Educators, chemists, and industrial workers can all agree: memorizing formulas isn’t just for passing exams. It underpins safety training, regulatory paperwork, and everyday troubleshooting. The more a team knows about what actually sits in each bottle or drum, the more time and money everyone saves later. Digital inventory systems or routine in-person checks can help spot formula mix-ups before they snowball into bigger problems. Anyone working around chemicals gains from making formula literacy part of their training. Precision in details like chemical formulas links directly to reliability, trust, and safety across the sciences.
Is 2-Ethylbutylamine hazardous or toxic?
Looking at the Chemical Up-Close
Most people have never heard of 2-ethylbutylamine unless they work in a lab or read safety data sheets for fun. The name sounds complicated, but it comes down to a simple aliphatic amine—a group of chemicals that usually smell fishy and get used in all sorts of niche manufacturing operations. You might find this stuff in research settings, industrial synthesis, or very specialized chemical applications. Its use isn’t widespread, so you won’t likely trip over a barrel of it at the corner store.
Risks You Shouldn’t Ignore
Toxicity always matters with industrial chemicals. Inhaling, swallowing, or just getting the stuff on your skin can prove risky. Studies and manufacturer data show amines like 2-ethylbutylamine irritate the eyes, nose, lungs, and skin. Acute exposure means you might end up coughing, sneezing, or feeling a burn on your hands. Long-term risks don’t get a lot of research since few folks handle this chemical daily, but the chemical cousins in this group can cause chronic issues if you don’t follow safety practices.
The chemical gives off vapors that sting. Labs require people to use gloves, goggles, and proper fume hoods to handle amines safely. I’ve seen coworkers try to skip the gloves once to move things along—and the burning sensation changed their minds pretty quickly. Exposure to even small spills left untreated causes redness or blisters within a few minutes. Emergency eyewashes get used more than you’d think, because a splash to the eye feels downright awful. These aren’t the kind of risks you want to learn about through experience.
Understanding Toxicity Data
Most industrial safety data for 2-ethylbutylamine points to moderate acute toxicity. Oral and dermal exposure data suggests that a certain amount—often only a few hundred milligrams per kilogram—can produce harmful effects in animals. There is little public human toxicity data since it isn’t something you’d find outside specialty chemical production, but it’s safe to assume handling without protection will lead to serious problems. Chronic toxicity data remain incomplete, which makes people rely on what’s known about similar amines. Allergic reactions, respiratory distress, and nervous system effects happen with high enough exposure.
Workers using chemicals without clear, up-to-date hazard labels put themselves at risk. That’s a lesson taught in every chemical handling course, and it holds true even for substances not famous for toxicity. For 2-ethylbutylamine, all the small print insists on protection because it attacks mucous membranes, skin, and eyes.
Steps Toward Safer Handling
Simple choices go a long way in chemical safety. Fume hoods, nitrile gloves, goggles, and proper storage keep accidental exposure close to zero. Facilities store amines in ventilated, cool rooms to stop vapor build-up. Anyone using this substance should understand spill control and emergency procedures. Eye washes and showers need to be kept clear, with clear signage so nobody loses time figuring out where to go if things go wrong.
The bigger picture calls for constant training and honest reporting about every accidental exposure. Most companies run routine risk assessments. Authorities like OSHA in the US require that handlers follow rules on labeling, personal protective equipment, and emergency response. By sticking to well-tested protocols, the chances of hazard drops way down.
If you don’t work with industrial amines, you won’t ever come across 2-ethylbutylamine in daily life. But in environments where people do handle it, the rules matter. Ignoring them risks painful, costly mistakes that no one enjoys cleaning up afterward.
How should 2-Ethylbutylamine be stored?
Understanding the Risks Behind the Chemical
2-Ethylbutylamine shows up in research labs and specialty manufacturing. Anyone who’s worked around amines knows one thing—they often have a strong, unpleasant odor. Even a small spill in the wrong place becomes unforgettable. This one isn’t much different. The liquid burns easily, releases fumes, and reacts with acids and oxidizers. It’s hard to forget that such small molecules can pack quite a punch if they don’t stay under control.
