2-Chloropropionic Acid: A Practical View on Yesterday, Today, and Tomorrow
Historical Development
Chemicals with small, simple structures often fly under the radar, but 2-chloropropionic acid has carved out a clear spot in the story of industrial chemistry. If you rewind to the era when early organic chemists started modifying acetic acid and its kin, you find a few folks uncovering new directions by adding just a single halogen atom. That’s pretty much where 2-chloropropionic acid first emerged—out of curiosity and a drive to harness new properties through small tweaks. Not every chemical born from those experiments made a lasting impression, but this one did, mainly because its unique structure proves useful in synthesis work. Over the decades, scientists have poked and prodded at its reactivity, hoping to make life easier for folks who need intermediates for crop protection, medical research, or materials development. While its heyday has never involved the limelight, its lineage runs deep, and chemists have kept finding reasons to stick with it.
Product Overview
2-Chloropropionic acid doesn’t sport much glamour. It serves a purpose in labs and industry because chemists trust its reliability. The molecule, with a chemical formula of C3H5ClO2, consists of a propionic acid backbone with a chlorine atom at the second carbon. That simple change creates major differences in how it reacts and what you can make from it. Its clear, colorless appearance and biting sour odor aren’t winning awards, but those who know chemistry see usefulness behind the plain looks. Unlike big-name compounds splashed across trade magazines, this acid quietly helps as a building block for herbicides, pharmaceuticals, and various performance materials. Each batch—whether sold as a reagent or as a reactant for custom synthesis—reflects years of trial and error.
Physical and Chemical Properties
Look at the numbers: the boiling point hovers a little below 200 degrees Celsius, and its melting point sits well below room temperature. Water solubility runs high, and it likes to pull moisture from the air, which means proper storage takes more than just leaving it in a flask on the shelf. Acidity makes itself obvious with a sharp bite, owed to the carboxylic group clinging tight to protons. Once that chlorine atom enters the picture, reactivity shifts. You end up with a molecule that responds eagerly to nucleophilic attacks, and that same chlorine makes it a decent candidate for substitution reactions. Chemists see these features and start thinking, “How do I make this reactivity work for me?”
Technical Specifications and Labeling
Labeling matters a lot in labs and production plants. When handling 2-chloropropionic acid, accuracy and transparency on the label keep people safe and uphold quality standards. Labs want to see a stated purity above 98%, and the acid content should meet precise measurement benchmarks. Residual solvents, color indicators, and water content all show up in the batch paperwork. Regulatory demands don’t stop at the doors: proper labeling extends into transportation, tied tightly to both hazard communication laws and practical safety on the job. Whether you buy it bottled or sourced in drums, clear labeling doesn’t just play a legal role—it marks the difference between routine work and a headline-making accident.
Preparation Method
Anyone who’s worked with halogenated acids gets familiar with two main approaches. You can start from lactic acid, converting a hydroxy group to a chlorine through the help of thionyl chloride or phosphorus trichloride, both classic reagents with long histories in industrial chemistry. Alternatively, some routes draw from propionic acid itself, turning primary alcohols into chlorides and then oxidizing the side chain. None of these methods run without fuss—byproducts like HCl gas and heat require careful handling, and scaling up production creates new headaches. Each route brings trade-offs in cost, yield, and safety, so operators run constant batch checks to keep output consistent.
Chemical Reactions and Modifications
This acid opens doors for more than one class of reactions. Nucleophilic substitution stands at the front, letting chemists swap out that chlorine for all sorts of functional groups, from amines to alkoxides. It’s a classic example in basic chemistry training, but in the real world, the product you get from a single swap forms the backbone of a herbicide, a drug, or a specialty chemical. You also see it in esterification, where the acid function turns into more complex esters—handy in pharmaceutical research. There’s always curiosity about how to make derivative compounds, so 2-chloropropionic acid shows up in textbooks and protocols that train the next generation of synthetic chemists. Anyone who’s spent hours at a bench knows that versatility remains king, and this acid brings that to the table.
Synonyms and Product Names
You might hear this compound called by names like DL-2-chloropropanoic acid, alpha-chloropropionic acid, or even racemic chloropropionic acid. Naming conventions sometimes trip up newcomers, since the structure stays the same, but end-use sectors—agriculture, pharma, basic chemical supply—tend to prefer one name over another. Picking the right synonym doesn’t just help on a spreadsheet. It’s essential in regulatory filings and when searching scientific literature for compatible compounds or reaction precedents. This simple acid, despite its size, has gathered a handful of monikers that reflect the diverse circles it travels in.