Why Storage Choices Make a Difference
Fires and hazardous vapors aren’t rare headlines when chemicals are involved. The stories don’t start with a dramatic event—they grow out of small lapses in how folks handle and store what’s on the shelf. I watched folks get headaches and double over coughing just because a seal failed and fumes spread through a room. This isn’t just annoying; chronic exposure can tear up the respiratory tract, eyes, and skin. Proper storage offers more than regulatory compliance or a checked-off safety talk; it keeps people and working spaces healthy.
Digging Into the Practical Steps
A science teacher once told me that a cool, dry, well-ventilated spot does a lot more for chemical safety than a locked door. I’d add that chemistry isn’t forgiving, so don’t store this near acids or oxidizing agents. Even a lazy squirt of acid in a shared fume hood can set off a dangerous chain reaction. Select a tight-sealing container made from compatible plastic or glass. Metal caps and cheap plastics risk cracking, corroding, or sparking reactions if fumes sit around long enough.
Most chemicals don’t announce themselves before acting up, but 2-Ethylbutylamine’s fumes do. If you catch a whiff, a leak likely hides somewhere. Never stash this liquid above eye level—lids pop open, people bump shelves, gravity works its magic. A chemical burn on the face or in the eyes doesn’t heal fast. Since it’s flammable, keep it far from any source of heat or spark. I’ve seen coworkers use makeshift storage rooms with knotted extension cords running everywhere. Even vapors near a faulty wire can spell disaster.
Attention Beyond the Basics: Labels, Inventory, and Training
In any workplace, sharp labels and up-to-date inventory make all the difference. Quick access to material safety data sheets shouldn’t fall by the wayside or get lost in a binder at the back of a cabinet. The best teams I’ve worked around posted clear instructions above storage areas. That meant less improvisation—and fewer mistakes. Regular checks for leaks, damaged containers, or signs of chemical breakdown help, because the simplest errors bring the worst surprises.
Real Solutions—Not Just Rules
No checklist or OSHA poster can replace daily habits. Spilled droplets left to dry on a counter, lids not twisted tight, bottles shoved together just because there’s no room—all these slip-ups put everyone in the space at risk. Training isn’t a ritual. Bring in real-life stories of close calls to drive the point home. Sometimes the story lingers long after the lesson. Store 2-Ethylbutylamine like the consequences matter, because they always do.
What are the safety precautions for handling 2-Ethylbutylamine?
The Hazards Nobody Should Ignore
Plenty of folks working in labs or plants run into chemicals like 2-Ethylbutylamine almost every day. From personal experience in industrial settings, this stuff demands respect. A colorless liquid with a sniff like ammonia, it will sneak up on you quick if you aren't careful.
Breathing in those vapors leaves eyes and lungs burning. Spilling it on skin or getting a whiff too close to your face brings headaches and a scratchy, irritated feeling that lingers. This isn’t the kind of chemical anyone wants floating around without good ventilation.
Personal Protective Equipment: Not Optional
Goggles, gloves, and lab coats aren't fashion accessories here; they’re armor. Nitrile gloves hold up best against 2-Ethylbutylamine since latex breaks down too fast. Experience says never cut corners with gloves, even on “just a quick job.” I learned this rule sweating through a rushed transfer one summer, and a minor splash left skin tingling for hours.
Face shields matter too if splashing seems likely. The vapors travel, so a well-fitted respirator with organic vapor cartridges just makes sense. Too often, the temptation is to skip a mask if working under a fume hood, but mix-ups happen and the consequences feel lousy.
Ventilation: Keep the Air Moving
Open air moves chemicals away from your lungs. Fume hoods make for a safer workspace. Pulling air away from the operator and out of the workspace keeps vapor concentrations low. I remember a day the hood was off for maintenance — the difference in air quality was clear right away. No one stayed in the room long out of fear or good sense.
Handling and Storage Tips Learned the Hard Way
2-Ethylbutylamine stays happiest in tight, labeled containers made out of glass or certain plastics, far from acids or oxidizers. Keep it cool and dry; hot, damp rooms set the stage for nasty surprises. I once saw a bottle left on a window ledge, light pouring in, and days later the pressure inside felt dangerous. Flammable liquids want respect, not sunbathing opportunities.