Safety and Operational Standards
Nobody wants a routine day in the lab turning difficult because of skipped safety steps. 2-Chloropropionic acid carries risks due to that ever-present chlorine atom and strong acidity. Direct contact sparks irritation; inhale it and you find respiratory discomfort pretty fast. Chemical handling protocols all stress gloves, goggles, lab coats, and solid ventilation. In industry, exhaust hoods draw off fumes, sensors keep tabs on levels in the air, and workers follow strict chemical hygiene rules. Beyond personal safety, environmental standards demand containment—spills shouldn’t leach into water or soil, given the persistent nature of halogenated organics. From the first time opening a bottle to transport and storage, every moment brings a set of clear standards, shaped by regulations but enforced because the stakes—for health and livelihoods—are too high to gamble.
Application Area
Agricultural science makes the most of this compound’s reactivity, transforming it into active ingredients in selective herbicides. Modern weed management depends on fine-tuning chemistry to target unwelcome plants, and 2-chloropropionic acid has powered several success stories on that front. It also helps in making intermediates for medical compounds, especially when controlled chirality matters in drug design. The textile sector dips into halogenated acids for novel polymers. Any researcher who’s pushed past simple alkane chemistry bumps into its usefulness as a stepping stone. Rather than grabbing headlines itself, it plays a foundational part in making more complex molecules available.
Research and Development
Everything in the chemical world moves forward on the back of research. Academics and industry groups alike have pressed to improve both the yield and environmental impact of making and using this acid. Some current work aims to swap harsh halogenation reagents for cleaner, less hazardous ones, while others hunt for catalysts that can lower energy input or selectively produce one enantiomer over the other. Building a better synthesis, often with greener chemistry at the center, matches the growing pressure for responsible manufacturing. Applied research doesn’t stop with producing the acid itself; plenty of attention focuses on making derivatives that bring selective effects in medicine or keep farmland more productive with less chemical runoff.
Toxicity Research
Toxicity always hovers over halogenated acids. Studies in animal models demonstrate clear impacts—central nervous system effects, respiratory tract irritation, and risks to aquatic life at higher exposures. Agricultural use brings special scrutiny since even small amounts leaking into irrigation water can harm wildlife. Occupational exposure studies underscore the importance of keeping airborne concentrations low, as repeated inhalation or skin contact triggers cumulative effects. Ongoing research looks for long-term data on ecosystem impact, but the caution flags run high already. This background knowledge shapes every decision on handling, from storage to waste disposal, and pushes regulatory bodies to keep requirements strict.
Future Prospects
Looking ahead, much of the future for 2-chloropropionic acid hangs on how the chemical industry adapts to stricter pollution controls and the ongoing search for cleaner alternatives. There’s persistent demand for reliable intermediates in pharmaceuticals and agriculture, but the old ways of making halogenated organics face a shrinking window as greener synthesis methods mature. Innovation hasn’t tapped out—bio-based routes and asymmetric synthesis offer hope for chipping away at some persistent safety and environmental risks. The path forward lies in refining efficiency, cutting waste, and proving that a long-established molecule can evolve without leaving a nasty chemical footprint behind. That challenge draws the best out of both chemists and engineers, with every win building safer, more sustainable systems for industry and the communities that border it.
What is 2-Chloropropionic Acid used for?
What Drives the Use of 2-Chloropropionic Acid?
People in labs and factories don’t always get to pick the perfect tool for the job, but some chemicals keep showing up because they simply work. 2-Chloropropionic acid is one of those. It’s a specialty chemical with a small but dedicated following, mainly because of what it brings to the table in organic synthesis and select industries. You probably haven’t seen it on a hardware store shelf, but its fingerprints show up in pharmaceuticals, agrochemicals, and a few unexpected corners.
Pharmaceutical Synthesis: Building Blocks and Beyond
Drug makers spend a lot of time hunting for efficient ways to build complex molecules. 2-Chloropropionic acid works as a useful intermediate in this world. That “intermediate” role can sound small, but think of it like carpentry: the best houses need reliable lumber, and this chemical acts as reliable lumber for active pharmaceutical ingredients. It’s a stepping stone towards amino acids, and some of the derivatives turn up in medicines people rely on every day.
Quality here can’t slip, not even a little. Every batch made for pharma gets tested to ensure it carries the right optical activity—otherwise, the medicine won’t do its job. In this space, regulatory oversight isn’t just a rulebook but a lifeline. Factories producing this compound for drug use operate under strict checks, because any impurity gets magnified as those molecules go through steps and end up in someone’s body.
Crop Protection Chemicals: Feeding the Modern World
If you eat bread, rice, or fresh vegetables, you benefit from chemicals like 2-chloropropionic acid, although you’ll never taste it. It pops up as a building block for herbicides. Farmers rely on these products because hungry weeds don’t care about a field’s bottom line. Without precise chemical controls, yields drop and grocery bills rise. Even so, the debate never ends on the risks and rewards of chemical weed killers. Advocacy groups and scientists keep pushing for safer run-offs, tighter application techniques, and transparency about what goes on crops, not just raw yield.
Specialty Chemistry and Research
Universities and private labs put 2-chloropropionic acid to work as a reagent. Researchers use it to tinker with reaction pathways, build new molecules, and even create specialty polymers. Every so often, a breakthrough makes its way from a benchtop experiment using this chemical into broader use. It’s the silent partner in discoveries—there in the reaction flask, rarely mentioned in headlines.
Risks, Safety, and Better Practices
Folks who handle this acid need to respect it. It causes irritation on contact and brings bigger risks if someone breathes too much of it. Chemistry shops build secure handling setups and enforce training, not because of paperwork, but because mistakes hurt people. Managing disposal also matters, as careless dumping would harm both water and soil. Industrial users partner with waste firms to ensure chemicals get broken down or incinerated far from drinking water.
Sustainability comes up in every review meeting now. The industry watches for new bio-based alternatives. Every year, researchers push boundaries, looking for cleaner feeds or greener solvents. Regulators encourage shifts to less hazardous processes, rewarding companies that cut the footprint of these specialty acids.
Looking Ahead
2-Chloropropionic acid continues to earn its keep for chemists and farmers. Its profile stays far from household names, but its impact stretches into medicine cabinets and kitchen tables. The call grows for safer handling, responsible disposal, and mindful synthesis. For now, expertise and vigilance will keep it useful and keep people safe.
What are the safety precautions when handling 2-Chloropropionic Acid?
The Unfriendly Side of 2-Chloropropionic Acid
Anyone who’s spent time working with organic acids like 2-chloropropionic acid knows that sharp smell and stinging sensation in your nose. I still remember the first time I opened a bottle in college—one careless sniff left me coughing, eyes watering. This chemical packs a punch. Direct skin contact can bring burning or blistering. Breathing in its fumes lands you a nasty headache and sore throat. Let it splash anywhere near your face and you’ll be blinking away tears for a week.
How Accidents Get Started
People make mistakes out of impatience or routine. Maybe you figure gloves aren’t needed for such a "quick transfer." Maybe you open a container beside an air register. Or set down an open beaker somewhere busy and walk away. I’ve seen even seasoned techs skip goggles because nothing “ever splashes.” That sort of attitude spells trouble every time.
No Substitutes for Basics
Wearing the right gear cuts down on accidents before they even have a chance to happen. Not just disposable gloves, but sturdy nitrile types that don’t tear so easily. Safety goggles mean your eyes stay safe even when a pipette jumps from your hand. A thick lab coat shields your skin and keeps sleeves from soaking up spills. Even a mask or face shield makes sense when you’re unsure how much will evaporate during transfers or mixing.
Work inside a fume hood if you can. That way, even if you knock over a flask, those vapors head out the vent instead of into your lungs. Make sure to check the ventilation—you can’t count on an old duct to do all the work if it’s clogged or the air handler’s off for repairs.
Storage and Handling: Small Steps Matter
2-Chloropropionic acid eats through most metals and plastics. Glass storage jugs with tight-fitting Teflon-lined caps stay reliable over time. Label everything with a permanent marker since sticky labels peel up from acid fumes, leaving an unmarked hazard on the shelf. Shoving the container in the coldest, driest cabinet slows down any breakdown and stops dangerous vapor leaks.
As someone who’s cleaned up more than one spill from a leaky bottle left loose on a countertop, I can vouch for keeping acids well away from bases or oxidizers. Even a small accident on the wrong surface means hours of scrubbing and lots of ruined tools.