Spills need quick, calm action. Absorb with sand or special spill kits, never sweep up barehanded. Small leaks disappear fast if everyone knows the drill and keeps cleanup supplies nearby. Regular training sessions help; there is no substitute for muscle memory in an emergency. I’ve seen confusion cost extra minutes that let fumes build up, and it never ends well.
Medical Response and Next Steps
If 2-Ethylbutylamine gets on skin or in eyes, water turns from a friend to a lifesaver. Rinse right away, keep at it for at least 15 minutes. The one time a coworker hesitated, a rash developed almost immediately. If anyone feels dizzy or sick, fresh air makes a difference, and calling for medical help shows you take safety seriously.
Workplace Culture Matters Most
Trust comes from seeing supervisors walk the walk, following the same safety steps they ask from the team. I’ve worked under leaders who shrugged off precautions and paid for it with accidents. Everyone breathes easier knowing rules protect real people, not just checkboxes on a safety audit.
Safe handling of 2-Ethylbutylamine isn't about paranoia. It's about sending everyone home healthy at the end of the shift. Get the gear, follow the steps, and look out for each other. That’s what keeps workplaces running smoothly, without drama or regret.
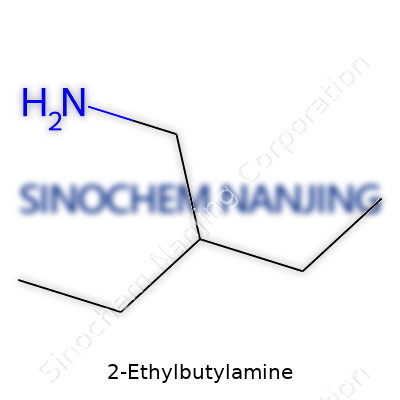
| Names | |
| Preferred IUPAC name | N-ethylpentan-1-amine |
| Other names |
sec-Butyl ethylamine 2-Ethyl-1-butanamine 2-Ethylbutan-1-amine |
| Pronunciation | /ˌtuːˌɛθ.ɪlˈbjuː.tɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 112-70-9 |
| 3D model (JSmol) | `2-Ethylbutylamine|JSmol|C(CC)CCN` |
| Beilstein Reference | 1209247 |
| ChEBI | CHEBI:16934 |
| ChEMBL | CHEMBL1603552 |
| ChemSpider | 16223 |
| DrugBank | DB04203 |
| ECHA InfoCard | ECHA InfoCard: 100_011_942 |
| EC Number | 202-425-8 |
| Gmelin Reference | 8287 |
| KEGG | C05744 |
| MeSH | C045651 |
| PubChem CID | 12009 |
| RTECS number | EK8575000 |
| UNII | 23M66K2A40 |
| UN number | UN2733 |
| CompTox Dashboard (EPA) | DJ1PLKQ3QK |
| Properties | |
| Chemical formula | C6H15N |
| Molar mass | 101.19 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | amine-like |
| Density | 0.771 g/mL at 25 °C (lit.) |
| Solubility in water | Miscible |
| log P | 0.97 |
| Vapor pressure | 1.6 kPa (at 20 °C) |
| Acidity (pKa) | 10.73 |
| Basicity (pKb) | 3.36 |
| Magnetic susceptibility (χ) | -5.43e-6 cm³/mol |
| Refractive index (nD) | 1.416 |
| Viscosity | 0.617 mPa·s (at 20 °C) |
| Dipole moment | 4.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 274.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -83.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4493.8 kJ/mol |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302, H314, H412 |
| Precautionary statements | P210, P261, P280, P301+P312, P302+P352, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 60 °C |
| Autoignition temperature | 255 °C |
| Explosive limits | 1.2% - 7.0% |
| Lethal dose or concentration | LD50 oral rat 189 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 555 mg/kg |
| NIOSH | PH4725000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 200 mg/kg |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
n-Butylamine sec-Butylamine iso-Butylamine tert-Butylamine 2-Ethylhexylamine |