Emergencies: Know What to Do Before It Counts
Plenty of folks in chemistry labs assume the eyewash and shower are there for someone else. Odds are you’ll turn to them yourself eventually. I’ve been in the room when a labmate splashed acid on his hand. He made straight for the sink and ran cool water over the burn, which probably saved him a trip to urgent care.
Always know the fastest way out and where the emergency kit lives. Spills start off small but panic can turn them into disasters. Neutralize minor spills with sodium bicarbonate, scoop it into a container, and leave the cleanup to those with the right know-how.
Building Smarter Habits
Working with tough chemicals like 2-chloropropionic acid never gets routine, no matter your experience. Regular training keeps safety at the front of everyone’s mind, especially newcomers who haven’t seen an accident up close. Nobody enjoys reading through pages of SDS, but those sheets hold clues—like safe exposure limits, what antidote works for inhalation, and which symptoms signal danger.
Better habits and consistent respect for these acids keep people, equipment, and the workspace in one piece. Taking those extra precautions lets you focus on your work rather than worrying about what could go wrong.
What is the chemical formula and structure of 2-Chloropropionic Acid?
Getting to Know 2-Chloropropionic Acid
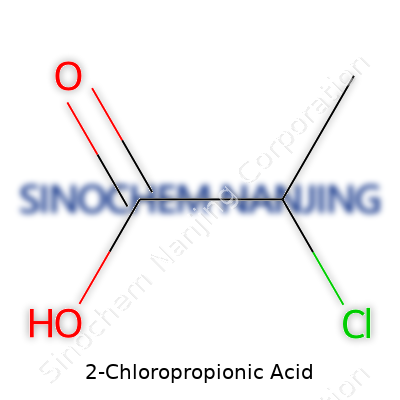
2-Chloropropionic acid carries the formula C3H5ClO2. The compound looks simple at first glance—a three-carbon skeleton with a chlorine atom and a carboxylic acid group pulling its strings. In chemical terms, we are dealing with a derivative of propionic acid, but the presence of chlorine at the alpha (2-) position changes the game in terms of its reactivity and behavior.
Peeling Back the Structure
Visualizing the structure helps. Imagine a chain of three carbon atoms. The first carbon forms the carboxyl group, COOH. The second carbon, the alpha carbon, bonds with a chlorine atom. The last carbon holds down a pair of hydrogens to complete the lineup. In skeletal terms:
HOOC–CH(Cl)–CH3
This setup brings an unusual twist: chlorine pulls electron density through the chain, affecting acidity. Chemists call this the alpha-halogen effect, and it’s not just trivia. In the lab, this means 2-chloropropionic acid turns out more acidic than plain old propionic acid, so it behaves differently in reactions and environmental breakdown.
Why Knowing the Details Matters
I spent part of my college years in organic chemistry labs, wrestling with bottles labeled with haloacids just like this one. The detail that stuck with me: mistakes with simple swapping can mean a failed synthesis or an unexpected reaction with common reagents. That chlorine atom isn’t just window dressing—it punches up reactivity and governs how the compound interacts with bases, metals, and even the human body.
Toxicity and Safety Overlooked
The mere addition of a chlorine changes a harmless acid into something requiring gloves and fume hoods. Sources at the US National Library of Medicine flag chlorinated carboxylic acids for potential toxicity. Smaller molecules like 2-chloropropionic acid may disrupt metabolic processes, especially in aquatic environments. This makes disposal and accidental spills a serious affair—for both the researcher and the environment.
Real-World Uses and Issues
2-Chloropropionic acid hasn’t made its way into day-to-day conversation, but it finds a home in synthetic chemistry. Manufacturers turn to it for preparing more complex molecules, sometimes pharmaceuticals. Given chlorine’s electron-withdrawing power, reactions that call for such acids look to halogenated options when fine-tuned reactivity is needed. This comes at a cost: handling protocols, disposal safety records, and material safety data sheets (MSDS) all need regular dusting off.
Solutions for Safer Chemistry
Experience has taught me the smallest errors with compounds like this can spiral. Training is the low-hanging fruit—teaching newer chemists to read structure diagrams, recognize risks, and treat every halogenated acid with full respect. Laboratories should use proper containment and disposal methods, relying on up-to-date environmental data. Substituting less harmful analogs can minimize exposure, but not every synthesis allows an easy swap. Open communication between academia, industry, and safety regulators keeps everyone on the same page.
Small Details, Big Impact
Understanding 2-chloropropionic acid comes down to seeing beyond a jumble of atoms. The presence of just one chlorine at the right place shifts properties and hazards—and also opens up a world of chemical possibilities. For anyone in the lab or anyone who reads the label on a chemical bottle, these details carry real weight.
How should 2-Chloropropionic Acid be stored?
Understanding Real Risks
2-Chloropropionic acid shows up in plenty of labs where folks handle chemicals that don’t forgive mistakes. As someone who’s spent years around flammable liquids, corrosive acids, and volatile solvents, I’ve learned to spot what makes a chemical tricky. This acid isn’t just another bottle on the shelf. If storage goes sideways, you could end up with dangerous fumes, leaky containers, or corrosive spills. Health risks rise fast because accidental contact and inhalation harm skin, eyes, and lungs, not to mention what happens with big leaks. It only takes one careless moment.
Choosing the Right Spot
At home or on a research team, I always head straight for a cool, dry, and well-ventilated spot. 2-Chloropropionic acid needs separation from bases, oxidizers, or strong acids. These combinations spark off hazardous reactions nobody wants to deal with. Moisture in the air speeds up decomposition or makes fumes. Quality storage matters, and cheap containers mean leaks, ruined labels, or worse.
When you pick out space in a shared chemical cabinet, keep this acid away from anything with nitrogen or amines, or any basic compounds. In my experience, most cabinets come with chemical-resistant shelves for a reason. Placing acid on bare steel or regular wood isn’t smart. Always check secondary containment trays—plastic pans work best since they catch stray drops and keep them from running across the counter. Neither glass nor regular plastic bottles always cut it, so reach for containers with tight seals, made for corrosive acids.
Real-Life Labeling and Handling
I like labeling every container in plain English, not just chemical codes. On busy days, one unreadable or faded label leads to the sort of confusion that causes serious accidents. Writable, chemical-resistant stickers work best. Include the full name, concentration, and clear hazard warnings so nobody grabs the wrong bottle. After seeing unlabeled acids cause a scary near-miss at a university lab, I double-check these labels at the end of every week. It’s worth the extra minute.
Acids like this should never get stored above shoulder level or in high-traffic spots. Even a small jar can leak vapor. Fume hoods or properly vented cabinets lower the risk. I’ve witnessed supervisors stash acids near a window on a summer day, only to find pressure warping the lid hours later—and fumes escaping. A steady, moderate temperature keeps the acid stable and prevents that.
Personal and Community Safety Matters
Safety gear makes all the difference. I’ve cleaned up enough chemical spills to insist on gloves, goggles, and sturdy aprons—not just lab coats—when transferring or opening the bottle. Even the best label doesn’t help if someone opens a container unaware of what’s inside. On big projects, everyone gets a quick rundown of where the acid rests and how to grab spill kits fast. Wash stations and clear signage become non-negotiable. Every prepared environment keeps both individuals and the wider community safe from costly, painful accidents.
For those running chemical inventories, I recommend monthly checks of storage and expiration dates, tossing containers that look corroded or out of date. Reporting leaks or spills straight away supports a culture where safety comes first, not second. That’s a lesson learned from hard experience.
What are the physical and chemical properties of 2-Chloropropionic Acid?
Getting Acquainted with 2-Chloropropionic Acid
2-Chloropropionic acid is a small molecule that scientists and manufacturers often encounter. Its structure is pretty straightforward: a propionic acid core with a chlorine atom attached to the second carbon. The molecular formula is C3H5ClO2. On paper, it might look simple, but the way this chemical behaves, both in nature and in the lab, deserves some real discussion.
Physical Properties Worth Attention
This compound shows up as a colorless, oily liquid at room temperature. The liquid's sharp, pungent odor becomes noticeable even during low-level handling. Anyone working in a lab remembers how quickly 2-chloropropionic acid evaporates; its boiling point hovers close to 162°C, and its melting point falls right around -22°C. The density sits at about 1.24 g/cm³, making it a bit heavier than water. The stuff mixes easily with water and organic solvents, which means accidental spills can spread quickly and create cleanup headaches.
Having spent some time in laboratories handling carboxylic acids, I can vouch for the strong sour smell clinging to your gloves after handling this one. The volatility and odor make good ventilation a must, not only for comfort but also to keep instructors and coworkers happy.
Chemical Behavior and Hazards
Chemically, this molecule brings more to the table than just its acidic proton. The chlorine atom increases its reactivity. Like other carboxylic acids, 2-chloropropionic acid reacts with bases to form salts and with alcohols to yield esters. Yet, the presence of that halogen opens up extra reaction pathways, including nucleophilic substitution. The molecule gets used in organic synthesis because chemists can swap out that chlorine atom, sometimes targeting new pharmaceuticals or agricultural agents.
Beyond its usefulness, the molecule presents safety issues. Many carboxylic acids deserve respect, yet the chlorine attached to the backbone usually indicates heightened toxicity and corrosiveness. The skin and eyes do not appreciate accidental splashes, and inhaling vapors causes irritation. The same reactivity that makes it helpful in a laboratory or factory can create health risks if someone overlooks personal protective equipment or ignores safety data sheets.
Application and Handling Solutions
Industries leverage 2-chloropropionic acid in herbicide production, organic synthesis, and even pharmaceutical development. Back during one of my college internships, I watched researchers use it as a starting material to build up complex molecules for testing. Working with reactive chemicals like this one taught me to keep safety lessons front-of-mind, especially when scaling up reactions.
Solutions for safer use do not require complicated interventions. Proper fume hoods, gloves rated for corrosives, and labeled secondary containers go further than fancy equipment ever could. In academic or industrial labs, regular safety training and clear signage remind everyone of the hazards. Disposal gets a mention too—neutralizing acidic waste, following local guidelines, and documenting every transfer eliminates headaches and regulatory trouble later.
Why Knowledge Matters
Understanding the properties and risks of 2-chloropropionic acid helps keep both workers and end-users safe. It's not just about memorizing a list of melting points or pKa values. It's about seeing how a small shift in chemical structure changes how you work, how you store materials, and how you protect the environment. Sharing these details makes chemistry less about rote memorization and more about thoughtful application—whether in a teaching lab or on the factory floor.
| Names | |
| Preferred IUPAC name | 2-chloropropanoic acid |
| Other names |
DL-2-Chloropropanoic acid 2-Chloropropanoic acid α-Chloropropionic acid Propanoic acid, 2-chloro- |
| Pronunciation | /tuː-klɔːr.oʊ-proʊˈpiː.ɒn.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 600-18-0 |
| 3D model (JSmol) | `load =CC(=O)O | Cl` |
| Beilstein Reference | 1720521 |
| ChEBI | CHEBI:48861 |
| ChEMBL | CHEMBL50696 |
| ChemSpider | 57918 |
| DrugBank | DB04162 |
| ECHA InfoCard | 08fab3ac-8c01-44eb-bfc6-ccd77c1e258a |
| EC Number | EC 201-198-9 |
| Gmelin Reference | 084223 |
| KEGG | C01007 |
| MeSH | D015639 |
| PubChem CID | 69729 |
| RTECS number | EO1575000 |
| UNII | Q145E04Y85 |
| UN number | UN3276 |
| CompTox Dashboard (EPA) | DTXSID6059244 |
| Properties | |
| Chemical formula | C3H5ClO2 |
| Molar mass | 108.54 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Pungent |
| Density | 1.44 g/cm³ |
| Solubility in water | Miscible |
| log P | -0.5 |
| Vapor pressure | 0.37 mmHg (25°C) |
| Acidity (pKa) | pKa = 2.83 |
| Magnetic susceptibility (χ) | -9.74 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.439 |
| Viscosity | 2.06 mPa·s (20 °C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 94.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –(537.6) kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -885.7 kJ/mol |
| Hazards | |
| Main hazards | Corrosive, harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H302, H314 |
| Precautionary statements | P210, P260, P280, P301+P312, P305+P351+P338, P308+P311, P501 |
| NFPA 704 (fire diamond) | 3-2-1 |
| Flash point | 87 °C |
| Autoignition temperature | 510 °C |
| Explosive limits | Lower explosive limit: 2.2%, Upper explosive limit: 10.8% |
| Lethal dose or concentration | LD50 oral rat 820 mg/kg |
| LD50 (median dose) | LD50 (median dose): 820 mg/kg (oral, rat) |
| NIOSH | GM3150000 |
| PEL (Permissible) | Not established |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Lactic acid Propionic acid 2-Bromopropionic acid 2-Iodopropionic acid 2-Fluoropropionic acid |

